Bis(Trichloromethyl) Carbonate: Beyond the Lab Bench
Tracing the Roots: How BTC Emerged
Chemical synthesis often mirrors a society’s ambitions and anxieties, and Bis(Trichloromethyl) Carbonate (BTC) tells its own story here. Researchers introduced BTC in the 1950s, mainly motivated by the search for better phosgene substitutes in acylation reactions. Early references in patent literature reveal a race to develop reactive agents that could avoid some hazards of phosgene gas yet still deliver on reliability and speed. In the decades since, BTC has carved out a place in organic chemistry toolkits, particularly as a reagent for synthesizing carbonates, carbamates, and ureas. Now, I see BTC showing up in the research journals I follow, often in conversations about clean synthesis and efficient route planning for pharmaceuticals—testament to its continuing appeal as chemists tackle today’s sustainability goals.
What BTC Really Is: Not Just Another Carbonate
On paper, Bis(Trichloromethyl) Carbonate comes across as a clear, colorless to light yellow liquid with the formula C3Cl6O3. Its strong, acrid odor usually makes an impression long before anyone starts measuring out quantities. BTC boils at around 94°C under atmospheric pressure, which means it volatilizes easily at room temperature if left uncapped. Its density, hovering near 1.63 g/cm³, and solubility, or lack thereof in water, have practical consequences in bench practice. Chemically, BTC behaves as a potent chloroformylating agent, reacting aggressively with water or alcohols and releasing phosgene as a byproduct in hydrolysis. Safety data sheets don’t mince words: use in a fume hood or face the real risk of acute toxicity.
BTC in Action: Performance, Labels, and What’s On the Bottle
Walking through a supplier’s catalog, you’ll spot BTC labeled under a clutch of names—Triphosgene, trichloromethyl carbonate, or even carbonochloridic acid, bis(trichloromethyl) ester. The bottles usually include hazard pictograms warning of acute toxicity, environmental ambition, and corrosiveness. Purity often exceeds 98% for lab applications, yet grades differ depending on use. The strong smell alone will remind anyone handling it to double-check seals and handling techniques. I’ve learned to keep BTC far from water lines; one careless splash, and you get a whiff of phosgene. Not the kind of mistake you want to make twice.
Getting BTC: Methods and Chemical Craft
The classic preparation route involves the chlorination of dimethyl carbonate or methyl chloroformate with chlorine gas in the presence of a catalyst. This process evolved as environmental restrictions around phosgene tightened, driving chemists to develop scalable routes that cut down on phosgene releases. Industrial producers favor batch or continuous reactors equipped with scrubbers to catch and neutralize byproducts. For laboratory-scale synthesis, the focus falls on controlling temperature and monitoring product purity by gas chromatography. Getting the process right can save hours dealing with side products or hazardous decompositions. Mistakes not only threaten yields but also expose staff to significant risk, so strict procedures are not just bureaucratic—they’re survival strategies.
Field Notes from the Bench: What BTC Does in Reactions
From a technical standpoint, BTC’s biggest draw comes from its ability to deliver carbamates and ureas without dragging phosgene gas into the equation. This property makes it a workhorse for chemists producing polymer precursors, bioconjugates, and agrochemicals. BTC reacts with alcohols to form carbonate esters, vital in protecting groups or as intermediaries for pharmaceuticals. Reactions typically release trichloromethyl chloride as a byproduct, demanding proper capture to keep side reactions in check. Folks working with amines or alcohols often find BTC easier to scale than phosgene-based methods, offering more manageable risk as long as nobody cuts corners with ventilation and PPE.
Behind the Nameplate: Synonyms and What They Mean in the Real World
BTC’s aliases, especially triphosgene, matter in day-to-day lab logistics. Chemists, regulatory officers, and buying agents must speak the same language, or Henry’s Law isn’t the only thing that breaks down. The term “triphosgene” really caught on because it signals BTC’s role as a safer alternative to phosgene. This isn’t just branding—the name guides storage protocols, risk assessments, and emergency plans. If someone asks for triphosgene, staff know to prep gas-detection monitors, check fumehood airflow, and review waste protocols. Within regulatory frameworks, clarity around product names can avoid shipment delays or accidental misclassification, both headaches in their own right.
Making Safety the Standard: Tackling Hazards Head-On
BTC may avoid some of phosgene’s dangers, but it stands as a harsh chemical in its own right. Acute inhalation leads to coughing, shortness of breath, and, in severe cases, pulmonary edema. Contact with skin or eyes rapidly causes burns or irritation. Spending years in the lab, I learned that working with BTC without a functioning fume hood is a ticket to trouble. Safety culture—annual training, spill drills, regular checklists—makes a concrete difference. Modern labs that track air flow, maintain spill kits, and respond quickly to alarms stay ahead in risk prevention. The standards reflected in GHS labeling, coupled with local employer mandates, set out clear operational rules: sealed, labeled containers stored in ventilated, temperature-controlled rooms, with no exceptions for “just a quick experiment.” Failures in this arena rarely end softly.
Where BTC Hits the Ground: Uses and Real-World Needs
BTC doesn’t get the press of a blockbuster drug or a splashy consumer product, yet its application range shapes key industrial and research frontiers. In pharmaceutical manufacturing, BTC activates amines for making carbamate drugs or peptide couplings. Agrochemical producers use it as a step in pesticide synthesis. Polymer chemists value its reactivity for building blocks in advanced materials, while analytical chemists rely on its derivatization features when prepping samples for mass spectrometry. Folks in academia push BTC’s envelope, using it in new routes to heterocycles and potential drug scaffolds. These applications keep demand solid, with manufacturers racing to improve quality, batch consistency, and, more recently, green credentials.
Research and Innovation: Chasing Better Results
Research teams continue to probe BTC’s potential, exploring catalytic alternatives and greener routes that ease environmental pressures. I’ve followed a few recent studies using alternative solvents or solid-supported catalysts, looking to lower waste and cut energy demands. Efforts also focus on recycling or recapturing chlorinated byproducts. Academic and industrial partnerships, often supported by public funding, advance these projects. The biggest advances I’ve noticed come from microreactor and flow chemistry systems, which let operators keep reaction volumes low and control hazardous intermediates with precision electronics. Lab automation and real-time monitoring further reduce exposure risk, letting researchers push the limits while keeping safety and quality in the spotlight.
Toxicity Under Scrutiny: What Science is Saying
BTC’s toxicity profile makes it an object of deep regulatory scrutiny. Chronic exposure triggers respiratory and skin sensitization, raising red flags for workers in production and lab environments. Research on animal models shows acute inhalation causes lung injury within minutes, compelling regulators in the US, EU, and Asia to set tight occupational exposure limits. Ongoing studies target the environmental fate of BTC and its breakdown products, since trichloromethanes persist and bioaccumulate in soil and waterways. In daily lab life, I’ve watched regulations shape disposal protocols: BTC and its residues end up in tightly sealed waste drums, never down the drain, and always earmarked for professional handling. These rules carry teeth, with hefty fines and shutdown risks for companies that slip.
Looking Forward: The Next Chapter for BTC
BTC’s story now stretches beyond basic chemical synthesis. Industry sees the need to reduce hazardous inputs and wastes in specialty chemicals, so greener synthetic routes attract sustained investment. Researchers chase catalysts that make reactions run faster, at milder temperatures, without complex downstream cleanup. Digital process control enables companies to meet ever-changing safety standards while maintaining throughput. Regulation will likely tighten, especially for chlorinated compounds, locking in higher demands for best-in-class handling and reporting. I expect BTC will survive by evolving: safer packaging, real-time monitoring, and biobased precursors could reframe its place in tomorrow’s chemistry. The heart of the issue is responsibility—chemists must balance BTC’s undeniable synthetic utility with a commitment to protecting people and the planet. A difficult line to walk, but one worth the effort for the advances this reagent supports.
What is Bis(Trichloromethyl) Carbonate used for?
Understanding Its Purpose
Bis(Trichloromethyl) carbonate, often called triphosgene, carries a weight in the world of chemicals that many outside the lab might not realize. It comes as a white crystalline solid, which makes it much easier to handle than its infamous cousin, phosgene gas. The solid form often feels safer for trained hands in the lab, which ends up protecting not just researchers but also the environment from unexpected release.
Where It Finds Work
Most of the buzz around triphosgene centers on its talent for making carbonates, carbamates, and isocyanates. These aren’t just fancy chemical names pulled from a textbook. They feed directly into daily products. Polycarbonate plastics, for example, rely on building blocks from reagents like triphosgene. Things like water bottles and protective gear on a construction site trace their origins back to chemical steps where triphosgene shines.
Lab chemists use triphosgene for peptide synthesis, an important process behind drug discovery and research. A peptide chain often needs a helping push to become stable, and triphosgene offers that, cutting reaction times and boosting yields. After working in research labs, one thing becomes clear: using triphosgene makes life easier compared to maneuvering around phosgene gas cylinders, which present real hazards.
The Safety Factor
Concerns swirl around chemicals with names that end in “chloromethyl.” Triphosgene holds a toxic punch, mostly because it releases phosgene if mishandled or exposed to heat. The chemical community demanded something easier to transport and safer to store. Regulations grew stricter, especially after high-profile lab accidents. Scientists adopted triphosgene as the solid alternative, reducing risk while retaining the usefulness of classic phosgene chemistry.
Problems and Possible Solutions
Triphosgene doesn't solve all the industry’s headaches. Once it interacts with moisture, phosgene forms, and that spells danger not only to workers but people downwind of a factory or warehouse. Reports show increased pressure for chemical manufacturers to adopt better containment strategies. Simple steps like sealed reaction chambers, better ventilation, and thorough emergency planning all make a difference. Investing in worker training pays off. After working setups where everyone knew the drill, accidents grew rare.
Some research teams push hard for alternatives that don’t trace back to harsh chlorinated chemicals at all. A few new synthetic pathways skip triphosgene altogether, focusing on greener reagents and recycling waste products. Switching these production lines over does take time and money. Companies that choose to invest often see fewer fines, fewer emergency clean-ups, and a real boost for their public reputation.
Looking Forward
Chemicals like triphosgene bridge a gap between safety, convenience, and efficiency. Until better reactions come of age, this solid carbonate keeps earning its place in pharmaceuticals, plastics, and laboratories. Focusing on safer practices and greener alternatives sets the tone for a new generation of chemists—one that balances the needs of progress with the health of people and planet.
What is the chemical formula of Bis(Trichloromethyl) Carbonate?
Why BTMC Matters in Modern Chemistry
Bis(Trichloromethyl) carbonate goes by the nickname triphosgene, and you'll spot it often in the organic synthesis world. Over time, its chemical shorthand—C3Cl6O3—has cropped up in patents, research journals, and safety training handouts. If you've spent time in a lab handling phosgene or working on carbonylation reactions, you've probably come across this compound as a safer, more manageable choice. Phosgene’s bad reputation isn’t exaggerated; its toxicity led chemists to hunt for reasonable substitutes, especially for reactions that call for a clean, effective carbonyl donor.
Triphosgene’s molecular makeup offers insight into its reactivity. Each molecule holds two trichloromethyl (CCl3) groups connected to a central carbonate group. This means that while it can pack similar punch as phosgene, it stores more easily—solid at room temperature, higher boiling point, and easier to handle without hazmat suits. Plenty of chemists can recall that first day unpacking a white crystalline bottle of triphosgene instead of wrestling with compressed phosgene gas. It matters a lot for academics, industrial chemists, and anyone expected to train students in safe chemical practices.
Safety, Practicality, and the Environment
Concerns over chemical safety and waste have been front and center in science circles for years—especially in university research and manufacturing. Triphosgene, with its distinct formula, offers a less volatile starting point for tasks that once required more hazardous chemicals. You get much of the same chemical function without betting the farm on a gas mask’s fit. With an increasing push for “green chemistry,” minimizing the dangers from chemical reagents factors into every research decision at schools, startups, and global manufacturing plants alike.
Of course, no chemical sits in a vacuum (so to speak). C3Cl6O3 will release phosgene under certain conditions—especially when heated or under acidic conditions. Proper ventilation, cold storage, detailed hazard training, and sharply supervised disposal still come with the job. But this step forward takes much of the fear out of reactions involving carbonates, carbamates, or isocyanates in a typical synthetic sequence.
Supporting Better Lab Practices
Everyone wants progress, but it shouldn’t come at the cost of safety, personal health, and the environment. In my own work, swapping out legacy reagents for smarter options like triphosgene streamlined synthesis and made the weekly safety meetings less ominous. Students working alongside me felt more comfortable learning classic transformations. Industry surveys highlight that training on triphosgene outpaces phosgene year after year because teaching facilities and manufacturers understand the lower risk profile.
Regulatory agencies and institutional review boards point to triphosgene as an example of how targeted chemical choices can shrink hazardous waste streams and reduce the likelihood of workplace accidents. It’s not the cure-all, but it reflects a practical path forward: blending good science, good stewardship, and common sense. By appreciating the structure and use of C3Cl6O3, those of us in the field shape education, process design, and even public trust in laboratory science.
Moving with Knowledge and Responsibility
Trained hands can wield powerful reagents for positive advances—if they recognize their tools. That means knowing a compound’s mechanics, risks, and wider role in science. With Bis(Trichloromethyl) carbonate, represented by C3Cl6O3, the chemistry community isn’t just chasing productivity or efficiency. We work toward a scene where science moves forward without leaving safety and sustainability behind. In my lab, that shift began with swapping old bottles for new, lighter, and safer options—shaped by both understanding and experience.
What are the safety precautions when handling Bis(Trichloromethyl) Carbonate?
The Risks Lurking in the Lab
Handling chemicals like Bis(Trichloromethyl) Carbonate brings a different kind of responsibility compared to working with something you’d find beneath a kitchen sink. Recognized by many as BTC or Triphosgene, this compound finds its way into organic synthesis, especially for those building pharmaceuticals or specialty chemicals in research and industry settings. From my own experience, even seasoned professionals can underestimate what a pinch of the wrong chemical might do when mixed with rushed steps or distractions. BTC catches folks off guard with its toxicity and its alarming ability to spread dangerous vapors. If unlocked from its secure container and left exposed, it releases phosgene gas—an infamous chemical warfare agent in World War I—so it doesn't allow for any shortcuts.
Why Proper Protection Comes First
Opening a BTC bottle kicks up a tension in the air. A whiff means serious lung damage, not just a sore throat or cough. You want goggles that seal against your face, not the ones that slide off with a lazy tug. Pair those with a solid pair of chemical-resistant gloves—preferably butyl rubber—since BTC punches through regular latex or nitrile a little too easily. Wearing a lab coat is only the foundation. The right look calls for full-length sleeves, pants, and solid shoes. In my lab days, an apron over the coat felt like overkill—until it saved my shirt and skin from a stray splash.
Ventilation: The Real MVP
Fume hoods aren’t just lab furniture. A good setup pulls away vapors before they sneak into your breathing zone. I once watched a colleague try to "just open the bottle quickly" on an open bench—he landed at occupational health with shortness of breath and weeks of recovery. Ventilation handles what even the best gloves and goggles can’t protect: the air you breathe. Always use BTC under a working, certified fume hood. A couple of minutes of patience at the hood beats a trip to the ER.
Emergency Plans Can’t Wait
Every lab day starts better after running through a mental checklist: equipment, emergency shower checked, eyewash station cleared, exits open. Spills, leaks, or accidental splashes happen quickly. Keep calcium chloride or soda ash nearby for neutralizing spills, and never reach for water, which reacts violently with BTC. Sheets with instructions for first-aid—eye flushes, skin rinses, rescue breathing—should hang close to the work area. During a spill, time blurs, so clear protocols prevent panic.
Training and Storage: No Shortcuts Allowed
No lab tech learns BTC safety from PowerPoint slides alone. It takes hands-on practice—opening the bottle inside a glovebox, double-checking container seals. Peer review keeps mistakes rare; calling a colleague to walk through the steps creates a habit few emergencies can shake. All BTC stocks stay locked in ventilated, corrosion-resistant cabinets, often inside secondary containers that catch leaks. I keep only a minimum amount on hand, lessening risk and headache.
Moving Toward Safer Alternatives
Research doesn’t stall in the hunt for safer reagents. Many facilities now swap out BTC for less volatile substitutes where the chemistry allows. Pressure grows for greener chemistry that delivers the same synthesis results without the specter of phosgene. Safer work means fresher air and less stress for all involved.
BTC has a role in advanced chemistry, but it leaves zero room for carelessness. Practice respect, wear the right protection, train with others, and treat emergencies as real possibilities. That discipline keeps the chemist, and everyone nearby, safe from lingering danger.
How should Bis(Trichloromethyl) Carbonate be stored?
Why Attention to Storage Matters
Bis(Trichloromethyl) carbonate, known in laboratories for its role as a phosgene substitute and reagent, brings specific risks that chemical handlers can’t afford to overlook. Its volatility lies not only in its chemistry but also in how it behaves outside of controlled reactions. Horror stories of poor storage often end with ruined research or, worse, health emergencies. Behind every drum or bottle, there’s a responsibility that follows handlers from lab benches to cabinets.
The Real Risks in Everyday Handling
Those who’ve handled this compound know the sharp scent, reminiscent of chloroform but with a more biting edge. Direct contact or inhalation irritates skin, eyes, and respiratory tract. Accidental spills will quickly demand respiratory protection, evacuation, and hours of cleanup. In storage, improper conditions can turn this hazard into a chemical accident waiting to happen: leaks, pressure buildup, and even fire become much more likely if care slips. Just keeping a lid on it isn’t enough.
What Works: Temperature, Containers, and Location
Out of habit—and after a lesson involving a sticky, noxious mess near a heat vent—temperature stands as the first stop for safety. Store it in a cool, dry place, preferably between 2–8°C if your workplace offers cold storage. Room temperature storage quickly becomes risky during summer months or if heating systems malfunction. Heat increases vapor pressure inside containers, which raises the risk of leaks.
Containers play a bigger part than most folks admit. I stick with tightly sealed glass bottles with chemical-resistant PTFE linings. Metal containers corrode and plastics sometimes soften over time. Don’t rely on makeshift lids or questionable seals. Anything meant for Bis(Trichloromethyl) carbonate must resist chemical breakdown as well as physical impact.
Placement goes far beyond “just on a high shelf.” Find a ventilated, locked cabinet, away from direct sunlight and far from incompatible chemicals like acids, alcohols, amines, and bases. This step fights complacency. In my experience, well-organized chemical storage not only prevents accidents but speeds up routine work. Label everything clearly, and keep updated logs so anyone who comes after you isn’t left guessing.
Addressing Human Error and Emergency Preparedness
Mistakes still find their way into even the best routines. Double-check that spill kits, fire extinguishers, and eyewash stations work and remain within easy reach of storage sites. Emergency numbers and instructions for dealing with Bis(Trichloromethyl) carbonate belong on the wall in plain sight. Periodic drills sound excessive until something actually goes wrong. More than once, I’ve witnessed a well-timed drill turn into a real-life save.
Training never goes out of style. Keep everyone who handles or stores chemicals up to date not only on what they work with, but also on the latest storage guidelines. If a new person steps onto the team, make chemical safety a conversation, not a handout. This builds trust and catches bad habits before they set in.
Better Storage Means Fewer Regrets
Safe storage of Bis(Trichloromethyl) carbonate isn’t just a box on a safety checklist. It’s the sum of habits, shared knowledge, attention to detail, and a recognition that every step—from lid placement to chemical separation—affects what happens next. A little discipline saves a lot of trouble, and the best workplaces make conscientious storage a point of pride, not a chore.
What is the purity and grade available for Bis(Trichloromethyl) Carbonate?
Paving the Way for Reliable Sourcing
Bis(Trichloromethyl) Carbonate shows up on the ingredient list for some of the most crucial reactions in pharmaceutical labs and specialty chemical plants. You’ll sometimes hear it called triphosgene, a safer, solid substitute for phosgene. Subtle differences in purity change a lot: efficiency of a reaction, need for purification, and even the safety of a workstation. Having seen several projects hit snags because someone grabbed the wrong grade, there’s no overstating how much the choice can impact cost or timeline.
Common Purity Levels in the Market
Purity matters in chemistry. For triphosgene, the broad baseline hovers around 98%. Labs that run on tight specs ask for material hitting 99% or even pushing beyond that mark. In most published papers, companies and researchers choose 98% or 99% pure material, backed by certificates of analysis. That extra one percent may sound minimal, but it’s the difference between an easy product isolation and hours of chromatography. Impurities like phosgene, chloroform, or unreacted trichloromethyl groups can introduce safety risks or lower yields. The highest quality stocks leave very little behind after a reaction—smooth sailing for teams chasing fine pharmaceuticals or optical materials.
Grades and Their Real Impact
Manufacturers split triphosgene into standard grades: technical, laboratory, and high-purity. Technical grade serves the production of bulk plastics and resins. These applications tolerate a degree of variability and impurities, keeping costs down for huge-scale synthesis. Lab grade usually steps up with tighter controls, often going beyond 98% purity. This is the bottle you’d expect in a research institution or smaller volume synthetic shop. The top shelf lies with high-purity (sometimes labeled “reagent” or “analytical” grade), where chemists and process engineers demand 99% purity and strict packaging to prevent degradation from moisture or heat. Using low-grade triphosgene inside a pharmaceutical workflow can prompt regulatory issues or even batch recalls, with entire teams hunting down sources of contamination. Discerning buyers check for up-to-date documentation: not just the label, but supporting HPLC or NMR trace, safety specs, and supplier reputation.
Securing Reliable Sources and Safer Workflows
Purity isn’t just a nice-to-have. Labs need stability in purity because regulatory agencies don’t pardon shortcuts. Traces of phosgene or unpredictable by-products risk exposing staff to dangerous gases. My own lab days taught me to spot signs of moisture contamination—a bottle that hisses or cakes on the inside often spells trouble. Suppliers worth their salt store triphosgene in airtight, amber bottles, with moisture indicators and tamper-proof seals. Large manufacturers such as Sigma-Aldrich, TCI, and Alfa Aesar consistently list both purity and typical certificate-of-analysis results online, letting buyers verify before purchase. For larger industrial batches, the practice runs on material safety data sheets, batch traceability, and transparent supply chains. Auditing suppliers and choosing those who meet ISO or GMP standards provides an insurance policy against out-of-spec shipments that could derail months of research or millions in products.
Maximizing Value and Safety by Grade Choices
Selecting the right purity starts with mapping out the reaction or process. For polymers, technical grade can pull its weight and save money. For API intermediates or fine structure analogues, every impurity creates trial and error. No two projects build up from the same foundation, but one truth remains: the lines between cost, purity, and reliability are never accidental. A little extra on a high-purity bottle can save headaches, hazmat incidents, and shutdowns. That lesson, learned the hard way in the early years, shows why specs shape every decision in a chemical workflow.
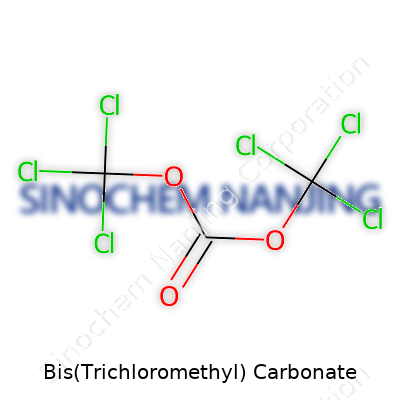
| Names | |
| Preferred IUPAC name | Trichloromethyl trichloromethoxyformate |
| Other names |
Triphosgene Bis(trichloromethyl) carbonate BTC |
| Pronunciation | /ˈbɪsˌtraɪˌklɔːroʊˈmɛθɪl ˈkɑːrbəˌneɪt/ |
| Identifiers | |
| CAS Number | 2215-35-2 |
| Beilstein Reference | 146366 |
| ChEBI | CHEBI:38627 |
| ChEMBL | CHEMBL504187 |
| ChemSpider | 58464 |
| DrugBank | DB13183 |
| ECHA InfoCard | 03c7d734-1f1d-4d67-99dd-f83c49cfd68f |
| EC Number | 204-118-5 |
| Gmelin Reference | 79057 |
| KEGG | C14322 |
| MeSH | D001666 |
| PubChem CID | 6587 |
| RTECS number | FG9625000 |
| UNII | VI8451J2ZL |
| UN number | UN 2576 |
| CompTox Dashboard (EPA) | DTXCID50418 |
| Properties | |
| Chemical formula | C3Cl6O3 |
| Molar mass | 277.75 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.66 g/cm³ |
| Solubility in water | Decomposes |
| log P | 1.98 |
| Vapor pressure | 0.04 mmHg (20 °C) |
| Acidity (pKa) | 15.8 |
| Basicity (pKb) | 13.55 |
| Magnetic susceptibility (χ) | −70.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 10.6 cP (20°C) |
| Dipole moment | 2.16 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 280.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -504.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1762 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if inhaled, swallowed, or absorbed through skin; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H317, H334, H335, H410 |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P302+P352, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-0-2-Health-2,Flammability-0,Instability-2 |
| Flash point | Flash point: 110 °C |
| Autoignition temperature | 230 °C |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 68 mg/kg (oral, rat) |
| NIOSH | WF9810000 |
| PEL (Permissible) | PEL: 0.1 ppm (1.4 mg/m3) as TWA |
| REL (Recommended) | REL: 0.1 mg/m³ |
| IDLH (Immediate danger) | 20 ppm |
| Related compounds | |
| Related compounds |
Phosgene Triphosgene Diphosgene |

