Bis(N,N-Dimethylthiocarbamoyl) Disulfide: A Real-World Look at Its Past, Use, and Future
Historical Development
Chemistry keeps pulling up surprises, and Bis(N,N-Dimethylthiocarbamoyl) Disulfide (sometimes called Thiram) shows how a simple compound can influence entire industries. People started producing this chemical in the 1940s, not long after research into dithiocarbamates exploded. Early farm researchers needed answers for crop protection and lasting fungicide, and these sulfur-rich molecules came forward as solutions. The first synthesizers understood that pests and disease could wreck grain harvests, and they saw how one compound could help farmers protect not just yields but the food chain that went with it. It was a time focused on survival, practicality, and innovation out of necessity.
Product Overview
Thiram stands out as a non-systemic fungicide and a key accelerator in the rubber industry. Makers and users call it by many names; it bounces between agriculture, manufacturing, and materials science. Farmers recognize it as a coating on seeds or young plants, a defensive layer against fungal rot that could otherwise decimate rows of produce. Inside factories, Thiram aids rubber vulcanization, shaping everything from tires to conveyor belts. In both worlds, the material’s identity shifts—sometimes a protective agent, sometimes an enabler—rarely front and center but always important in the background.
Physical & Chemical Properties
Bis(N,N-Dimethylthiocarbamoyl) Disulfide typically lands on the workbench as a white crystalline powder. Scratch it, and the powder holds a faint sulfur odor, tipping you off to its chemistry right away. Its melting point sits around 155 °C, and it barely dissolves in water, but mixes more easily with acetone, ethanol, or ether. The chemical structure packs sulfur and nitrogen closely, which gives it the properties that keep fungi at bay or help rubber reach the right strength. Store it too long in moist air, and you’ll smell decomposition. Too much heat, and it breaks down into smaller thiocarbamates and even gases that need ventilation.
Technical Specifications & Labeling
Each bag or drum of Thiram carries a specific guarantee—often purity listed as not less than 97% by weight, moisture content under 0.5%, and sometimes a breakdown of dust content or mesh size. Labeling requires hazard pictograms under GHS guidelines, along with first aid instructions, proper storage advice, and batch identification. Factories print specifications in fine point, but users rely on clear symbols and warnings. In transport or storage, operators follow strict weight limits and spill contingencies, a hard lesson learned after past workplace accidents. As regulations change, so do the label standards, now often including QR codes for instant access to detailed safety data sheets.
Preparation Method
To create Bis(N,N-Dimethylthiocarbamoyl) Disulfide, chemists start with dimethylamine and carbon disulfide in the presence of a base, mixing at low temperature to form the intermediate dimethyldithiocarbamate salt. Oxidation comes next, often using hydrogen peroxide or chlorine, fusing two dithiocarbamate molecules to craft the disulfide bond. Each step needs careful pH adjustment and temperature monitoring, or impurities creep in. Byproduct gases and washoff need proper handling; otherwise, they risk environmental contamination. Experienced operators keep a close watch on every reaction shift—one mistake and the yield drops or a safety incident erupts. Modern plants use continuous reactors, but old batch setups still pop up in smaller specialty runs.
Chemical Reactions & Modifications
In the lab, Thiram serves as more than a stable powder. It undergoes reduction to give back dimethyldithiocarbamate anions, or alkaline hydrolysis to create methylamines and carbon disulfide again, sometimes used to recycle the parent materials. It reacts quickly with metals, forming complexes valued in analytical chemistry as colorimetric reagents. Under UV light or heat, the disulfide bond fragments, meaning storage outside sunlight is a must. I’ve watched researchers test new analogs by tweaking the alkyl groups, chasing improved biological activity or more stability in soil. These studies often shape future generations of seed treatments and rubber accelerants.
Synonyms & Product Names
This molecule answers to many names, a common problem in older chemicals. Thiram remains the name most growers and manufacturers pick, but Mertect, TMTD, and commercial blends like Arasan or Nomersan pop up depending on market or region. In regulatory filings or analytical journals, the full IUPAC name or the CAS number (137-26-8) sometimes appears, helping chemists avoid confusion with similar dithiocarbamates. Such a split personality often slows down research or international logistics, demanding cross-checking labels and paperwork.
Safety & Operational Standards
Few chemicals balance utility and risk like Thiram. Industry users keep to strict handling guidelines—gloves, goggles, chemical-resistant suits in high-dust areas, shower stations near any place the powder might hit skin or eyes. Air monitoring matters most in dusty mixing rooms, and well-trained staff can spot Thiram’s acrid smell from meters away. Farms limit exposure by treating seeds offsite and letting them dry before storage or transport. Regulatory agencies push for personal exposure below 0.05 mg/m³ over an 8-hour day in most countries, based on long-term toxicity studies. In case of a spill, procedures demand prompt cleanup, sealed waste containers, and reporting to environmental agencies.
Application Area
Agriculture dominates Thiram’s story. Farmers add it as a seed treatment to stave off fungal blights and damping-off disease during the first crucial days after planting. Golf course keepers spray it onto turf to fight snow mold, and fruit packers sometimes use it on apples or pears to keep storage rot away. In the rubber trade, Thiram accelerates curing, helping create flexible, durable materials used across the world in tires, shoe soles, hoses, or gaskets. Oddly, Thiram also steps into non-obvious spaces, like animal repellent formulas for fruit trees or flower beds, adding bite to leaves and keeping deer and rabbits away. Each end use depends on the safety measures and rules governing food or consumer contact.
Research & Development
Labs still chase better reliability and less environmental fallout. Researchers study how Thiram breaks down in soil and water, tracking metabolites for weeks or months. Universities look for new formulations with less off-target toxicity, aiming for “smart” coatings or slow-release blends that release just what’s needed, at just the right time. Rubber chemists tweak processing conditions to shave waste and lower energy costs, sometimes using Thiram sparingly to cut down on final product residues. Collaboration has grown, with agriculture, toxicology, and environmental groups challenging companies to measure, monitor, and limit unintended side effects.
Toxicity Research
Animal and lab data both point to real hazards. Acute exposure causes skin and eye irritation, allergies, and hormone disruption in some tests. Chronic exposure—like in untreated workers—is tied to changes in liver enzymes, red blood cell function, and, more rarely, nerve problems. On the environmental side, fish and aquatic invertebrates show strong reactions at lower doses. Wider field studies monitor residues in harvest crops and track how fast the compound breaks down in soil and water, pushing regulators to clamp down on both workplace exposure and what traces remain in food. In practice, the way to limit risk centers on containment, limiting dust, protective gear, and clear instructions. As a parent and a consumer, I always check how much remains on food and pay close attention to reports about drinking water or livestock feed.
Future Prospects
Thiram’s outlook ties into the future of crop protection and materials science. More countries target lower pesticide residues, and sustainable farming pushes for alternatives, either in precise formulations or replacements with less ecological impact. Companies race to develop compounds that keep key parts of Thiram’s performance, but fade away faster once the job is done. Synthetic biologists test plant-derived coatings, while others chase bioremediation strategies to break down residues left in the field. Inside rubber manufacturing, engineers aim to offset Thiram use with new sulfur-based accelerators. The tension between safety, yield, and profit keeps the debates fresh; society demands food and materials but shows less patience for old methods that carry extra health or environmental risks. I see opportunity for consumer input—in labeling, supply chain choices, and funding new R&D—to drive this transition. Each step depends on listening to the science, open communication, and a willingness to change.
What is Bis(N,N-Dimethylthiocarbamoyl) Disulfide used for?
Looking Closer at a Key Rubber Chemical
Most people never hear about Bis(N,N-Dimethylthiocarbamoyl) Disulfide, but a lot of workers in the rubber industry know its punchy abbreviation: THIRAM. That’s because this chemical plays a part in making things that touch everyday life, from tires gripping the road to the soles under sneakers. Thiram belongs to a group of compounds known as thiuram sulfides, which help push along the process of curing rubber. With the world’s appetite for rubber products—tires, belts, hoses—still huge, so is the need for chemicals like this one.
Why Farmers and Factories Turn to Thiram
Thiram carries a dual identity. In a manufacturing plant, its main job comes as a rubber accelerator. Workers use it to speed up vulcanization, a process that transforms sticky sap into a solid, durable material. Without accelerators such as Thiram, rubber stays soft, stretches out, and falls apart quickly. With Thiram in the mix, tires roll longer, boots last through more storms, and belts keep conveyor lines moving.
If you walk out of the factory and into the fields, Thiram changes hats. Farmers spread it over crops as a fungicide. Fungi can devastate harvests—potatoes, onions, apples, peanuts. Instead of letting those crops rot, Thiram provides crops with a line of defense. Some might argue the chemical goes overboard, but it’s hard to argue with the farmers facing down late blight in potatoes or apple scab. Without fungicides, past food shortages taught painful lessons about just how much trouble a fungus can cause.
Concerns From Both Sides
Experience with Thiram isn’t all smooth. Rubber workers and those spraying crops see the other edge of its use: health concerns. People exposed on the job may notice skin rashes or breathing problems. Long-term research still continues, but environmental groups sound alarms about its impact on water sources and wildlife—especially aquatic life.
Regulators act with caution, tightening rules for workplace safety and food residues. The Environmental Protection Agency in the United States marks it as moderately toxic. Some international markets, including parts of the EU, restrict or ban the chemical in food production out of precaution. These actions don’t come out of nowhere; they follow studies tracing its run-off into waterways and questioning its long-term impact if overused.
Balancing Benefit and Risk
Balance feels tough here. Factories rely on Thiram for affordable, reliable rubber. Farms depend on it to protect crops and ensure enough food reaches stores. Both groups keep eyes open for better alternatives. In the rubber industry, researchers look for new accelerators that work just as well without health drawbacks. In agriculture, growers test integrated pest management and natural processes to control fungi. Still, Thiram sticks around, thanks to its history of delivering solid results.
As with most chemicals, the key will be careful handling and responsible use. It’s up to manufacturers and farmers to follow rules, wear the right gear, and use as little as possible to get the job done. Better education, upgraded safety standards, and ongoing research matter if society wants to enjoy the benefits without trading away health or environmental quality. That puts responsibility on both industry and regulators—not just for today, but for the fields and factories of tomorrow as well.
What are the safety precautions when handling Bis(N,N-Dimethylthiocarbamoyl) Disulfide?
A Chemical that Deserves Respect
Working with Bis(N,N-Dimethylthiocarbamoyl) Disulfide, or simply thiram, sticks in the mind because of its sharp sulfur smell and the immediate urge to wash up after handling it. Used in everything from agriculture as a fungicide to rubber manufacturing, thiram has plenty of useful qualities. That same usefulness comes with a set of risks no one should brush off.
Hazards Are Real—No Room for Complacency
Skin doesn’t like thiram. Direct contact can irritate and even cause allergic reactions, especially if it gets trapped under gloves. Inhaling the dust or fumes causes coughing or throat irritation. People who skip masks or gloves sometimes end up with headaches or nausea. That’s more than just an inconvenience—it’s a signal your body gives you to steer clear.
The dust lingers in the air. Eating or drinking after touching thiram without washing up isn’t smart—a small slip-up can foster stomach cramps, dizziness, or far worse symptoms if exposure continues over time. Chronic contact linked with thiram might push liver and kidney issues, according to studies from regulatory agencies like the EPA and European Chemicals Agency.
Real Steps That Protect
Common sense works best when it’s turned into a habit. It always helped me to mentally walk through the job before opening a container. Gown up in a lab coat or coveralls. Gloves—nitrile, not latex—hold up to the chemicals. Always check for pinholes; broken gloves defeat the point.
Face shields or at least goggles stand between eyes and the chance of a splash. Even a few droplets sting or lead to a hospital visit. Respirators with particulate filters do the trick for the airborne threat, especially when pouring or mixing powders. Folks with facial hair might take an extra minute to get a proper seal—a loose mask wastes everyone’s effort.
Clean Workspaces and Good Habits
Keep thiram in tightly sealed containers, away from food, water, and open flames. Spills get tricky—neutral absorbents make cleanup safer; avoid sweeping with a dry broom, since that just sends dust clouds into the air.
After the job, always wash up thoroughly—face, hands, all surfaces that had any contact—before grabbing lunch or touching a phone. Contaminated gear should go straight into the laundry, not tossed with regular street clothes. Facility managers and supervisors do well when they keep gloves, lab coats, and eye protection in stock, not just on paper. It isn’t just regulations forcing this; one close call resets assumptions for good.
Dealing with Exposure
Mistakes happen, even for careful folks. If thiram splashes onto skin, soap and lots of water help. Eyes need a full rinse at the eyewash station, never just a quick wipe. Breathing trouble or odd symptoms after exposure should send someone to seek medical help on the spot, not hours later. Early intervention curbs long-term health impacts.
Solutions from Experience
Experience taught me that safety instructions work best when everyone buys in, not just new workers being told what not to do. Regular training, open conversations about close calls, and easy access to safety data build a stronger culture than posted rules ever could. Investing in ventilation, personal protective equipment, and clear labeling doesn’t just meet standards—it keeps people confident and focused so mistakes stay rare.
People who work around chemicals like thiram share responsibility. Backing each other up, speaking up about shortcuts, and making it simple to follow safety routines ensures everyone goes home healthy. That’s not just advice—it’s the difference between routine and regret.
What is the chemical formula and structure of Bis(N,N-Dimethylthiocarbamoyl) Disulfide?
Breaking Down the Name
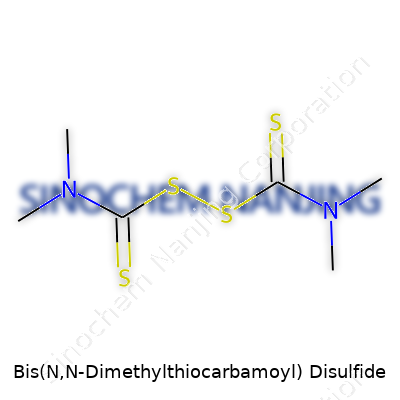
Bis(N,N-Dimethylthiocarbamoyl) disulfide sounds complicated right away. In reality, it just pieces together basic building blocks in organic chemistry. Picture two dimethylthiocarbamoyl groups joined by a sulfur-sulfur (disulfide) bridge. That’s the backbone of this compound. Chemically speaking, its formula is C6H12N2S4. You can find it in textbooks as tetramethylthiuram disulfide, or by the trade name thiram.
Diving into Structure and Design
Looking up the structure, you’ll notice a central S-S bond, each sulfur atom linked to a thiocarbamoyl group. Each of those groups features two methyls attached to the nitrogen. The skeletal formula displays this arrangement as:(CH3)2N-C(=S)-S-S-C(=S)N(CH3)2
Years of laboratory work show how sulfur bridges stabilize the molecule, letting it play a unique role in real-world applications. Strong sulfur bonds do more than hold the compound together—they foster the chemical’s antifungal and pesticidal properties, which farmers and industrial processors have relied on since the early 20th century.
Uses and Real-World Relevance
Farmers apply thiram as a fungicide, especially for seed treatment. Science journals from the 1950s onward confirm effectiveness against seed-borne and soil-borne diseases. That means higher crop yields and less loss before harvest. Its value also stretches beyond farming. Rubber manufacturers use thiram to speed up vulcanization—the process that strengthens and stabilizes rubber for tires, footwear, seals, and hoses.
Though thiram brings benefits, mismanagement can create risk. Long-term exposure raises concerns about toxicity for workers and the environment. Researchers at the U.S. National Institutes of Health report that ingestion or repeated skin contact, especially in poorly ventilated spaces, triggers nausea, skin irritation, and in rare cases, neurological symptoms. Awareness of these risks led regulators in Europe and North America to tighten rules on thiram residues in food and field usage.
Balancing Safety and Innovation
Science supports caution with chemicals like bis(N,N-dimethylthiocarbamoyl) disulfide. Personal experience working in research and development exposed me to the strict protocols in chemical labs—always wearing gloves, keeping the space ventilated, and never skipping safety data sheets before handling these powders. My colleagues know from hard lessons that shortcuts in safety never pan out. We rely on safety training and evidence-based standards not only for compliance but for peace of mind.
Industries have responded by adopting safer alternatives and engineering controls. Modern treatment facilities use closed systems to limit airborne dust. Farms now count on newer biologically derived fungicides where economic and agronomic conditions allow. But there is no substitute for solid chemical literacy. Technicians need education that goes beyond memorizing formulas. Real progress means understanding how a structure like C6H12N2S4 interacts with its surroundings—be it soil, air, water, or human tissue—so that value can be delivered without unintended harm.
Moving Forward
Better solutions rest on good science and shared responsibility. Chemists work hand-in-hand with farmers and manufacturers to prioritize transparent information and smarter risk management. That fostered trust in the marketplace and set a model for future innovations. Knowing both the power and limitation of chemicals like bis(N,N-dimethylthiocarbamoyl) disulfide keeps everyone on solid ground.
How should Bis(N,N-Dimethylthiocarbamoyl) Disulfide be stored?
Hazards Taking Center Stage
Anyone who’s spent real time working with organosulfur chemicals recognizes the hazards without needing much reminding. Bis(N,N-Dimethylthiocarbamoyl) disulfide, better known to some as Thiram, comes with its own grab bag of risks—skin and respiratory irritation, and, under certain conditions, possible fire or toxic gas release. Trust me, I’ve seen a rushed colleague skip proper storage; the stench and hassle that followed made a classroom lesson painfully real. Simple rules, followed with discipline, prevent trouble most of the time.
Rule One: Keep It Cool and Dry
Every chemical textbook says the same thing for a reason: cool, dry areas matter. Water and high temperatures can start degradation pretty fast, creating both safety and quality problems. South-facing windows? Forget it. Hot storage rooms in summer? Equally bad call. Air-conditioned, low-humidity storage rooms give you an extra buffer, so you can come back weeks or months later to a chemical that hasn’t changed its behavior or created new hazards.
Containers Matter More Than We Admit
Glass makes the go-to for many organics, but for Thiram, shatterproof options and chemically compatible plastics (think HDPE) add safety where it counts. I once saw a glass jar nicked during handling; soon after, it was leaking powder onto the shelf. The right container and a tight seal do more than preserve shelf life—they keep the storeroom safe for the next person. Label your containers the way you want your neighbor to—clear, correct, nothing left guessing. GHS pictograms and proper hazard language go on every label, every time.
Avoid Mixing with Incompatibles
It takes only a quick look at the Safety Data Sheet to see a list of chemicals you don’t want near Thiram: oxidizers, strong acids, and alkalis. Fumes or leaks can trigger reactions, so close proximity turns minor mistakes into big problems. Without separation, I’ve seen accidental contact cause headaches for both the lab team and emergency response.
Ventilation and Spill Planning
Good air exchange in storage rooms pays dividends. Just last winter, we cleaned up a minor spill because the room’s negative pressure system kicked in, pulling suspicious odors away before anyone felt sick. Spill kits always sit near the door, with appropriate absorbents and gloves—nobody finds themselves hunting for gear mid-crisis.
Tight Inventory, Tight Controls
Counting your stock and keeping detailed logs adds one more line of defense. Misplaced or expired chemicals surprise nobody if you track things with monthly checks. This is the reality inside every smart facility—records, routine, and respect for the materials that put us in business. Staff handle transfers with secondary containment trays, never alone, and always with eye-wash and showers tested weekly.
Lessons That Stick
Storing chemicals like Bis(N,N-Dimethylthiocarbamoyl) disulfide safely doesn’t call for heroics, just good habits and a bit of common sense. Gear up with the right containers, keep your area cool and dry, separate the troublemakers, and never assume a spill won’t happen. Real-world experience teaches faster than any pamphlet: careful storage means fewer emergencies, less waste, and a safer team.
What are the potential health hazards of Bis(N,N-Dimethylthiocarbamoyl) Disulfide?
Understanding the Chemical
Bis(N,N-Dimethylthiocarbamoyl) disulfide pops up in the industrial world, especially where rubber goods and chemicals get their shape. Many people outside the factory never hear its name, but those who handle it shouldn’t shrug off its potential dangers. Years ago, I visited a chemical plant as a young reporter. The sense of caution among workers handling unknown powders left me curious for good reason—many workplace stories have ended with unforeseen illness from chemicals once thought harmless.
Health Hazards
Contact with this compound primarily threatens the skin, lungs, and eyes. The stuff can irritate anything it touches. Even a whiff as you open a drum might bring on coughing, sneezing, or a burning feeling in the nose. Folks mixing or loading it often see red, itchy rashes that stick around for days. Eye contact brings watering, pain, and even blurred vision in heavier exposures.
Occupational health officers warn that bis(disulfide)s can leave workers with chronic health problems. The National Institute for Occupational Safety and Health (NIOSH) flags compounds like this as possible allergens and respiratory irritants. Long-term, repeated exposure may link up with asthma-like symptoms or lasting lung sensitivity. That’s not speculation—doctors treating factory workers have seen these symptoms appear after months or years in chemical plants.
Risks Beyond the Workplace
Exposure isn’t just about the worker. Improper storage or disposal spills chemicals into the air and water outside the plant fences. In the past, leaks landed in nearby water supplies, putting both wildlife and folks living down the road at risk. Parents in those communities often wonder about the long-term impact on their kids’ health.
Scientists found that this compound tends to break down into even nastier molecules—some linked to broader environmental risks. For example, carbon disulfide, a breakdown product, has shown nerve and reproductive system effects in laboratory animals. Though most health issues come from big, repeated exposures, nobody welcomes chronic headaches, fatigue, or chance of future illness in return for a paycheck.
Precautions and Solutions
Real protection starts with information. Employers need to be upfront about the risks and keep up-to-date safety protocols. Workers who see labels, safety sheets, and good personal protective gear handle chemicals with more confidence and less risk. Gumboots, gloves, and eye shields are not negotiable in environments where spills or splashes can surprise you.
Regular air monitoring and better ventilation bring toxin levels down. I once spoke with a plant manager who installed a local extraction system. It cost the company a chunk of change but cut airborne dust by nearly half and eased complaints of headaches on the shop floor. An investment in safer handling pays off quickly in peace of mind and fewer sick days.
Community oversight also matters. Regulatory agencies keep factories honest with regular inspections and public reporting. Making sure the neighbors know what’s stored and how emergencies get handled keeps everyone in the loop and builds trust.
Preventing harm calls for vigilance at every level. People deserve straight talk about risks and a real effort to keep both workers and the environment protected. Through better education, investment in engineering, and steady oversight, industries using bis(N,N-Dimethylthiocarbamoyl) disulfide can do right by the people counting on them.
| Names | |
| Preferred IUPAC name | N,N,N′,N′-Tetramethylimidodithiocarbonic diamide disulfide |
| Other names |
Thiram Tetramethylthiuram disulfide Dimethylthiocarbamoyl disulfide Thiuram TMTD |
| Pronunciation | /ˌbɪs ɛn ɛn daɪˌmɛθəlˌθaɪ.əˈkɑːr.bəˌmɔɪl ˈdaɪˌsʌl.faɪd/ |
| Identifiers | |
| CAS Number | 137-26-8 |
| Beilstein Reference | 127873 |
| ChEBI | CHEBI:38646 |
| ChEMBL | CHEMBL330024 |
| ChemSpider | 25906 |
| DrugBank | DB08657 |
| ECHA InfoCard | 100.038.281 |
| EC Number | 216-591-8 |
| Gmelin Reference | 71307 |
| KEGG | C08209 |
| MeSH | D003997 |
| PubChem CID | 8530 |
| RTECS number | NT9275000 |
| UNII | 1N03JQ6OYD |
| UN number | UN2773 |
| Properties | |
| Chemical formula | C6H12N2S4 |
| Molar mass | 272.5 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | slight amine odor |
| Density | 1.32 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.39 |
| Vapor pressure | 1.03E-6 mmHg at 25°C |
| Acidity (pKa) | 12.48 |
| Basicity (pKb) | 3.6 |
| Magnetic susceptibility (χ) | -22.24×10^-6 cm³/mol |
| Refractive index (nD) | 1.653 |
| Viscosity | 70 cP (20°C) |
| Dipole moment | 1.26 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 252.2 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -146.8 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -860.7 kJ/mol |
| Pharmacology | |
| ATC code | S01XA09 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "Harmful if swallowed. Harmful in contact with skin. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Suspected of damaging fertility or the unborn child. |
| Precautionary statements | P261, P273, P280, P304+P340, P302+P352, P312, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | Flash point: 138°C |
| Autoignition temperature | 200 °C (392 °F; 473 K) |
| Lethal dose or concentration | LD50 oral rat 151 mg/kg |
| LD50 (median dose) | 177 mg/kg (rat, oral) |
| NIOSH | WN0875000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.3 mg/m³ |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Dimethyldithiocarbamate Ziram Thiram Ferbam Mancozeb |

