Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane: Opportunity and Responsibility in a Changing Chemical Landscape
Historical Development
The story of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane began decades ago, when advances in organic chemistry opened doors for tailored halogenated compounds. As the saying goes, necessity breeds invention. Industrial and agricultural shifts in the 20th century pressed chemists to build molecules that could handle more than just the standard wear and tear of daily use. The drive behind the scenes often focused on pushing the boundaries of environmental persistence and biological resistance—metrics that, at the time, represented success. Scientific journals from the 1960s and 1970s would occasionally mention new synthetic pathways and novel compounds like this one. Looking back, the period holds value not only for its discoveries, but also for teaching us that progress carries consequences.
Product Overview
Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane sits between tradition and disruption. Its presence in specialty chemical portfolios often ties to niche uses, such as intermediates in dye manufacture, flame retardants, and biocides. For those who’ve worked in chemistry labs filled with the odor of dense phenolic powders and solvents, talking about this compound feels familiar. You remember the way small tweaks in chlorination patterns could tip the balance from useless byproduct to vital precursor. The reputation of these chemicals walks hand in hand with both their effectiveness and the shadow of unintended impacts.
Physical & Chemical Properties
Chemists know a molecule by its quirks. Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane carries weight, with a molecular structure that resists easy breakdown. Solid at room temperature, this aromatic structure contains multiple chlorine substitutions. These atoms lend both stability and stubbornness. Water solubility sits on the low side, giving it a tendency to linger, whether in an industrial vessel or, less fortunately, in the environment. Anyone who has handled chlorinated phenolics can recall the challenge of fully removing the last traces from glassware. The chemical properties tell a story: resistance to acid and base, reactivity with strong nucleophiles, and a lingering presence long after its work is done.
Technical Specifications & Labeling
Labels on bags and barrels once seemed like an afterthought in chemical plants. Over time, the industry learned that precision matters. There’s value in knowing exactly what you’re dealing with. The technical data for Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane usually includes purity percentage, trace contaminant levels, and recommended storage instructions. Older containers sometimes lack the rigor now demanded by international standards. Legitimate operations now pay close attention to batch numbers, hazard pictograms, and expiration dates. This stricter attention doesn’t show up just at the manufacturing stage; it follows every drum that leaves the factory floor and lands in research or production. You can thank decades of hard lessons for this shift—a move from raw production to real accountability.
Preparation Method
Manufacturing this compound draws on a straightforward route—condensing chlorinated phenols with methanal under acidic or basic control. Those who’ve run these reactions know the safety gear isn’t just for show. Reflux setups, meticulous control over pH, and a steady hand all count. The process often runs into side reactions, with temperatures and times needing constant attention. Even small temperature swings can tilt yields or generate persistent byproducts. The story of scale-up moves beyond the handbook’s pages; it unfolds on the plant floor with every batch, as operators balance tradition, cost, and constant vigilance.
Chemical Reactions & Modifications
Chemistry offers many avenues for making small molecules dance. Functional groups on Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane invite substitution, oxidation, or coupling reactions. Lab notebooks from my graduate days show experiments where phenolic hydrogens acted as points of entry for further derivatization—sometimes yielding more potent, sometimes more benign, alternatives. Whether aiming at increased solubility or modified biological activity, modifications require technical know-how, plenty of patience, and a willingness to accept unexpected results. Each new route or reaction profile might open new uses, or might reinforce the need for caution with compounds this persistent and reactive.
Synonyms & Product Names
The tangle of chemical names reflects an era’s approach to innovation. Researchers and manufacturers have used various synonyms—trichlorinated bisphenol derivatives, or more systematic IUPAC formulations. Many in the industry started with the commercial or trade names before grappling with the full systematic nomenclature in academic papers. This sometimes created confusion across national lines and industries, which led to contamination events and legal disputes. Such naming confusion drove the movement toward harmonized global databases and tighter regulatory reporting.
Safety & Operational Standards
Hard-won lessons shape operational standards. Chlorinated compounds like this one show up on watchlists in environmental and occupational safety circles for a reason. Exposure risks can range from skin and eye irritation to more serious, long-term health concerns. Safety data sheets have grown thicker over the years as fresh research spells out new hazard statements and workplace exposure limits. My experience walking a floor lined with emergency showers, gloves, and constant air monitoring equipment gave me a real appreciation for what’s at stake. Modern regulatory frameworks call for continual monitoring, detailed worker training, and evolving emergency response plans. Companies that try to cut corners usually find themselves hit both financially and legally by lapses or leaks. Staying ahead requires not just compliance but active vigilance.
Application Area
In specialty manufacturing, Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane once held a prized spot—used in inks, dyes, flame retardants, and biocides. The chemical’s stability and resistance to microbial growth built its reputation. A look at old purchasing records or technical manuals makes clear how much these properties drove demand, despite growing concern about downstream persistence. Product managers once celebrated shelf life and resistance without always asking what happened after disposal. As regulations tightened and consumer awareness increased, the material found itself on a shifting playing field. Many have moved to phase out or restrict its use in sensitive areas, driven both by new testing data and advocacy from environmental health researchers.
Research & Development
The past decade has brought more scrutiny and, with it, a rethink of how research teams approach persistent organic chemicals. Grants and journal articles now focus on alternatives or green chemistry solutions, rather than only tweaking known molecules. Discussions at industry conferences I attended have shifted from, “What’s the next tweak?” to, “How do we minimize lasting impact?” Tools like high-throughput screening and computational modeling allow chemists to predict, far earlier, the fate of new derivatives. Still, labs worldwide carry supplies of older compounds, necessary for comparative studies or to bridge a gap no new solution yet fills. R&D isn’t just about invention—sometimes it’s about responsible reinvention or, even harder, a final farewell to something that once worked so well.
Toxicity Research
Widespread use brought closer study of environmental and toxicological impacts. Decades ago, researchers started to find traces downstream—sediments, wildlife tissues, even human biomonitoring samples. Long-term toxicity studies in both lab animals and real-world environmental circumstances started to paint a picture. Repeated low-level exposures could disrupt endocrine function, impact reproduction in wild species, or influence developmental processes. Academic and regulatory experts increasingly weighed persistence and bioaccumulation, not just acute short-term effects. Looking back, it’s evident that science occasionally lagged behind real-world consequences—and catching up has been a painstaking process marked by public health advocacy, litigation, and eventually, regulation. The message is clear: chemical progress must start with a long view of safety.
Future Prospects
Looking forward, Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane faces an uncertain role. The future lies in developing safer, less persistent alternatives that match the function without the problems. Green chemistry has shifted from buzzword to mandate, driving global companies to invest in cradle-to-cradle design and full life-cycle analysis. Regulatory bodies continue to review acceptable uses, often tightening allowable limits or outright banning certain applications. Collaboration between regulatory bodies, industry, and academia holds the best hope. I’ve seen promising university-industry partnerships work: chemists sharing data early, engineers providing process insight, toxicologists guiding screening, all aiming to prevent harm before it starts. This approach costs time upfront, but spares the public and environment much more down the line. If the history of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane teaches anything, it's that responsibility and innovation must walk together, or neither gets far.
What are the main applications of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane?
A Deep Look at Its Main Jobs
In industrial chemistry, a few compounds seem small until you see how many products depend on them. Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane, better known to some as hexachlorophene, stands out in this way. At first, people see the long name and think of a niche lab chemical. In truth, its reach stretches through healthcare, agriculture, and even the work of keeping factories safe from microbes. Over the years, the story of this compound speaks to both its power and the caution that responsible science and manufacturing require.
Antibacterial Power in Health Settings
Just about every hospital keeps infection under control partly because of antiseptics and cleaners. Hexachlorophene’s claim to fame comes from its strong grip on bacteria, especially Staphylococcus aureus. Long before alcohol-based hand rubs filled every dispenser, many surgical teams scrubbed up with soaps featuring this compound. It provided a solid safeguard against common pathogens. Some studies tracked hospital infection rates and found real drops after switching to hexachlorophene-based products. Overuse led to safety concerns, but no one disputes that it changed the rules of infection control over decades.
Controlled Use and Regulation
Any discussion about hexachlorophene has to touch on the major turning point—its restriction. Multiple cases in the 1970s raised alarms about risks to newborns exposed to high concentrations, especially in baby powders and skin creams. Fatal cases put safety at the center of every future decision. Today, regulations keep the use of hexachlorophene under strict oversight. Healthcare workers use it for hand-washing and pre-surgical preparation but always at levels proven safe. Manufacturers and regulators both learned from earlier mistakes, showing why rigid safety standards give real protection for the public.
Defending Materials: Preservative Uses
Beyond hospitals, products with water content or organic ingredients face a constant struggle with bacteria and fungi. Hexachlorophene once played an important role in keeping cosmetics, creams, and even some textile finishes free from microbes. It helped products last longer and lowered the risk of spoilage in storage. Its effectiveness meant less need for harsh chemical cocktails, which some sensitive users couldn’t tolerate.
Farming and Veterinary Protection
Livestock and plants benefit from the same qualities. Hexachlorophene gives some agricultural disinfectants their reliable track record. Barns, animal enclosures, and processing tools pick up bacteria fast. Surfaces cleaned with solutions containing this compound cut down cross-contamination. This made a real difference in herds prone to mastitis and other tough infections. Farms, vets, and large pet operations saw real improvements in their bottom line when chronic infections started to drop.
The Way Forward: Safer Alternatives and Responsible Use
Public health depends on antimicrobial agents, but every success brings new questions. After the hexachlorophene controversies, researchers took a closer look at substitutes. Chlorhexidine and povidone-iodine became common replacements, but none matched the exact profile of the old standard. That shows the trade-offs scientists have to weigh: balancing powerful action with minimal harm. Today, professional organizations call for ongoing monitoring, careful ingredient selection, and clear labeling for every product. The lesson from hexachlorophene’s story is that even trusted compounds need honesty and caution in every link of the supply chain.
What is the chemical structure and molecular formula of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane?
Understanding the Compound’s Makeup
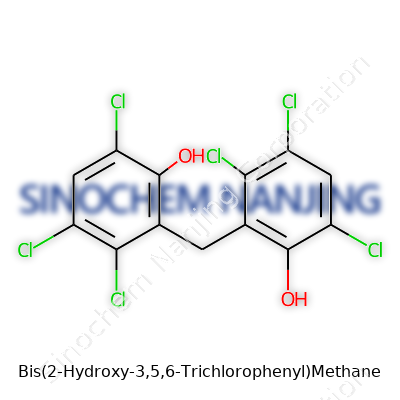
Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane is a bit of a mouthful, but breaking it down makes its meaning clearer. The name gives a map for the structure. Start with "bis," meaning two. Each of these "six-ringed" units is a phenol ring—think of the basic framework in substances like aspirin or BPA. With every phenol ring here, there’s a trio of chlorine atoms replacing hydrogen on positions three, five, and six. A hydroxy group sits on the second carbon. Connecting these two rings, a single methane carbon (just a one-carbon bridge) pulls them together at the spot right next to each phenol’s hydroxy group.
This direct construction puts the molecule in a class that includes a lot of environmental legacy chemicals. Take PCBs or DDT; their structures include several chlorines substituted onto aromatic rings. Such dense chlorination makes the molecule resistant to breakdown. While that means stability, it also makes clean-up and environmental persistence a fact of life.
Molecular Formula and What It Means
Stack up all those functional groups and what’s left is the formula C13H6Cl6O2. Each phenol adds six carbons, five chlorines, and a hydroxy group; the methane bridge supplies a single carbon. Learning where each atom goes becomes more than an academic exercise when tracking environmental exposure, health risk, or designing laboratory syntheses. Researchers and regulators both look at these formulas to predict toxicity and reactivity.
The specific arrangement of atoms makes the difference between a biologically benign molecule and something bioaccumulative in the food chain. Now, in the case of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane, the high number of chlorines points toward high fat solubility and persistence. Not much breaks these bonds quickly.
Real-World Impact and Opportunities for Safer Chemistry
Chemicals in this class, especially with several chlorines, don’t just vanish after use. They stick around in the environment, sometimes far from where they got released. This persistence raises concerns about long-term effects. People with experience in environmental testing know that tracking even small amounts gives a hint at larger trends. Years of environmental sampling have shown these molecules can turn up in soil, water, and sometimes even in wildlife far upstream from any source.
So the conversation isn’t just about what’s in a name or a formula. It’s about how we use synthetic molecules and how we can encourage jobs in green chemistry. Every example of a persistent, bioaccumulative molecule brings a chance to rethink the design. There’s solid promise in approaches that replace chlorinated aromatics with structures that break down faster in the environment. Some researchers suggest swapping out the chlorine for less persistent halogens or designing molecules with "toggle switches" that make them easier to degrade after their useful life.
Learning the structure and molecular formula matters—inside labs, out in the field, and at the policy table. The lesson is straightforward: build for function, but keep a sharp eye on fate and persistence. The structure of Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane, with all those chlorines concentrated on aromatic rings, stands as a reminder of how design choices echo well beyond their chemical equation.
What safety precautions should be taken when handling Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane?
Understanding the Risks
Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane isn't a name you hear around the dinner table. This chemical, sometimes found in specialty labs or used in particular industrial applications, brings its baggage of health hazards. Chlorinated compounds can mess with organs, ignite allergic reactions, or irritate skin. If inhaled, dust or fumes from a powder or granule can stick in the lungs. Splash it on your skin or get it in your eyes? Prepare for redness, pain, and maybe worse if you let it sit too long. Constant handling can make a real impact on health, especially without strong precautions.
Personal Protective Gear: Not Just for Show
Forget the simple gloves you use for cleaning. Tackle this chemical with the same gear you’d wear for any toxic substance. That means acid-resistant gloves—not the vinyl kind you get in a box of 100. Full goggles block chemical splashes from your eyes, because regular glasses won't save your cornea from permanent damage. Toss in a splash-proof lab coat, not just an apron, since this stuff can soak right through casual clothing. If your workplace has a fume hood, don’t hesitate: use it. Breathing fumes or dust, even for a minute, brings trouble. Respirators rated for organic chemicals become essential whenever a risk of airborne particles pops up.
Setting Up a Safe Workspace
Labs that skip on proper storage set folks up for pain. Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane fits best in tightly sealed glass containers, shelved away from acids, bases, and oxidizers. Moisture control matters—a leak or a broken stopper can flood the space with toxic vapors or mess up the chemical’s stability. Every time I’ve seen someone skip out on spill trays, clean-up takes twice as long and doubles the risk. Trap potential spills with secondary containment. Designated work areas fight cross-contamination with food or personal items, and a strong, clear label squashes confusion about contents.
Protocols During Use
Safe handling starts with a plan, not just quick reactions. Always keep a clean workspace—dumping powder onto a cluttered bench invites accidents. Work in small portions to limit exposure, since it’s easier to control a little mess than a big one. A friend in industrial safety once told me, “Treat every spill as serious,” and it stuck with me; sweeping up without a mask or using bare hands amplifies the harm. Grab disposable towels, proper waste containers, and a ready-to-go neutralizing agent before you begin. No one beats luck forever.
Emergency Know-How Saves Lives
Every handler should know his or her routes—frankly, I’ve seen panic erase memory during chemical splashes. The eyewash station and emergency shower must sit close, uncluttered, and clearly marked. Instructions shouldn’t collect dust: everyone should drill the steps to rinse eyes, wash off skin, or call for help. First aid kits with up-to-date supplies shield against panicked improvisation. Incident reporting helps track close calls and teach the next crew better habits.
Supporting Safety Culture
Trust builds through repetition and openness. Regular safety briefings, access to updated data sheets, and a clear message from managers get the team on board. If someone skips gear or regular checks, peer reminders should step in before habits bend out of shape. I’ve seen the most success where curiosity meets respect—workers question old habits, ask for upgrades to PPE, and management listens. That back-and-forth sharpens awareness, cuts down sick days, and shrinks cleanup costs.
Solution-Focused Steps
Outdated protocols don’t serve anyone. Demand investment: proper ventilated hoods, replacement PPE, and continuous training. Promote anonymous reporting for near misses, breaking any stigma around error. Digital checklists guard against memory lapses in busy labs.Move with intent, study each step, and never treat chemical work as routine—there’s always something new to learn.
How should Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane be stored?
Looking Beyond Labels
When you think about chemicals with names as long as Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane, most folks figure this stuff belongs in science labs and nowhere near a backyard shed. That instinct is right. This chemical, known for its use in certain plastics and specialty materials, carries some risks. It’s not the sort of thing you want to treat lightly. A lot of people don’t realize how much the way you store a compound affects your well-being, but proper care protects people, property, and even animals that might cross paths with the container.
Why Storage Calls for Caution
I’ve seen what happens when people take shortcuts. A cracked lid or wrong shelf can spell real trouble. Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane stands out because, despite its value in manufacturing, it’s not something you want in the air, soil, or close to your skin. Persistent organic chemicals sometimes hang in the environment. They also pose risks to workers and anyone else who might handle them during logistics or disposal. Those risks only grow if a container leaks or heats up.
Common Sense Approach to Chemical Storage
Keep it simple. This compound stays safest in cool, dry locations, away from sunlight and anything that heats up—like radiators or motors. Moisture or strong light may trigger changes that up the risk, either by breaking down the compound or creating hazardous byproducts. Some storage rooms have ventilation specifically because even a slow leak from a poorly closed drum raises concerns. Good airflow does more than keep the area comfortable. It gives fumes nowhere to linger.
From what I’ve seen working with supply teams in research settings, the sturdiest containers have tight, chemical-resistant seals. High-density polyethylene or amber glass works better than metal or thin plastics. Regular checks for damaged seals, rust, or bulging matter just as much as following the rules printed on the safety data sheet. Clear labels help, but attention to detail carries a lot more weight.
Personal Responsibility and Preparedness
Everyone—from floor supervisors to new hires—needs to learn the right way to handle chemical storage. Whether you’re running an operation or doing research, training pays off. It’s easy to get careless and let things slide after years on the job, but complacency leads to spills and serious headaches. Having spill kits, goggles, gloves, and eye wash stations nearby goes a long way if trouble pops up. Nobody expects an accident, but fast, informed action keeps an incident small.
Solutions That Make a Difference
Companies have found that double-containment shelving and regular safety reviews solve storage problems before they cost time or health. Using locked, ventilated cabinets and electronic logs to track who checks on the inventory also discourages shortcuts. Neighbors and employees trust businesses that prove their safety is a top priority. Government agencies reward good practices with fewer fines and smoother audits.
Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane doesn’t belong on the list of shelf-stable, easy-going chemicals. Risks may look remote on paper, but real-world experience has shown me: taking shortcuts on storage turns a manageable compound into a real threat. That’s something nobody wants on their conscience—or their hands.
What are the potential health and environmental hazards associated with Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane?
The Unseen Layers of Chemical Exposure
Spend enough time around manufacturing, and you'll hear whispers about certain chemicals that everyone tries to dodge. Bis(2-Hydroxy-3,5,6-Trichlorophenyl)Methane, commonly popping up in specialty plastics and old-school flame retardants, rarely makes headlines. That silence doesn't erase the real threat it can pose to health and the wider environment.
How Chemical Exposure Reaches Everyday Life
I remember the first time tracking industrial dust samples in a plastics molding facility. Nobody thought twice about the residue, yet later we discovered a slew of synthetic byproducts—including this one—clinging to work boots, hair, and breakroom tables.
This compound, a mouthful to say and toxic to boot, belongs to the chlorinated phenols group. These chemicals have a bad track record. They don’t slip quietly from our surroundings but prefer to stick around. Human exposure sneaks in through the air, dust, and even leaching from old consumer products. You’ll see some impact on skin and eyes from direct contact, but what can’t be seen or felt right away demands closer attention.
Health Risks Backed by Research
Workers in production lines inhale or touch residues more often than anyone else. Studies have connected such compounds with hormone disruption. Some members of the chlorinated phenol family link up with thyroid problems, developmental delays for kids, and immune function changes. The EPA and European Chemical Agency have both raised concerns over similar substances. Chronic exposure could tip the risk scale closer to cancer, though not every case draws a straight line.
Households living near dumping grounds or outdated landfills bear the brunt too. Water samples from areas near old chemical disposal sites reveal higher traces than you’d expect—a sharp reminder that contaminants don’t respect property lines.
Environmental Fallout: Persistence and Bioaccumulation
What worries me most isn’t just short-term toxicity, but how this compound travels and lingers. It binds to soil and river sediments instead of breaking down. Fish and birds pick up these residues, carrying them higher up the food chain. Catch-perch in a polluted river, and the chemical could end up in the kitchen. Ecosystems without much margin for error take the hardest hit.
Rain doesn’t wash it away effectively, and UV light only gnaws at it slowly. That long life turns a temporary spill into a lasting problem. Watching environmental monitoring crews haul water and sediment samples year after year, it’s clear recovery takes patience and public vigilance.
Chasing Solutions
Possible solutions grow from collective action. Manufacturers can swap in safer alternatives or tweak processes to contain airborne dust and effluent. Stronger oversight and better waste management limit leaks and dumping. Everyday folks help, too, choosing safer products and asking tough questions about ingredient transparency.
Regular health screening for plant workers and active monitoring near chemical dump sites protects the most vulnerable. Information matters—a simple label, a public database, a better substitute—these steps, though modest, keep pressure on the right side of change.
Any chemical left unchecked risks growing roots in places we least expect. Awareness spreads fastest when communities share findings, demand safety, and refuse to treat health like an afterthought.
| Names | |
| Preferred IUPAC name | Bis(2-hydroxy-3,5,6-trichlorophenyl)methane |
| Other names |
Chlorophenylmethane Methylenebis(trichlorophenol) Hexachlorophene |
| Pronunciation | /ˈbɪs tuː ˈhaɪˌdrɒk.si ˈθriː ˈfaɪv ˈsɪks traɪˌklɔːrəˈfiː.nɪl ˈmɛθ.eɪn/ |
| Identifiers | |
| CAS Number | 29232-93-7 |
| Beilstein Reference | 1462202 |
| ChEBI | CHEBI:34502 |
| ChEMBL | CHEMBL2103839 |
| ChemSpider | 21171300 |
| DrugBank | DB13467 |
| ECHA InfoCard | 45a4cb5e-c67a-442b-9298-60fcee7a0e3b |
| EC Number | 202-621-7 |
| Gmelin Reference | 77122 |
| KEGG | C18122 |
| MeSH | Dichlorophenylmethanes |
| PubChem CID | 13480282 |
| RTECS number | SL8225000 |
| UNII | NJ1M18F55V |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID7018577 |
| Properties | |
| Chemical formula | C13H6Cl6O2 |
| Molar mass | 358.99 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.65 g/cm³ |
| Solubility in water | insoluble |
| log P | 2.98 |
| Vapor pressure | 3.2E-9 mmHg at 25°C |
| Acidity (pKa) | 7.64 |
| Basicity (pKb) | 9.52 |
| Magnetic susceptibility (χ) | -0.00016 |
| Refractive index (nD) | 1.6890 |
| Viscosity | 1200 mPa.s |
| Dipole moment | 3.8894 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 607.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -819.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1136.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AE09 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H410 |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | > 244.4 °C |
| Autoignition temperature | > 470°C |
| Lethal dose or concentration | LD50 Oral Rat: 780 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral > 14 gm/kg |
| NIOSH | SN3500000 |
| PEL (Permissible) | 0.01 mg/m3 |
| REL (Recommended) | 0.01 mg/m3 |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Bis(2-Hydroxy-3,5,6-trichlorophenyl)methane Triclosan Hexachlorophene Bisphenol A Bisphenol S |

