Bis(2-Chloroethyl)Methylamine: A Comprehensive Commentary
Historical Development
Bis(2-Chloroethyl)Methylamine, known to many chemists and historians as a cornerstone of chemical warfare research, entered the scientific landscape in the early twentieth century. Its significance ramps up in times of military conflict, with World War I providing the original stage for studies into nitrogen mustards. Names like “HN2” and “nitrogen mustard” evoke more than simple curiosity; they conjure up decades of strategic study, government secrecy, and medical exploration. Scientists in wartime settings weren’t simply theorizing in ivory towers—they worked feverishly, creating substances thought to offer dramatic tactical advantages. The dual-use nature of such chemicals, drifting between weapons and medicine, often provokes a wave of ethical debates that echo through laboratories and legislative chambers even today.
Product Overview
The compound finds uses beyond the murky world of warfare. Researchers discovered early on that bis(2-chloroethyl)methylamine can alter the growth cycle of rapidly dividing cells, pushing labs to investigate its potential in cancer therapy. It’s found a role as an alkylating agent, carving a niche in both lab research and limited clinical applications. Its molecular structure—methyl attached to a di-chloroethylamine skeleton—lets it react with nucleic acids, a fact that’s led to both breakthroughs and headaches for chemists who need precision and safety in dosing and handling.
Physical & Chemical Properties
Tangible stuff matters in a lab: this compound sits as a colorless, oily liquid with a specific, almost sharp, odor. High volatility under the right conditions raises eyebrows for those working on ventilation and safety planning. With a boiling point in the ballpark of 216°C and a density near 1.1 g/cm³, it’s heavier than water but equally stubborn to contain once released. Solubility often determines the protocol, and bis(2-chloroethyl)methylamine doesn’t dissolve especially well in water, though organic solvents make short work of it. These quirks drive home the importance of grounded, streetwise lab safety—a lesson anyone handling it never forgets.
Technical Specifications & Labeling
Clear and accurate labeling roots itself as an operational cornerstone. Hazard pictograms, GHS signal words, and risk codes jump out from the registration documents. Purity levels tend to run high, above 98%, as impurities can alter reactivity and hazard potential. The batch number and production date, together with detailed storage guidelines, often occupy prime real estate on a bottle, reducing room for error. Technicians and suppliers alike recall stories where a missing digit or faded label spelled disaster, reminding everyone that chemical management isn’t just about paperwork—it’s a matter of lives and property.
Preparation Method
Synthesis routes tend to rely on the reaction between methylamine and ethylene oxide, followed by subsequent chlorination. Industrial-scale setups demand efficiency and safety, using reactors built to withstand the corrosiveness of chlorine while optimizing conversion rates. I’ve sat with plant engineers tweaking stir rates and reactor temperatures, all hoping to squeeze the last drop of yield without risking leaks or unexpected side reactions. The palpable tension in these moments—balancing production goals with safety—makes loss-mitigation planning more urgent than any classroom lesson ever could.
Chemical Reactions & Modifications
The molecule’s two chloroethyl arms behave almost like hands eager to grab onto DNA bases or proteins, a feature exploited in anticancer agents but dreaded in toxic exposure scenarios. Hydrolysis breaks it down in water, especially under alkaline conditions, an old-school trick for decontamination. As researchers fiddle with side chains or swap the methyl group for other alkyl fragments, fresh analogs appear—each one bearing slightly different reactivity, toxicity, and medical potential. Chemists face the constant challenge of predicting which tweaks will boost effectiveness while dialing back risk, a reminder that chemistry rarely hands out one-size-fits-all answers.
Synonyms & Product Names
Names carry baggage. Besides bis(2-chloroethyl)methylamine, industry veterans reference HN2, methyl-bis(2-chloroethyl)amine, or even the more clinical-sounding mechlorethamine. Every name signals different historical or technical contexts, swinging the perception of the molecule from feared war agent to lifesaving medication. It’s not just paperwork and bureaucracy that makes these synonyms matter—the different names show up in regulatory lists, transport protocols, and old journal articles, sometimes tripping up even experienced chemists searching for critical data.
Safety & Operational Standards
Strict protocols reign supreme here. Laboratories and factories alike enforce airtight containment procedures, and handling often requires full-face respirators, chemical-resistant gloves, and diligent air monitoring. Emergency showers and eyewash stations can't sit idle when engineers start pumping solvents. Regular audits and staff training go beyond regulatory box-ticking; they save skin, lungs, and sometimes lives. From my own experience in safety committees, knowing that secondary containment trays and sealed hoods stand ready provides practical confidence—actual readiness to respond to spills or unexpected exposures. Failure in this arena rarely leads to simple inconvenience; it can mean hospitalization or worse, making inattention a luxury no one enjoys.
Application Area
The molecule’s story doesn’t settle for a single track. Beyond chemical defense studies, labs explore cancer treatment properties, particularly in Hodgkin lymphoma and other blood-related cancers. Its tendency to disrupt DNA means doctors and researchers remain both hopeful and cautious, squeezing therapeutic value from danger. In the industrial world, alkylating agents feature in polymer and agricultural chemistry, and even scientific authors occasionally stumble onto references tucked away in niche industrial patents. Growing public and regulatory scrutiny means these varied application areas worry about more than profits—they face frequent, sometimes heated, public conversations about acceptable risk.
Research & Development
Scientists and clinicians push to understand how the molecule’s alkylating power can both harm and help. Structure-activity relationship studies fill volumes, mapping each tweak’s effect on toxicity, stability, and medical usefulness. Researchers have spent whole careers investigating how to make these agents more selective—aiming fire at cancer cells without destroying healthy tissue not far away. It’s common to find large, multinational teams working on analog design, delivery systems, and evidence synthesis about long-term outcomes. Competitive grants come and go, but dedication to unraveling the molecule’s complexities never really slows down.
Toxicity Research
Toxicity defines the molecule’s darkest legacy. Exposure burns skin, trashes airways, and can lead to lasting organ damage. From rat studies to hospital case reports, the literature swells with warnings. Acutely, the compound causes vesication and even genetic mutations. Regulatory bodies in Europe and North America lay down occupational exposure so low that trained professionals sometimes need to push analytical limits just to confirm compliance. Doctors, toxicologists, and public health researchers recalibrate limits as new evidence emerges. Survivors of accidental or deliberate exposure speak plainly about suffering and long-term rehab, which shifts public attitudes, sometimes sharply, toward risk intolerance.
Future Prospects
Many expect the role of bis(2-chloroethyl)methylamine to shrink in warfare contexts thanks to international treaties and public revulsion, but its place in medicine and lab research keeps evolving. With precision oncology gaining steam, companies and labs target ways to build in selectivity, so that alkylating agents move closer to “magic bullet” status with fewer nasty surprises. Green chemistry drives development of safer synthesis and decontamination approaches, as laboratories and industry respond to both consumer demand and regulatory clampdowns. Watching regulators, patient advocates, and scientists wrestle with the old challenge—can a dangerous tool be harnessed for good—one thing becomes clear: the debate isn’t cooling off any time soon.
What is Bis(2-Chloroethyl)Methylamine used for?
Unmasking the Purpose Behind the Chemical
Anyone raised on stories about the World Wars probably heard about mustard gas. The chemical behind it, Bis(2-Chloroethyl)Methylamine, better known as HN2 or Nitrogen Mustard, holds a grim place in history. Unlike most industrial chemicals you’d find in the corner of a garage or warehouse, this one was never made to build anything, clean up messes, or keep factories humming. Its purpose was to hurt, disable, and kill humans. Military labs across the world churned out a variety of these compounds in the twentieth century, not for progress, but for destruction.
From Battlefields to Hospitals
Growing up, I never thought something as terrifying as a chemical weapon would wind up in the world of medicine. After the horror stories of World War I and World War II, scientists noticed that soldiers exposed to mustard agents showed changes in their blood, particularly in their white blood cells. Doctors realized these compounds had a powerful effect on rapidly dividing cells. That’s the mechanism behind today’s use in chemotherapy. Oncologists use derivatives of Bis(2-Chloroethyl)Methylamine to slow or halt cancers that feed off quick cell division.
The American Cancer Society confirms that mechlorethamine, a close cousin of HN2, shows real effectiveness against lymphomas, some leukemias, and even testicular cancer. Of course, nobody celebrates the way these drugs make patients feel. Nausea, immune suppression, and hair loss aren’t easy to bear. The benefits tip the scale in favor of treatment. We’re talking about a poison from the battlefield, tamed and resuited to fight disease rather than fuel war. It’s a strange sort of progress, turning a weapon into medicine.
Keeping a Tight Grip: Security and Regulation
Government agencies like the CDC and the World Health Organization keep these chemicals on close watch. The United States tightly controls manufacturing, storage, and sale, tracking every gram through regulation and paperwork. I know folks who work in healthcare logistics and they don’t take these rules lightly. Mishandling or theft remains a real fear, especially in a world where rogue states and non-state groups show interest in chemical weapons. The risk doesn’t just lie in intentional attack, but in accidents and environmental contamination.
Risks and Reminders
The chemical lingers in some places long after wars end. Even decades later, you’ll hear of old munitions found in fields or on the seabed, leaking toxins into the environment. Cleanup teams put their health on the line every day, and the job often takes more time and money than people realize. Exposure can scar lungs, burn skin, and cause long-term cancer risk. The environmental hazards alone stretch the ethical cost of producing such substances in the first place.
What Can Be Done?
Education has power here. People deserve honest accounts about the risks, not just dry technical facts. Disposal programs deserve support to keep these chemicals far from weak hands. In hospitals, cancer patients rely on these drugs, but research into less toxic and more targeted treatments continues. If we don’t take these stories seriously, the consequences could repeat in ways we regret.
Bis(2-Chloroethyl)Methylamine’s story cuts across science, politics, and ethics. Some inventions carry more weight than others—sometimes in ways that haunt us, sometimes in ways that save a life.
Is Bis(2-Chloroethyl)Methylamine hazardous or toxic?
Looking Beyond Chemical Names
Bis(2-Chloroethyl)Methylamine often hides behind its technical label, but many know it for a darker chapter in history: it’s the core of nitrogen mustard, once developed for chemical warfare. That fact alone offers a big clue about its risk level. In everyday life, it’s rare to run across this substance—few workplaces, hospitals, or labs handle it outside of tightly controlled settings. Still, the legacy of nitrogen mustard continues in modern treatment as a foundation for some cancer-fighting drugs, although those are tweaked forms, designed with strict safeguards.
How Dangerous Is It, Really?
Exposure stories catch attention because the hazards are not minor. Touching Bis(2-Chloroethyl)Methylamine can burn skin or eyes quickly. Breathing in its vapors stings the lungs, brings coughing or choking, and can cause lasting lung trouble. I’ve worked with a range of lab chemicals over the years, and clear guidelines always stand out for compounds like these. Splash protection, powerful exhaust systems, and sealed containers become the norm whenever anything with “mustard” in the name comes on the inventory list.
Much of the concern also comes from how this molecule behaves inside the body. At the cell level, it scrambles DNA and blocks repair, which is why tiny doses have found a home in chemotherapy. Outside of medicine, uncontrolled exposure creates cancer risk, not just for lab folks, but for anyone who comes in contact during spills or improper disposal.
Why The Bigger Conversation Matters
Stories about chemical hazards highlight gaps in public trust and understanding. After big industrial accidents or reports of chemical misuse, people want clear answers: how dangerous is it, and are we protected? It’s not enough for scientists or companies to publish complex safety data. Communication has to bring facts to the ground—for instance, explaining that the severe risks of this chemical come from direct, unprotected contact. You won’t face it at the grocery store, but stories occasionally surface from forgotten military dumps or lab mishaps.
Facts Back Up The Worry
Researchers in toxicology and occupational medicine point to Bis(2-Chloroethyl)Methylamine as a textbook example of a chemical that demands caution. The National Institute for Occupational Safety and Health (NIOSH) lists it as a potential occupational carcinogen. Regulatory groups like the EPA and World Health Organization keep it on hazard lists. Those with compromised immune systems or chronic lung conditions are even more at risk if exposed, and animal studies confirm long-term exposure links to cancer and organ failure.
What Works for Prevention
Health and safety always start with reliable information. Chemical handlers need hands-on training—not just reading manuals. Proper labeling, specialized storage, and disposal routines really save lives. Local first responders need alerts about what sits in facilities nearby, so they arrive ready if a spill or fire happens. Strong legal oversight, including regular inspections and stiff fines, backs up these safety steps. Digital inventory systems help labs or factories know exactly what’s on the shelf, avoiding forgotten stockpiles that deteriorate and cause trouble years down the line.
Anyone who’s watched a workplace review or a public safety drill feels the unease mixed with relief—most mistakes can be prevented with enough layers of vigilance. Bis(2-Chloroethyl)Methylamine doesn’t show up in everyday life, but its risks remind us why rules, transparency, and community awareness can’t take a day off.
What is the proper storage method for Bis(2-Chloroethyl)Methylamine?
Real-World Dangers Call for Real Storage Solutions
Anyone who has ever spent time in a laboratory—whether academic, industrial, or military—knows chemicals like Bis(2-Chloroethyl)Methylamine don’t come with room for carelessness. Known in some circles by its notorious alias, "HN2," this compound causes harm to people and the environment. Without strong storage habits, its risks can sneak up on even cautious hands.
Ignoring Storage Risks Is Not an Option
A lot of people worry about the mess of paperwork involved in chemical handling, but paperwork fades in importance the first time you smell something pungent when you walk into the storeroom. With Bis(2-Chloroethyl)Methylamine, leaks or degraded containers spell trouble. Exposure can lead to blistering injuries and respiratory problems, and the impact reaches past the building walls to threaten soil and water.
The Role of Environment: Not Every Room Works
Long-term safety means more than tossing this compound onto the nearest shelf. Standard practice involves keeping it in airtight, chemically resistant containers that can handle leaks and won’t corrode. From long days in chemical storerooms, I’ve seen plastic degrade and glass containers shatter when humidity and temperature climb out of the safe range.
The temperature matters a lot. Bis(2-Chloroethyl)Methylamine stays put at a cool 2–8°C. Any warmer, and the potential for decomposition or vapor build-up grows. Too cold, and some containers get brittle. If space doesn’t have environmental controls, somebody will pay for it. Chemical refrigerators and isolated storage cabinets labeled with clear hazard warnings aren’t window dressing, they’re there because past mistakes taught hard lessons.
Why Separation and Security Matter
The worst feeling comes from realizing incompatible chemicals sat together for months. Strong oxidizers turn a Bis(2-Chloroethyl)Methylamine spill into a fire hazard. Security against theft matters, too. Many chemicals have stories tied to criminal misuse, and Bis(2-Chloroethyl)Methylamine sits near the top thanks to its origin as a chemical warfare agent. Locked doors, record logs, and regular checks keep accidents and intentional damage at bay.
People Need to Understand the Chemical, Not Just Fear It
Training goes further than showing someone where goggles are stored. Misunderstanding dangerous chemicals leads to shortcuts. In my experience, honest talks about historic accidents leave an impression—often more lasting than any poster warning in a hallway. Safety data sheets and training sessions shouldn’t gather dust.
Reducing Risk with Good Habits
Regular inspections, inventory updates, and real accountability for storage save lives. Cleanup gear—neutralizers, absorbent pads, emergency showers, and eyewash stations—must always be nearby, never locked away or blocked by boxes. If people walk by safety equipment every day, muscle memory starts kicking in, making the right response second nature during an emergency.
Building a Culture, Not Just Rules
Any storage method for a hazardous chemical only works if the whole team believes in the system. I’ve seen management pay for the best cabinets and monitoring sensors, but if tired staff forgets the basics or hesitates to speak up, disaster gets its chance. Open communication keeps standards high, and a shared sense of responsibility ensures history won’t repeat itself on your watch.
How should accidental exposure to Bis(2-Chloroethyl)Methylamine be handled?
Every Second Counts
Real life rarely sends warnings before danger arrives. Bis(2-Chloroethyl)Methylamine, better known as HN2 or nitrogen mustard, belongs to a family of chemicals with a nasty reputation. This compound causes damage the moment skin or lungs meet it. Nothing about an accidental spill or inhalation feels ordinary, and waiting for official instructions only wastes time you might not have.
Getting Away from Danger
The first move after exposure: leave the area. This stuff clings to skin and clothes, and vapor fills a room before you notice. Doors and windows open fast, people move upwind, and the urge to “take just a second” for a phone or bag must get ignored. Walking out, not running, keeps panic down and cuts the risk of falling or spreading the chemical farther.
Skin and Eyes: Immediate Actions
HN2 burns tissue quickly, but water works better than nearly anything else for a fast initial rinse. Strip off every piece of clothing, including shoes and accessories. Throw those into a plastic bag, seal it, and move it away from others. For every exposed inch of skin, flush with flowing, clean water. A shower beats a sink because chemicals collect in hair and folds. Soak for at least 15 minutes. Lenses in the eyes trap the chemical, so take them out. Rinse both eyes with gentle, low-pressure water and blink on purpose during washing. Do not rub the skin or eyes during this—for chemicals like nitrogen mustard, friction pushes it in deeper.
Mouth, Lungs, and Fear
Breathing in HN2 feels like swallowing smoke. Coughing, shortness of breath, and a metallic taste mark exposure. Getting to fresh air, sitting upright, and keeping calm help. Doctors warn that panic increases oxygen demand, which can worsen breathing problems. Swallowing or inhaling the liquid form raises the risk of life-threatening harm. Don’t induce vomiting or drink anything in an attempt to “wash it out”—medical support does more than home tricks.
Why Urgent Medical Care Matters
HN2 doesn’t just burn the outside; it damages DNA, cripples bone marrow, and sets off organ failure if left unchecked. Visible blisters show up hours after exposure, not immediately. Lowered white cell counts, unexplained fevers, or bleeding can appear days later. Emergency care teams bring more than bandages—they monitor blood, supply oxygen, and treat blisters in controlled settings. No at-home measure matches a proper hospital’s resources.
Prevention Over Panic
Hospitals and first responders already practice for chemical spills, but most accidents happen because somebody forgot gloves, goggles, or a fume hood. Anyone using HN2 needs gear listed on the safety data sheet: face shields, nitrile gloves, closed shoes, and tight workspaces with proper airflow. Training sessions save more lives than warning labels—so does having a spill kit and emergency shower nearby. Posting emergency numbers near lab benches reminds those on site that help is never a phone call away.
Building Trust through Accuracy
What you do in the first few minutes after accidental exposure can decide outcome days later. No one should feel embarrassed about seeking help. I once watched a seasoned researcher fumble a bottle and instantly shout rather than hide the mistake—minutes later, the team stood in showers, doctors arrived, and, most importantly, nobody suffered long-term effects. Preparation and honesty beat wishful thinking every time, and they start with sharing honest accounts, not just checklists.
What are the chemical properties and structure of Bis(2-Chloroethyl)Methylamine?
Chemical Character and Structure
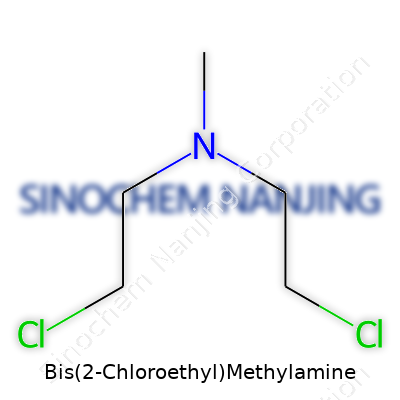
People working in labs probably know Bis(2-Chloroethyl)Methylamine under another name: HN2, or as a nitrogen mustard. This compound stands out for its role in both science and, historically, in less savory places. Structurally, its formula reads C5H11Cl2N. Picture a methylamine backbone—one nitrogen atom bonded with one methyl group and two 2-chloroethyl arms swinging from the sides. The key functional parts are these two chloroethyl chains. Each chain takes a hydrogen from methylamine and sticks on a chloroethyl group, leading to its infamous reputation as both a chemotherapeutic and a vesicant.
Looking at this from a chemist’s perspective, those nitrogen and chlorine atoms define the game. The chloro group makes each chain very reactive. Drop this molecule into the right environment—say, a slightly alkaline solution—and you see those chlorines split off easily, leaving behind a positively charged nitrogen ready to form new bonds with other molecules. That reactivity drives almost everything HN2 does, both in industry and in biological systems.
Properties That Matter Outside the Textbook
If you sniff around a lab, you probably won’t ever forget the faint fishy odor this compound gives off. Bis(2-Chloroethyl)Methylamine shows up as a colorless, oily liquid at room temperature. It dissolves easily in water, ethanol, and many organic solvents. Its boiling point sits lower than a typical organic solvent. Leaving the lid off, you risk it drifting away and causing harm, especially if you get a whiff. The air around it doesn’t just smell—it can harm tissue at very low concentrations.
This chemical reacts quickly and aggressively with many nucleophilic compounds. That ability is why it earned past (and sometimes ongoing) use in warfare and cancer treatment. Its reactions with DNA are what both made it a weapon and a therapy. Once inside a cell, Bis(2-Chloroethyl)Methylamine’s reactive nitrogen forms cross-links with DNA, jamming up cell division. That leads to cell death or, in controlled doses, to the death of cancer cells. Unlike many chemicals, its impact shows up right away—and not just on paper, but in people’s lives.
Risks, Regulation, and Smart Use
My own experience, and that of my colleagues, underscores the danger. Even low exposures to this compound require heavy protective gear and strong ventilation. Skin contact causes painful blisters. Toxic fumes fill a room in moments if you spill even a small amount. Several countries regulate and track this chemical closely because of its dual identity: valuable in medicine, hazardous as a weapon.
Science has to find a balance—making the most of Bis(2-Chloroethyl)Methylamine’s ability to fight disease, while guarding against its risks. Proper training, chemical hoods, and careful labeling help keep chemists and the public safe. Research continues on alternatives that break apart faster in the environment or target only cancer cells, reducing the shadow of harm. In my work, that responsible approach matters more than anything. Understanding why this molecule reacts the way it does—and how that shapes everything from cancer treatment to historical conflicts—keeps us from repeating mistakes and encourages smarter, safer science.
| Names | |
| Preferred IUPAC name | N-methyl-N-(2-chloroethyl)ethanamine |
| Other names |
HN2 Nitrogen Mustard Mechlorethamine Chlormethine |
| Pronunciation | /ˌbɪs.tuː.klɔːr.oʊˈɛθ.ɪlˌmɛθ.əlˈæm.iːn/ |
| Identifiers | |
| CAS Number | 51-75-2 |
| Beilstein Reference | 63553 |
| ChEBI | CHEBI:63625 |
| ChEMBL | CHEMBL1059 |
| ChemSpider | 7284 |
| DrugBank | DB06717 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.003.222 |
| EC Number | 205-793-9 |
| Gmelin Reference | 82887 |
| KEGG | C02197 |
| MeSH | D001726 |
| PubChem CID | 15686 |
| RTECS number | BZ9625000 |
| UNII | T7V2R6K4FS |
| UN number | 2810 |
| Properties | |
| Chemical formula | C5H11Cl2N |
| Molar mass | 153.05 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 1.14 g/cm³ |
| Solubility in water | soluble |
| log P | 0.71 |
| Vapor pressure | 1.72 mmHg (at 25 °C) |
| Acidity (pKa) | 8.1 |
| Basicity (pKb) | 4.35 |
| Magnetic susceptibility (χ) | -30.31·10^-6 cm³/mol |
| Refractive index (nD) | 1.4960 |
| Viscosity | Non-viscous liquid |
| Dipole moment | 3.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 285.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -85.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4824 kJ/mol |
| Pharmacology | |
| ATC code | L01AA06 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause genetic defects; may cause cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H350, H351, H341, H319, H315, H317, H335 |
| Precautionary statements | P261, P280, P301+P310, P305+P351+P338, P307+P311, P405, P501 |
| Flash point | 65°C |
| Autoignition temperature | 215 °C |
| Lethal dose or concentration | LD50 oral rat 20 mg/kg |
| LD50 (median dose) | 10 mg/kg (rat, oral) |
| NIOSH | NL3675000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.001 mg/m3 |
| IDLH (Immediate danger) | IDLH: 15 mg/m³ |
| Related compounds | |
| Related compounds |
Nitrogen mustard Chlormethine Cyclophosphamide Ifosfamide Melphalan |

