Bis(2,3-Epoxypropyl) Ether: A Look Beyond the Beaker
Tracing the Path: Historical Development
Chemical discovery usually begins with curiosity and fills out with purpose. Bis(2,3-epoxypropyl) ether didn’t land in labs as the star of the show. It came along during the remarkable mid-20th century stretch when researchers dug into epoxide chemistry, seeking compounds with practical industrial benefits. Early explorations pointed to strong cross-linking potential, which opened doors for fields like polymer manufacturing and epoxy resin technology. As application matured, so did our understanding of safer production, better stability, and a more responsible approach to its risks.
Understanding the Product Landscape
Anyone who’s spent time in a synthetic lab can appreciate how a compound like bis(2,3-epoxypropyl) ether changes the conversation. It falls under the general umbrella of glycidyl ethers, notable for the distinct pair of oxirane rings. Unlike single-ring epoxides, this structure brings added flexibility to chemists seeking reactive intermediates. It’s clear why people reach for this compound in making resins tougher or adhesives more versatile. Names don't always tell the full story, but you’ll see it labeled with terms like diglycidyl ether or DGE, all nodding to its reactive backbone.
Diving into Physical and Chemical Details
Life in the chemical plant revolves around the quirks of substances. Bis(2,3-epoxypropyl) ether sinks in as an oily, nearly colorless liquid. That doesn’t seem remarkable, but its boiling point and miscibility speak volumes for anyone working on process design. Two reactive epoxide rings mean you have plenty of chances to modify, cure, or cross-link—all central to the role it plays in making adhesives stick longer or coatings last in tougher conditions. Its chemistry stands up: stable if bottled properly, quick to form networks in the hands of a skilled formulator, and never shy about reacting when given the right trigger.
Technical Specifications and Clear Labeling
Lab workers rely on labeling that gets right to the point. Accurate details matter. Purity percentage, allowable water content, and inhibitor presence all shape reaction outcomes. Safety labeling should match global hazard standards—GHS pictograms, signal words, and hazard statements make a real difference in keeping workers alert. From years spent managing chemical inventories, I know how much attention a clear, honest label saves when situations get tense.
How it’s Made: Preparation Method
Behind every bottle of bis(2,3-epoxypropyl) ether, there’s a multi-step dance of reactants that requires careful balance. Often, the synthesis starts with epichlorohydrin, which hits the mark for both reactivity and availability. A condensation with glycerol or a related diol, often steered with a base, stretches out the carbon chain and brings both epoxide rings into play. Controlling pH, temperature, and water content in the reactor isn’t about ticking boxes—it’s about avoiding runaways, off-spec product, or worse. Coming from a pilot-plant background, attention to these variables often separates a clean batch from a hazardous mess.
Inside the Flask: Chemical Reactions and Modifications
People value bis(2,3-epoxypropyl) ether for more than just raw reactivity. The compound cracks open all kinds of possibilities with its dual epoxide groups. Direct addition and ring-opening reactions lend themselves to making polyethers or grafting onto polymers for new properties. Cure chemistries that use hardeners or acid catalysts give finished materials—like floor coatings or fiber-reinforced plastics—a resilience that stands up in tough settings. Modifying or extending the core molecule spins out hundreds of resin recipes, each fine-tuned to end-use demands in aerospace, electronics, or construction.
Other Names in Use: Synonyms and Product Names
Sifting through chemical catalogs, you’ll meet alternative names, including DGE and diglycidyl ether. There’s value in learning the aliases; missing a synonym can derail a literature search or complicate regulatory checks. In practice, recognizing these variants shortens the time it takes to find material safety data, trace studies in journals, or check for legal restrictions.
Staying Safe: Operational Standards and Handling
Chems like this aren’t toys. Past experience has drilled in the need for solid ventilation, good gloves, and full-face protection when mixing or transferring bis(2,3-epoxypropyl) ether. It’s not acute toxicity that gets you—it’s the repeated contact, the low-level exposures, and the slow build-up of risk that comes with shortcuts. Following OSHA and REACH guidelines isn’t a matter of filling compliance checklists; it keeps workers healthy and lawsuits out of the headlines. Proper training, updated emergency protocols, and a habit of double-checking container seals all play a bigger role than most people think.
Where the Work Happens: Application Areas
Industry insiders know bis(2,3-epoxypropyl) ether appears wherever tough, long-lasting polymer systems are needed. Epoxy resins based on this compound hold up bridges, line wind turbine blades, and protect electronics from moisture and shock. Its broad adoption in adhesives, coatings, and sealants points to how manufacturers value rugged, chemical-resistant networks. Emerging uses in composite materials and high-tech electronics design suggest the compound will keep riding the wave of advanced engineering sectors. Anyone who’s laid down a two-part flooring epoxy or watched engineers patch composite aircraft fairings knows the practical payoff of chemistry done right.
New Science: Research and Development
Ongoing research rarely sits still with legacy compounds. New projects focus on reducing environmental footprint during manufacture and use, dialing up biodegradability, and finding safer curing methods to slash emissions. Work in universities and corporate labs often circles back to the same core question: Can we make things lighter, stronger, and more sustainable without pushing up cost or toxicity? My own past collaborations with polymer scientists taught me how big the rewards can be when innovation threads the needle on safety and performance.
Toxicity: Hidden Risks and Ongoing Study
Long-term studies continue to ask tough questions about the safety of bis(2,3-epoxypropyl) ether. Its potential to irritate skin and cause sensitization remains a concern, especially for workers with high exposure rates. Some animal studies flagged genotoxic potential, pressing regulators and companies to tighten monitoring and exposure limits. As with most legacy chemicals, the aim now is to track, reduce, and, where possible, substitute less hazardous alternatives. Real change relies not on wishful thinking, but on direct investment in better fume controls, hands-on safety training, and honest reporting of workplace incidents.
Looking Down the Road: Future Prospects
As regulation gets tighter and public awareness rises, demand for safer, greener chemical technology grows. Interest continues to shift toward renewable epoxide sources and formulas that leave smaller environmental footprints. The challenge won’t just come from regulatory pushback—it’ll come from customers who want transparency in how things are made and what ends up in the air and water. Smart research leaders already focus on greener synthesis and smarter recycling of epoxide-based materials. It’s hard to predict exactly what the next ten years will bring for bis(2,3-epoxypropyl) ether, though history says the most adaptable compounds, and the most responsible firms, will be the ones shaping what’s next.
What is Bis(2,3-Epoxypropyl) Ether used for?
A Surprising Player in Industrial Chemistry
Bis(2,3-Epoxypropyl) Ether—sometimes known as diglycidyl ether—hardly makes headlines, yet for anyone who has ever mixed up epoxy glue or admired the finish on a sturdy floor, this chemical plays a key role. It springs up most in the world of epoxy resins, where it helps connect molecules together and harden things for the long haul.
Foundation of Strong Epoxy Resins
Think about the toughest adhesives you have at home, like the glues that fix broken ceramics or set coat hooks on walls. Bis(2,3-Epoxypropyl) Ether brings chemicals together, sparking the reactions which create the solid plates and coatings industry depends on. Epoxy resins aren't just about glue—they shield garage floors, help circuit boards survive heat, and line pipes so water doesn’t rust metal from the inside out.
Protection and Practical Uses
Many manufacturers put this ether to work in coatings and adhesives designed for long-term toughness. In construction, resins made with it keep concrete strong against water and salt. The same kind of process goes into the paint on ships that keeps rust at bay, and the finish for your tabletops that resists spills and scratches.
If you look at electrical gadgets, you’ll spot the footprint of Bis(2,3-Epoxypropyl) Ether in the insulation around wires and the protective shell of circuit boards. Modern electronics count on stable, long-lasting surfaces, so the material science behind them gets tested in real-world use every day.
Balancing Safety and Production
It’s not all smooth sailing. Diglycidyl ether comes with health risks for workers and anyone exposed without enough protection. Skin contact can cause irritation and allergies, and breathing in its vapors poses even steeper risks. Experience, both in the lab and on factory floors, taught many of us how easy it is to underestimate these dangers. For years, some workplaces skimped on gloves or ventilation, which led to rashes, eye problems, or respiratory irritation.
Solutions to these safety challenges exist, but following them takes real effort. Industries now often use closed systems, meaning the ether doesn’t escape into the air. They also keep strict checks on protective gear and train workers on safe handling. People who actually mix resins or coat surfaces know it’s not enough to just read a label. Frequent training, real-time air monitoring, and prompt cleanup after spills—these small steps save health in the long run.
Looking Forward with Responsible Use
As researchers and consumers, we all want safer, greener chemistry. There's a push to develop alternatives with fewer health risks. Until those solutions arrive, careful oversight keeps things safer. Transparency from companies and stronger workplace rules go a long way. I’ve seen both sloppy and careful companies in action—the careful ones end up saving money, time, and well-being, proving that responsible use isn’t just a legal box to check.
So, next time you trust a strong bond or admire a crisp, durable floor, there’s a decent chance Bis(2,3-Epoxypropyl) Ether helped make it happen. Taking its risks as seriously as its benefits might not grab headlines, but it keeps both products and people in good shape.
What are the safety precautions when handling Bis(2,3-Epoxypropyl) Ether?
Working with a Potent Chemical
Bis(2,3-Epoxypropyl) ether shows up in epoxy resins, paints, and some lab work. Most folks don’t bump into it at home, but for those of us in labs or workshops, the risks are pretty clear once you see the safety label. Even brief contact with skin or lungs can set off burning, rashes, or worse. I remember the sting after a splash hit my wrist years ago, and how fast the red patch spread. Safety guidelines got a lot more real to me then.
Getting the Right Gear in Place
Direct contact causes problems, so gloves, snug goggles, and a lab coat come first. Latex and nitrile don’t always block every solvent, so check for gloves rated for organic chemicals. I learned early to double up if my hands would stay near a spill or splash zone. A fitted respirator or solid ventilation keeps vapors out of your lungs. The smell can be faint—never trust your nose; always trust the fume hood.
Preparing Space and Watching the Air
Rooms handling Bis(2,3-Epoxypropyl) ether call for serious airflow toward exhaust fans, not across your face. Long ago, a sloppy setup in a crowded workspace sent fumes straight to where new students were working. The coughing fit that followed showed how easy it is for exposure to get overlooked. Air monitors for volatile organics aren’t just for big companies. Even small labs gain peace of mind with a decent detector on the wall.
Spill Plans and Fast Reactions
Spills happen, even with steady hands. Oversight leaves residue on benchtops, but forgetting to decontaminate allows skin exposure hours later. Keep a spill kit near—granules for soaking up, disposable towels, and an acid-resistant bucket for waste. Everyone working nearby must know how to sweep in, contain the mess, and report the incident. I learned to trust my habits only after I saw how quickly an unnoticed drip could eat through plastic gloves.
The Small Stuff Adds Up
Every touch, every breath counts with this ether. Rashes build up quietly; allergies flare after years of small exposures. The science is clear: repeated contact links with chronic skin and respiratory problems. Health checkups in work environments make a real difference—ignoring them out of overconfidence leaves lasting scars. Posting clear signage does more than check a legal box; it picks up tired eyes before a mistake turns into an injury.
Simple Routines Make the Difference
Waste disposal matters almost as much as handling. Draining this stuff into sinks risks water contamination and health. Secure all waste in tight-sealing drums, keep detailed logs, and use labeled bags for contaminated gloves and cloths. I’ve seen what happens when someone tries to cut corners here; fines pile up, and cleanup becomes a much bigger issue.
Teaching Never Stops
Nothing replaces hands-on walkthroughs and real stories. People learn from each other. Newcomers should shadow veterans on their first encounters. We all carry the lessons, scars, and the weight of minor mistakes. Honest talk about risks and respect for Bis(2,3-Epoxypropyl) ether builds a much safer workspace.
What is the chemical formula and structure of Bis(2,3-Epoxypropyl) Ether?
Taking a Closer Look at the Chemistry
Bis(2,3-Epoxypropyl) ether stands out in the world of synthetic chemistry. This compound serves as an important ingredient in making things like industrial adhesives, certain plastics, and coatings found on a range of familiar products. If you handle materials science, knowing its details isn't just trivia — it's foundational for safely developing, using, and regulating materials that enter the market and environment.
Chemical Formula and Molecular Structure
Its molecular formula is C6H10O3. The structure forms by linking two glycidyl (epoxypropyl) groups to either side of an oxygen atom, creating a basic “ether” skeleton. If you sketch it, the core has an oxygen atom bridging two identical arms, each carrying an epoxide ring. Chemists often call this shape “diglycidyl ether.” Pin down the molecule, and you’ll see two oxirane (three-membered cyclic ethers) rings, crucial for its chemical reactivity and usefulness.
Why Structure Drives Reactivity
Speaking from time spent experimenting with epoxy systems, the real impact comes from those small, tense oxirane rings. Each ring stores substantial chemical energy. Introduce the molecule to hardeners or certain catalysts, and it “opens” those rings in a reaction known as curing or crosslinking. This property lets manufacturers customize material hardness, flexibility, or chemical resistance, shaping how products like floor coatings and sealants perform in everyday life. The backbone offers ether stability, so the molecule doesn’t fall apart under heat or light — not all synthetic chemicals hold up so well.
Health, Safety, and Environmental Issues
Using Bis(2,3-Epoxypropyl) ether has its downsides. The very feature that brings value — high reactivity — creates risks, too. Skin exposure can set off allergic reactions and irritation. Breathing vapors in enclosed spaces poses respiratory issues. I’ve seen some workers in small shops underestimate these hazards or skimp on gloves, learning the hard way after rashes and coughing spells. Realistically, most people outside chemical manufacturing won’t encounter this compound, but those who do benefit from strong education and clear labeling at every step.
Environmental persistence can’t be ignored. Because this ether stands up to heat and chemical attack, spills stick around and may affect water supplies or soil. Regulation mandates careful storage and disposal. The industry moves forward with standards for proper ventilation and containment, yet slips and accidents still occur. That makes basic education and accountability central to minimizing harm.
Finding Smarter Solutions
A safer path often rests on substituting less hazardous chemicals, refining production to avoid byproducts, and using personal protective equipment without exception. Transparent communication around hazard data and prompt spill response both keep workplaces safer. Efforts to develop “greener” epoxies receive support from both governments and corporate buyers — there’s strong incentive from regulations and consumer demand. I've noticed more manufacturers shift toward sourcing alternatives with less environmental impact, proving that innovation and responsibility can move together.
A Chemical That Deserves Respect
Bis(2,3-Epoxypropyl) ether, with formula C6H10O3, delivers unique performance in synthetic materials but also calls for thoughtful handling due to its high energy rings and strong chemical resistance. Knowing both structure and consequences underpins smart use and points the way to practical improvements in safety and sustainability wherever this powerful molecule shows up.
How should Bis(2,3-Epoxypropyl) Ether be stored?
The Real Risks of Mishandling
Anyone who works with chemicals knows the dangers that come with Bis(2,3-Epoxypropyl) Ether (BEE). One incident in a poorly ventilated storeroom can teach you a tough lesson. Ethereal scents don’t just float in the air—they can irritate your lungs and burn your skin if you brush up against a leaking drum. For most people outside the lab, these warnings sound overblown. For folks who mix and handle this stuff every day, stories about mishaps hit close to home. Emergency showers, skin rash, and ruined boots stick in your memory.
Temperature Strikes Back
One fact you keep close—this chemical reacts to temperature changes. Warm rooms ramp up its vapor pressure, meaning fumes can build up even if you think you’ve sealed everything tight. Cool, dry spaces matter a lot. Keeping temperatures below 30°C won’t just prolong shelf life, it also cuts down on dangerous vapors. Inflamed faces and stinging eyes make you believe it. Sometimes storerooms get ignored in budget talks, but even inexpensive temperature controls pay dividends in workplace safety.
Separation Isn’t Just a Convenience
Shelving matters. I’ve sorted through crowded shelves, searching for a missing bottle, only to find incompatible chemicals jumbled together. Acids, strong bases, oxidizers—none of these belong near BEE. One knocked-over jug could trigger a fire. Segregated storage might seem like common sense, but it’s one of the first corners cut as workers get busy. Fire-resistant cabinets and clear labeling keep daily chores from becoming headlines.
Keep It Tight—Seal It Right
A worn cap or a poorly sealed drum turns into a headache fast. Leaks get sticky, equipment corrodes, and surfaces become slippery. Every maintenance shift means wasted time and risk. Investing in airtight, chemical-resistant containers always pays off. Rags, gloves, and eye protection aren’t luxuries in this environment. Basic housekeeping—cleaning up spills and checking seals—cuts down on health risks and supply loss.
Information and Real Training
Reading a safety data sheet once doesn’t mean you’re set for life. I’ve seen labels get smudged, paperwork vanish, and procedures slowly drift from what’s printed. Real training sticks when you repeat it, clarify it, and chase down rumors. Regular safety tours and clear signage keep everyone on track, whether they started this month or have decades of experience. Nobody can afford shortcuts, and those small acts build real expertise over time.
Solutions to Common Weak Points
Bringing up safety feels repetitive, but it’s exactly how incidents get reduced month after month. Storage rooms protected by lock and key give you control over who handles chemicals. Smart placement of exhaust fans pulls away harmful vapors. Routine maintenance—checking containers for wear and making sure inventory stays up to date—stops small issues from turning into emergencies. Even something as simple as routine checklists has kept people out of the clinic. With BEE, attention to detail separates high-risk environments from dependable workplaces.
Everyone Has A Stake
Safe storage isn’t someone else’s job. I remember how serious people got in safety meetings after one minor spill. No one rolled their eyes when new protocols came in. Bis(2,3-Epoxypropyl) Ether doesn’t give second chances to the careless. Every well-stored drum, clear label, and working vent matters to the people who’ll clock in tomorrow. Vigilance in storage means we go home healthy—and that’s not something to take lightly.
What are the potential health hazards of Bis(2,3-Epoxypropyl) Ether exposure?
A Chemical With a Heavy Reputation
Bis(2,3-Epoxypropyl) ether pops up in industries where workers handle resins, adhesives, and some plastics. Its sharp, almost ether-like odor signals its presence long before any symptoms start, but most people don’t recognize that warning. If you’ve walked past workers applying strong glues in a factory, you’ve probably smelled chemicals from this same family. Most of us don’t pause to consider what repeated exposure can do.
What Personal Experience Teaches
I used to work in a lab where organic solvents got handled daily. We wore gloves and masks, but fumes seemed to find their way through the tiniest gaps. Colleagues complained about headaches and dry coughs by lunchtime. Most people downplayed the sore throats and skin itch. Over time, I watched as a few friends developed skin rashes and respiratory issues. They shrugged it off at first, blaming air conditioning or dust. Later, some got sent for skin allergy tests, only to find sensitivity to epoxy compounds—Bis(2,3-Epoxypropyl) ether among the culprits.
What Science Reveals
This chemical takes a heavy toll on skin and airways. Direct contact with the skin may cause burning, redness, or blistering. Inhalation brings on coughing and shortness of breath. Even low doses over several years ramp up the risk of sensitization—once sensitivity develops, even a tiny amount triggers a reaction. Researchers found links between regular exposure and asthma-like symptoms in workers. The National Institute for Occupational Safety and Health (NIOSH) and OSHA both flag this type of ether as hazardous; both agencies urge employers to use controls like ventilation and protective gear, though not every job site keeps up with these recommendations.
Diving into Long-Term Effects
Scientists warn that effects go beyond the immediate discomfort. Some studies connect bis(2,3-epoxypropyl) ether to an increased risk of cancer with long-term, high-dose exposure. Animal tests raise concerns over changes in DNA when cells come in contact with this compound, making its carcinogenicity a real worry. Long-term exposure has even been tied to liver changes in rats, hinting at the possibility for subtle but dangerous effects in humans that could fly under the radar for years.
Finding Solutions—Not Just Labels
Safety talks and warning labels help, but change comes from how workplaces control chemical use and how workers protect themselves. In places where ventilation improved and exposure times got shortened, the number of complaints dropped. Small changes—better gloves, splash goggles, and real air exchange—paid off. If I had known about the full risks years ago, I would have spoken up about the headaches sooner or insisted on better protective measures.
At the policy level, health agencies encourage regular health checks for workers handling such chemicals. Employers can go beyond minimum guidelines, rotating tasks to minimize chronic exposure. Regularly checking air quality and listening to worker complaints keeps small problems from turning into career-ending illnesses. Workers who speak up about symptoms help everyone by highlighting emerging hazards before they become tragic stories.
The best defense comes from shared vigilance—workers knowing the risks, managers staying accountable, and medical professionals checking for signs before they become lifelong scars. A strong team mindset turns health guidelines from suggestions into real, practical steps that keep everyone from suffering the hidden tolls of bis(2,3-epoxypropyl) ether exposure.
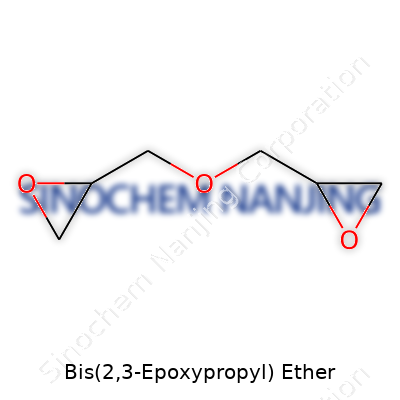
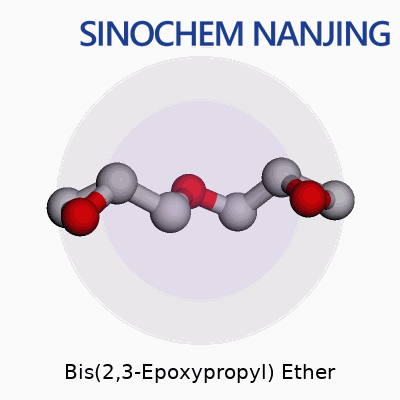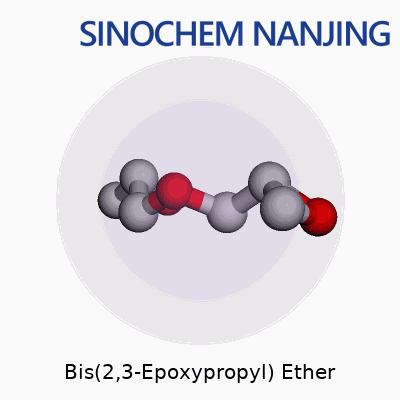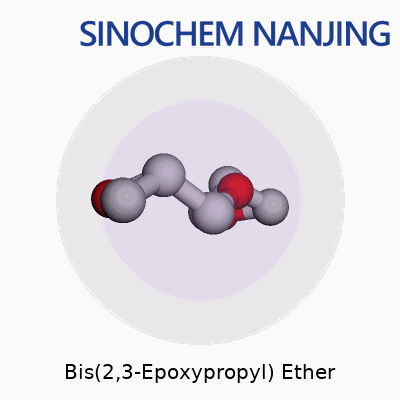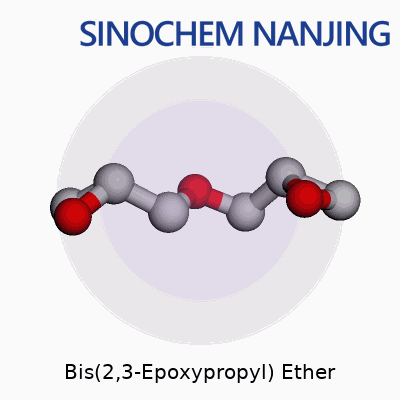
| Names | |
| Preferred IUPAC name | 2,2′-Oxybis(methyloxirane) |
| Other names |
BSEE Diglycidyl ether DGE Alpha,Omega-diglycidyl ether |
| Pronunciation | /ˈbɪs tuː θri ɪˈpɒksiˌprəʊpɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 2167-51-3 |
| Beilstein Reference | 1841242 |
| ChEBI | CHEBI:48037 |
| ChEMBL | CHEMBL22241 |
| ChemSpider | 21256 |
| DrugBank | DB14006 |
| ECHA InfoCard | 03eaf7db-dfb8-4b6d-b184-e1d5eaa5036a |
| EC Number | 203-439-8 |
| Gmelin Reference | 77477 |
| KEGG | C19573 |
| MeSH | D001943 |
| PubChem CID | 7906 |
| RTECS number | SL6540000 |
| UNII | 6J9B8Y366Q |
| UN number | UN2310 |
| CompTox Dashboard (EPA) | DTXSID4020702 |
| Properties | |
| Chemical formula | C6H10O3 |
| Molar mass | 130.17 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 1.18 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.1 |
| Vapor pressure | 5 mmHg (20°C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | 1.74 |
| Magnetic susceptibility (χ) | -75.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4390 |
| Viscosity | 10 mPa·s (25 °C) |
| Dipole moment | 4.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 385.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -291.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4094.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS06, GHS08, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H311, H314, H317, H319, H341, H351, H411 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 Health:1 Flammability:2 Instability:0 |
| Flash point | > 110 °C |
| Autoignition temperature | 215°C |
| Explosive limits | Explosive limits: 1.8–6.9% |
| Lethal dose or concentration | LD50 oral rat 11300 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Bis(2,3-Epoxypropyl) Ether: 1130 mg/kg (oral, rat) |
| NIOSH | B603 |
| PEL (Permissible) | PEL: 50 ppm (parts per million) |
| REL (Recommended) | REL (Recommended Exposure Limit) of Bis(2,3-Epoxypropyl) Ether is "0.1 ppm (0.6 mg/m³) TWA". |
| IDLH (Immediate danger) | IDLH: 75 ppm |
| Related compounds | |
| Related compounds |
Epoxy Glycidol Ethylene glycol Epichlorohydrin Polyethylene glycol |

