Bis(1-Methylethyl) Fluorophosphate: A Down-to-Earth Look at an Unforgiving Compound
Historical Development
Bis(1-Methylethyl) fluorophosphate slips into chemical history during an era when organophosphorus compounds attracted attention for their potent biological effects. Chemists in the early 20th century, driven by curiosity and wariness, saw how phosphorus, fluorine, and organic groups could come together to form molecules with stark power. Researchers noted the efficiency of these compounds in inhibiting enzymes essential for nerve function, and they took keen interest in tailoring toxicity for both pest control and, during darker chapters, warfare applications. Society saw both the benefits and horrors tied to these chemicals, leading to strict oversight and public concern. Over time, nations built frameworks around production and research of organophosphates, shaped by hard-learned lessons stretching from laboratory discovery to field misfortune.
Product Overview
Bis(1-Methylethyl) fluorophosphate belongs to a class of chemicals that rarely leaves the lab without strong controls and competent oversight. This molecule stands out due to its strong enzyme-inhibiting capability—mainly aimed at acetylcholinesterase. Its structure, with two iso-propyl groups attached to a phosphorus atom that connects to a fluorine atom and an oxygen, gives it unique behaviors seen especially in chemical research and industrial processes. Its commercial use remains tightly restricted, and for good reason. Laboratories value it not for broad industrial application but for its consistent action and known performance characteristics.
Physical & Chemical Properties
This compound typically appears as a colorless to slightly yellow liquid at room temperature. A sharp, chemical smell gives early warning to those in its presence. With modest volatility, it can evaporate and present inhalation risks, a reminder of why chemical hoods and tight protocols matter. Bis(1-Methylethyl) fluorophosphate resists easy breakdown in water but can slowly react to form alcohols and acids in moist environments. Its density and low viscosity call for care in transfer and measurement, as spills quickly spread. The phosphorus-fluorine bond makes for a molecule that clings tightly to its construction, holding together under typical lab conditions but showing reactivity with strong bases and nucleophiles.
Technical Specifications & Labeling
Labeling for this chemical pulls no punches. Standard bottles signal risk through bold hazard symbols and detailed handling instructions. Purity usually reaches above 95% for technical-research use. Most suppliers cover bottles in tough secondary containment and record lot numbers, expiration dates, and production origins. Safety Data Sheets (SDS) stretch for several pages, outlining symptoms of exposure, decontamination steps, and emergency procedures. Regulations in the United States, Europe, and Asia converge on requirements for secure shipping, user licensing, and mandatory training before receipt. Even for seasoned chemists, the label’s caution triggers extra diligence during every handling.
Preparation Method
Chemists generally start with phosphorus oxychloride and react it with isopropanol to produce a diester. Controlled substitution of the final chlorine with a fluorine source—often using potassium fluoride—produces Bis(1-Methylethyl) fluorophosphate. Each synthesis step brings its own challenge, from dealing with corrosive reagents to handling flammable solvents. Yields can reach respectable numbers, but impurities demand careful distillation. Many researchers note the importance of keeping water away, to avoid product hydrolysis and loss. Waste from production contains toxic by-products, which must be tracked and treated by professional disposal teams.
Chemical Reactions & Modifications
Bis(1-Methylethyl) fluorophosphate stands out for its keen reactivity at the phosphorus center. It acts as a substrate in nucleophilic substitution reactions, especially with strong nucleophiles that displace the fluoride group. Reaction with water kicks off a slow hydrolysis, releasing isopropanol and exposing the core phosphoric acid. Lewis acids, bases, or metallic catalysts can push the molecule toward further breakdown or rearrangement. Modification efforts in research often target making the compound either less toxic or easier to decompose during neutralization. Few labs attempt wholesale redesign, given the compound’s touchy nature and regulatory scrutiny.
Synonyms & Product Names
Names for this chemical trace its roots and chemical structure: Diisopropyl fluorophosphate, DFP, Isoflurophate, or Phosphorofluoridic acid, diisopropyl ester. Pharmaceutical-grade sources may use legacy names from nerve agent research, but most scientific catalogues settle on Diisopropyl fluorophosphate. Central registries and export-control lists typically group these all together, due to the shared fluoride-linked phosphorus unit and their shared risk profiles.
Safety & Operational Standards
Handling Bis(1-Methylethyl) fluorophosphate requires a full suite of protective gear: double gloves, face shields, long-sleeved lab coats, and often a supplied-air respirator mask. Storage happens in locked vaults or chemical safes, with strong secondary containment. Emergency response plans must detail antidote use—such as atropine—as well as immediate wash facilities for exposed skin or eyes. Operations limit time and quantity handled, with team work and constant supervision expected. Standard lab fume hoods can provide basic protection, but many procedures call for specialized gloveboxes or totally enclosed systems. All work must be documented for regulatory compliance, with special logs required for acquisition, use, and disposal.
Application Area
In the research world, this compound functions as a trusted tool for enzyme inhibition studies. By reliably blocking acetylcholinesterase, it lets neuroscientists simulate nerve agent effects or probe chemical pathways that impact brain and nerve health. Medical research teams turn to small-scale uses to model conditions like Myasthenia gravis and test antidote strategies for chemical poisoning. Regulatory authorities watch this application area closely, to prevent drift from laboratory utility into harm. Outside specialist labs, few have cause to interact with this molecule, making it well suited to its narrow niche.
Research & Development
Current research looks for ways to neutralize Bis(1-Methylethyl) fluorophosphate swiftly, reducing risk for accidental exposure or environmental contamination. Chemists have explored new antidote cocktails, along with enzyme scavengers that pull the toxin from bloodstreams before damage sets in. Development teams work on detection systems—small badges and test strips—that might catch airborne vapors at parts-per-billion levels. In pharmaceutical circles, a few groups hunt for less hazardous analogs that can gently probe biological systems without sharp toxic profiles. Academic partnerships push open-source data on this and similar molecules, helping peers recognize risks before accidents shape the lesson plan.
Toxicity Research
Studies confirm just how dangerous Bis(1-Methylethyl) fluorophosphate stands even in small doses. Exposure leads to overstimulation of nerves, uncontrolled muscle contraction, and, without medical intervention, possible fatality. Animal research, performed under tight controls, shows just a fraction of a milligram per kilogram can trigger these effects. Reports over the decades highlight the pain and difficulty in treating accidental exposure; some cases left victims with lingering neurological symptoms for years. Toxicology work continues to map safe exposure thresholds, inform cleanup strategies, and build global best practices for responders and clinicians tackling emergencies involving this compound.
Future Prospects
As research tools develop and regulations tighten, the future for Bis(1-Methylethyl) fluorophosphate likely stays bounded inside specialist labs and regulatory oversight. Interest in safer enzyme inhibitors grows year over year, driven by the biotech industry’s hunger for targeted chemical tools that carry a much lower human risk. Governments and industry partners collaborate on real-time sensor networks and advanced filtration that might stop accidental releases before harm occurs. Some forward-looking research reviews suggest engineered enzymes or nanomaterials could help break down organophosphate toxins in the environment, turning hard-learned chemical lessons into real protection for the next generation of scientists and first responders. With every project, the balance between scientific utility and community health guides difficult decisions about how and why such compounds deserve a place at the research bench.
What is Bis(1-Methylethyl) Fluorophosphate used for?
Not Just a Chemical Name—What Are We Talking About?
The name Bis(1-Methylethyl) fluorophosphate doesn’t roll off the tongue. For plenty of folks, it probably sounds like background noise from chemistry class. In plain language, we're looking at an organophosphate compound. That group of chemicals has earned a complicated reputation, mainly because it includes everything from certain pesticides to nerve agents. Getting into why this compound matters calls for cutting through some thick science jargon to see what kind of footprint it leaves on industries and everyday life.
Weaponization and Safety Concerns
One can’t tiptoe around it—Bis(1-Methylethyl) fluorophosphate has a dark history. It sits in the family tree of nerve agents related to sarin and soman. These substances disrupt the nervous system by messing with the enzyme acetylcholinesterase. The result is overstimulation of nerves, with tragic outcomes if someone gets exposed. Stories from the news and history books have made many people anxious about how these compounds wind up outside the lab.
The main reason this chemical shows up on watchlists is its potential use in chemical warfare. International agreements like the Chemical Weapons Convention target organophosphates, putting real pressure on governments and scientists everywhere to handle them responsibly. Nobody wants to see history repeat itself with modern twists.
Scientific Research and Testing
Researchers do find themselves working with Bis(1-Methylethyl) fluorophosphate. In places like government labs or pharmaceutical development centers, scientists use it to test antidotes and treatments against nerve agent poisoning. Having a chemical in controlled conditions allows for a better understanding of how these agents attack the body and what might stop them. Most of the work goes on under heavy regulation and careful safety procedures. Access stays limited for good reason.
Learning more about this compound has pushed medicine and toxicology forward. For example, work with organophosphates has paved the way for emergency treatments like atropine and pralidoxime, which show up in ambulances and military kits worldwide. By tackling something dangerous head-on, science has come up with ways to save lives in the event of accidents or attacks.
Safeguarding Against Accidents and Misuse
No talk about Bis(1-Methylethyl) fluorophosphate can dodge the issue of accidents. Production, storage, and disposal all demand strict safety measures. Past chemical leaks and improper disposal have poisoned soil and water, putting communities at risk. Strong oversight isn’t just a bureaucratic hurdle—it protects everyday people from invisible dangers. Community awareness, regular audits, and transparent practices shape how chemicals like this get handled. It makes sense to push for even tougher enforcement and education, especially in regions where oversight might slip.
What Needs to Change?
Placing public health and safety first has to drive decisions about organophosphate chemicals. Technology could help track storage and movement better—think real-time sensors or blockchain records for chemical shipments. Education remains a cornerstone. Giving first responders the right tools and training helps communities face threats from both accidents and intentional misuse. We can’t wish away the history of nerve agents, but using what we’ve learned can keep risk at bay and encourage responsible, informed action.
Is Bis(1-Methylethyl) Fluorophosphate hazardous or toxic?
Looking Beyond the Chemical Name
People see a name like Bis(1-Methylethyl) Fluorophosphate and might glaze over, but skipping over this chemical could be a mistake. Unlike basic cleaning products, this compound draws direct connections to some of the most hazardous legacy chemicals ever made. It closely relates to organophosphorus compounds—a group better known for their role in insecticides and even nerve agents like sarin. News like this hits differently if you’ve ever followed the story of chemical spills, or have lived near industrial sites where even a tiny leak causes real panic.
Bis(1-Methylethyl) Fluorophosphate works as a potent acetylcholinesterase inhibitor. What this means for people isn’t just science jargon—this kind of interference can disrupt how nerves talk to muscles, which carries harsh consequences. Exposure can trigger symptoms like twitching, respiratory distress, and sometimes convulsions. Larger doses can end up being fatal. These risks aren’t theoretical; agencies like the CDC and World Health Organization repeatedly flag substances like these as high risk.
Poking at the Real Risks
Unlike table salt or vinegar, you won’t find Bis(1-Methylethyl) Fluorophosphate on a grocery shelf. Production runs tightly controlled because it doesn’t take much to be dangerous. Even low-level exposure through skin or eyes can spell trouble. In training, professionals treat this kind of compound with the same caution as other chemical weapons. I remember walking through a chemical safety exercise—no one took their gloves off for a second while handling organophosphates. That alone should say plenty about the stakes.
These risks don’t stop at acute toxicity. Some research hints at longer-term neurological effects, especially if someone faces repeated, low-level exposure at a job site. In places where oversight slips or regulation weakens, stories come out about workers facing chronic health troubles. The body keeps score, especially when nerves get hacked at a chemical level.
Seeking Safer Grounds
Hazardous chemicals like this spark big questions. Why does industry keep producing them? With organophosphorus compounds in particular, agriculture has sometimes leaned hard on their bug-killing power. Some countries banned or heavily controlled their use as safer options came online. The road to safer chemistry runs long, though. For every old chemical phased out, others creep in, and sometimes regulators play catch-up.
Strong safety guidelines and real enforcement offer the best buffer against trouble. Training, protective equipment, and airtight labeling save lives. Anyone who works with these chemicals ought to have easy access to decontamination tools and up-to-date safety sheets. Many companies have moved to substitute less hazardous alternatives, but some sectors still depend on the old standbys.
Consumers play a part, too. Public pressure helped push global treaties limiting the spread and trade of the worst offenders. Those agreements don’t stop every accident, but do keep awareness high and force governments to explain their choices. If you hear stories about industrial incidents, or a product recall over a chemical like this, there’s a reason the headlines keep coming back: people want to know their air, water, and workspaces won’t poison them. That need for transparency and accountability doesn’t fade with time.
What are the storage and handling requirements for Bis(1-Methylethyl) Fluorophosphate?
Why Proper Storage and Handling Really Matter
Bis(1-Methylethyl) fluorophosphate doesn’t enjoy casual treatment; its toxic nature has created enough safety debates in many laboratories. When a compound brings serious health risks—neurotoxicity lands high on the list for this one—the responsible response is a plan rooted in respect and transparency. Safe storage procedures shield both workers and the environment from catastrophic mistakes.
Tight Storage: Not Just a Suggestion
Proper containment reduces the risk of leaks or accidental exposure. This isn’t just about putting bottles behind locked doors. The compound reacts with moisture, releases acids, and fumes that can harm airways and skin. Sturdy chemical cabinets—acid-resistant, non-reactive shelving, solid secondary containment—set the standard. Labels need bold clarity, including manufacturer and hazard warnings. Staff should never rely on memory.
Temperature swings ruin stability. Room temperature storage often encourages slow degradation, so controlled cool environments—think dedicated flammable solvent refrigerators—help extend shelf life and reduce pressure build-up. Avoid food-use refrigerators; never allow confusion or contamination.
Direct sunlight causes problems too since UV light can accelerate breakdown. Rooms with covered, shaded interiors and closed-circuit air handling offer smart choices. Ventilated cabinets prevent buildup of hazardous fumes but some chemical hoods pull vapors directly into lab air, a problem for anyone not wearing proper PPE.
PPE: Everyday Necessity, Not Occasional Option
Direct skin and eye contact can turn minor mistakes into emergencies. Nitrile gloves, chemical splash goggles, and fitted lab coats form the basic kit, backed by powered respirators or full-face shields during transfer or spill cleanup. In my experience, forgetting PPE even for a moment—like leaving hands exposed to open bottles—means taking on more risk than anyone needs. Clean up spills swiftly using absorbent materials rated for concentrated acids, and always have shower and eyewash stations in clear sight and easy reach.
Documented Protocols, No Shortcuts
Some of the worst mistakes come from skipping steps. Always log the receiving, usage, and disposal records. Facilities with solid SOPs—reviewed at least annually—do a much better job at keeping everyone on the same page. Teach new staff with supervised practice, not just procedure handbooks.
Waste management demands attention, not just basic labeling. Segregate fluorinated waste streams and keep acids and organophosphates in separate, clearly marked containers. Coordinate removal with certified hazardous materials providers, not routine waste services. Even small traces left on glassware need specialty cleaning—not tap water and soap.
What Can Go Wrong—And Can Be Fixed
Complacency causes preventable injuries and environmental releases. One forgotten open lid or a quarter-inch of standing water can create hydrolysis and toxic fumes overnight. Regular checks matter—a monthly walk-through to inspect inventory, lids, and signage beats any written rulebook.
Strong safety cultures get results upstream and downstream. Companies and universities leading the way invest in meaningful chemical safety training. They keep extra kits and spill response supplies on hand, not locked up. Risk reduction doesn’t arrive by accident; it grows from everyone expecting and demanding high standards.
In my own work, tight controls, double-checks, and honest communication have saved more than one day from turning into disaster. Mistakes happen, but preparation and clear heads prevent tragedy. It’s not glamorous, but respect for substances like bis(1-methylethyl) fluorophosphate protects futures, lives, and reputations all at once.
What is the chemical structure and molecular formula of Bis(1-Methylethyl) Fluorophosphate?
Chemical Structure and Formula
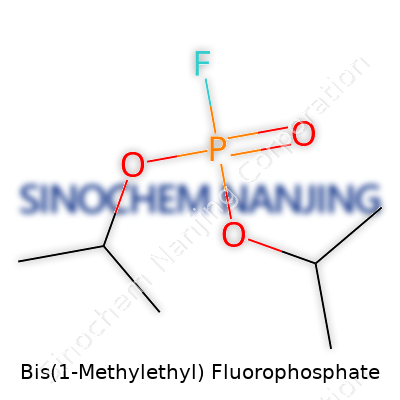
Bis(1-Methylethyl) fluorophosphate, also known as diisopropyl fluorophosphate (DFP), carries the molecular formula C6H14FO3P. The arrangement shows a phosphorus atom sitting at the core, surrounded by two isopropyl oxygen groups, a fluorine atom, and a double-bonded oxygen atom. Writing it out, the skeletal formula looks like this: (C3H7O)2P(O)F. Here, each isopropyl group branches away, attached through an oxygen atom, almost like outstretched arms. These features give DFP a very precise role in chemistry and biology.
Why Structure Drives Its Behavior
Structure in chemistry often dictates behavior, and DFP gives a prime example. The phosphorus-fluorine bond reflects high reactivity, not something to treat lightly. Add the bulky isopropyl groups, and the way it acts with other molecules changes even more. Scientists have learned that DFP, in the hands of researchers, will target enzymes called acetylcholinesterases. These are the same enzymes nerves rely on to keep messages moving at the right speed. Once DFP is in the system, it attaches firmly, blocking the enzyme’s action.
This kind of behavior isn’t just a footnote in chemical textbooks. Early in my lab training, one of the first rules laid out was simple: treat organophosphorus compounds with respect. A drop in the wrong place or on unprotected skin carries outsized risks. DFP isn’t just a “lab chemical”—it’s a nerve agent at its core, and its effects can move quickly from cellular disruption to whole-body trouble.
Practical Applications and Historical Use
People often overlook how many research breakthroughs have come from uncomfortable discoveries. DFP, built for chemical warfare studies in the 20th century, soon found use in controlled neurological research. Neuroscientists used it to study nervous system signaling, and toxicologists have used it to develop antidotes and test hazards.
Laboratories working with DFP typically reinforce safety precautions. Chemical fume hoods and specialized gloves turn into daily gear. The risk of accidental exposure comes up in safety briefings, again and again. One errant droplet can impact the nervous system within minutes—symptoms like muscle twitching, sweating, and even tremors follow soon after.
Data Supporting Responsible Handling
The CDC has classified DFP as a toxic chemical with risks similar to other organophosphates. LD50 data show that a dose of just a few milligrams per kilogram can be fatal to many lab animals. The reactivity of the phosphorus-fluorine bond leads to permanent enzyme inhibition, so decontamination procedures call for strong alkaline solutions and quick response. I remember stories of old-time researchers who took short cuts, leading to accidents—each one emphasized that trusting intuition, not proper handling, brought trouble.
Potential Solutions for Safer Use
Advances in lab design and protective equipment have changed the outlook for researchers. Automatic dispensing, new sensor technologies, and improved waste disposal now help people cut down on risk. Updated protocols encourage better training and stronger oversight, ensuring that everyone who handles DFP knows exactly what’s at stake.
Bis(1-Methylethyl) fluorophosphate stands as both a cautionary tale and a tool. Its chemical structure gives it an outsized impact in chemistry labs and hazardous materials studies. Treating it with diligence and modern safety approaches continues to protect both researchers and the broader public.
Are there any safety guidelines or protective measures when working with Bis(1-Methylethyl) Fluorophosphate?
Why Extra Precautions Matter
Bis(1-Methylethyl) Fluorophosphate serves a function in chemical synthesis and some niche research fields, but there’s a reason most discussions about this compound turn almost immediately to personal safety. Its toxicological profile, which shares some traits with nerve agents, means small mistakes can lead to big consequences. Even folks who don’t rattle easily get a little more careful around organophosphates. The dangers don’t just involve skin contact or inhalation, but a whole-body risk that boils down to severe neurological damage after a single slip-up.
PPE: The Everyday Shield
Chemists and technicians who use this compound know gloves are just the start. Nobody trusts standard latex or vinyl here. Nitrile or butyl gloves give a stronger barrier. A heavy lab coat with close-fitting sleeves and long pants leaves no room for skin exposure, and side-shield safety glasses keep particles and fumes from reaching the eyes. Face shields give another layer of defense during transfers or handling large samples. Working this close to danger, splash resistance isn’t optional, it’s basic self-respect.
Engineering Controls: Beyond the Basics
No fume hood, no entry — that’s most chemists’ rule. Fume hoods with strong air flow help cut down inhalation risk that can knock someone out fast. Enclosed glove boxes keep the compound away from direct handling, especially in labs with repeated or long-term tasks. Negative-pressure rooms limit the area of possible contamination. Labs that use this compound keep spill kits designed for organophosphates right there in the workspace, not in a storage closet down the hall.
Procedural Habits That Save Health
Researchers work slow and methodical, planning out every step before opening a container. Dry runs matter. Labeled containers and clear work areas leave nothing to chance. Buddy systems aren’t just about comfort — if something goes wrong, a lone worker has a slim window for antidotes like atropine or pralidoxime to take effect. Regular wash breaks, even if no contact seems likely, add another barrier. Double-bagging waste for safe disposal, and logging every bit of use in detail, keeps surprises down and makes it easy to find the source if problems occur.
Emergency Response: Drills and Treatments
Training for emergencies gets real fast in places that even consider using Bis(1-Methylethyl) Fluorophosphate. Team members learn to spot early symptoms: pinpoint pupils, muscle twitching, weakness. Immediate access to emergency decontamination showers, eyewash stations, atropine auto-injectors, and protocols for summoning emergency crews shortens reaction time. Records track exposures and help doctors make fast calls on treatment, improving odds when every second counts.
Product Storage: Keeping the Chain Unbroken
This isn’t a compound to keep on open shelves. Locked, ventilated, and signed storage cabinets stop accidental mixing or theft. Temperature control keeps degradation or accidental volatility in check. Routine inventory checks and dedicated containers limit accidental contamination. Only trained staff touch the jars. After every use, logs track date, time, and condition to flag problems before they grow.
Solutions and Smarter Protocols
No lab operates safely just by buying equipment or drafting paperwork. Frequent drills, honest after-action reviews and a culture where even young students can call out problems keep everyone sharp. Peer-to-peer training, not just reading manuals, helps embed lessons deep. Replacing the compound with safer alternatives wherever possible cuts the risk, showing that commitment to safety isn’t a badge — it’s a daily choice. Regulations from bodies like OSHA and the CDC give a framework, but it’s the team’s real habits that keep accidents from happening in the first place.
| Names | |
| Preferred IUPAC name | Diisopropyl fluorophosphate |
| Other names |
DFP Diisopropyl fluorophosphate Fluorophosphoric acid, diisopropyl ester Isopropylfluorophosphonate Isofluorophate Isopropyl fluorophosphate |
| Pronunciation | /ˈbɪs wʌn ˌmɛθ-əl-ˈɛθ-əl flaʊˌrɒf-ə-speɪt/ |
| Identifiers | |
| CAS Number | 55-11-6 |
| Beilstein Reference | 1538739 |
| ChEBI | CHEBI:38811 |
| ChEMBL | CHEMBL504243 |
| ChemSpider | 109787 |
| DrugBank | DB02354 |
| ECHA InfoCard | 41bc6f10-7047-4f59-b16d-259b2d3a6ed8 |
| EC Number | 202-011-6 |
| Gmelin Reference | 67845 |
| KEGG | C19002 |
| MeSH | D017601 |
| PubChem CID | 65704 |
| RTECS number | OZ5950000 |
| UNII | IL9450G005 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID7041264 |
| Properties | |
| Chemical formula | C6H14FO3P |
| Molar mass | 200.172 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent odor |
| Density | 1.044 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.7 |
| Vapor pressure | 0.0133 hPa (20 °C) |
| Acidity (pKa) | 1.7 |
| Basicity (pKb) | 1.7 |
| Magnetic susceptibility (χ) | -51.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.406 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1256.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7261 kJ/mol |
| Pharmacology | |
| ATC code | Nerve agent |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS06,GHS03 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H311: Toxic in contact with skin. H331: Toxic if inhaled. H411: Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P330, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 1, Instability: 2, Special: - |
| Flash point | 82 °C (180 °F) - closed cup |
| Autoignition temperature | 321°C |
| Lethal dose or concentration | LD50 oral rat 8 mg/kg |
| LD50 (median dose) | LD50 (median dose): 8.6 mg/kg (rat, oral) |
| NIOSH | WF9025000 |
| PEL (Permissible) | PEL: 0.01 ppm (0.08 mg/m3) as TWA |
| REL (Recommended) | 3 mg/m³ |
| IDLH (Immediate danger) | IDHL: 10 mg/m3 |
| Related compounds | |
| Related compounds |
Diisopropyl phosphite Diisopropyl chlorophosphate Diisopropyl phosphorochloridate Diisopropyl methylphosphonate Dimethyl fluorophosphate |

