Beryllium Nitrate: A Hard Look at a Critical Compound
Historical Development
Beryllium nitrate didn’t pop up overnight. The race to understand and use beryllium’s unique properties started in the early twentieth century. Chemists explored the nitrate form as industrial and military needs demanded lighter, stronger materials. Early on, research teams in Europe and America worked to isolate and purify beryllium compounds, hunting for new opportunities in specialty metallurgy and energetics. Its development shadowed the broader story of beryllium—more demanding than other metals, always calling for cleaner and safer methods. By the 1950s, beryllium nitrate had entered scientific circles that focused on rocket propellants, specialty catalysis, and research reagents, paving the way for the advanced synthesis routes we know today. Shaped by military contracts and academic curiosity, its legacy isn’t just in the chemistry, but in the hard-fought knowledge earned by those who handled it, often at real risk.
Product Overview
Beryllium nitrate stands out for its rare combination of high reactivity and unique solubility. In its purest laboratory form, the compound usually appears as a colorless, crystalline solid. Specialists value it for the way beryllium can act as both a catalyst and a high-energy oxidizer. The product is not your standard chemical barrel—you’ll find it locked tight, labeled with hazard warnings, and usually handled by folks who understand the stakes. Its major buyers include research laboratories, specialized aerospace contractors, and universities, not mass manufacturing plants. Beryllium nitrate is never an off-the-shelf chemical; it’s usually made to order and shipped in small quantities under careful controls.
Physical & Chemical Properties
This compound shows off a sharp, almost geometric appearance in its crystalline state. The solid dissolves easily in water, resulting in a highly acidic solution owing to the nitrate ion. Beryllium nitrate doesn’t just sit quietly on the shelf—it reacts significantly as a strong oxidizer. The molecular weight clocks in at around 133 grams per mole, but it packs a toxic punch in much smaller doses. Unlike more pedestrian nitrates, beryllium nitrate releases dense, white fumes when it hits open flame or strong heat, a signal of the dangerous gases at play. Its melting and decomposition bring unpredictability, demanding serious respect from anyone who handles it.
Technical Specifications & Labeling
Strict labeling backs up every bottle or drum of beryllium nitrate. Hazard diamonds, UN numbers, and chemical hazard statements cover every surface. Technical sheets list purity, common impurities, particle or crystal size, and storage requirements—airtight and moisture-free containers, kept far from combustibles or anything organic. Shipping documentation lines up with international standards, serving both regulatory needs and the real need for accountability if there’s a spill or an exposure. The specifics aren’t just bureaucratic—they’re life-saving guides, a must for any lab that uses this compound.
Preparation Method
Most beryllium nitrate forms in the lab by carefully reacting beryllium oxide or hydroxide with concentrated nitric acid. The reaction rewards precision, as too much heat or acid can send toxic vapors billowing out. Chemists typically prefer using a controlled dropwise addition, chilling the apparatus to minimize runaway reactions. Because of the toxicity, every step demands heavy ventilation and protective equipment. After completing the reaction, careful crystallization and washing remove any unreacted starting materials. The final solid is collected, dried in a vacuum, and then sealed up tight—never left out in the open.
Chemical Reactions & Modifications
Beryllium nitrate’s real power comes from its reactivity. In a lab, it doubles as a strong oxidizer, able to push organic and inorganic syntheses that call for a jump-start. Add a reducing agent, and the beryllium drops out of solution as hydroxide or oxide. Mix with ammonia, and you’ll build complex coordination compounds, which researchers explore for electronics and catalysis. The nitrate group allows substitution reactions and opens up the compound to further modification, though few dare to make or modify it in bulk because of the known hazards. In energetic materials development, beryllium nitrate serves both as a study target and as a chemical intermediate, showing up in patents and journal articles that chase new high-performance explosives and fuels.
Synonyms & Product Names
Chemists know beryllium nitrate by several names, depending on who’s buying or making it. You’ll see it listed in catalogs as "beryllium dinitrate", "beryllium(II) nitrate" or sometimes as "Be(NO3)2." Different manufacturers have used house codes and numbers over the years, but the chemical registry numbers—the CAS label—do most of the heavy lifting when it comes to verifying you have the right material. Any confusion here can spell disaster down the line, so most suppliers insist on triple-checking orders and certificates of analysis.
Safety & Operational Standards
There’s no making light of the safety side. Beryllium nitrate is toxic, and inhaling even a tiny amount can lead to chronic beryllium disease. Labs and workshops set strict standards: fume hoods, gloves, face shields, and double containment are standard—not suggestions. Safety training happens before the first bottle is opened, and chemical spill protocols hang on every wall. Real-world health records show that laboratories with slack procedures have suffered long-term injuries and regulatory fines. International guidelines from OSHA, NIOSH, and Europe’s REACH map out what’s required, but it’s the daily diligence of scientists and technicians that actually keep the risks in check.
Application Area
Beryllium nitrate plays an outsized role in high-level R&D, especially for aerospace and military technology. Work on solid rocket propellants and advanced explosives in the 60s and 70s tapped into its oxidizing power, and those studies still crop up in declassified papers and engineering handbooks. Today, it hangs around as a specialty reagent in inorganic chemistry labs, where the beryllium ion’s coordination habits lead to new catalytic systems. Some medical imaging research flirts with beryllium-containing compounds, though safety and toxicity tend to keep them in early-phase study. The market remains small and closed, bound by strict regulations and an enduring respect for the dangers involved.
Research & Development
Research into beryllium nitrate reflects the lure of new materials and the need for better safety. Chemists experiment with it to unlock coordination chemistry or to design nitro-based energetic materials with improved stability. Metallurgists explore its place as a dopant in beryllium alloys or for surface treatments that push the boundaries of what lightweight structures can handle. Analysis tools—X-ray diffraction, mass spectrometry, and others—have improved traceability and understanding of its reactions since its first discovery. Even as public interest in the compound ebbs and flows, specialist research soldiers on, driven by academic curiosity and the possibility of new breakthroughs in both material science and reaction engineering.
Toxicity Research
No discussion of beryllium nitrate gets far before toxicity comes up. Inhaling dust or vapor, even in small amounts, can scar lungs for life or trigger systemic disease long after exposure. Modern toxicity research measures airborne beryllium with ultrasensitive instruments, mapping out how little it takes to do harm. Animal studies, industrial hygiene records, and disaster response drills all underline the point—exposure adds up over time, and cumulative impacts outweigh acute poisoning risks. Medical surveillance tries to catch early signs, but prevention through airtight process controls and respirator programs outpaces any cure. The real-world lesson: nobody who works with beryllium nitrate gets careless twice.
Future Prospects
Tight regulations and ongoing safety concerns put a cap on widespread use. But beryllium’s unique properties aren’t matched by many other elements—so demand persists for its compounds in corners of science where performance trumps cost and difficulty. Advances in safety engineering and digital process controls raise the possibility that one day, broader application could be less risky. Meanwhile, scientists keep exploring new synthesis routes that might tame the hazards and open the door to wider adoption, especially in catalysis and materials science. That said, any future for beryllium nitrate will depend on a culture that never lets down its guard against its well-documented risks.
What is Beryllium Nitrate used for?
How Beryllium Nitrate Shapes Industries
Beryllium nitrate sounds like one of those chemicals tucked away in research closets, but it reaches into a surprising number of areas. In my college days, chem labs kept small, secure bottles on high shelves—no one touched them without gloves and goggles. Looking back, that kind of handling wasn’t just over-cautious; it reflected the serious work this compound pulled in both research and wider industry.
A big part of its story unfolds in chemistry labs. Beryllium nitrate allows researchers to make pure beryllium compounds, serving as a key source for other beryllium-based chemicals. That trait means chemists use it when they need high reactivity and precision, especially for experiments investigating the element’s unique properties. Anyone who’s cracked open advanced inorganic textbooks recognizes the importance of getting those pure starting points. The compound acts almost like a bridge, helping scientists move from raw materials to cutting-edge discoveries.
Electronics and Specialized Manufacturing
People sometimes overlook the role of beryllium nitrate in electronics. The electronics industry uses beryllium compounds to improve ceramics and glass, especially when a product needs both strength and thermal stability. Beryllium nitrate’s contribution helps those working on x-ray windows, high-tech sensors, or certain types of fiber optics. The reason is simple—components with beryllium stand up to tough environments, keeping signals clear and distortions low. Industries making those parts depend on precise chemical processing, and that’s where beryllium nitrate often comes in.
Years ago, I met a glass engineer who specialized in analytical devices. He described how adding certain chemicals allowed for stronger, clearer glass in equipment like spectrometers. Beryllium wasn’t always top of mind, but he pointed out that a small tweak at the chemistry stage made an instrument last longer and keep readings more reliable over time.
Rocket Fuels: A Cautionary Note
Beryllium nitrate sometimes enters discussion about rocket propellants. This isn’t the side touted in brochures because of the risks involved. It can release oxygen when heated, driving more vigorous reactions. That property has seen historical use in some experimental rocket systems. Still, safety concerns have taken the front seat. Inhalation or skin contact brings serious health risks—beryllium compounds can cause severe disease, and nitrate salts can trigger their own set of problems. As a result, the aerospace world has mostly shifted toward less toxic alternatives where possible.
Health, Safety, and Responsible Use
If there’s a lesson learned from working near materials like beryllium nitrate, it’s that expertise and respect matter. Proper handling safeguards both workers and the wider community. Regulations now require strong ventilation systems, personal protective gear, and closed-process equipment when using the compound. Many facilities invest in frequent air monitoring, and medical surveillance for staff who work with beryllium substances. These aren’t just rules on paper—they’re lived practices designed to keep the benefits without exposing people to harm.
With tighter controls and new research, industries keep finding ways to harness the best of specialty chemicals while reducing hazards. Progress hasn’t been perfect, but looking at how far safety protocols have come since I first saw those locked bottles on the shelf, it’s clear that science, health, and industry progress together.
Is Beryllium Nitrate hazardous to health?
Hazards Hidden in Plain Sight
Working around chemicals, you start to notice which ones folks eye with caution. Few people talk about beryllium nitrate, though, even though it's nothing to shrug off. Most people know about asbestos or lead; fewer know about the risks tied to beryllium, especially in the form of beryllium nitrate. It comes as a pale crystalline solid, and its hazards track much deeper than skin level.
How Beryllium Nitrate Affects the Body
Beryllium compounds don't just irritate—they can wreak havoc on your lungs if you breathe in dust or fumes. The nitrate form brings a double-whammy. Not only does the beryllium pose a threat, but nitrates also add a layer of toxicity. Think heavy coughing, chest pain, and shortness of breath after exposure—those are not just stories in a textbook. Workers who’ve spent time in environments with poor ventilation have developed chronic beryllium disease, which scars the lungs and doesn’t let up.
The risk doesn’t only live in old factories. Research by the U.S. National Institute for Occupational Safety and Health (NIOSH) and the Occupational Safety and Health Administration (OSHA) shows that even small doses carried by airborne dust can set off severe immune responses years after exposure. One grain of beryllium dust, too small to see, can be all it takes for a sensitive person.
Short-Term and Long-Term Health Concerns
Getting beryllium nitrate on your skin may cause rashes or burns—painful, but easier to notice. The real trouble starts when it sneaks into the lungs. Acute inhalation causes inflammation that looks a lot like pneumonia, and doctors can miss it if they’re not looking for a chemical cause. Over the long run, exposure scars the lungs, which leads to ongoing coughs, shortness of breath, and, for some, a sentence to a lifetime of steroid treatments. The American Lung Association has shown how even trace exposures raise the risk for lung cancer, particularly for people with a decade or more of contact.
Who Needs to Pay Attention?
Technicians, research scientists, or anyone dealing with specialty ceramics or electronics run into beryllium nitrate sometimes. It hides out in places where people rarely read the fine print on Safety Data Sheets. A few years back, a lab friend of mine forgot to put on gloves and masks because he “only needed a minute inside.” He coughed for weeks, learning the hard way that there are no shortcuts with these chemicals.
How to Cut Down the Risks
Factory managers and safety officers have their work cut out. It's not enough to post warnings. Regular air sampling, top-of-line ventilation, and personal protective equipment remain the foundation of workplace safety. Handling should always use fume hoods or gloveboxes. Frequent training helps new staff recognize the hazards, not just senior chemists. It takes a culture that looks out for newcomers and veterans alike.
For any company working around beryllium nitrate, medical surveillance isn’t optional. Blood tests for beryllium sensitivity and chest X-rays, at least yearly, have saved lives by catching chronic beryllium disease early. OSHA limits exposure to 0.2 micrograms per cubic meter in the air—barely a whisper, but even a small overstep results in big consequences.
Final Thoughts
If you're reading labels or consulting Material Safety Data Sheets, pay close attention anytime beryllium nitrate pops up. A single lapse can transform a routine workday into a lasting health fight. Respect for these hazards doesn’t come out of nowhere; it comes from experience, science, and paying attention to warnings before it’s too late.
How should Beryllium Nitrate be stored?
A Chemical With a Reputation
Beryllium nitrate doesn’t often grab headlines, but anyone who’s spent time in a research lab or industrial chemical store respects its risks. On the shelf, it looks like a regular container, sometimes a fine powder or a colorless solution. Under the surface, though, it carries hazards that go beyond mere irritation. Inhaling it, even touching it, can unleash reactions that lead to berylliosis or worse in those not properly equipped. Beryllium's toxicity is well-known among chemists and safety officers, yet accidents still crop up—sometimes just because someone slapped the container in the usual spot beside less treacherous reagents.
Getting Serious About Containment
Many folks have watched colleagues store chemicals side by side, even after clear warnings spanning decades. Beryllium nitrate demands something different. It reacts with organic material and moisture, releasing nitrogen oxides and beryllium salts. That means humidity and heat make a risky mix. Experience has taught me, and many organizations, that airtight glass or high-quality plastic bottles help prevent leaks and contamination. Airtight seals, solid labeling, and desiccant packs in storage cabinets make a difference. No casual shelving next to acids, bases, or oxidizable compounds—this stuff benefits from a bit of isolation.
Ventilation Beats Regret
Locking up beryllium nitrate in a cupboard isn’t enough. Even small escapes, through micro-leaks or old caps, pose a risk. A well-ventilated, preferably negative pressure storage space reduces the buildup of dust or vapor, keeping the rest of the room safer. Working for years in shared laboratories, I’ve seen how air-handling upgrades change the whole vibe—fewer headaches, less coughing, and a firmer grip on potential disaster. Air currents in the right direction make a huge difference, especially if a bottle fails or someone spills while reaching for it.
The Right Gear Saves Lungs and Skin
Personal experience tells me most incidents happen not because people ignore rules, but because they underestimate a compound. Proper gloves, goggles, and lab coats keep beryllium out of lungs and off hands. Storing spare protective gear near the storage space helps—sometimes all it takes to avoid disaster is seeing a pair of gloves right next to the door. Emergency eyewash bottles and showers nearby offer an extra line of defense, turning a near-miss into a lesson rather than a hospital trip.
Respect in Training, Not Just on Paper
Written policies carry weight only if folks take them seriously. Some of the best safety programs I’ve worked with revisit dangerous compounds like beryllium nitrate at least twice a year, mixing in hands-on drills. Clear instructions taped near storage spaces—what to do, not just what to avoid—keep the focus fresh. Regular inventory checks mean everyone knows if a bottle’s gone missing or if a container’s cracked. Reinforcement builds a culture where new team members realize early that beryllium’s storage is no box-checking exercise.
Looking Ahead: Smarter Storage Solutions
Storing beryllium nitrate may never be completely risk-free, but technology chips away at hazards. Sealed, lockable cabinets wired to alarms or ventilation systems can catch problems before they worsen. Digital temperature and humidity monitors send alerts when things drift out of range. Combining experience, hard science, and real respect for the compound’s firepower lays a foundation that newer workers will hopefully build on, not repeat mistakes. For anyone working with such materials, safe storage isn’t about following orders—it’s about looking out for your colleagues, your own health, and the integrity of the lab itself.
What is the chemical formula of Beryllium Nitrate?
Understanding the Chemical Formula
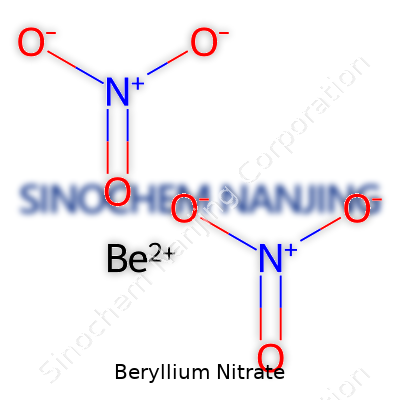
Beryllium nitrate comes with the chemical formula Be(NO3)2. So, there’s one beryllium atom connected with two nitrate groups. If you’ve spent time in a chemistry classroom, you’ll notice the logic behind this combination. Beryllium brings a 2+ charge, and each nitrate holds a single negative charge. So, two nitrates balance out one beryllium, making for a neutral compound.
Why Beryllium Compounds Deserve Attention
Getting the formula right isn’t just about quizzes or exams. Beryllium nitrate belongs to a group of beryllium salts that carry real stakes. The element beryllium itself is notorious in industry. Breathing dust or fumes can lead to berylliosis, a dangerous lung disease. The nitrate version, thanks to its solubility and toxicity, means working with this compound isn’t a small matter. The increased use of precision electronics and aerospace parts has brought beryllium-based materials to the shop floor and research lab much more often. It sits in a strange spot—necessary for cutting-edge tech, but increasingly controversial for worker safety.
Science, Industry, and Learning from Experience
Conversations with colleagues in research labs leave clear memories. We always treat beryllium compounds with special caution, more so than many metals. Over the years, I’ve met several people trained in handling beryllium, right down to controlling airflow and using personal safety gear that seems excessive until you hear about those rare but devastating cases of exposure.
Mistakes around chemical formulas often show up in surprising places, from expired safety signage to poorly labeled bottles on a shelf. Beryllium nitrate’s formula isn’t just a trivia point. It tells us how to react, how it behaves under heat, what happens when it meets water or acids, and how much of a risk it poses if something spills.
Supporting Claims with What We Know
Research published in peer-reviewed journals, including OSHA and NIOSH reports, continues to flag beryllium’s risks. Inhaling dust—even at low concentrations—can cause lasting harm. The EPA lists beryllium compounds as hazardous air pollutants, while the International Agency for Research on Cancer names beryllium a human carcinogen. These aren’t distant, academic concerns. They play out in factories, labs, and communities close to manufacturing sites.
Misspelling a chemical formula might sound small, but for beryllium nitrate, it sets the stage for real hazards. Safety protocols, emergency response guides, and even shipping paperwork rely on chemical formulas to avoid accidents.
Practical Steps Toward Safer Handling
Every place that stocks beryllium nitrate should keep paperwork up to date—labels, Material Safety Data Sheets, and training programs. Regular refresher courses make a difference, and relying on current, reliable information outweighs convenience. Chemists, lab managers, and even students have a stake in keeping their workspace safe.
Since regulations shift with new research, workplaces should stay in touch with industry groups and health agencies. Cutting corners to save time or cash never pans out if you weigh the risks. Worn gloves or missing signage invite disaster. The more accurately we treat beryllium nitrate—from formula to storage—the safer science and industry become.
What precautions should be taken when handling Beryllium Nitrate?
Why Beryllium Nitrate Demands Respect
Beryllium nitrate isn’t the sort of chemical people keep in the back of a garage. It finds use in research labs and some industrial settings, mainly for making specialty ceramics or in the study of nuclear materials. Its danger isn’t only about being toxic if swallowed or inhaled. The real trouble starts when fine dust or fumes get loose, often unnoticed, and can lead to serious lung disease—including lifelong, sometimes fatal, conditions.
Understanding the Health Risks
Exposure brings risks that go way beyond shortness of breath or cough. Long-term inhalation may cause chronic beryllium disease, an incurable lung condition. Just a tiny bit of airborne beryllium can pose trouble. Getting it on your skin isn’t harmless either. Inhaling dust, touching contaminated surfaces, even skin contact—these all provide pathways for harm. The International Agency for Research on Cancer (IARC) has classified beryllium compounds as carcinogenic to humans. That speaks for itself.
Personal Protective Equipment and Workspace Controls
Experience in the lab taught me that good gloves do more than keep your hands clean. Nitrile or neoprene gloves, goggles with side protection, and a lab coat that doesn’t get worn outside the workspace help stop contamination at the source. Respirators should be part of the plan, not an afterthought, especially anywhere dust or mist might form.
Leaving safety to chance is not a good idea. Busy days and distractions lead to short-cuts. I remember seeing someone wipe their face with a lab sleeve—they forgot gloves stay on until finished and hands have been washed. Consistent habits matter. Negative pressure enclosures and fume hoods keep particles or vapors from spreading. Good airflow, exhaust systems, and HEPA filtration give everyone a better chance of staying healthy.
Hygiene and Decontamination
Anything that touches beryllium nitrate—benches, tools, even notebooks—should stay within one controlled space. Double bagging waste, sealing it before disposal, and never using compressed air to clean work surfaces, prevents dust movement. Regular washing of hands and forearms is a must, not a suggestion. Locker rooms and showers keep work contamination from coming home.
Training and Awareness
Easy to skip the training or assume people remember protocols. That’s when mistakes happen. Everyone involved needs training that covers risks, symptoms of exposure, and the right way to handle emergencies. Having material safety data sheets posted helps, but talking about risk out in the open makes it real—not just words on a poster.
Medical Surveillance and Monitoring
Workplaces should offer regular health checkups. Blood tests, lung function screening, and even medical removal if results look off. I’ve seen coworkers catch early symptoms only because routine monitoring flagged something nobody would have noticed otherwise. Early intervention often makes a difference.
Solutions for Safer Handling
Minimize how much is used—never open more than needed. Substitute with less hazardous chemicals if possible. Automate transfers using closed systems wherever it makes sense. Emergency eyewash stations and spill kits must be in arm’s reach, not locked in a distant office.
Good safety culture means speaking up if something looks wrong. It’s better to slow down than regret a hasty job with the wrong chemical. Getting everyone on board—cleaners, supervisors, lab users—cuts risk across the board, not just for people in lab coats.
| Names | |
| Preferred IUPAC name | Beryllium dinitrate |
| Other names |
Nitric acid, beryllium salt Beryllium dinitrate Beryllium(II) nitrate |
| Pronunciation | /bəˌrɪli.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13597-99-4 |
| Beilstein Reference | 3563905 |
| ChEBI | CHEBI:84959 |
| ChEMBL | CHEMBL1201578 |
| ChemSpider | 22917 |
| DrugBank | DB14512 |
| ECHA InfoCard | 100.032.988 |
| EC Number | 236-850-6 |
| Gmelin Reference | 1045 |
| KEGG | C18708 |
| MeSH | D001592 |
| PubChem CID | 15255 |
| RTECS number | DS1750000 |
| UNII | OUU7K6QJ09 |
| UN number | UN2549 |
| CompTox Dashboard (EPA) | `DTXSID4036867` |
| Properties | |
| Chemical formula | Be(NO3)2 |
| Molar mass | 133.02 g/mol |
| Appearance | White deliquescent crystals |
| Odor | odorless |
| Density | 2.13 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.0 |
| Vapor pressure | 7.5 mm Hg (25 °C) |
| Basicity (pKb) | -3.62 |
| Magnetic susceptibility (χ) | '−13.7×10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.497 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -583.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AN03 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; causes severe skin burns and eye damage; may cause cancer; may cause genetic defects; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H301, H312, H314, H332, H334, H350, H360, H372 |
| Precautionary statements | P260, P261, P264, P271, P280, P284, P301+P330+P331, P304+P340, P305+P351+P338, P310, P320, P342+P311, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-3-W |
| Autoignition temperature | 410 °C |
| Lethal dose or concentration | LD50 oral rat 42 mg/kg |
| LD50 (median dose) | LD50 (median dose): 15 mg/kg (oral, rat) |
| NIOSH | Not Established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Beryllium Nitrate: 0.002 mg/m³ |
| REL (Recommended) | Moderate |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Beryllium Beryllium nitrate tetrahydrate Beryllium sulfate Beryllium carbonate Beryllium chloride |

