Beryllium Chromate: Unpacking the Science, The Risks, and The Future
Historical Development
Take a look at the story of beryllium chromate, and you find a vivid illustration of how science chases both opportunity and caution. Scientists in the mid-1900s first managed to synthesize this compound, motivated by a growing curiosity about how uncommon elements like beryllium interact with transition metal ions. Laboratories, mostly in the US and Europe, reported initial successes by blending beryllium salts with chromate sources, and quickly noted the striking nature of this substance. The discovery itself mirrored the era’s broader excitement for pushing boundaries in inorganic chemistry, racing towards new compounds with unique arrangements. The growing interest in beryllium compounds during the postwar boom meant that chromate chemistry seemed like a promising direction, especially since chromates already served vital roles in pigments and corrosion resistance. That hope started to dim as the hazards associated with beryllium became public. The scientific literature thinned out, with researchers moving cautiously, recognizing the real health costs attached to this work, yet the curiosity hasn’t disappeared.
Product Overview
Beryllium chromate, usually written as BeCrO4, stands out for its rare pairing of two quite reactive metals. The material often appears as a yellowish solid, echoing the bold colors typical of chromium compounds. Its purpose hasn’t ever been widespread, in large part because beryllium’s risks discourage large-scale handling. Some early studies dreamed of unique catalyst roles or specialized research tools, but that potential has mostly stayed in the pages of academic journals. It remains a specialty chemical—one whose story gets shaped more by laboratory curiosity than by any sense of commercial promise.
Physical & Chemical Properties
Put beryllium and chromate together, and the resulting compound won’t surprise anyone familiar with inorganic chemistry: you get a powdery solid, yellow like many chromium(VI) compounds. It dissolves in water, showing off a greenish tinge, thanks to the chromate ion. Temperature affects its stability, and heating can split the compound back into its components, sending beryllium oxide and chromium oxides into the air. It acts as both an oxidizer and a source of highly toxic dust, so the chemistry invites both fascination and a good deal of wariness. With beryllium’s low atomic mass, this compound is lighter than a casual glance might suggest, but the bulk physical traits remain similar to other chromates: fine grains, risk of airborne particles, intense color.
Technical Specifications & Labeling
Scientific suppliers approach beryllium chromate with more warning symbols than sales notes. Any bottle worth its label will list not just its chemical formula, but the absolute need for respiratory and dermal protection, signaling the twin dangers of beryllium and chromate exposure. Handling instructions look a lot like those for radioactive materials—lab coats, gloves, goggles, and fume hoods or glove boxes are not optional. While purities above 98% have been recorded in some syntheses, most labs would balk at working with anything that risky outside the smallest quantities. Labels should warn about dust, potential for cancer, chronic lung damage, and the need for government reporting, underscoring how far we are from everyday use.
Preparation Method
Synthesis follows the logic of basic inorganic chemistry, but the execution requires respect for danger. Small quantities of beryllium sulfate or chloride get mixed with a stoichiometric amount of sodium chromate, usually in distilled water, and the beryllium chromate precipitates out. The process itself creates fine particles, and a whiff of the mixture’s dust could mean lasting health trouble. Precipitation often unfolds at a controlled pH, close to neutral, as extremes encourage either dissolution or more hazardous byproducts. After filtration, the yellow material dries under gentle warmth, inside closed systems. The simplicity of the chemistry sits in sharp contrast with the headaches attached to every stage of handling, disposal, and storage. Even universities with a taste for exotic compounds often decide the risks outweigh the benefits.
Chemical Reactions & Modifications
Beryllium chromate doesn’t hide its reactive streak. Expose it to reducing agents, and chromium(VI) ions drop back to the green chromium(III) state, giving visual proof of a reaction designed for demonstration but not for the fainthearted. Acids decompose the solid almost immediately, spitting out toxic beryllium ions and the familiar deep yellow-orange of chromate in solution. The compound can also serve as a beryllium donor, with both cation exchange reactions and destructive oxidation available, but the need for specialized containment keeps such chemistry almost entirely theoretical. Experimenters willing to tackle modifications often step away after reckoning with the implications for waste disposal and environmental impact.
Synonyms & Product Names
Within the research literature and chemical supply catalogues, beryllium chromate appears as BeCrO4, beryllium(VI) chromate, and the less formal "beryllium yellow chromate." The lack of commercial scale means few brand names exist, and each synonym points back to the core risks: beryllium’s toxicity, chromate’s status as a heavy metal, and the shared legacy of occupational illness shaped by both elements over decades.
Safety & Operational Standards
Beryllium chromate’s dangers call for a strict set of protective routines. Beryllium dust, even in microgram amounts, can scar lung tissue for life, leading to chronic beryllium disease and in severe cases, cancer. Chromium(VI) presents its own dire risk as a carcinogen, and together, these hazards make this compound a red-flag item in every chemical safety guide. Regulatory agencies like OSHA and the EPA have hammered out exposure limits for both components. Realistically, anyone working with this material keeps airborne particles far below the legal limits and enforces containment with HEPA-filtered cabinets and secure storage. Decontamination after use involves both wet-wiping surfaces and careful chemical neutralization, followed by lengthy documentation. Waste ends up sealed and sent to secure hazardous disposal sites, skipping the regular trash bins altogether. These rules aren’t theoretical, and history tells the story of labs and workers who paid the cost for ignoring proper controls.
Application Area
Opportunities for practical use of beryllium chromate have always been hemmed in by its twin reputations as a laboratory curiosity and a health menace. Small pockets of inorganic chemistry research have explored its role as an oxidant in the synthesis of other rare compounds, but nothing commercial has survived the transition from bench to industry. Its inclusion in catalysis studies falls into the "niche use" category, and even here, most progress now avoids beryllium wherever possible. The strongest argument for its continued presence in labs comes from those studying the intersection of less-familiar elements rather than any specific application in manufacturing, medicine, or electronics. Science will always have space for these kinds of exotic compounds, but their circle remains small, defined largely by risk and specialization.
Research & Development
Innovation involving beryllium chromate spends most of its energy on understanding mechanisms rather than making products. Experimental chemists in academia have poked at questions involving the ion-exchange behavior, the chromate’s role in redox chemistry, and subtle shifts in crystal structure. None of these studies lean toward making devices or materials for real-world deployment. Instead, they build out our picture of how these rare compounds behave, filling in details that could guide safer or more useful relatives in the future. Even so, interest is shrinking. Funding agencies tend to sidestep risky research that doesn’t offer a clear path toward safer substances with societal value, especially as public awareness of occupational hazards rises.
Toxicity Research
Anyone who’s ever read about occupational lung disease, heavy metal exposure, or toxicology knows why beryllium chromate’s hazard profile leaves little room for optimism. Beryllium exposure costs lives and livelihoods, with decades of research linking it to chronic beryllium disease and lung cancer at very low levels. The chromate portion piles on extra threat; chromium(VI) compounds feature on every public health agency’s watchlist as proven carcinogens. Toxicology research now focuses on understanding biological pathways, mapping out how both ions disrupt enzymes, DNA, and immune systems. Current thinking leaves no doubt: even minute exposure carries risk, and there’s no safe route for routine or casual handling. Animal studies back this up, showing respiratory, urinary, and immune impact even at parts-per-million concentrations. One lesson comes through clearly—clean procedures, well-maintained engineering controls, and strict regulatory oversight have to define every stage of use.
Future Prospects
Looking at where science is heading with beryllium chromate, it’s hard to picture a revival in industrial or commercial use. Environmental and occupational health research keeps reminding us of the care required for beryllium and chromium compounds, so most universities steer students toward less-hazardous challenges. Future directions will most likely sharpen our understanding of how trace metals drive chemical change, rather than expanding the substance’s practical reach. Synthesis might shift toward safer analogues, using computational chemistry and green chemistry to sidestep the problems inherent in beryllium and chromium(VI). As environmental regulations tighten every year, labs face more scrutiny when using legacy hazardous compounds. Some might say there’s value in deepening our knowledge of rare, reactive chemicals; the wider world will be watching to see that such curiosity walks hand-in-hand with responsibility.
What is Beryllium Chromate used for?
Trying to Understand Beryllium Chromate
Beryllium chromate comes up in chemistry textbooks or niche online discussions, mostly as an interesting compound rather than a widely-used material. The formula is BeCrO4. Chatting with folks in lab environments or folks who work in specialty metals, I rarely hear practical stories about beryllium chromate showing up on the shop floor.
Industrial Interest and the Lab Bench
On paper, someone could expect beryllium chromate to show potential for high-tech manufacturing. Both beryllium and chromium find work in aerospace, reactors, or electronics, thanks to their special physical and chemical properties. Beryllium resists heat and holds up under stress. Chromium compounds carry signature colors and oxidation power for reactions. But bring them together—there’s not much evidence of beryllium chromate landing a spot in routine production.
Most mentions drift into hypothetical use: pigments or catalysts. Even here, experts steer away due to safety risks and smooth options elsewhere. University chemistry classes might talk about beryllium chromate for reaction demonstrations or to explain relationships and trends in chemical families. Actual lab shelf samples are rare. If someone has handled it, odds are it happened during a tiny-scale experiment, wrapped in gloves and masks.
Safety, Cost, and Reality Check
Here’s the real anchor—beryllium and chromium (especially as chromate) are not friendly to people or the planet. Beryllium dust can scar lungs for life. Inhaling crumbles of chromate brings cancer risk. Safety instructions for either element stretch longer than a grocery list. Put them together, and the red flags go up.
Any project using this compound comes under tough regulatory watch. Hazard waste rules in Europe or the US practically freeze out beryllium chromate from entering paint, plastics, or electronics. Getting a permit, handling approvals, and disposal all cost real money. Researchers don’t line up to jump these hurdles unless there’s a unique payoff, and so far, nobody has written up a commercial breakthrough.
Looking Elsewhere for Solutions
The industries that might look at compounds of beryllium and chromium lean toward safer options. Pigments now use organic dyes and less hazardous metals like titanium or iron oxides. Catalysts in chemical plants favor less toxic metals, unless only a rare mineral can do the job. If a company needs heat-resistant ceramics, it’ll often start with alumina or zirconia. For specialty electronics or reactor parts, purified beryllium metal, not the chromate salt, carries the load.
Environmental and worker health groups have a point: “ultra-hazardous” substances usually cost more than they give back. Companies cut legal risk, insurance, and disposal bills by picking safer choices. Even in research, finding less toxic proxies opens more doors for collaboration and reduces lab stress.
Responsible Chemistry
My own experience working with high-hazard materials taught me the limits—and toll—of chasing theoretical properties without considering the real barriers of safety and law. Beryllium chromate may show up as a quiz question or in a chemical catalog, but few, if any, can name a job where it earns its keep in day-to-day work. Out of concern for people and the earth, that’s probably a good thing.
Is Beryllium Chromate toxic or hazardous?
Two Hazardous Elements in One Compound
Beryllium chromate sits in a rough corner of the chemical world. On one side, beryllium causes big headaches for health and safety folks. On the other, chromium’s toxic forms give environmental regulators plenty to worry about. Beryllium itself gained a reputation not for its utility, but for its danger. Even small amounts in the workplace air can find their way into the lungs. For some people, it triggers berylliosis – a disease that turns lung tissue into scar tissue. Over time, breathing gets harder, energy drops, and working’s almost impossible. There’s no simple cure, and symptoms can hit years after exposure. The big risk isn’t some rare lab accident; it comes from everyday handling and dust.
Chromium compounds complicated the picture. In certain states, chromium causes allergy, skin ulceration, and cancer. Factories used to ignore these warnings, but legal payouts and stricter codes forced a closer look at the colorants and plating agents built from chromium. If the chromium in beryllium chromate happens to be in the hexavalent form (Cr(VI)), workers might not notice the risk until hands break out in sores, or lab reports flag tumors. Millions spent on cleanup and medical bills often follow.
My Own Glimpses Into Lab Hazards
Back in the research days, I never trusted compounds with either beryllium or certain chromium forms listed on the bottle. Gloves, masks, and ventilation fans only helped so much. A compound like beryllium chromate demanded a whole set of protocols: double bagging the waste, signing off on access logs, reviewing the MSDS before even cracking the seal. Colleagues who ignored these steps usually did so just once – a scare or a rough cough set most people straight. Seeing the cloud of fine dust rising off a fresh bottle gave me more respect for the old-timer’s strict warnings.
Workplace Exposure and Public Health
Beryllium chromate rarely pops up in everyday life, which keeps the general public fairly safe. The trouble mostly appears in specialty industry spaces — research labs, niche electronics work, or maybe in certain aerospace projects. Tradespeople encountering unfamiliar powders can’t always tell what lurks inside a jar or panel. Rushed safety training makes matters worse. Even a few micrograms per cubic meter of airborne beryllium can start trouble.
Government agencies haven’t left this to chance. OSHA and NIOSH drew up strict exposure limits for beryllium and hexavalent chromium. Many companies run air tests and blood screens for certain job categories. The Environmental Protection Agency keeps a watch on how these substances get dumped or spilled outside, with superfund sites paying a steep price for earlier shortcuts. These limits were drawn from decades of tracking sick workers, autopsies, studies in animals, and ongoing lawsuits. The fact that such tough rules exist tells its own story.
Dealing With the Problem in Practice
Safer chemistry often means swapping out problem compounds for tamer options. In practice, beryllium chromate’s nasty profile sends most companies searching for alternatives long before a safety review team gets involved. If certain applications require it, workers use fume hoods, sealed gloves, and respirators—plus they log every gram used. Crowded labs don’t mess with it unless absolutely necessary. Waste heads straight to hazardous disposal, never a regular dumpster or sink.
People who work around risky compounds know that paperwork and permits aren’t just bureaucracy. They form a real line of defense, making sure that losers from decades past don’t get repeated, and that young workers go home healthy. Beryllium chromate, for all its chemical interest, stands as a classic example of a material that’s best left in the bottle, unless thorough protections are in place. If you’re ever in doubt, double-check the labels and reach for the right gear. It’s much easier to prevent harm than to fix it later.
What is the chemical formula of Beryllium Chromate?
Beryllium Chromate: Fact Versus Fiction
Beryllium chromate isn’t a name that shows up often in daily talk, but folks who spend time around chemistry know that both beryllium and chromate compounds can cause headaches in a lab. Beryllium itself is sort of the “problem child” among metals—useful, lightweight, tough, but toxic even at low levels. Chromate compounds also sit in the danger zone because of their history linked to health issues during handling and waste disposal.
So, What’s the Actual Formula?
Anyone who learned basic chemistry probably saw the formulas for regular chromates and beryllium salts. Chromate anion has a charge of 2– (CrO42–), and beryllium usually shows up as Be2+. Pair one Be2+ with one CrO42–, and the math checks out. So, the answer you see in textbooks: BeCrO4.
Here’s where people can trip up. Open up reputable databases—Merck Index, CRC Handbook, PubChem—and few, if any, will list detailed properties for beryllium chromate. Researchers rarely report actually isolating the stuff. From my years browsing chemical supply catalogs and pulling material for experiments, beryllium chromate just never turned up like beryllium sulfate or potassium chromate.
Why Is This Important?
Accuracy in chemical formulas isn’t nitpicking for the sake of ego. Messing up a formula can lead to costly mistakes, from failed syntheses to exposing a lab to more risk than anticipated. If someone expects to find a stable BeCrO4 for demonstration, they’re likely to run into dead ends, wasted budget, or worse—unexpected hazards. According to the CDC, both beryllium compounds and hexavalent chromium exposures lead to lasting health problems. Getting facts clear from the start helps everyone understand what they're truly dealing with.
Chemists love validation. The reactivity and behavior of beryllium chromate have barely seen any confirmation in peer-reviewed literature. Without solid evidence and synthesis, most teachers use it as a theoretical salt—fine on paper, but not a bottle you’ll see on a shelf. I’ve had students ask whether they could order it for demonstration. Every time, I’ve pointed them to safer, well-characterized salts for learning about chromates or beryllium ions.
Finding Real-World Solutions
So, what can people do if they want to understand or work with beryllium or chromate chemistry? Stick to compounds with full safety profiles, clear synthesis routes, and regulatory guidance. Potassium chromate and sodium chromate check those boxes. For beryllium chemistry, the sulfate and chloride salts handle most standard needs. Balancing hands-on science with real safety demands isn’t just good practice; it’s what keeps doors open for learning without regret.
Many newer resources stress limiting exposure and even restricting chromate use to essential situations. I’ve seen schools swap out older “classic” experiments for ones using less toxic ions without losing the learning value. The main thing is not to chase after a formula based only on charge balance or textbook lists. Trust sources built on reproducible research, not just theory.
Bottom Line
“BeCrO4” works on paper, but don’t expect to get your hands on an actual sample with reliable data. Precision and fact-checking count for everyone, from high school students to full-time lab techs. Keeping attention on real, practical chemistry makes the field safer and more rewarding for every learner and professional.
How should Beryllium Chromate be stored and handled?
Why Attention to Safety with Beryllium Chromate Counts
Beryllium chromate doesn’t show up in most everyday lives, but I’ve worked with similar hazardous compounds in labs before. Skipping over safety steps just isn’t an option. Beryllium and chromate both carry heavy risks. Beryllium dust causes chronic lung disease after even small exposures, while chromium (especially in this form) is tied to cancer and severe allergic reactions. People who ignore this reality have found out the hard way: a reckless moment can mean coughs that never go away, ruined skin, or something much worse. Handling beryllium chromate requires respect for its hazards.
Getting Storage Right for Health and Compliance
Walking into a chemical storage room, I want to see clear labels, organized shelves, and no mystery jars tucked in the corners. Beryllium chromate belongs in a locked, corrosion-resistant cabinet, separate from acids and organics. Containers must stay tightly sealed, clearly labeled with hazard warnings, and sturdy enough not to crack under minor bumps. Security rules are not just for show—only trained, authorized people should reach these shelves. I’ve checked Safety Data Sheets often, knowing that regulations like OSHA and EPA have strict rules for this exact reason.
Using the Right Gear
Messing around with beryllium chromate without gear is like jumping into a storm without a raincoat. I’ve always suited up: gloves made of nitrile or rubber (not the cheap stuff), splash goggles, a face shield, and a full lab coat. If dust or fumes come up, I reach for a properly fitted respirator. This isn’t about paranoia—it’s a response to facts. NIOSH research proves long-term exposure leads to permanent lung damage or worse. Changing gloves promptly and washing hands after handling chemicals like this just makes sense. Leaving the lab, you should never take any dust or traces with you—showers and clean clothing become non-negotiable.
Spill and Waste Protocols
I’ve dealt with spills, and things get tense fast. Never sweep or compress beryllium chromate powder. Wet methods or HEPA-filtered vacuums prevent tiny particles from floating into the air. Disposal isn’t as simple as chucking it in the trash: proper disposal containers with tight lids, labeled as hazardous waste, and scheduled by a licensed contractor. No shortcuts. The law is clear and the environmental toll of chromium compounds hitting soil or water is huge.
Training and Vigilance
Complacency breeds accidents. Regular training on handling and emergency protocols keeps everyone sharp. I’ve learned to run through drills of eyewash station use, spill containment, and escape routes. Managers have a responsibility here: keep safety gear stocked, provide updated info, and watch for shortcuts creeping into daily routines. Peer checks help—my colleagues and I watched each other’s backs every day. I’ve seen how a culture of accountability limits accidents.
Room for Technology to Help
Improvements in storage monitoring have changed the game. Environmental controls, like automated ventilation and real-time sensors, give instant alerts if there’s a leak or change in air quality. These aren’t luxuries; they catch problems before health risks grow.
The Stakes Remain High
Beryllium chromate isn’t just another chemical on the shelf. Mishandling can leave lives and ecosystems permanently damaged. Following established guidance isn’t just about rules—it’s about protecting real people. Sticking to proven precautions, enforcing tough standards, and staying watchful every day lets science and industry keep moving forward without preventable harm.
Where can I purchase Beryllium Chromate?
A Rare Request with Bigger Implications
As odd as it sounds, plenty of people—whether students looking for science project help, collectors, or amateur chemists—sometimes type questions about buying substances like Beryllium Chromate. Before even heading to the internet, it’s worth knowing this isn’t just a curiosity. My high school chemistry days taught me curiosity can lead to research, and sometimes, to dead ends for good reason.
Facts about Beryllium Chromate
The compound in question, Beryllium Chromate, holds chemical hazards that go beyond “just another salt.” Beryllium itself can cause awful lung issues. Even seasoned lab workers use special protection when handling it. Add on top the chromate part, which comes from chromium(VI), known for its cancer risks, and the mix really doesn’t belong anywhere near open-to-consumer sales. This isn’t a fear tactic—just years of research and incident reports.
Legal and Safety Restrictions
If you’ve ever tried placing an order from chemical suppliers like Sigma-Aldrich, Alfa Aesar, or Fisher Scientific, strict vetting applies. These companies don’t ship high-hazard substances to private addresses. You’ll get asked for licenses, research justifications, workplace certifications. I remember a friend in grad school who waited weeks for nitric acid; Beryllium Chromate stands on an even higher level in terms of control. In the U.S. and many other countries, you’ll run straight into walls set by occupational safety and environmental laws.
Why Such Strict Rules?
Both personal and public experience tells us accidents happen. The stories of chronic beryllium disease, or of chromate-induced cancers in industrial workers, aren’t rare in the history of science and manufacturing. In places where standards faltered for even a year or two, the results were permanent health impacts. It’s not just about protecting individual users; it’s a matter of community safety and liability too.
So, Where Does That Leave the Curious?
For educational purposes, alternatives exist. Universities, accredited labs, and research collectives often work with hazardous chemicals but under the guidance of experts, within equipment built to keep people safe. Science museums sometimes organize demonstrations that let you observe reactions involving risky chemicals—but behind glass, with all the proper protocols. If you need Beryllium Chromate for genuine industrial or research purpose, start by talking to accredited chemical vendors, get safety training, and follow compliance documentation. As a private individual, buying such substances isn’t an option—and for good cause.
A Better Way Forward
Instead of searching for banned or dangerous materials, focus on understanding their chemistry, impacts, and why they’re tightly regulated. Schools and online resources now offer safe, legal routes for in-depth study. There’s always value in learning, but safety rules exist for a reason. Most chemical suppliers stick closely to laws built from hard-learned lessons. For anyone frustrated by these limits, speaking directly with science instructors or professional chemists can point you to experiments that deliver the same excitement without the risk.
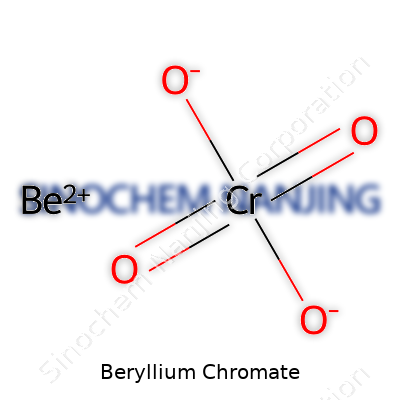
| Names | |
| Preferred IUPAC name | beryllium dioxido(dioxo)chromium |
| Other names |
Beryllium chromate(VI) Chromic acid, beryllium salt Beryllium dichromate |
| Pronunciation | /bəˈrɪliəm ˈkroʊmeɪt/ |
| Identifiers | |
| CAS Number | [13598-15-7] |
| Beilstein Reference | 3581248 |
| ChEBI | CHEBI:84933 |
| ChEMBL | CHEMBL1201720 |
| ChemSpider | 20370474 |
| DrugBank | DB14636 |
| ECHA InfoCard | 100.016.726 |
| EC Number | 246-120-1 |
| Gmelin Reference | Gm 20/2 132 |
| KEGG | C18975 |
| MeSH | D001583 |
| PubChem CID | 24588 |
| RTECS number | QS9625000 |
| UNII | F2TL01403B |
| UN number | UN1544 |
| CompTox Dashboard (EPA) | DTXSID4044406 |
| Properties | |
| Chemical formula | BeCrO4 |
| Molar mass | 152.02 g/mol |
| Appearance | Yellow solid |
| Odor | Odorless |
| Density | 3.11 g/cm³ |
| Solubility in water | soluble |
| log P | -0.28 |
| Vapor pressure | 0 mm Hg (approx.) |
| Basicity (pKb) | 11.2 |
| Magnetic susceptibility (χ) | +1150e-6 |
| Refractive index (nD) | 1.72 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 69.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -844.0 kJ/mol |
| Pharmacology | |
| ATC code | B01AX06 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs; may cause genetic defects; causes severe skin burns and eye damage; may cause allergic skin reaction. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H350, H301, H314, H317, H330, H334, H340, H341, H350i, H360, H372, H410 |
| Precautionary statements | P201, P202, P220, P261, P264, P270, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P314, P321, P330, P333+P313, P363, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-OX |
| Lethal dose or concentration | LDLo orally in rat 200 mg/kg |
| NIOSH | RN0123 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Beryllium Chromate: 0.002 mg/m3 |
| REL (Recommended) | REL: Ca See Appendix A |
| IDLH (Immediate danger) | 0.05 mg/m3 |
| Related compounds | |
| Related compounds |
Chromium trioxide Beryllium nitrate |

