Beryllium Carbonate: A Critical Look from Origin to Outlook
Historical Development
Interest in beryllium compounds goes back to the early 1800s, when scientists wrestled with unknown minerals and curious green gemstones. Figuring out beryllium carbonate started with the isolation of beryllium itself, which Friedrich Wöhler and Antoine Bussy pulled off in 1828. Beryllium carbonate slipped into the spotlight later, not as a naturally occurring mineral, but crafted through deliberate chemical reactions. Its history ties directly to the push for lighter, stronger materials across science and industry. As aviation and electronics made demands that old metals and minerals couldn’t meet, chemists kept searching for new compounds with promising properties. Beryllium’s lightness and unique behavior under high temperatures always caught their attention, and that curiosity keeps the compound relevant today. Whenever historians talk about the advance of specialty metals, beryllium carbonate shows up on the timeline, reminding us how far the periodic table’s hidden prize has come.
Product Overview
What you usually find in a lab or catalog labeled as beryllium carbonate refers to a white or sometimes off-white powder. It’s not something you find in the wild, so every grain derives from lab work or industrial setups. People in research, manufacturing, or chemistry don’t use beryllium carbonate for its looks, but for its role as a precursor or an ingredient in making other beryllium products—especially in ceramics, specialized glasses, and as a reagent in chemical analysis. If you’re in the rare earth route, this compound may crop up where purity matters and when every atom counts.
Physical & Chemical Properties
Holding a vial of beryllium carbonate, you'd see a solid that seems nondescript and powdery, dissolving a little in water—though not nearly as easily as some other carbonates. Its chemical formula, BeCO3, looks simple, but the structure proves less stable than what you’ll find with magnesium or calcium. This instability shows up if you leave the compound exposed to the atmosphere: moisture and airborne carbon dioxide start shifting the equilibrium, leading the powder to break down, and beryllium hydroxide appears. Many chemists notice the decomposition if storage doesn’t focus on dryness. The compound displays basic behavior—it reacts with acids to bubble out carbon dioxide and with bases in more niche, complicated reactions.
Technical Specifications & Labeling
Anyone handling or buying beryllium carbonate expects strict specifications. Purity matters—impurities can sabotage downstream reactions or final ceramic performance. Labs typically demand assay values of 99% or higher, with limits on heavy metals (especially lead, cadmium, and iron). Product labels call out grade—research, technical, or premium grades—plus storage and handling instructions, batch numbers, and warnings citing its significant toxicity. Regulatory compliance isn’t optional; shipping papers list hazard classes and signal wording, and every container carries clear hazard pictograms to comply with global standards like GHS (Globally Harmonized System).
Preparation Method
The go-to route for making beryllium carbonate stays pretty straightforward: React soluble beryllium salts—often beryllium sulfate or chloride—with sodium carbonate in aqueous solution. This prompts precipitation as beryllium carbonate. After filtering and washing, the compound stays delicate, prone to breaking down if not kept anhydrous. Some specialist syntheses tweak this route, using ammonium carbonate or bubbling carbon dioxide through beryllium hydroxide suspensions to achieve specific hydration levels or increase particle uniformity. Controlling temperature, rates of addition, and agitation, chemists get dramatically different grades and characteristics, so the process leaves room for skill.
Chemical Reactions & Modifications
Beryllium carbonate doesn’t stay untouched in practice. It reacts briskly with dilute mineral acids, fizzing as it produces carbon dioxide and beryllium salts—an everyday step in analytical chemistry. Heat it in the open, and the powder releases carbon dioxide at a couple hundred degrees Celsius, going straight to beryllium oxide. This thermal decomposition interests those making high-purity beryllium ceramics or exploring heat-resistant compounds. Substitution reactions don’t run rampant here, since beryllium’s small ionic radius and strong polarization keep many ions from swapping places without struggle. Yet, advanced researchers keep finding ways to modify the compound with ligands or stick it onto polymers for research purposes.
Synonyms & Product Names
Reference books and supplier catalogs don’t stick to one name. Along with beryllium carbonate, you’ll see synonyms like carbonic acid beryllium salt, or even “BeCO3” as a catch-all. Some contexts use basic beryllium carbonate, which technically means the Be2CO3(OH)2 version, not straight BeCO3. Some brands toss their own proprietary code on the label, but be wary: only regulatory-quality grade and clear chemical identity count for safety and legal use.
Safety & Operational Standards
Beryllium compounds, including the carbonate, demand respect for their toxic potential. Inhalation, dust exposure, or skin contact can trigger chronic lung disease (berylliosis) and, in some cases, cancer. Regulatory limits set airborne concentrations of beryllium at very low thresholds—current US OSHA rules target 0.2 micrograms per cubic meter averaged over eight hours. Employers and workers must use fume hoods, double-layer containment, and personal protective equipment—nitrile or neoprene gloves, splash goggles, and certified respirators. Operating procedures in research or industry environments focus on keeping the work area free from dust, managing waste as hazardous, and avoiding cross-contamination. Training, documentation, and regular air monitoring reflect that one mistake or oversight puts lives at risk, and regulators take enforcement seriously.
Application Area
Most beryllium carbonate gets used as an intermediate, helping make beryllium ceramics or acting as a reagent in specialty syntheses. Engineers in electronics and nuclear applications value ceramics made from beryllium compounds for their capacity to withstand high temperatures, boast low neutron absorption, and dissipate heat rapidly. These features make beryllium-based shields critical in fusion reactors and advanced aerospace projects. Analytical chemists, though that’s a niche set, continue to use the compound to prepare beryllium standards or separate rare earths in mineral analysis. Broader adoption remains capped by cost, handling risk, and the drive to replace hazardous substances wherever possible.
Research & Development
Curiosity hasn’t run out for beryllium carbonate. Today’s research papers look at new synthesis routes, aiming for higher purity or nanostructured forms that could unlock both better performance and safer handling. Some teams investigate ways to stick beryllium carbonate nanoparticles into polymers or other matrices, chasing better sensors, lightweight battery casings, or responsive composite materials. Efforts in computational chemistry keep probing the coordination chemistry of beryllium to predict new uses or cost-effective substitutes. Progress often comes slowly, slowed by strict safety regimes and the challenge of developing non-toxic methods for beryllium handling.
Toxicity Research
Few chemicals show such a challenging toxicity profile as beryllium carbonate. The dust, even at low concentrations, can sensitize lungs and create a persistent immune reaction—that’s why chronic beryllium disease remains a concern decades after first exposure. Lab studies and occupational health tracking still detail the long latency and severity. Animal models and cell culture research continue to clarify the biochemical mechanisms for carcinogenicity. Regulators and industrial hygienists demand constant review of safety data; every new study influences public policy, permissible exposure limits, and the push for alternatives.
Future Prospects
Over time, the world will keep wrestling with the tradeoffs of using beryllium carbonate. The physical properties promise breakthroughs in ceramics, advanced optics, and fusion reactors. That said, industrial and academic minds keep searching for replacements or safer derivatives, since regulatory pressure and public health costs rise alongside new discoveries about toxicity. Advances in materials science may eventually shadow beryllium carbonate’s high spots, but for now, its performance in heat-resistance and electronics leaves it relevant. For those who work with it, only rigorous safety and clear-sighted respect keeps research and manufacturing moving ahead without incurring irreversible health costs.
What is Beryllium Carbonate used for?
Looking at Beryllium Carbonate in Real Life
Beryllium carbonate rarely pops up in everyday conversation, but it plays a part in areas where materials with specific qualities are needed. From my days tinkering in the university’s chemistry lab, I learned pretty fast that beryllium compounds don’t get picked for big consumer products. They’re usually reserved for settings where their unique properties make a genuine difference.
Where Beryllium Carbonate Makes a Difference
This compound comes up most often on the research side of things. Researchers in the chemical industry use beryllium carbonate as a starting point when making other beryllium compounds. Since beryllium itself has some impressive qualities—like being lightweight, strong, and having a high melting point—its compounds sometimes feed into processes where standard materials fall short.
Beryllium carbonate can help make certain ceramics better at handling heat. Most folks will never see it, but some high-performance ceramics in aerospace rely on this sort of specialty ingredient during development. It’s not just the aerospace field, either. Electronic parts need materials that keep their shape and power under intense pressure, and beryllium compounds have a knack for holding up where others break down.
Safety Takes Center Stage
I want to address something important here. Working with anything containing beryllium takes strict attention to safety. It’s not up there with table salt or chalk dust—beryllium compounds have been linked to serious health issues. Breathing in even small amounts of beryllium dust over time can cause a chronic lung condition called berylliosis. Every chemistry lab I’ve set foot in keeps beryllium work behind heavy ventilation and protective equipment. Workers who handle these materials wear respirators and protective clothing to stay safe.
The U.S. Occupational Safety and Health Administration (OSHA) lists clear exposure limits. The Centers for Disease Control and Prevention (CDC) follows these as well. Companies with these materials on site train their staff how to work safely with them and handle disposal with care. Even small mistakes get taken seriously.
Alternatives and Solutions
People keep looking for alternatives to beryllium carbonate, mostly because of the health risks. In the electronics and ceramics industries, researchers experiment with other compounds that could step in with less worry attached. For most mainstream uses, the industry sticks to alternatives unless there’s no substitute for what beryllium carbonate can offer. This keeps exposure low and reserves its use for applications where it really makes sense.
Better technology also means safer handling. Modern labs feature enclosed workstations and more accurate measuring tools now. This lowers the risk compared to setups from decades ago. If an industry still needs what beryllium brings to the table, companies work hard to minimize any danger to both employees and the environment.
Why It Matters
I’ve seen firsthand how niche compounds like beryllium carbonate can move things forward in science and industry. They don’t belong in the average household, but their role in research and advanced manufacturing can't be ignored. Careful use and attention to health will keep them useful, but not hazardous.
If you’re ever working near a lab that mentions beryllium, just remember: there are reasons for the rules, and safety matters as much as the science.
Is Beryllium Carbonate toxic or dangerous?
Beryllium’s Reputation: Not Without Reason
People hear the word “beryllium” and often think about safety warnings in industrial settings. Experience dealing with chemical handling tells me those warnings aren’t overblown. Beryllium as an element isn’t something you want to handle lightly—its toxic effects are well-known, and the risks carry over to beryllium compounds, including beryllium carbonate. Even if the precise form changes, the underlying danger doesn’t just disappear.
Why Beryllium Carbonate Gets Attention
Beryllium’s danger comes mostly from its ability to cause chronic lung problems. Breathing dust or fumes from any beryllium compound opens the door to something called chronic beryllium disease. CDC data shows lung inflammation, scarring, and even an increased risk of cancer. Acute exposure irritates skin, eyes, and mucus membranes. With beryllium carbonate, inhaling or even touching the powder can mean hauling around these risks.
Health Effects: It Doesn’t Take Much
Tiny exposure amounts add up. Chemical handling guides generally flag the safe limit for beryllium in air at less than one microgram per cubic meter. Symptoms sometimes start with coughing, chest pain, or shortness of breath, and the problem can keep smoldering for years before people realize the root cause. I once worked with a tech whose exposure happened over a decade, and it took even longer for the health effects to make sense. Not even the so-called “insoluble” forms of beryllium—such as beryllium carbonate—get a pass, because the body can still absorb and move them around.
Environmental and Workplace Risks
Proper ventilation, sealed processes, and correct disposal matter a lot with this stuff. The Environmental Protection Agency doesn’t mince words when describing beryllium pollution—spilled powders can seep into the ground or travel on wind. In workplaces, personal protective equipment is standard, and periodic air monitoring turns into a regular fire drill. Factories keep strict rules for storage and accidental spills. From what I’ve seen, forgetting to label a bucket or shortcutting safety training can lead to costly errors.
Safer Alternatives and Smart Protections
The tricky part with beryllium carbonate is that it sometimes pops up in specialty ceramics, scientific work, or metallurgy. Most fields have switched to less hazardous materials where possible, but old habits linger. Training never stops; new workers often underestimate fine powders and their tendency to float into the air. Personal experience taught me the difference between working in a lab that treats these compounds with respect and one that cuts corners—illness rates don’t lie.
Every workplace dealing with beryllium must commit to regular health screening. Medical checks can catch early signs before problems get serious. Good ventilation, sealed equipment, and sturdy gloves form the front line of defense. It pays to respect labels and material safety data sheets. No short-term gain makes up for long-term lung damage or worse.
Understanding the risk attached to beryllium carbonate matters for anyone handling it—even if its use has shrunk over the years. As science has shown again and again, a lack of visible symptoms doesn’t mean the danger isn’t building under the surface. In my view, the best solutions combine smart precautions with a healthy dose of caution, never letting familiarity breed carelessness.
What is the chemical formula of Beryllium Carbonate?
Basic Science, Real Impact
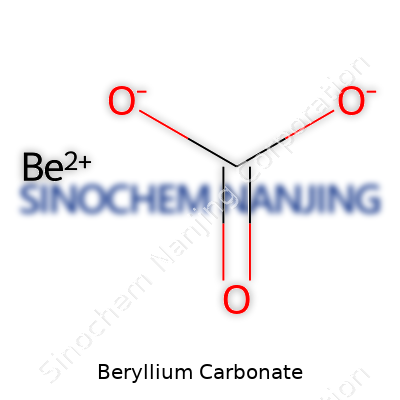
Beryllium carbonate has a chemical formula of BeCO3. This isn’t just a trivia answer. The way each element comes together means something for anyone working with minerals or studying chemistry. Beryllium stands out in the periodic table, not just as a lightweight metal, but as a material with unique properties and practical uses. Combine that with the carbonate ion (CO32−), and you land on a chemical that can teach a lot about stability, reactivity, and safety.
Why Beryllium Carbonate Rarely Shows Up
Believe it or not, finding beryllium carbonate in the wild isn’t likely. It barely hangs together at room temperature—moisture brings it down, converting it into beryllium hydroxide and releasing carbon dioxide. In my years sweating through lab safety seminars, the rarity of this compound never went unnoticed. People asked why you rarely see it outside certain chemical reactions or controlled setups. Its instability makes it a poor candidate for anything outside laboratory research or specific industrial processes.
Health Risk Is No Joke
Beryllium has a toxic reputation. Doctors and industrial workers learned this the hard way in years past, dealing with dust that causes chronic, sometimes fatal, lung conditions. The carbonate form still carries the dangers tied to the element, even if it doesn’t persist long in nature or regular air. Safety data puts beryllium compounds among the strictest in labs, and I remember whole shelves segregated just for these bottles. Proper ventilation, gloves, and disposal procedures aren’t just best practices. They’re the only way to stop accidents.
Education and the Curriculum Gap
I saw enough blank stares in introductory chemistry lectures to realize that students don’t get enough hands-on experience. The formula BeCO3 may land on tests, but it often shows up as just another entry in a long list. Adding examples rooted in real risk helps concepts stick. Beryllium’s health warnings and the instability of its carbonate form can drive home bigger lessons about respect and responsibility in handling chemicals—even the ones that seem obscure on paper.
Responsible Use and Innovation
Some companies look at beryllium’s properties and see possibilities in aerospace and electronics. Its low mass and high melting point appeal to engineers chasing better performance. Still, the path from the lab to the factory means taking every precaution. Substitutes for beryllium are always under development, and I’ve seen more organizations turning to less hazardous options, both due to regulatory pressure and a genuine shift toward safer workplaces. Even chemistry teachers now use digital simulations to show reactions that can’t be done in class because of the danger.
Building a Safer Future
Information about formulas, stability, and health risks needs to travel beyond textbooks and reach anyone deciding what chemicals to use or teach. Clear rules, updated safety training, and a real commitment to innovation can protect people without slowing progress. Beryllium carbonate’s story reminds researchers and teachers how discovery and healthy skepticism go hand in hand. If we keep sharing hard lessons and supporting better choices, the next generation stands a better chance of handling both opportunity and risk wisely.
How should Beryllium Carbonate be stored?
Understanding What’s at Stake
Beryllium carbonate isn’t something folks run into every day, but anyone working in a lab knows it carries some weight in the world of chemical safety. Health experts and toxicologists mark it as a hazardous material due to both its toxicity and the physical risks connected to beryllium. Exposure, even at low levels, can trigger lung issues that nobody wants to deal with—chronic beryllium disease does not play around. That’s not fearmongering; it's drawing the line between caution and serious trouble.
Storage Starts with the Right Container
Plastic, glass, or high-density polyethylene containers with tightly fitting lids work best. Metal might corrode, so steering clear makes sense. These containers block out moisture, which beryllium carbonate tends to soak up. Moisture increases the risk of a chemical reaction or makes the powder clump. An airtight seal keeps air and water vapor out. If you use a secondary containment tray, spills get caught and cleanup takes less effort.
Get the Storage Environment Straight
Temperature control can make all the difference. I’ve worked in labs where someone forgot to keep the cabinet cool—clumping and sweating chemicals followed. Beryllium carbonate prefers a cool, dry space out of direct sunlight. Light and heat push chemicals toward instability, not something anyone wants in the workplace. No flammable storage cabinets or spots near acids; mixing those chemicals up could lead to a dangerous reaction, which messes with both safety and lab equipment.
Label Everything Clearly
Don’t rely on memory or scraps of tape. Store beryllium carbonate in containers that sport clear, chemical-resistant labels. Include the name, hazards, and the date it went in. This way, no one grabs the wrong thing. Safety data sheets should live nearby so anyone handling the chemical can double-check safety measures. Clear labeling means the right steps happen during an emergency or routine checks—no guesswork needed.
Limit Access and Keep a Record
Not everybody in a lab should get near beryllium compounds. Only staff with proper training and protective equipment should handle the stuff. Logging use and storage helps track inventory, which matters if regulations ever come into play or someone needs to check who accessed the chemical last. Good records make it easier to spot any issues before they cause problems.
Protect People, Not Just Chemicals
I’ve learned that personal experience and expert guidelines agree: gloves, goggles, and lab coats go on before you pop open any container. Dust masks or respirators should also be in reach. Even with the best planning, accidents sometimes happen. Posting contact information for emergency services and keeping spill kits nearby turns panic into action.
Disposal Matters, Too
Never toss unused beryllium carbonate in the regular trash or pour it down the drain. It goes into a designated hazardous waste bin. Waste teams need to know what they’re handling—otherwise the chain of safety breaks right there.
Anyone in a lab finds out fast that storing chemicals isn’t about rules for rules’ sake. It is good science, protecting both people and the environment. Safe storage means fewer close calls and healthier lives. That’s the only way chemical handling should work.
Is Beryllium Carbonate soluble in water?
The Curiosity Behind Solubility
Beryllium doesn’t make the headlines in most households. Yet, its carbonate form grabs attention for chemistry students and industry professionals who like to dive deeper into why certain chemical compounds either dissolve in water or stay stubbornly undissolved. I remember sitting in my undergraduate chemistry class, puzzled by the periodic behavior of alkaline earth metals, especially the quirks in their carbonates. The chart says calcium and magnesium carbonate barely budge in water, but what about beryllium carbonate?
Strong Bonds and Reasons to Stay Solid
Beryllium carbonate steps into the picture with a rather unique trait. It barely dissolves in water. This comes down to the tiny size of the beryllium ion and its high charge density. In chemistry circles, this is known as polarizing power. Beryllium grabs onto those carbonate ions with such force that water molecules can’t pull them apart easily. The result? You won’t get much beryllium or carbonate floating around unless you mess with the chemistry a bit—like bubbling carbon dioxide through a cold beryllium hydroxide suspension. But even then, you’re forming a different, less stable compound.
Why Solubility Matters Beyond the Lab
This seemingly obscure property shifts from a classroom curiosity to a real-world concern in several ways. Beryllium sits in factories, aerospace labs, and environmental tests. Its rarity in soluble carbonate form means that in water supplies, you won’t suddenly stumble across toxic levels just by mineral leaching. The Environmental Protection Agency keeps its eye on beryllium, but the bigger risk comes from inhaling dust—not from drinking carbonate-rich water. As somebody who’s read incident reports and safety datasheets, I know most beryllium hazards circle back to workplace environments, not dissolved chemicals in rivers or lakes.
For researchers, this sets beryllium apart. Standard water treatment systems focus on substances that dissolve and spread fast. Beryllium carbonate’s stubbornness in water means fewer alarms for water engineers but reminds everyone to double-check their methods if they’re testing for beryllium. Water treatment that targets dissolved metals won’t have much to do unless highly acidic or specific laboratory scenarios enter play.
Supporting the Science
Textbooks like Atkins’ “Inorganic Chemistry” lay out these trends—solubility of carbonates drops as you move from magnesium to beryllium on the periodic table. You also find journal studies confirming beryllium carbonate’s almost theatrical resistance to water, showing solubility levels close to zero under standard conditions. That’s been confirmed by both experimental chemists and environmental scientists looking for rogue elements in the field.
Room for Improvement in Handling and Communication
While beryllium carbonate rarely jumps into water, the bigger lesson comes from how we treat rare and hazardous substances. Even rare chemicals can surprise when conditions change. Handling beryllium in any form calls for careful controls: masks, gloves, proper training. The science tells us beryllium carbonate isn’t a water-solubility problem, but transparency in safety data and clear communication keep both workers and researchers safe. Industry guidance improves all the time, with more focus on preventing dust and airborne exposure rather than chasing dissolved particles in water. If anything, this helps us see the value of nuanced chemical knowledge—real safety starts with genuinely understanding the materials in play, not just following generic rules.
| Names | |
| Preferred IUPAC name | Beryllium carbonate |
| Other names |
Carbonic acid, beryllium salt Beryllium carbonate, anhydrous |
| Pronunciation | /bəˌrɪli.əm ˈkɑː.bə.neɪt/ |
| Identifiers | |
| CAS Number | [1312-36-9] |
| 3D model (JSmol) | `"BeCO3"` |
| Beilstein Reference | 358941 |
| ChEBI | CHEBI:86437 |
| ChEMBL | CHEMBL1201574 |
| ChemSpider | 21569703 |
| DrugBank | DB01307 |
| ECHA InfoCard | 100.007.069 |
| EC Number | 208-566-1 |
| Gmelin Reference | 60478 |
| KEGG | C18871 |
| MeSH | D001594 |
| PubChem CID | 10668421 |
| RTECS number | DS1925000 |
| UNII | LL60O88Y2I |
| UN number | UN1566 |
| CompTox Dashboard (EPA) | DTXSID7023075 |
| Properties | |
| Chemical formula | BeCO3 |
| Molar mass | 68.026 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.63 g/cm³ |
| Solubility in water | Decomposes in water |
| log P | -1.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 11.4 |
| Basicity (pKb) | 11.18 |
| Magnetic susceptibility (χ) | -1.32e-6 |
| Refractive index (nD) | 1.500 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 73.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -970 kJ/mol |
| Pharmacology | |
| ATC code | B05CX05 |
| Hazards | |
| Main hazards | Toxic if inhaled, ingested, or in contact with skin; may cause skin and respiratory sensitization; carcinogenic; may cause irritation to eyes, skin, and respiratory tract. |
| GHS labelling | GHS05, GHS08, Danger, H350, H360, H372, H302, P201, P202, P260, P264, P270, P280, P308+P313, P314, P405, P501 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H350: May cause cancer. H372: Causes damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P302+P352, P304+P340, P308+P313, P312, P321, P333+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LDLo oral rat 50 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Beryllium Carbonate: "0.002 mg/m3 (as Be) |
| REL (Recommended) | Beryllium carbonate is not recommended for use due to its high toxicity. |
| IDLH (Immediate danger) | 0.005 mg Be/m³ |
| Related compounds | |
| Related compounds |
Beryllium carbonate hydroxide Beryllium oxide |

