Beryllium Acetate: An In-Depth Look
Historical Development
The story of beryllium acetate winds its way back to the earliest chemistry labs where scientists hungrily explored the behavior of rare-earth elements. In the twentieth century, researchers recognized beryllium’s unique place among light metals. By the mid-1900s, beryllium compounds gained traction in both academic research and specialized manufacturing, driven by atomic energy pursuits and advanced materials programs. Early curiosity about beryllium salts set the stage for the synthesis of several acetates. People have been both wary and intrigued by this substance throughout the decades, given its notorious toxicity and remarkable physical properties. The search for robust, reliable methods to handle and analyze beryllium-based salts continues steadily, spurred on by defense and electronics industries hungry for compounds with niche traits.
Product Overview
Beryllium acetate belongs to a selective family of organometallic chemicals with a central beryllium ion bound to acetate ligands. The salt shows up most often as a hydrated crystalline powder, often light in color and highly soluble in water or alcohol. Its reputation comes from the blend of lightweight metal and carboxylate, useful in specialty synthesis and scientific projects. The product finds its market niche in laboratories focused on advanced inorganic chemistry, materials science, and analytical development. With ongoing demand for ever more specialized chemical tools, this compound keeps a small but loyal following among researchers dealing with beryllium technologies, even as stringent safety rules tighten across the world.
Physical & Chemical Properties
This acetate compound has a distinct look—crystalline granules or powder, typically white or off-white. Beryllium acetate dissolves quickly in water, reflecting the ionic nature of the salt, and holds up well in most standard organic solvents. The material’s melting point runs relatively low, usually in the range of 190–200°C, and it decomposes with the release of acetic acid. In the open air, the chemical remains stable under dry conditions, but moisture in the air can cause it to clump and degrade, losing active content over time. Key parameters for the industry include purity, hydration state, and solubility, which can impact both the yield and safety profile in applied settings.
Technical Specifications & Labeling
Specifications for beryllium acetate lean on both purity and moisture content. The typical product label calls out beryllium content by percentage weight, clear hydration state, and trace impurity limits. Most suppliers package the powder in small quantities, ranging from grams up to a few kilograms, using airtight containers to slow down hydrolysis and contamination. Labels emphasize beryllium’s toxic profile with hazard symbols and explicit instructions for handling, requiring users to take personal protection seriously. Given the dangers, distribution of this chemical faces oversight from multiple regulatory bodies in Europe, North America, and Asia. Proper record keeping and material traceability form part of the shipment process, reflecting the compound’s tight regulatory environment.
Preparation Method
The synthesis involves reacting beryllium salts, usually beryllium sulfate or beryllium chloride, with sodium acetate or glacial acetic acid in aqueous conditions. The pathway produces beryllium acetate through double decomposition, with clear separation and crystallization as the solution evaporates. Once filtered and dried under vacuum, the resulting solid is tested for purity through spectroscopy, gravimetry, and sometimes X-ray diffraction. Lab-scale production remains the standard as few firms operate facilities for large-batch manufacturing due to regulatory costs and workplace safety demands. Preparation protocols rarely shift, as most commercial systems stick closely to the methods put down in chemical handbooks and peer-reviewed articles over the past fifty years.
Chemical Reactions & Modifications
Beryllium acetate keeps chemists busy with its reactivity. In solution, the compound can swap out the acetate group for other carboxylates, phosphates, or halides through metathesis. Heating leads to breakdown, producing beryllium oxide—an essential ceramic precursor—and releasing acidic vapors. In more elaborate synthesis, beryllium acetate can act as a mild catalyst or as a source for beryllium in organometallic cluster formation. Over time, synthetic chemists have developed routes to tweak the acetate ligands with functional groups for use in targeted molecular assembly. The field remains niche, but the chemistry unlocks new approaches in materials science and coordination chemistry.
Synonyms & Product Names
Beryllium diacetate and beryllium(II) acetate crop up in catalogues and journals alongside the common name. Some texts use the moniker “acetic acid, beryllium(2+) salt,” a nod to traditional nomenclature. Storage containers and technical datasheets may also reference CAS Number 543-60-6 for industrial tracking. This string of names reflects both scientific convention and the patchwork of regulatory and commercial systems that circulate the compound through different markets and research communities.
Safety & Operational Standards
Handling beryllium acetate calls for top-tier safety practice. Dust from the powder represents a critical inhalation hazard, with well-documented risk of chronic beryllium disease and cancer after long-term exposure. Labs enforce strict control measures: chemical fume hoods, full-face respirators, gloves, and specialized waste disposal protocols. Many countries ban or tightly control uses outside licensed facilities. Frequent training forms part of workplace protocols, with real-time air monitoring and routine health screening for chemical workers. Local and federal laws frame every aspect of its use, from shipment to storage to end-of-life recovery. Even a moment’s slip can result in contamination, which underscores the level of vigilance needed.
Application Area
Most work with beryllium acetate rings out in the laboratories, both academic and industrial. It shows value in synthesizing beryllium ceramics, nuclear fuel elements, and in the preparation of advanced composites where beryllium’s low weight and high thermal resistance matter. Chemists look for beryllium acetate when purity and solubility call the tune in catalyst development or as a source of beryllium ions in solution-based experiments. Some legacy uses in electronics linger in specialized sensor design and as dopants in certain crystal growth processes. The tools of modern materials science may move on, but a small core set of applications holds steady, as alternatives don’t always deliver beryllium’s suite of physical properties.
Research & Development
Contemporary research pushes the boundaries, focusing on uncovering new roles for beryllium compounds in lightweight, heat-resistant composites. Scientists experiment with its use in advanced aerospace and defense materials, where extreme conditions destroy lesser substances. Innovations in analytical chemistry call for options with unique solubility or catalytic behaviors, and beryllium acetate fits this bill while forcing teams to take new levels of safety precautions. R&D projects around the world periodically come back to this compound when looking at state-of-the-art energy capture, transducer development, or the manipulation of rare metal ions in complex media. The need for high-performance, low-mass materials brings interest back to beryllium, even amid the ongoing search for safer alternatives.
Toxicity Research
The dangers tied to beryllium acetate run deep. Decades of toxicology study show even small amounts can cause serious health problems, especially in lung tissue. Inhalation brings the greatest threat, prompting granulomas and possibly chronic beryllium disease—a slow, brutal immune reaction. Long latency periods mean doctors often diagnose well after first contact, complicating public health tracking. Modern toxicity research leans on cell-based models and animal studies, confirming early findings while searching for better ways to detect exposure. Occupational standards continue to evolve, cutting legal limits almost to zero in some jurisdictions. Unlike many industrial chemicals, there’s no real safe threshold. All meaningful research with beryllium acetate faces intense oversight, and the drive for alternative compounds in commercial use has never moved faster.
Future Prospects
Beryllium acetate’s future will be decided by the balance between safety needs and the thirst for new high-tech materials. Trust in tight regulatory systems will keep the compound on the books in scientific circles where there’s no substitute for its properties, but broad industrial adoption faces an uphill struggle. Worldwide, businesses and labs keep hunting for less toxic ways to match what beryllium compounds can do, looking to magnesium, aluminum, or synthetic composites to fill the gap without the same health concerns. New research into controlled handling techniques, medical monitoring, and bio-monitoring offers a guardrail for working chemists, letting the field move forward without repeating mistakes from previous generations. The need for top-tier materials isn’t fading, but the spotlight now shines on safety, driving both innovation and caution in beryllium acetate’s place in the modern world.
What is Beryllium Acetate used for?
The Role of Beryllium Acetate in Materials Science
Beryllium isn’t a name that pops up often outside science labs. Its shimmering salts, like beryllium acetate, play a quiet but important role in research and cutting-edge industry. Beryllium acetate often gets used in ceramics, nuclear reactors, and specialty glass production. Scientists like it because it dissolves well in water, making it easy to mix into formulas or coat surfaces. In my time researching advanced materials, working around beryllium compounds always brought an extra layer of care and respect due to the toxicity — always double gloves and fume hoods. That extra effort exists for good reason.
Why Researchers Rely On Beryllium Acetate
Beryllium acetate serves as a handy source of beryllium ions during the creation of ceramics that need to withstand high temperatures. Think of components for rocket nozzles, satellites, or precision x-ray windows. The acetate form helps distribute beryllium more evenly throughout the material during manufacturing. This distribution improves thermal conductivity and keeps things running cooler for longer, which matters if you’re building tools for space or high-energy physics.
Beryllium-based ceramics have another perk — incredible strength for their weight. If you need something strong, light, and resistant to breaking down, there aren’t many other elements or compounds that compare. Beryllium acetate, with its convenient solubility, offers a practical way to introduce beryllium into these complex mixes without making things overly complicated for technicians or scientists.
Energy, Electronics, and the Nuclear Field
The nuclear industry has a unique relationship with beryllium compounds. Beryllium acetate finds its way into production processes for neutron moderators or reflectors. These specialty parts shape and reflect neutrons inside reactors, tailoring the chain reactions that keep power plants running or research going. It’s not about abundance, it’s about control and reliability. That’s the value scientists look for in specialty chemicals like these.
In electronics, researchers sometimes reach for beryllium acetate to make certain thin films or to study beryllium’s effect on conductivity. The field doesn’t use much — cost and health risks push teams toward alternatives most of the time. Still, where there’s a clear technical win, no one ignores a compound just because it takes work to handle safely.
Health and Safety Risks
Beryllium carries health risks that no one can skip over. Inhaling even a little dust can cause berylliosis, a severe lung disease. I remember material safety days in the lab and the posters warning about chronic beryllium disease. No shortcuts, not even if you’re short on time. Trained staff, sealed containers, and specialized disposal — all are required by law and common sense. The U.S. Occupational Safety and Health Administration (OSHA) sets strict exposure limits. Companies put big money into protective gear and training, because safety violations can lead to lawsuits and damaged reputations.
Better Solutions: Is There a Safer Road?
With all the risk, researchers are always looking for ways to replace or reduce beryllium use. Sometimes, switching to aluminum or titanium in ceramics can work, but only if you can trade off some of beryllium’s special properties. For nuclear technology, the options narrow quickly. Government and industry are putting pressures on developing safer alternatives, but the combinations of light weight, strength, and heat resistance aren’t easy to match all at once.
Responsible use grows in importance as technology advances. Labs train new scientists not just in chemistry, but in safe practices and environmental responsibility. Beryllium acetate stays vital for research and industry, but the costs — not just in dollars, but in safety and lives — push everyone toward innovation that protects both people and planet.
Is Beryllium Acetate hazardous or toxic?
Beryllium Acetate’s Risks Are No Small Matter
Beryllium acetate isn’t something you’re likely to run into at the supermarket, but it plays a role behind the scenes in science labs and some corners of industry. Too often, people see a chemical formula and glance past the safety sheets. Having spent years handling chemicals in academic labs, I learned to pay close attention to any compound linked with beryllium. That habit has paid off more than once — especially with substances overlooked by those new to the bench.
Direct Health Risks from Exposure
Beryllium compounds have a track record for causing health trouble. Beryllium acetate lands among them. Breathing in even tiny bits can leave the lungs scarred — what experts call chronic beryllium disease or berylliosis. A painful, irreversible condition, berylliosis doesn’t need years of heavy exposure to leave a mark. The U.S. National Institute for Occupational Safety and Health (NIOSH) warns about beryllium dust and salts: skin rashes, lung issues, weight loss, even increased cancer risk. Every reputable handling guide for beryllium acetate flags toxic inhalation and warns of possible organ damage.
My own experience with emergency room visits after a chemical spill (not beryllium, but a similarly hazardous compound) left a lasting memory. No hazard sheet ever feels like extra paperwork after seeing what skin and lungs can go through. The risk may be low if the powder or solution stays in a sealed bottle, but a dropped flask or poorly fitted respirator changes everything. Accidents don’t care about confidence or intent — only about preparation.
Occupational Exposure and Community Impact
Some people assume beryllium acetate matters only in big manufacturers or national labs. But the compound can show up in university research settings, materials testing, or specialty electronics work. Workers often say they follow every protocol, but history repeats itself when safety funding gets cut or training slips. Children near beryllium operations in Pennsylvania, where processing plants have operated for generations, have shown signs of exposure. This isn’t just a lab problem.
Best Practices Can Make a Big Difference
Protective gear changes the whole story. A well-fitted N95 or P100 mask, gloves that stand up to caustics, and solid ventilation—they all protect lab workers. There is no excuse for skipping the basics. Labs can install local exhaust hoods and invest in chemical training for every employee, not just for show but to save lives. Supervisors don’t always like spending time or money on prevention until someone gets hurt. But constant reminders, spot checks, and access to shower stations can reduce accidents. Policies need teeth—reliance on employee memory or good intention doesn’t cut it where beryllium acetate is concerned.
Personal Responsibility and Wider Solutions
Legislation and company policy dictate a lot, but individual choices matter too. If you don’t understand the risks or the first step for spill response, don’t open the bottle. Signs and labels work only if read and understood. Industry could take a lesson from groups working with asbestos—people need ongoing health checks and public reporting about beryllium incidents, not just after a lawsuit lands. Research is pushing toward less toxic substitute materials in some areas, and that process deserves all the support it can get.
As someone who’s watched close calls upend careers, one thing stays clear: Respect for the chemical means respect for your own future. Tools and training, not shortcuts or luck, tilt the odds in favor of safety when beryllium acetate comes out of the cabinet.
What is the chemical formula of Beryllium Acetate?
Beryllium Acetate: Chemical Identity
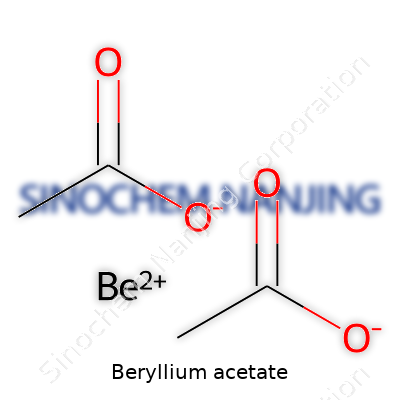
Beryllium acetate looks pretty unremarkable on a shelf, but its composition carries unique value in the world of chemistry and industry. The chemical formula, Be(C2H3O2)2, reveals both the make-up and real-world considerations around its use. One beryllium atom pairs with two acetate groups, each one known from common vinegar chemistry as the functional part of acetic acid. That means beryllium acetate falls under the family of organo-beryllium compounds, bridging metals with organic chemistry in a single bite-sized formula.
The Unique Nature of Beryllium and Acetate
Beryllium stands out for being light and strong at the same time. Very few metals can claim that sweet spot. In the form of beryllium acetate, people sometimes use it to introduce beryllium into chemical reactions when they need it to dissolve and react more readily than plain beryllium metal. The acetate part acts as a small, portable group easily swapped in coordination chemistry; acetic acid backs the household vinegar, but the scientific version helps metals become more versatile in lab work. Be(C2H3O2)2 combines both, unlocking new options for researchers.
Health, Safety, and the Risks That Come with Use
Using beryllium compounds, including beryllium acetate, always brings up serious safety issues. This is not just a matter of paperwork or regulations. Exposure to beryllium dust or fumes can trigger chronic beryllium disease, which damages lungs and can even lead to fatal outcomes. I remember working in a lab where strict protocols ruled every step, right down to gloves, masks, and fume hoods anytime we handled any beryllium salts. Even a tiny amount feels like a risk when weighed against the potential for lifelong health issues.
Stakeholders in both research and manufacturing have to follow best practices set out by agencies like OSHA and the CDC. Professional training makes a big difference. The smallest error—forgetting to properly seal a bottle or letting acetates mix with glove powder—can open up avoidable exposure. Over time, many laboratories adapt specialized tools and monitoring systems to keep tabs on airborne beryllium. Responsible organizations test employee air samples and use wet chemistry approaches to minimize the dust risk. The research shows that keeping contamination under control is not just about a single product, but about stable, ongoing vigilance.
Factoring In Environmental Responsibilities
Disposal raises questions, too. Beryllium compounds accumulate in waste streams unless handled the right way. Hazardous waste regulations treat them as threats to both human and environmental health. Labs and factories work closely with certified waste companies to neutralize or contain leftovers. The average chemist does not pour this stuff down the drain, and responsible manufacturers coordinate with local authorities to keep aquifers and soil clean.
Seeking Better Alternatives and Safe Handling
Researchers always look for safer ways to achieve the same chemical transformations. Sometimes substitutes for beryllium come into play, though none truly match its unique properties in high-temperature alloys or as catalysts. In many cases, using smaller amounts, keeping exposures to a minimum, and upgrading ventilation form the backbone of best practices. Investing in real-world safety—beyond what the rulebooks say—matters for everyone involved, from those in the lab coat to those overseeing waste transport.
How should Beryllium Acetate be stored?
No-Nonsense Safety for a Potent Chemical
Beryllium acetate shows up in research aimed at catalysis, ceramics, and specialized glass. Anyone with experience around this material knows it doesn’t demand attention for no reason. Exposure can cause serious health problems, especially to the lungs and skin. Each time I open a box labeled “toxic,” Beryllium compounds remind me why strict rules matter as much as technical skill. Following best practices isn’t just about keeping regulators happy; it protects everyone around—from the intern shelving bottles to the seasoned technician measuring out milligrams.
Location is Everything
To store Beryllium acetate, I look for a cool, dry space, tucked away from chemicals that might trigger a reaction. No one wants acid vapors or oxidizers near this stuff. A locked, marked cabinet works best, preferably one designed for toxic or carcinogenic materials. Lab signs keep confusion to a minimum—there’s no benefit in a colleague grabbing the wrong container by mistake. About once a year, someone asks, “Is this shelf safe for Beryllium?” Airflow and temperature become just as important as labels. Room temperature is fine, as long as humidity stays controlled.
Original Packaging: Don’t Get Creative
Most of the time, the original bottle does the job. Manufacturers choose chemical-resistant material for a reason. Tampering with that only raises the risk. Screw caps must stay tight. After decades around chemicals, I’ve seen enough leaks and crusty residue to know that sloppy storage raises the chance of contamination. It’s all about keeping everyone out of harm’s way, even if it means double-bagging or secondary containment. Plastic bins or tubs help stop broken glass from turning into a disaster cleanup. That routine step can save a whole bench from getting shut down.
Segregate to Stay Safe
Beryllium acetate doesn’t belong with acids, peroxides, or flammable solvents. I store it with other heavy metal compounds, each on a separate tray. Clear separation keeps fire, toxic vapors, and accidental mixing off the table. Having worked through two surprise audits, I can say: Nothing ruins a week like explaining an unlabeled shelf of incompatible chemicals.
Active Ventilation and Spill Planning
Even in storage, dust from Beryllium acetate can pose a threat. I make sure the area has good ventilation, or at least a spill kit nearby so accidents get contained fast. PPE stays close, including gloves and goggles. Investing in a small respirator means no one scrambles during an emergency. Years ago, a coworker skipped that step and ended up red-faced in the office after a spilled scoop. Respecting a strong safety routine means those slips get handled with less stress and no hospital trips.
Disposal and Inventory Matter
Unused Beryllium acetate doesn’t simply get tossed in the trash. Hazardous waste rules say it goes to a certified disposal site. Our lab logs every intake and outgoing container, double-checking for leaks and dates. Early disposal beats letting old bottles become fragile and dangerous. Every year we audit—not just for compliance, but so no forgotten bottle sits waiting for an accident.
Knowledge Protects People
Experience around Beryllium acetate leads to habits that protect both people and research. Clear labels, original packaging, smart segregation, and honest assessments create a safer environment. The science stays exciting, and everyone heads home healthy. That’s the gold standard in any lab worth its salt.
What are the handling and safety precautions for Beryllium Acetate?
What is Beryllium Acetate?
Beryllium acetate is mainly used in scientific research, some ceramics, and in the making of specialty alloys. It’s not something found in everyday settings. Most people working with it handle it in labs, factories, or certain industrial applications.
Why Precautions Matter
The big reason for caution comes down to one fact: beryllium compounds don’t mess around. The U.S. Occupational Safety and Health Administration (OSHA) classifies beryllium as a highly toxic substance. Breathing in even tiny particles can trigger a lung disease called chronic beryllium disease. It acts slow, shows up years after exposure, and doesn’t go away. In some cases, it can even turn deadly. Safety isn’t a suggestion—it’s a requirement grounded in real consequences.
Key Handling Practices
Before opening a container, it makes sense to check that every detail is in place. Air filtration should run smoothly. The workstation stays uncluttered to reduce the risk of accidental spread. If spills do happen, a trained team must know the steps for dealing with dust and wet residues. Beryllium acetate has a habit of sticking to gloves or clothing, so it helps to double-check before moving away from the work zone. Nobody wants to track toxic material out of the lab or workshop.
Personal Protective Equipment Makes the Difference
Casual work gear won’t cut it. Lab coats, gloves, and goggles create the basic barrier. A NIOSH-approved respirator fits into the picture in any situation where dust or vapors could escape. I learned this in graduate school, watching a careless moment lead to hours of decontamination. Shoe covers and disposable clothing also help keep the risk contained. The less skin exposed, the better off a person stays.
Good Habits Save Lives
Everyone working with beryllium acetate should get in the habit of washing hands with soap and water before eating or drinking. Food, drinks, and even open water bottles belong nowhere near the work area. It’s easy to forget, but bad habits around the lunch table have led to serious exposures. Cleaning surfaces, using wet wipes, and vacuuming only with HEPA filters keeps particles out of the air where people could breathe them.
Proper Storage and Disposal
Beryllium acetate goes in tightly sealed, clearly labeled containers—no exceptions. Containers stay in locked cabinets, away from acids and bases. All waste, including rags and disposable gloves, counts as hazardous waste and should follow special chemical disposal protocols. Mixing with regular trash or pouring solvent down the drain can taint water and land for years, hurting both people and the environment.
Training and Awareness
Every workplace should give regular training on the risks and best practices for handling beryllium acetate. People just entering the job need more than a quick talk—they need real hands-on practice. I’ve seen that clear, honest conversations save more accidents than the thickest manual. Questions need honest answers. No one benefits from shortcuts or silence when handling a toxin this serious.
Better Solutions for the Future
Switching to less toxic materials, improving ventilation, and upgrading engineering controls help reduce risks. Artificial intelligence, robotics, and remote handling systems keep people further from direct exposure—a trend that’s gaining ground in modern labs. By investing in better practices and safer alternatives, we shape a world with fewer workplace illnesses tied to dangerous materials like beryllium acetate.
| Names | |
| Preferred IUPAC name | Diberyllium tetraacetate |
| Other names |
Acetic acid beryllium salt Beryllium diacetate Beryllium(II) acetate |
| Pronunciation | /bəˈrɪli.əm əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | [543-80-6] |
| Beilstein Reference | 1718733 |
| ChEBI | CHEBI:91258 |
| ChEMBL | CHEMBL1232573 |
| ChemSpider | 23055 |
| DrugBank | DB14636 |
| ECHA InfoCard | 100.027.899 |
| EC Number | 208-755-9 |
| Gmelin Reference | 4024 |
| KEGG | C18643 |
| MeSH | D001594 |
| PubChem CID | 24864870 |
| RTECS number | DS1925000 |
| UNII | 2QD09P0HLL |
| UN number | UN1325 |
| CompTox Dashboard (EPA) | DTXSID8045275 |
| Properties | |
| Chemical formula | Be(C2H3O2)2 |
| Molar mass | 185.06 g/mol |
| Appearance | White solid |
| Odor | Acetic acid-like |
| Density | 1.92 g/cm³ |
| Solubility in water | Soluble |
| log P | -1.0 |
| Vapor pressure | 0.025 mmHg (25°C) |
| Acidity (pKa) | 2.8 |
| Basicity (pKb) | 6.09 |
| Magnetic susceptibility (χ) | −14.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.45 |
| Viscosity | 20 cP (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1018.3 kJ/mol |
| Pharmacology | |
| ATC code | B01AX10 |
| Hazards | |
| Main hazards | Toxic if inhaled, swallowed, or in contact with skin; causes skin and respiratory irritation; may cause cancer. |
| GHS labelling | GHS02, GHS06, GHS08, Danger, H272, H301, H315, H319, H330, H335, H350, H360, H372 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H350: May cause cancer. H372: Causes damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P210, P260, P262, P280, P302+P352, P304+P340, P308+P313, P314, P405, P501 |
| Lethal dose or concentration | LD50 (oral, rat): 12 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | 2 µg/m³ |
| REL (Recommended) | 0.02 mg/m³ |
| IDLH (Immediate danger) | 4 mg/m3 |
| Related compounds | |
| Related compounds |
Beryllium carbonate Beryllium nitrate Beryllium oxide |

