Benzyloxy Sulfonyl Chloride: At the Crossroads of Chemistry and Innovation
Historical Development
Chemists have explored aromatic sulfonyl chlorides for generations, but benzyloxy sulfonyl chloride really stepped into the spotlight as organic synthesis took off through the twentieth century. I remember digging through stacks of old patents and journals in grad school, noticing how much attention academic groups gave to derivatives like this compound. Researchers across Europe and the US tinkered with these reagents, hoping to find new ways to protect functional groups in tricky molecules. Back then, benzyloxy sulfonyl chloride drew interest for its solid reactivity and straightforward preparation—offering skills to mediate transformations where older, fussier compounds often sputtered out. It’s this kind of slow, collaborative progress that often builds the backbone for bigger discoveries later on.
Product Overview
Benzyloxy sulfonyl chloride fills a niche that’s surprisingly valuable: activating, protecting, or modifying substrates in both industrial synthesis and academic research. You see this name pop up in labs focused on pharmaceuticals, namely because it gives chemists a nimble tool to play with functional group protection. What sets it apart isn’t just reactivity but consistency—batch after batch, it lets researchers chase predictable reactions in an uncertain world. That reliability gives people confidence that a long, complex preparation won’t get derailed by a moody reagent.
Physical and Chemical Properties
Benzyloxy sulfonyl chloride draws its usefulness from a blend of stable and reactive traits. In the bottle, it presents as a pale liquid, sometimes tinged yellow by trace decomposition. Its aroma isn’t subtle—chlorinated aromatics rarely are—and anyone who’s opened a bottle in a small lab has stories about stubborn odors that linger on gloves, lab coats, and even paperwork. Chemically, the compound stands out for a sulfonyl chloride group hanging off a benzyloxy ring. This combination makes the compound eager to react with nucleophiles—amines, alcohols, even water, if the humidity runs high. That rapid hydrolysis reminds chemists to treat storage and handling as non-trivial. If you’ve ever left the bottle open near a sink, you’ll discover sticky residues by the next morning.
Technical Specifications and Labeling
Specialty chemical suppliers aim for purity high enough to serve research and pilot-scale work. In practical terms, that means analysts monitor residual solvents, secondary byproducts, and water content. For many organic transformations, chemists demand at least 98% purity—anything less risks introducing side products in downstream reactions. The labeling, at least in my experience, gets crowded with hazard information: pictograms warning about skin and eye irritation, sometimes a note about respiratory risks, details about proper gloves and goggles, and the typical CSDS code language about adequate ventilation. While these labels sound routine, they reflect honest past challenges chemists face in keeping workspaces safe.
Preparation Method
Synthesizing benzyloxy sulfonyl chloride feels almost old-fashioned in its simplicity—sulfonylating agents like chlorosulfonic acid meet benzyloxy-containing substrates under careful temperature control, with the whole process watched like a hawk for exothermic bursts. If scale-up becomes necessary, attention shifts to venting and moisture management, since excess fumes and accidental hydrolysis can turn a routine batch into a safety training anecdote. Purification depends on precise fractionation, pulling away unreacted chlorides and isolating the pure product in a dry, sealed flask. Lab protocol now leans on fume hoods with solid airflow and glassware that never sees a bead of water until the operation is finished.
Chemical Reactions and Modifications
This compound shines as a sulfonylating agent, transferring its sulfonyl group to targets ranging from simple alcohols to complex diamines. It enables chemists to manipulate molecular frameworks, often through the creation of sulfonamides and sulfonate esters, that show up in pharmaceuticals, agrochemicals, and modern materials. The chloride group makes for a willing participant, slicing off in the presence of stronger nucleophiles and opening routes to further derivatization. The world of protecting group chemistry leans on these features; anyone who’s tried wrangling an ornery polyfunctional molecule sees the advantage in a reagent that reacts fast, then lets go cleanly in a deprotection step. Sometimes minor mods—swapping the benzyl ring for a differently substituted aromatic—set the stage for new physical properties, broadening the library of custom sulfonylating reagents.
Synonyms and Product Names
Lab books and trade catalogs don’t always settle on one name. You might see “benzyloxy sulfonyl chloride,” sometimes “benzyloxybenzenesulfonyl chloride,” or even just cryptic abbreviations in reaction schemes. That patchwork of names occasionally slows down literature searches or leaves newcomers second-guessing if they’ve got the right compound. Other languages add their own twists, but the core remains: a benzyl group, an oxygen linkage, and a sulfonyl chloride moiety. Most chemists learn to spot the relevant molecular fingerprint by eye, even when the names wander.
Safety and Operational Standards
Respect for chemical safety shapes every step of handling benzyloxy sulfonyl chloride. Anyone who’s worked in synthesis knows how splashy, pungent, and corrosive these reagents can be. Labs enforce strict standards—working under fume hoods, storing in airtight bottles, segregating from bases and nucleophiles, and wearing nitrile gloves rated for strong acids and chlorinated materials. If spilled, the standard approach involves neutralizing with sodium bicarbonate and grabbing spill pads before the fumes travel. Ventilation keeps low-level exposure from irritating airways over a workday. These details seem mundane until a lapse leads to chemical burns or lingering asthmatic symptoms. In industry, automated dispensing units and process monitoring shave down the risk even further, but most stories about “learning experiences” happen on the bench, not the plant floor.
Application Area
Benzyloxy sulfonyl chloride finds its way into everything from pharmaceuticals to agrochemicals to specialty polymers. Academic chemists prize its ability to slip a sulfonyl group onto a complex molecule without major fuss. In drug discovery, this functional group sometimes modulates biological activity or improves solubility. Material scientists reach for it to tweak the conductivity or surface properties of polymers. In my lab days, a single bottle might support three or four projects at once: protecting amino groups during peptide assembly for one person, sulfonylating an aromatic core for another, and troubleshooting side reactions for a third. That’s a testament not just to the compound’s versatility, but also to the persistent creativity of people chasing new and useful chemical space.
Research and Development
As organic synthesis becomes more reliant on automation and high-throughput screening, chemists keep hunting for reagents that work quickly and cleanly. Benzyloxy sulfonyl chloride lands squarely in this arena. Several research groups look for analogs with slight tweaks—bulky substituents, for example, that improve selectivity or stability. There’s also a push to improve green chemistry metrics, whether it’s through more benign chlorinating agents or methods that cut out hazardous waste. This work doesn’t always make headlines, but it steadily improves both the sustainability and scope of fine chemical synthesis. Over time, successful tweaks ripple out to manufacturing, where savings on time, energy, or waste add up quickly.
Toxicity Research
Toxicologists classify sulfonyl chlorides as respiratory and skin sensitizers, and benzyloxy sulfonyl chloride fits this pattern. Acute exposure risks include coughing, eye irritation, and even chemical pneumonitis at high concentrations. Chronic risks remain less studied, but any regular handler keeps a healthy respect for the possibility of cumulative effects—a splash on bare skin leads to burning, and inhaled fumes prompt evacuation and medical checks. Animal studies often inform regulatory thresholds, but researchers keep agitating for more human-relevant toxicology data. Better exposure controls sprang up in labs once chemists realized how even low-level contact can contribute to long-term health issues, especially in crowded academic settings.
Future Prospects
Benzyloxy sulfonyl chloride won’t replace every sulfonylating agent any more than glycerol replaced all other alcohols, but its balance of reactivity and manageability sets it up for a lasting role. Chemists will keep looking for ways to run these reactions under milder conditions, with less hazardous waste and more control over selectivity. Miniaturized and flow-based systems could minimize human exposure, turning benzyloxy sulfonyl chloride and its cousins into invisible cogs within safer, more efficient research pipelines. At the same time, demand for new medicines, advanced materials, and functional foods continues to drive interest in reagents with reliable track records and creative potential. If the pace of fundamental chemical discovery holds up, we’ll likely find new uses and safer ways to harness compounds like benzyloxy sulfonyl chloride for decades to come.
What is the chemical formula and CAS number of Benzyloxy Sulfonyl Chloride?
The Formula and CAS Number
Benzyloxy sulfonyl chloride packs a simple formula: C7H7O2SCl. The CAS number is 16140-26-4. It’s not a household name, but in the right labs, that string of numbers opens doors to a world of synthetic routes.
What Purpose Does Benzyloxy Sulfonyl Chloride Serve?
You don’t run into benzyloxy sulfonyl chloride at the corner pharmacy. Instead, it’s tucked away on the shelves of research labs and specialty chemical suppliers. Folks reach for it during the preparation of protected sulfonamide groups, or when the benzyloxy moiety needs installing. A few years ago, I remember paging through procedure notes from an older chemist, and this compound gave them a solid route for making tailored intermediates. Without it, their options would have been a lot messier and more toxic.
Why Pay Attention?
The world loves to chase after new molecules, but the bedrock of progress often sits with these “supporting actors.” Benzyloxy sulfonyl chloride gives chemists a tool that cuts down on unnecessary waste, both in terms of time and byproducts. In the search for new pharmaceuticals, the industry can burn hundreds of hours and liters of solvents on protection and deprotection steps if the chemistry doesn’t cooperate. With a stable, well-characterized compound like this one, development speeds up, and risks drop. The fewer steps in a synthesis, the better your odds of reaching the next breakthrough a little faster and safer.
Think about the older methods: some used gas-phase chlorination or required high energy input. Both options bring hazard and cost. This compound offers a more targeted way to get those sought-after sulfonamide bonds—a foundation for many drugs. More control in the lab means fewer toxic side streams and reduced chance of dangerous byproducts. The green chemistry movement leans hard on choices like this, aiming to trade out yesterday’s methods for smarter, safer ones.
Working With It—Facts and Precautions
No chemical deserves mindless handling. Benzyloxy sulfonyl chloride can sting your airways, irritate skin, and wreck your unprotected eyes. I’ve seen enthusiastic younger chemists leap into work without thinking about fume hood airflow or splash protection; they learned fast, sometimes painfully. Always check your gloves, and keep the bottle secure when not in use. Anyone who’s spent time at the bench has a story about a minor spill that turned into an hourlong cleanup. Good habits help prevent those tales from happening at all.
Transport requires tightly sealed glass bottles, kept dry and cool. The compound doesn’t play nice with water—hydrolysis eats it up, and no one likes cleaning up that acidic residue. It doesn’t need to complicate waste streams if you keep the basic safety rules in mind: label containers, segregate sulfur chlorides from alcohols and bases, and double-check waste cans before disposal. Safety data sheets spell this out, and it pays to read them before opening the bottle.
Moving Forward—Smarter Practices in the Lab
So many bottlenecks in chemical synthesis vanish with the right starting material. Benzyloxy sulfonyl chloride brings reliability. If suppliers keep standards high and users follow practical safety advice, research keeps moving forward without avoidable accidents or delays. Responsible sourcing matters, too. Labs should insist on documentation and traceability—both for safety and for reproducibility. When the pressure is on to make smarter drugs or cleaner materials, there isn’t much room for carelessness at the fundamental level.
This compound isn’t flashy, but it earns its spot in the toolkit. Care for it, respect its reactivity, and it’ll help the next generation of chemists make things cleaner and faster.
What are the common applications or uses of Benzyloxy Sulfonyl Chloride?
The Tool Behind Many Organic Syntheses
In organic chemistry, benzyloxy sulfonyl chloride carries a reputation for versatility. Students and researchers learn early that progress often relies on clever building blocks—this compound delivers in that department. Its main draw rests in its ability to transfer a benzyloxy sulfonyl (often known as “besyl”) group onto amines and other nucleophiles. In real-world terms, this gives chemists a straightforward way to modify molecules, isolate specific functions, or protect delicate groups during complex syntheses.
Making Better Pharmaceuticals
The pharmaceutical world always searches for ways to build drugs with accuracy. Benzyloxy sulfonyl chloride steps in as a reagent, helping to protect free amine groups during the construction of antibiotic, anticancer, or antiviral compounds. In a busy laboratory, the difference between a successful drug candidate and a failed experiment sometimes comes down to such protection steps. Errors in this stage can ruin months of work. The reliability of benzyloxy sulfonyl chloride for forming stable sulfonamide intermediates makes it invaluable for medicinal chemists seeking efficiency and purity.
Streamlining Peptide Synthesis
Anyone who works with peptide chemistry knows the headaches involved with side reactions. Benzyloxy sulfonyl chloride helps sidestep many of those problems, specifically by masking amine groups when stringing together amino acids. During stepwise peptide synthesis, chemists need to control reactivity—exposing specific groups one at a time. The benzyloxy sulfonyl group stays intact under many reaction conditions, then comes off easily when needed, allowing the final peptide to emerge pure and ready for testing. This reliability increases yields and saves materials, which means lower costs and fewer hazardous byproducts.
Growing Role in Agrochemical Research
Synthesizing new crop protection agents—herbicides, insecticides, or fungicides—often hinges on having the right synthetic tools. Benzyloxy sulfonyl chloride finds a spot here thanks to its ability to help produce sulfonamide derivatives, which play critical roles in many agrochemical formulas. Researchers value predictable reagents; if plant safety screens fail or an intermediate proves unstable, the result is lost resources and missed deadlines. This sulfonylating agent supports reliable routes to innovation in greener and more selective agricultural chemicals.
Looking Ahead: Safety, Sustainability, and Alternatives
Like many specialty chemicals, benzyloxy sulfonyl chloride brings concerns about safe handling and environmental impact. It reacts strongly with water and produces fumes that call for good ventilation and solid lab safety practices. Waste management also keeps users cautious. The chemistry community recognizes the pressure to find less hazardous reagents or processes that cut down on hazardous waste and energy use. Greener chemistry journals devote attention to alternatives that might reduce the risks or environmental burden, but so far, few options match the utility of benzyloxy sulfonyl chloride on the bench.
Supporting Evidence and Trust in Research
The continued use of benzyloxy sulfonyl chloride in both academic and industrial settings speaks to its proven track record. Numerous peer-reviewed studies detail its roles in synthesis, and leading suppliers offer tightly controlled product quality backed by rigorous analysis. My own work in graduate school relied on trusted reagents like this, and most successful research teams build up their own experience with these building blocks over time. Responsible use draws on that collective wisdom, balancing opportunity with honest attention to risk.
What is the recommended storage condition for Benzyloxy Sulfonyl Chloride?
Putting Safety First for Chemical Storage
Benzyloxy sulfonyl chloride falls into the group of chemicals that demand extra respect in the lab and warehouse. Its value in organic synthesis depends not just on how you use it, but how you hold onto it between deliveries and experiments. I’ve spent enough time handling sensitive reagents to know that carelessness leads to spoiled materials and safety scares. Unless everyone is clear about proper storage, an expensive order can go to waste, or worse—cause an avoidable accident.
Temperature and Environmental Conditions Matter
Benzyloxy sulfonyl chloride reacts quickly with moisture, and its fumes don’t treat lungs kindly. According to supplier safety data, this chemical keeps best at cool temperatures, away from direct sunlight. Most guidelines recommend a dry, well-ventilated area, using a sealed container that doesn’t allow vapors or water to get in. A fridge between 2 and 8°C usually works, but make sure the storage place stays far from food or break rooms. Colleagues swapping stories about ruined batches due to fluctuating room temp convince me that a consistent, low temperature is worth the effort.
Why Moisture Control Can't Be Ignored
Sulfonyl chlorides love to react with water, releasing hydrochloric acid and toxic gases in the process. I’ve seen what happens when a reagent bottle sits open too long on a humid day—fumes sting the nose and throat, and crystals in the neck of the bottle clog up future uses. Keeping containers tightly capped, using desiccators for backup, and never pipetting directly from the stock will prevent contamination. It only took one ruined reaction for me to double up on silica gel packets and double-check every lid every time.
Storage Containers Make a Difference
Glass is usually the best bet for these types of reagents, as plastics sometimes degrade or get brittle over time. Dark amber glass helps block out light that can speed up decomposition, so supplies last longer and keep their punch for critical protocols. Labels should show hazard symbols and storage notes. My old mentor insisted on large, bold markings after a misplaced bottle once ended up next to peroxides—a near miss that left a strong impression.
Ventilation and Emergency Access Save Lives
Strong ventilation keeps accidental inhalation risks low during retrieval or repackaging. I prefer chemical storage rooms with a fume hood nearby and clear spill kits on hand. Beyond keeping compliant with local rules, these steps stop minor slipups from turning into injuries or exposure. Emergency eyewash and showers need to stay a short jog away from any spot where you might open a reactive bottle.
Simple Habits Prevent Big Problems
Every time I open a bottle, I remember advice from veteran chemists: check the label, date, and seal. Don’t trust memory with hazardous materials. Make storage checks routine, and brief new team members on hazards before they even think about handling benzyloxy sulfonyl chloride. Safety starts with shared knowledge and practical habits, not paperwork alone.
What are the safety precautions and handling measures for Benzyloxy Sulfonyl Chloride?
The Risk Is Real
Benzyloxy sulfonyl chloride may not sound like much to folks outside a chemistry lab, but this chemical demands respect. Working with it, I quickly traded curiosity for caution. Getting hit by its vapors or coming into direct contact can burn skin, eyes, and lungs. Just cracking open the bottle releases fumes that sting the nose. So before even thinking about handling it, preparation matters more than anything else. Arming yourself with information is just as important as wearing gloves.
Protection Starts With Gear
Anyone dealing with this chemical grabs goggles, a splash-resistant lab coat, and thick nitrile gloves, plus a face shield for good measure. I remember standing nervously under a fume hood the first time, goggles fogging up, but I would never trade a clear view for the risk of a chemical splash. Ordinary latex gloves fall apart under serious chemical stress. Nitrile or laminate gloves last longer and create a better barrier. Lab coats covered my arms and buttoned tight at the wrist. The habit stuck with me—too many accidents happen when someone rolls up their sleeves.
Ventilation Is Not Optional
No one opens benzyloxy sulfonyl chloride at a desk. Strong ventilation isn’t up for debate. Fume hoods don’t only filter; they keep vapor away from your lungs. A room with airflow alone won’t cut it. The right setup protects nearby coworkers, too. My own sore throat and watery eyes taught me not to trust any shortcut. Investing in a tested fume hood keeps the lab safer and keeps chemical smells stuck behind glass.
Right Storage Makes a Difference
This chemical doesn’t play nice with humidity or most metals. It belongs inside a tightly sealed, labeled container, separate from water, alcohols, or anything flammable. Chemical-resistant cabinets go a long way. Some places keep a spill kit nearby, packed with absorbent mats and neutralizing agents. In my view, a few well-placed warning signs make people think before reaching in. Clean hands and clear labels spell less trouble down the road.
Handle Spills With Respect
Even careful hands slip up sometimes. Spills need speedy and calm action. No one charges in without already knowing what to do. Small spills get covered with an inert material—think sand or special absorbent powder. The waste goes into a sealed bag, which then goes straight to hazardous disposal. Big spills call for evacuation, and then bringing in trained emergency crews. Spraying water is off-limits, since it triggers dangerous fumes. I’ve never had to deal with a big spill myself, and I hope I never do. Still, knowing what to do ahead of time beats panic every time.
Training and Teamwork Count
People make the biggest difference. Routine safety drills, up-to-date safety data sheets, and open conversations about hazards save more lives than any expensive cabinet ever could. I find that colleagues who don’t rush and double-check everything create a safer environment. Simple habits matter: never work alone, always share your plans, and keep communication open. Respect for the dangers of benzyloxy sulfonyl chloride makes the lab a place for discovery, not disaster.
What is the purity specification and available packaging size for Benzyloxy Sulfonyl Chloride?
The Real Story about Chemical Purity
Chemistry leaves little room for sloppy work. With substances like Benzyloxy Sulfonyl Chloride, my experience in the lab taught me that quality depends greatly on purity. Anything less than a high-purity product creates headaches—from unpredictable yields to spectral noise. Leading suppliers generally commit to purity levels at or above 98%. This purity is not simply a suggestion; it reflects rigorous batch testing using methods like HPLC and NMR, confirming that each order matches its certificate of analysis.
I’ve worked with batches both pure and questionable. Low-grade product adds mystery to synthesis, and that’s not the kind of suspense anyone wants. Impurities can trigger side reactions, eat up time, waste solvent, and drive up costs for everyone from pharma researchers to material scientists. A lot of us watch the fine print for that minimum 98% label before we start work.
Why Purity Specification Isn’t Just a Number
Reactive reagents demand close attention. Raw materials like Benzyloxy Sulfonyl Chloride find their place in specialized organic synthesis, sometimes as a protecting group or for sulfonation steps. An impurity percentage above 2 drags down efficiency. The market reflects this demand: leading catalogs and distributors publicize clear specifications, publishing details about water content, melting point, and impurity profiles so nobody gets an unpleasant surprise.
I once ordered a reagent from a lesser-known source. The purity read 95%. The outcome: a two-day cleanup exercise that ate up valuable grant money and put my research timeline in jeopardy. It didn’t take much convincing after that—my team stuck to wholesalers who posted transparent specs and batch-to-batch consistency.
The Practical Side of Packaging Size
In the real world, package size sets limits on lab planning. I’ve seen 5g and 25g sealed glass bottles as the common choices, with 100g containers available for industrial users. Smaller vials support one-off tests or early-stage exploration. Bulk buyers, such as pharmaceutical scale-up groups, lock in supplies in much larger containers, sometimes by the kilogram. Still, most shelf-stable packaging keeps to moisture-resistant glass, secured under an inert atmosphere since Benzyloxy Sulfonyl Chloride reacts aggressively with water.
Selecting the right size streamlines storage and reduces waste. Buying more than needed results in expensive leftovers, especially when shelf life depends on protecting the compound from humidity and decomposition. The cost-effective approach calls for knowing your process and estimating needs with a realistic eye; you don’t want to skimp and risk running out, but overbuying for a compound that degrades in air chips away at budgets.
Quality and Safety Go Hand in Hand
Chemicals with this kind of reactivity come with hazards. I’ve seen careless handling end in damaged PPE and hasty calls to the lab safety office. Packaging now often includes extra labeling and tamper-evident tops. Handling instructions arrive with the product: store in a cool, dry place, reseal tightly after each use, and avoid prolonged exposure to air. MSDS data matter more than ever—too many researchers learn the hard way that a splash or leak has immediate consequences.
For anyone seeking Benzyloxy Sulfonyl Chloride, remember that a clear understanding of purity specs and packaging sizes not only advances your work but also protects your team and the integrity of your results. Cutting corners on quality or choosing the wrong packaging quickly introduces more risk than most want to accept.
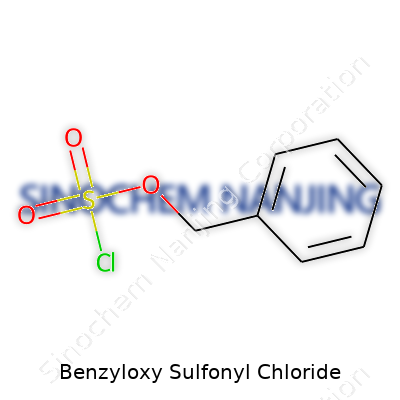
| Names | |
| Preferred IUPAC name | Benzyloxy(sulfonyl) chloride |
| Other names |
Benzyloxy sulfonic acid chloride Benzyloxybenzenesulfonyl chloride Benzylsulfonyloxy chloride |
| Pronunciation | /ˌbɛn.zɪˌlɒk.si ˌsʌlˈfəʊ.nɪl ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | [1623-24-3] |
| 3D model (JSmol) | `3D model (JSmol)` string for **Benzyloxy Sulfonyl Chloride**: ``` CC1=CC=CC=C1O[SH](=O)(=O)Cl ``` |
| Beilstein Reference | 3511044 |
| ChEBI | CHEBI:134209 |
| ChEMBL | CHEMBL4151617 |
| ChemSpider | 23974572 |
| DrugBank | DB08365 |
| ECHA InfoCard | 03dbe378-940f-484f-8bbd-e44ceec43392 |
| EC Number | 619-711-4 |
| Gmelin Reference | 5295 |
| KEGG | C19221 |
| MeSH | C009708 |
| PubChem CID | 15736706 |
| RTECS number | DM8575000 |
| UNII | BZV5XQ067H |
| UN number | UN3261 |
| CompTox Dashboard (EPA) | DTXSID20894854 |
| Properties | |
| Chemical formula | C7H7ClO3S |
| Molar mass | 234.68 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.35 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 0.2 mmHg (20 °C) |
| Acidity (pKa) | 7.1 |
| Basicity (pKb) | 7.63 |
| Magnetic susceptibility (χ) | -57.40·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5840 |
| Viscosity | 0.678 cP (20°C) |
| Dipole moment | 3.6352 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 385.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| Main hazards | Corrosive, causes burns, harmful if inhaled, toxic by ingestion, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H334 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P310 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 179 °C |
| Lethal dose or concentration | LD50 (oral, rat): 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 640 mg/kg |
| NIOSH | BZ4825000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg |
| IDLH (Immediate danger) | IDLH not listed |
| Related compounds | |
| Related compounds |
Benzyl Chloride Benzenesulfonyl Chloride Benzyl Alcohol Benzyloxy Sulfonic Acid Tosyl Chloride |

