Benzyl Chloroformate: More Than Just a Reagent
Historical Development: Following the Science Down the Years
Benzyl chloroformate didn’t just pop onto the scene overnight. The journey traces back to advances in organic chemistry during the late nineteenth and early twentieth centuries, when chemists started understanding and experimenting with carbamate chemistry. The search for robust protecting groups in peptide synthesis turned the spotlight on benzyl chloroformate. Once researchers figured out that this compound could shelter the amino function in building proteins, things really started to accelerate. Its recognition as the 'Cbz' protecting group dates back to the earliest days of solid-phase synthesis, setting the stage for modern pharmaceutical and peptide work. This was a time when benchtop syntheses often filled the laboratory with pungent fumes, and practical, reliable reagents like this changed the way chemistry could be done at scale.
What is Benzyl Chloroformate?
Commercially, benzyl chloroformate carries a few different names—sometimes you hear 'Z-Cl', 'CBZ-Cl', or even 'Phenylmethyl chloroformate.' All those names point to the same clear, slightly yellow liquid. This molecule packs both the reactivity of an acid chloride and the stability offered by the benzyl group. It’s not just a bottle in a storeroom. I’ve seen scientists depend on it while developing peptide chains, linking up small molecules, or introducing temporary protection to functional groups, especially in medicinal chemistry and peptide production.
Physical and Chemical Properties: More Than Meets the Eye
Anyone who’s handled benzyl chloroformate remembers the distinct, pungent odor—a warning in itself. The liquid boils a bit north of 200 degrees Celsius at atmospheric pressure. Its density hovers close to that of water, but its behavior could not be more different. Benzyl chloroformate decomposes with water, producing benzyl alcohol, carbon dioxide, and the infamous hydrogen chloride gas, which reinforces just how cautiously it should be treated. Its chemistry thrives in nonpolar organic solvents, and it steers clear of water at all costs if one wants to preserve the molecule for use.
Technical Specifications and Labeling: Standards that Matter
Labeling is more than a regulatory box-check. Understanding a bottle labeled with the UN number for toxic organochlorides or the signal word ‘Danger’—not just ‘Warning’—signals the need for chemical fume hoods, eye protection, and PPE. Good manufacturing practice is not academic detail; these standards make a difference when it comes to personal safety. Purity often lands somewhere above 98% in reputable labs. If a scientist has ever ended up with cloudy product after a peptide coupling, it’s not hard to trace the root cause to subpar material or improper handling.
Synthesizing Benzyl Chloroformate: Getting Down to Chemistry
Synthesizing benzyl chloroformate doesn’t take exotic materials. The classic route involves reacting benzyl alcohol with phosgene, usually in an inert solvent and with careful cooling because phosgene reacts violently and unpleasantly. These days, safer phosgene substitutes see more use, but the fundamentals haven’t shifted much. The real test, though, lies in meticulous temperature control, ventilation, and patience. Poor separation or trying to shortcut purification steps leads to byproducts and contaminants. That’s a lesson learned from real mistakes in real laboratories.
Reactions and Modifications: The Workhorse of Protection
Benzyl chloroformate’s claim to fame grew out of protecting amino groups. In peptide chemistry, it’s a workhorse for introducing the 'Cbz' group—reacting with amines to tuck away the reactive hydrogen and stop unwanted side reactions. Down the line, hydrogenolysis brings everything back, removing the Cbz group with gentle catalytic hydrogenation. Beyond peptides, chemists use it for making carbamates, tweaking polymers, or stabilizing reactive sites in custom molecules. Each one of these transformations adds layers to the drug development and material science industries—fields that touch lives in hospitals and factories far from the lab bench.
Alternate Names: Benzyl Chloroformate’s Many Hats
The world of synonyms in chemistry can be a headache for the newcomer—benzyl chloroformate, benzyl carbonochloridate, and Cbz-Cl all land you at the same place. Early mistakes taught many that searching via ‘phenylmethyl chloroformate’ in a catalog turns up the same compound. This duality of naming isn’t trivial; regulatory databases and supply chains need precision. Grainy old literature sometimes refers to 'Z-protecting group' work, but CBZ dominates modern papers.
Safety Practices and Operational Standards: Staying Safe with a Potent Reagent
If you’ve ever had a whiff of the vapors from benzyl chloroformate, it becomes obvious how important safety really is. The compound irritates skin, eyes, and mucous membranes; the fumes should never be underestimated. Lab safety culture stresses working in a chemical fume hood, wearing splash goggles, gloves, and using proper waste disposal—never pouring down the drain. Even minute contact, in my experience, brings sharp discomfort and motivates a commitment to PPE. Regulatory bodies such as OSHA and the European Chemicals Agency agree: this isn’t a kitchen-sink chemical.
Application Areas: From the Lab to Life
The reach of benzyl chloroformate stretches well beyond academic chemistry. Pharmaceutical companies depend on it in peptide synthesis, especially for active pharmaceutical ingredients used to treat disease. Agricultural chemistry, custom dye synthesis, and some flavors and fragrance work all rely on temporary protection groups like Cbz. Its ability to create carbamate esters gives agricultural researchers routes into new crop protectants, while industrial labs appreciate its reliable reactivity. Every tablet, every diagnostic peptide, tells a small part of this story.
Research Trends: Keeping Up with the Science
Recent years brought a wave of greener synthesis routes. The questions that keep researchers up at night include: Can we do without phosgene and its relatives? Can we swap out large-volume solvents for something less harmful? Improvements in catalytic hydrogenation now allow deprotection with less waste. Modifications of Cbz chemistry, like introducing tailored benzyl derivatives or exploring non-traditional solvents, keep the literature lively. Peptide development for novel therapies still leans heavily on Cbz, showing the old method keeps pace with evolving pharmaceutical needs.
Toxicity: Risks that Need Respect
Benzyl chloroformate doesn’t shy away from risk. It’s classified as toxic, and exposure can irritate lungs, skin, and eyes, leading to serious symptoms. Inhalation leads to coughing, tightness of chest, and in high exposures, even pulmonary edema. Chronic exposure hasn’t been studied as deeply as some other industrial chemicals, but European authorities have flagged possible mutagenic or carcinogenic risks. Real-world accidents keep reinforcing the hazards—quick response, proper engineering controls, and rigorous training save more than just time in regulatory compliance.
The Road Ahead: Prospects for Benzyl Chloroformate
Benzyl chloroformate may be a staple, but it isn’t finished evolving. The appetite for safer, more sustainable syntheses grows every year. Researchers keep searching for alternatives to phosgene-based manufacturing. In peptide chemistry, greener reagents and solvent-free methods are gathering momentum. The enduring utility of the Cbz group keeps the door open for new coupling reagents and milder deprotection strategies. If we take my own experience as a guide, benzyl chloroformate will keep adapting—just as the demands of medicine, materials science, and green chemistry keep changing. Progress often grows out of adapting reliable tools for a changing world, and this old stand-by continues to earn its shelf space.
What is Benzyl Chloroformate used for?
What Makes Benzyl Chloroformate Stand Out
Growing up with a pharmacist in the family, I learned early on that chemistry shapes nearly everything we use daily, from medicines to cleaning agents. Benzyl chloroformate ranks among the go-to reagents for organic chemists. Its sharp smell is memorable, and its usefulness even more so. Laboratories and industries reach for it not because it’s flashy, but because it gets the job done—consistently.
The Backbone of Peptide Synthesis
Anyone who’s tried building peptides in a lab knows that protecting amino groups is a constant headache. Benzyl chloroformate brings real relief here as a carbobenzoxy (Cbz, Z) protecting group. Chemists attach it to amino acids, shielding their reactive parts from side reactions. Later, this group comes off cleanly when needed, leaving everything else untouched. This step saves weeks of troubleshooting and reduces wasted materials, which directly impacts research speed and cost.
Pharmaceuticals, Not Just Theory
Nearly every modern drug, especially those built from delicate building blocks, owes something to processes using carbobenzoxy groups. Many antibiotics, antiviral agents, and even some specialized cancer drugs depend on benzyl chloroformate in their development. Pfizer, Merck, and smaller startups keep this compound in stockrooms because it makes drug development less prone to failure.
Making the Chemistry Safer
Safety comes up fast with benzyl chloroformate. It’s reactive and toxic. I remember the first time I helped open a bottle in the lab—the gloves, goggles, and strong ventilation mattered more than anything else. There have been cases where mishandling brought on burns and breathing issues, so anyone working with it must train and respect the risks. Industry regulations track every shipment closely to lower the odds of leaks.
Protecting More Than Amino Acids
Outside pharmaceuticals, benzyl chloroformate pops up in the synthesis of flavors, dyes, catalysts, and specialty chemicals. Researchers fine-tune battery components or even molecular sensors with its help. In all these cases, the idea stays the same—use it to temporarily block a chemical group, do the main transformation, then gently peel the protection back.
Sustainability and Alternatives
Some critics point out that the process produces waste and sometimes releases harsh byproducts. Adoption of greener chemistry practices is growing, such as recycling solvents and switching to cleaner cleavage methods. Academic labs and industrial giants now run pilot projects focusing on alternatives like Boc (tert-butyloxycarbonyl), which can sometimes provide similar protection with different safety and environmental trade-offs. Still, for now, benzyl chloroformate keeps its place because of reliability and proven track record.
Room for Improvement
There's a real push to create new molecules that deliver protection with even fewer hazards and less cleanup. Grants and research dollars now chase these goals. Open discussion among chemists, regulatory authorities, and safety experts could spur breakthroughs that keep essential chemistry moving forward, without all the baggage. Experience in the lab shows that the safest path comes from combining practical know-how, up-to-date safety guides, and honest evaluation of every new substitute.
What is the chemical formula and structure of Benzyl Chloroformate?
Chemical Formula and Structure
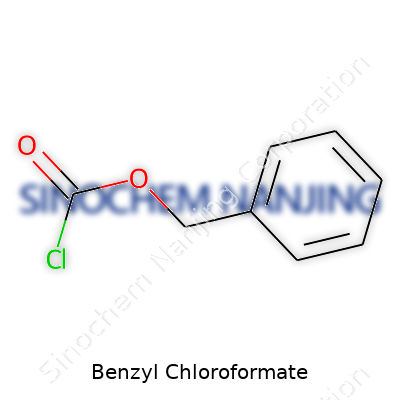
Benzyl chloroformate goes by the formula C8H7ClO2. You’ll see its structure drawn as a benzyl group (a benzene ring with a CH2 linker) attached to a carbonyl group, which itself connects to a chlorine atom. To describe it simply, imagine a benzene ring, then a CH2 group, then an O-C(=O)-Cl portion. Chemists shorthand this as C6H5CH2OCOCl. In the lab, it looks like a clear, colorless liquid, but its smell—a sharp, almost choking odor—makes its presence felt right away.
Why Benzyl Chloroformate Matters
I remember prepping for a peptide synthesis project and making my way through a haze of protection group chemistry. Benzyl chloroformate, also called "Z-Cl" or "Cbz-Cl," came up more than once. It’s a classic "blocking agent" for amine groups on amino acids. Protecting functional groups like this keeps reactions cleaner and reduces unwanted side products. Students see this molecule on reaction schemes just as often as Bunsen burners in the lab.
Industry’s reliance on benzyl chloroformate is hard to ignore, since it helps produce pharmaceuticals, agrochemicals, and specialty chemicals. Introducing the carbobenzyloxy (Cbz) group preserves the core structure of biologically active compounds when certain modifications need to take place. Researchers in drug development have used benzyl chloroformate to protect amino acids during the multi-step process of building complex peptides, which might end up as new antibiotics or promising cancer treatments.
Handling and Safety
Benzyl chloroformate doesn’t play nice in the air: it reacts easily with water, releasing hydrochloric acid. During graduate studies, I learned that even a few drops spilled on a lab bench turn the air acrid and hard to breathe. Labs require fume hoods and proper gloves because skin contact burns and eye exposure means a quick trip to emergency eyewash stations. Accident reports tell the same story again and again: respect the chemical, use it only with protective equipment, and store it in tightly sealed, dry containers.
Potential Concerns and Safer Practices
Chemical waste disposal has challenged labs using benzyl chloroformate. You don’t just pour leftovers down the drain—waste must go into specially labeled containers for later neutralization and processed under strict environmental controls. Some chemists have started looking for greener or safer alternatives. Companies encourage the use of automated dispensers, so fewer hands touch pure benzyl chloroformate. Some research points to solid-supported or less volatile chloroformate reagents, which cut down on fumes and splashes.
Solutions for a Safer Laboratory
Better engineering controls—high-powered hoods, local exhaust—are now standard. Digital inventory management helps spot leaks early, since sensors raise alerts long before the nose picks up trouble. High school and college labs, guided by new chemical safety standards, keep benzyl chloroformate use limited to trained professionals. Shared experience makes one fact stand out: every bottle carries risk, but teaching respect for the chemistry leads to safer scientists and more reliable experiments.
Final Thoughts
Benzyl chloroformate’s C8H7ClO2 structure isn’t what draws people in—it’s how this molecule fits into the bigger story of discovery and scientific care. Each generation of chemists learns safety and precision, treating this useful reagent both as a tool and a reminder of the need for respect in every experiment.
What are the safety and handling precautions for Benzyl Chloroformate?
Understanding the Risks
Benzyl chloroformate pops up in chemical synthesis, especially when making protecting groups during pharmaceutical manufacturing. In a university lab, I’ve watched chemists approach this clear, sharp-smelling liquid with a sense of wariness. The chemical can cause eye, skin, and respiratory irritation—one accidental whiff while transferring it, and you remember to double-check the fume hood every time. My colleague learned quickly after a pinhole in his gloves turned a routine cleanup into an urgent trip to the sink; redness and tenderness showed up almost instantly.
Volatility brings real challenges. Benzyl chloroformate releases hydrochloric acid and carbon dioxide during hydrolysis. That means any spill or exposure to water can cause a small cloud of corrosive gas. For folks in labs or production plants, this isn’t a distant concern; cases of chemical burns and respiratory issues are backed up in safety training. Most chemical suppliers distribute it with warnings in bold letters, not just fine print. So respect for this chemical grows through experience, not just through reading safety data sheets.
Practical Steps For Safe Handling
Folks who work with benzyl chloroformate get trained to use fume hoods, chemical splash goggles, and double-layered nitrile gloves. Cotton lab coats don’t cut it—the chemical will chew through both fabric and skin with enough exposure. My own lab keeps calcium carbonate or sodium bicarbonate handy, just for neutralizing small spills quickly. Anyone who has tried to clean up a leak without proper absorbent knows the stinging in your nose and eyes lingers unless you manage the chemicals properly.
Handling doesn’t stop at mixing and reaction. Disposal and cleanup can trip up even experienced technicians. Pouring leftover benzyl chloroformate directly into a sink causes dangerous fumes. Instead, sealed waste containers lined with absorbent material get used. My team always checks containers for leaks, as we’ve faced incidents where improper sealing resulted in overnight gas buildup—corrosive enough to pit metal waste bins.
Preparedness Matters
It’s easy to feel immune with safety gear on, but even small mistakes hurt. One memorable incident involved a researcher ignoring a split in a glove “just for this one transfer.” They ended up with a chemical burn. This teaches the lesson: don’t shortcut safety checks. Always test fume hoods, inspect gloves, wear goggles, button up coats, and keep neutralizers ready. For anyone new, running a mock transfer with water builds confidence and muscle memory before real chemicals come out.
Changing Culture and Accountability
Facilities that handle benzyl chloroformate can’t leave safety up to individual memory. Routine safety audits, real-time air monitors, and mandatory spill drills turn good habits into team culture. My workplace started monthly review sessions where everyone, even senior staff, share near-miss stories. Instead of singling people out, the group discusses solutions—like improved labeling or changing glove supplier. I’ve seen that kind of open dialogue not only prevent injuries, but also cut down the number of accidental releases by half in a single year.
Benzyl chloroformate demands respect, not just compliance. Treat it casually and sooner or later, you or your colleagues end up hurt. Shared stories, visible safety gear, and fast response resources don’t just meet regulatory checkboxes—they let researchers focus on innovation instead of injury recovery or cleanup.
How should Benzyl Chloroformate be stored?
Getting Real About Storage Risks
Benzyl chloroformate doesn’t tolerate mistakes. Just a few drops can send strong fumes through a room. My first hands-on experience with it involved more nerves than usual, even in a lab. The smell triggered a strong warning in my mind: This stuff means business. Over time, it’s become clear that treating benzyl chloroformate like an everyday lab chemical carries real consequences. Reports from chemical safety boards outline burns, eye injuries, and even fires when it gets mishandled. That's no exaggeration. According to the CDC, even brief exposure can set off coughing, eye pain, and more if ventilation falls short or spills go unchecked.
Conditions That Make a Difference
Use a cool, dry place — not the regular shelf where solvents sit. Benzyl chloroformate breaks down with moisture or warmth, and that breakdown doesn’t just make a chemical weird, it makes it dangerous. Unsafe storage sometimes means corrosive hydrogen chloride floating unchecked through storage areas. Even a small leak can trigger lung-wrenching reactions. Bear in mind a locked, ventilated chemical cabinet works better than any makeshift arrangement.
Materials Matter
Store it in the container it arrived in. Glass holds up better than most plastics. Metal containers spell trouble, especially when the metal reacts with the compound, speeding up decomposition and releasing gases that nobody wants to inhale. Tight-fitting lids keep both evaporation and accidental moisture at bay. One time, I watched a loose cap ruin a whole batch in a matter of hours. Replacing a container costs far less than handling an accident.
What Goes Wrong Without Care
OSHA records show that improper storage leads to more spills than actual use in experiments. The patterns show up everywhere—someone heads home early, someone else misreads the label, a container ends up in sunlight. Heat and light speed up breakdown, which means more fumes and more risk. Anyone in charge of chemical safety checks the thermometer and reviews the placement. Labels fade quickly, so updating them makes practical sense.
Response Plans Save More Than Paperwork
Having a nearby eyewash station isn’t just compliance; it’s about walking away from accidents with eyesight intact. Good labs keep chemical spill kits close. Teams drill regularly, so firefighters or paramedics don’t handle surprises. Training cuts down the odds of panic when a jar falls or a spill spreads across the bench top.
A Safer Approach, Backed by Experience
Many overlook simple details, like double-checking cabinet ventilation or inspecting seals. I’ve seen how easy distractions lead to close calls—busy labs mean people rush. But storing hazardous chemicals like benzyl chloroformate in the right spot, with the right people trained, and the right equipment within arm’s reach, creates a work culture that limits harm. Safety isn’t about fear; it’s about lasting respect for chemicals that keep the world moving but never let their guard down.
What are the common applications of Benzyl Chloroformate in organic synthesis?
The Workhorse Behind Amino Acid Protection
Benzyl chloroformate has a way of popping up in many organic chemistry labs, and for good reason: it serves as a trusted protecting group for amines, especially when working with amino acids. Researchers looking to build peptides often reach for benzyl chloroformate to create carbobenzoxy (Cbz or Z) groups, giving them control over which end of the molecule will react next. Anyone who’s tried hand at peptide synthesis knows that unexpected side reactions can ruin a whole week’s work. With benzyl chloroformate, you can block trouble before it starts. This approach makes it possible to assemble complex peptides, one building block at a time, and later remove the Cbz group without damaging those delicate peptide bonds.
Building Blocks for Pharmaceuticals
Benzyl chloroformate finds its way into pharmaceutical labs for more than just peptides. Medicinal chemists rely on it to protect nitrogen atoms during the journey from simple starting materials to valuable drug candidates. It opens the door for multi-step synthesis, helping keep parts of the molecule safe while chemists modify other regions. Many early-stage drug candidates would stall without such reliable protection. In my experience, cleaning up a synthetic route with benzyl chloroformate often saves on purification headaches down the road. Drug researchers tend to trust what’s proven by time and thorough testing, and this reagent consistently delivers in those areas.
Streamlining Preparation of Urethanes and Carbamates
Beyond amine protection, benzyl chloroformate plays a crucial role in making carbamates, urethanes, and other nitrogen-containing compounds. By introducing a benzyl group, chemists can create intermediates needed for plastics, agricultural chemicals, and dyes. These intermediates serve as platforms for even bigger transformations. Some specialty polymers and dyes only come together smoothly when benzyl chloroformate is involved, offering reliability that alternatives often lack. Working with this reagent provides a kind of assurance—especially for those who value straightforward troubleshooting in the lab.
Cleavage: Efficient Recovery Without Harm
One big advantage to the benzyl group: it comes off cleanly. Hydrogenation with a palladium catalyst snips away the Cbz group, freeing the amine without leaving behind scars on the main molecule. This predictable deprotection step means chemists can focus on their targets instead of cleaning up unexpected messes from harsh conditions. Anyone who’s run a large-scale reaction appreciates that kind of consistency, especially with sensitive compounds that would otherwise fall apart under strong acids or bases.
Why Care About Reliable Protecting Groups?
The market for pharmaceuticals and custom chemicals demands ever longer, more intricate molecules. With each extra step, risks multiply. Protection strategies anchored by benzyl chloroformate fight off unwanted side reactions while opening the door to creativity. Chemists have built trust in this molecule over decades. Extensive studies back up its safety profile, likely contributing to its regulatory acceptance. Factoring in accessibility, cost, and a solid literature base, it’s easy to see why researchers still lean on benzyl chloroformate in high-stakes synthesis. Careful sourcing and good ventilation, combined with standardized disposal, keep its use sustainable in both academic and industrial settings.
| Names | |
| Preferred IUPAC name | Benzyloxycarbonyl chloride |
| Other names |
Benzyloxycarbonyl chloride Z-chloride Chloroformic acid benzyl ester |
| Pronunciation | /ˈbɛn.zɪl klɔː.rəˌfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 501-53-1 |
| Beilstein Reference | Beilstein Reference: 1919227 |
| ChEBI | CHEBI:74734 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 14118 |
| DrugBank | DB14006 |
| ECHA InfoCard | 100.005.250 |
| EC Number | 205-638-2 |
| Gmelin Reference | Gmelin Reference: **83268** |
| KEGG | C00741 |
| MeSH | D001570 |
| PubChem CID | 24507 |
| RTECS number | CU5950000 |
| UNII | N8BX73007F |
| UN number | UN1738 |
| CompTox Dashboard (EPA) | DTXSID4044369 |
| Properties | |
| Chemical formula | C8H7ClO2 |
| Molar mass | 170.59 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | fruity |
| Density | 1.21 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.99 |
| Vapor pressure | 0.45 mmHg (20 °C) |
| Acidity (pKa) | 13.8 |
| Basicity (pKb) | Basicity (pKb): 13.2 |
| Magnetic susceptibility (χ) | -60.5e-6 cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 1.804 cP (20 °C) |
| Dipole moment | 3.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -261.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -687.8 kJ·mol−1 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H317: May cause an allergic skin reaction. H335: May cause respiratory irritation. |
| Precautionary statements | P280, P261, P301+P330+P331, P305+P351+P338, P308+P310, P404+P233 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | Flash point: 110 °C |
| Autoignition temperature | 380°C |
| Explosive limits | 3.3–11.4% |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 1880 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 825 mg/kg |
| NIOSH | CN8400000 |
| PEL (Permissible) | PEL: 0.2 ppm |
| REL (Recommended) | 50 μg/m³ |
| IDLH (Immediate danger) | IDLH: 20 ppm |
| Related compounds | |
| Related compounds |
Phenyl Chloroformate Methyl Chloroformate Ethyl Chloroformate Isobutyl Chloroformate tert-Butyl Chloroformate |

