Benzoyl Hydroperoxide: Past, Present, and Beyond
A Story Rooted in Chemistry
Looking back at the history of benzoyl hydroperoxide, it’s clear that its early journey got shaped by the industrial push for better polymerization methods and a search for safer, more convenient oxidizers. In the 20th century, as plastics like PVC and polystyrene hit markets worldwide, chemists realized traditional initiators fell short. Benzoyl hydroperoxide stepped in, offering high reactivity with a lower impact on the temperature sensitivity of processes. Its value comes from being a solid at room temperature, which makes storage and weighing much less of a headache compared with volatile liquid peroxides that posed more obvious hazards. This combination of safety and strong initiating power earned benzoyl hydroperoxide a fan base among manufacturers and researchers intent on transforming the way plastics and elastomers got made. The fact that it shows up today in fields ranging from dentistry to acne treatment illustrates how innovation often relies on repurposing chemical workhorses forged during the core years of industrial chemistry.
Product Overview Through Everyday Eyes
Staring at a tub of acne cream in a pharmacy or watching a laboratory technician pull a white, flaky powder from a shelf, someone unfamiliar with benzoyl hydroperoxide probably wouldn’t guess it’s the same compound. Yet, its roles are shaped by the trust users have built in its properties over time. Manufacturers focus attention on the stability and purity of this oxidizing agent, selling blends customized for polymerization or for medical use. Straight benzoyl hydroperoxide ranks high in reactivity, easily breaking down into radical initiators. While the raw compound stores its energy in a relatively stable form, it doesn’t take much encouragement — heat, friction, or certain impurities — for it to release that energy, which is why safety always rides shotgun wherever it’s used. The fact that both a dentist making custom mouth guards and a dermatologist treating acne would look at the same base ingredient speaks to its remarkable versatility and reliability across the decades.
Physical and Chemical Properties: Walking the Line Between Utility and Risk
Holding benzoyl hydroperoxide, you get a fine, white powder or sometimes slightly granular chunks. It melts at moderate temperatures, sitting unassumingly at room temperature, though behind this calm appearance lurks a readiness to decompose when triggered. Its chemical structure shows why. Built around a peroxide bond flanked by benzoyl groups, the compound offers an ideal launching point for free radicals — energetic fragments that drive polymerization and oxidation. That peroxide bond wants to split, and if mishandled, the split comes with rapid heat and the sort of pressure build-up that has shattered more than a few flasks across research labs. Benzoyl hydroperoxide isn’t soluble in water but finds a home in many organic solvents. That sort of selectivity drives its usefulness but feeds directly into storage concerns. Any chemist worth their salt learns quickly to keep it cool, dry, and away from accidental knocks or stray contamination.
Technical Standards: Bottling a Volatile Force
What sets reputable suppliers apart involves their control over product consistency and labeling accuracy. Pharmaceutical and industrial grades share similarities — low impurity levels, consistent particle size — but meet different standards. Labels carry vital warnings about explosive potential and instructions about ventilation and protective equipment. Details like recommended storage temperature and water content (many forms are dampened with a small percentage of water to cut down on sensitivity to shock) make the difference between safe use and disaster. Regulatory frameworks such as the GHS (Globally Harmonized System) force users to pay attention, with pictograms and hazard statements that can't be ignored without risk. The actual numerical purities and moisture contents merit respect, not because they read as technical jargon, but because behind every decimal sits years of accidents and hard lessons.
Making Benzoyl Hydroperoxide: Simplicity and Precision
Preparing benzoyl hydroperoxide might seem straightforward to those steeped in synthesis. Still, the chemistry behind it demands a careful hand. The most common route takes benzoyl chloride, reacting it with hydrogen peroxide in the presence of an alkali, often sodium hydroxide. The key lies in watching temperature and mixing rate. Go too quickly or miss a setpoint, and the process can spiral out of control, releasing gas, heat, or — worst-case — an unexpected detonation. The yield depends on careful addition, neutral pH management, and cleaning out byproducts (especially benzoic acid and unused reagents) through repeated washes. Industrially, engineers design closed reactors and remote handling systems to sidestep the risks that smaller labs face. For anyone preparing the compound, cleanliness, slow addition, and patience amount to the holy trinity of safe hydroperoxide work.
Modification and Chemical Reactions: Breadth of Possibility
Benzoyl hydroperoxide’s real claim to fame rides on its chemical reactivity. It opens the doorway to radical polymerization, turning simple monomers into the long chains that build plastics people use daily. By tweaking reaction times, amounts, and the nature of monomers, innovators have tailored materials for every conceivable application — from packaging to medical devices. The oxidizing power also gets borrowed for selective reactions in organic synthesis, where it brightens faded dyes, cleaves stubborn molecules, and tacks on oxygen atoms exactly where chemists want them. Modifications, such as blending with inert materials or stabilizing agents, can stretch the shelf life, reduce dusting, or limit the potential for shock or heat-triggered accidents. Those who build better peroxides through molecular tinkering chase safer, more controllable reactions, all while pushing the performance envelope for targeted industries.
Synonyms, Product Names, and the Maze of Nomenclature
In technical literature, benzoyl hydroperoxide doesn’t always show up under the same name. Depending on the context or the brand, you’ll spot synonyms like dibenzoyl peroxide or simply BPO. Trade names vary, shaped by applications — as an acne medication, a dental paste, or a composite initiator, the labeling reflects its intended field and purity level. Navigating this maze asks for careful attention, given that similar-sounding compounds (such as benzoyl peroxide, lacking the hydroperoxide functionality) play dramatically different roles. Clear labeling helps researchers and practitioners avoid cross-mixups that could prompt unexpected results or safety violations.
Raising the Bar on Safety and Operations
Working with benzoyl hydroperoxide draws a hard line between safe practice and recklessness. Safety standards grow from tragic experience: explosions from friction, static discharge, or accidental blending with metals or strong acids. Regulations from OSHA, the EPA, and international authorities focus on personal protective equipment, storage in cool, ventilated spaces, and restrictions on quantities kept in one place. Good technique means setting procedures that treat every ounce of material with respect, ensuring staff get regular training on handling spills or unexpected decomposition. Engineers design process equipment from the ground up with venting and emergency controls factored in. The cost of skipping these steps shows up in hospital logs and incident reports, not just lost product or equipment.
Diverse Application Areas
The reach of benzoyl hydroperoxide stretches across fields instead of just nestling in a chemistry storeroom. In manufacturing, it drives the polymerization of resins, the backbone of plastics and composites people depend on daily. In healthcare, it forms the front-line active in topicals for acne, using oxidative stress to knock out unwelcome bacteria and unplug pores. The dental industry finds it irreplaceable in curing acrylics and making custom prosthetics. Its power as a bleaching agent gives textiles a cleaner look, and as an initiator, it sets off chain reactions in adhesives that hold everything from cars to electronic gadgets together. Each field finds tradeoffs; medicine demands purity and tolerability in topical formulations, while polymerization plants eye triggers for safe handling on a kilogram scale. What ties it together is the need for safe, reliable initiation of controlled chemical change, no matter the surface trappings.
Research and Development Keeps Pushing
Every university or corporation chasing edge in material science or pharmaceuticals watches for ways to squeeze more utility, safety, or selectivity out of this old workhorse. Scientists go after better stabilizers, new co-initiators, and ways to package the powder or paste to blast through logistical hurdles. Clean-label formulations for consumer products drive cleaner, lower-residue blends. The drive to minimize environmental impact threads through patents and open literature, as green chemistry takes precedence and regulatory scrutiny gets tighter. The frontiers of photoinitiation and controlled-release blends mean benzoyl hydroperoxide still has legs in engineering tomorrow’s materials and treatments, even as upstarts arrive on the scene.
Toxicity and Where Risk Meets Reward
Handling benzoyl hydroperoxide responsibly means understanding both its immediate hazards and its long-term toxicity profile. Acute exposure can lead to skin irritation, burning sensations, and, with inhalation, respiratory distress. Environmental spills prove toxic to aquatic life, and long-term studies track its breakdown products for any carcinogenic or mutagenic effects. Regulators force close attention to waste disposal, limiting discharge to sewage and keeping close tabs on occupational exposure. Decades of clinical and industrial exposure have pushed product guidelines to err on the side of caution, especially in medical applications where trace residues can linger. The task for toxicologists and policymakers boils down to balancing real benefit with controllable risk, not bowing to alarmism but not dismissing red flags either.
Where the Road Leads Next
Benzoyl hydroperoxide continues to underpin many technical leaps across disciplines, yet its future rests on how researchers and industries adapt its strong points while dialing down dangers. Looking at next-generation initiators, greener manufacturing, and higher-purity consumer blends, I see a compound that acts as a sort of bellwether — reflecting broader chemical trends. Efforts at solvent-free processing, shelf-stable micropellet formulations, or reduced-residue acne treatments all trace back to a single goal: preserving function, upping safety, and lowering the health or environmental cost. The future will bring tighter regulations and deeper understanding of its behavior in diverse contexts. At the same time, the weight of experience with benzoyl hydroperoxide gives chemists a better map to steer new discoveries, using lessons learned in factories and labs to avoid past mistakes and look out for the next big breakthrough.
What is Benzoyl Hydroperoxide used for?
Why Benzoyl Hydroperoxide Lands in Medicine Cabinets
Ask anybody who has dealt with teenage breakouts, and they’ll probably recognize benzoyl hydroperoxide. Used in countless over-the-counter creams and gels, this ingredient doesn’t just fight surface bacteria—it goes deeper. In my high school years, acne hit hard and nothing seemed to help until a doctor recommended a face wash with this stuff. The swelling, the redness, the painful cysts all started to fade. That change wasn’t instant, but it stuck with me: a science-backed solution can help even when hope starts to thin.
What sets benzoyl hydroperoxide apart is its ability to break down and release oxygen into skin pores. Bacteria like Cutibacterium acnes hate oxygen, so the chemical creates an environment where breakouts struggle to survive. There’s no single trick that beats acne, but studies published in the Journal of Clinical and Aesthetic Dermatology show benzoyl hydroperoxide cuts pimple count by up to 60% in just twelve weeks.
Benzoyl Hydroperoxide’s Day Job in Industry
Surprising as it sounds, this stuff isn’t just hanging out in bathroom cabinets. Factories use benzoyl hydroperoxide to kick-start chemical reactions during plastic production. It turns raw materials into solid, durable goods—a service that touches construction, furniture, and even the makers of everyday utensils. I remember working in a small workshop where we repaired fiberglass boats. Without benzoyl hydroperoxide, resins refused to harden, leaving us with sticky, unusable surfaces. It’s a real-world solution to a practical problem.
The chemical’s dual purpose highlights a trend I’ve witnessed: everyday products borrow heavily from industrial-stage science. Safe handling calls for respect. In clinics, dermatologists recommend small amounts and highlight risks, like skin irritation. At the repair shop, we needed gloves and good airflow to avoid rashes and headaches. Safety isn’t a side note—it’s part of using strong chemistry responsibly.
Risks, Rewards, and Smart Use
Most people see the “benzoyl” on a cream and worry about side effects, and there’s reason for that. Applied too often or in high concentrations, benzoyl hydroperoxide can leave skin flaky and raw. A review from the American Academy of Dermatology flags this: irritation follows overuse. Switching to low strengths or applying a moisturizer can cut down on peeling. My own experience tells me patience works wonders—start with less, then build up as the skin adapts.
Out in the lab or on shop floors, the stakes rise. Mishandling large batches can spark fires or explosions. Regulations require storage in cool, dry places and careful handling, not because of red tape, but because real accidents happen. OSHA tracks industrial incidents involving poorly stored chemicals, so there’s a track record for why guidelines aren’t just suggestions.
Lowering risk takes a smart approach: choose trusted brands for skin care, stick with instructions, and don’t mix with other strong exfoliants. In workplaces, ongoing training helps teams remember safety steps. These solutions let the benefits shine—clearing up acne and powering industry—without the downsides that come from rushing or cutting corners.
Benzoyl hydroperoxide has offered relief in both my personal routine and my time on shop floors. Its versatility is impressive, but respect is key. Benefit comes through careful use, backed by research and routine experience.
How should I apply Benzoyl Hydroperoxide?
Understanding What You're Working With
Benzoyl hydroperoxide gets a lot of attention as a treatment for acne. You’ll spot it as an ingredient in creams, gels, and washes at almost any drugstore. I remember sneaking into my older brother’s medicine cabinet for his white tube of acne gel, hoping for a quick fix. Instead, those first weeks came with both frustration and curiosity—some redness, dry patches, and a new appreciation for patience.
The Importance of Preparation
Straightforward habits lead to better results. Start by washing your hands and face gently with lukewarm water and a mild cleanser. Over the years, I’ve learned the hard way that scrubbing only makes matters worse—irritation ramps up, and breakouts can drag on. Pat your skin dry, never rub. Giving the skin a clean, dry surface sets up benzoyl hydroperoxide to work properly.
How to Apply Without Overdoing It
Dermatologists agree—using more product doesn’t mean faster clearing. Squeeze out a small, pea-sized amount for the entire face if you’re spot treating or spreading it thinly over breakout-prone zones. Too much will just strip the skin and create peeling. My own first attempts, dabbing thick dots around my chin, taught me that moderation saves you a lot of discomfort.
Apply the treatment only to affected areas. If you’re new to benzoyl hydroperoxide, try once daily application in the evening. Everyone’s skin needs an introduction phase to adjust since stinging or dryness can develop early on. I stuck with every other night at first, slowly building up as my skin toughened up. If all goes well after a week or two, you can bump it up to twice a day.
Spotting and Managing Side Effects
The product works by killing acne-causing bacteria and loosening build-up in pores, but it can irritate. Common issues include dryness, redness, peeling, or mild itching. Having lived through a few peeling cycles myself, I’ve learned to use a fragrance-free moisturizer daily and stick with gentle non-alcohol-based toners if at all. Avoid layering other strong topical treatments, especially retinoids or acids, unless cleared with a healthcare provider.
If you notice burning, swelling, or intense redness, scale back or pause use. Persistent reactions may signal an allergy—see a doctor for advice. Keep the product away from sensitive spots like eyes and mouth. Some of my old bedsheets fell victim to bleach stains, another reason to let it soak in fully before bedtime and use a white pillowcase if needed.
Tips That Make a Difference
Always wear sunscreen in the daytime. Benzoyl hydroperoxide can make skin more sensitive to the sun, raising the risk of burns and lasting marks. Foundations with SPF or mineral-based powders do double duty when days get busy. Stubborn acne might not vanish overnight, but sticking with a consistent routine for at least four to six weeks gives the best shot at seeing real improvement. If nothing budges, a dermatologist can offer stronger solutions.
Benzoyl hydroperoxide isn’t magic, though it often feels like a lifeline during a stubborn breakout. Using it thoughtfully keeps side effects down and results visible, helping skin recover without wrecking confidence along the way.
What are the side effects of Benzoyl Hydroperoxide?
Redness and Dryness: The Everyday Struggle
Anyone who has faced breakouts knows that reaching for products like benzoyl hydroperoxide feels like taking action against stubborn acne. The flipside, though, might seem relentless: bright red patches, peeling skin, and that tight, dry feeling. These side effects aren’t rare. Redness pops up fast on sensitive faces, and for many, the dryness rivals the original problem. When I first tried this ingredient as a teenager, the flaking around my nose drove me to wear less makeup, not more.
Itchiness and Burning: Hard to Ignore
Itchy, burning skin often follows after using benzoyl hydroperoxide, especially in high doses or on already-irritated skin. The sensation pushes you to scratch, which only adds to the irritation or leaves small wounds prone to infection. It isn't surprising that studies like those in the Journal of the American Academy of Dermatology point to irritation as a leading reason people give up on this kind of acne care.
Bleaching Stains and Unexpected Surprises
Benzoyl hydroperoxide doesn’t just stick to skin. It sneaks onto towels, pillowcases, and collars, leaving faded spots in its wake. Plenty of people, myself included, have ruined a shirt or two before realizing that skincare can double as a laundry challenge. Manufacturers warn about it, but the surprise never hits home until a favorite t-shirt turns blotchy.
Allergic Reactions: More than a Little Irritation
Though less common, allergic responses can knock someone off their feet. Swelling, severe redness, blisters—these need quick attention. Patch testing on a small skin area before regular use could spare someone a rush to the pharmacy. Dermatologists point out that benzoyl hydroperoxide breaks down into benzoic acid, which triggers reactions in people with certain allergies. Stories about folks giving up on acne treatments altogether stem from these more severe run-ins.
Turning Side Effects Around
Side effects don’t mean people need to swear off benzoyl hydroperoxide forever. Dermatologists often recommend starting slow: a thin layer, low concentration, and no doubling up with other strong ingredients like retinoids. Taking breaks, using a mild moisturizer, and looking for gentle cleansers can take the edge off the harshness. Some find overnight spot-treating works better than smearing the ingredient all over. In my experience, skipping it one day out of three trimmed the dryness without sacrificing the acne-fighting benefits.
Open Dialogue Matters
People often turn to the internet for answers when their skin rebels. Dermatologists and pharmacists offer trustworthy advice grounded in years of treating these flare-ups. Building a trusting relationship with a professional can turn a frustrating trial-and-error process into a clear plan that delivers results. Real-world stories add perspective, but science builds the foundation for working through these side effects.
Care that Goes Beyond Products
Managing acne isn’t only about the strongest products on the shelf. Diet, stress, sleep, and gentle skincare habits all play a role. For some, swapping pillowcases more frequently or tweaking a morning routine matters as much as switching creams. Respecting skin’s signals and giving it a break from excess products can calm redness and itching over time. That’s often the missing piece that helps healing stick.
Can Benzoyl Hydroperoxide be used with other acne treatments?
Understanding Benzoyl Hydroperoxide’s Place on the Bathroom Shelf
Ask anyone who’s stood in the skin care aisle, and you’ll probably hear a story about benzoyl hydroperoxide. It’s almost a rite of passage: the fresh sting, the flaky skin, the memories of hoping overnight that breakouts would fade by morning. This ingredient punches hard against acne, reducing bacteria and clearing up pores. But treatments don’t always work on their own, which leads people to experiment and combine products, hoping for faster, better results.
It's Tempting to Layer Solutions, But the Risks Are Real
There’s a natural urge to speed up the process. Spot cream here, foaming cleanser there, maybe add a little retinoid. It can seem harmless; each product comes with its own promises. Dermatologists see the mix-and-match adventure play out a lot—young people, adults, everyone searching for an answer. But there’s a catch. Many prescription or over-the-counter options target acne through different paths. Benzoyl hydroperoxide attacks surface bacteria and helps with inflammation. Retinoids, like adapalene, adjust cell turnover. Salicylic acid clears dead skin out of pores. Layering them gets tricky since they each pack a punch in their own way.
Stacking such ingredients without guidance leads to burning, peeling, and redness. Research from the American Academy of Dermatology highlights that rash decisions with combinations often result in irritation strong enough to push people away from treating their acne—making the problem worse in the long run. A survey of acne patients showed about 50% stopped medication early because of side effects, many tied to over-mixing treatments.
The Case for Doctor Guidance
In a world full of influencers and internet tips, the urge to experiment stays strong. Nothing beats advice from a skin doctor. A board-certified dermatologist brings experience and real understanding. They’ve seen the angry reactions on cheeks and the raw patches on foreheads. They know that using benzoyl hydroperoxide and adapalene in one routine can sometimes work, particularly when introduced slowly, a few days apart. Often, doctors suggest starting with one medication for a few weeks before easing another into the routine, watching for irritation each time.
Simple Steps Make All the Difference
Even with guidance, patience wins over speed. Those who go slow with adding products tend to stick with their routine—and see better results. Some people tolerate benzoyl hydroperoxide well in the morning, while a retinoid at night does more good than harm. Others rely on gentle cleansers and moisturizers to buffer their skin and keep the barrier healthy. Trying everything at once almost always backfires.
My own run-ins with aggressive combinations taught me that more medicine isn’t always better. Peeling skin made me self-conscious, sometimes even worse than the original pimples. Swapping stories with others, I hear similar regrets. Simple routines, tweaked with a professional’s help, lead to fewer side effects and better progress.
Looking Ahead: Education Over Guesswork
As more people have access to dermatologists online, opportunities to get safe, individualized plans increase. Schools, social media, and primary care offices can do better by sharing clear information about mixing acne treatments. Instead of anxious guesswork, everyone stands to benefit from honesty about what these powerful ingredients can—and cannot—do together.
Is Benzoyl Hydroperoxide safe for sensitive skin?
A Closer Look at the Ingredient
Benzoyl hydroperoxide turns up on many shelves, promising hope for anyone tired of stubborn breakouts. Dermatologists often recommend it for tackling acne, with its main claim to fame being how it kills acne-causing bacteria and unclogs pores. Yet, if you’ve got sensitive skin, the first time using it can feel like stepping through a minefield. Tingling and redness don’t always signal that it’s working—sometimes, those sensations mean your skin’s struggling to keep up.
What Sensitive Skin Folks Experience
My teenage years brought plenty of deals with swollen pimples, and I bought a benzoyl product following advice from a pharmacist. The first application left my cheeks stinging and peeling. Turns out, people with sensitive skin often notice burning, dryness, or even flaking after using something strong like benzoyl hydroperoxide. These side effects aren’t rare—studies published by the American Academy of Dermatology show a lot of folks quitting these treatments because irritation just gets too tough to handle.
Dermatologists suggest patch testing before diving in. Rubbing a pea-size dab onto your jawline and waiting a day can help flag problems early. If redness or swelling sets in, that’s your cue to ease up or skip it entirely. Some manufacturers offer lower concentrations, such as 2.5% instead of 10%, which might help reduce irritation.
The Science and Balancing Benefits With Risks
Benzoyl hydroperoxide’s power stems from its ability to release oxygen in pores, making the environment tricky for the bacteria Propionibacterium acnes. But it can rough up the protective layer that keeps moisture inside your skin. Without that barrier in good shape, sensitive skin just gets angrier.
Published research from the Journal of Clinical and Aesthetic Dermatology points out that irritation risk increases with both the concentration and frequency of application. Using stronger formulas often leads to more complaints about dryness and peeling, especially in those with delicate skin types. Moisturizers play a big role in helping manage these effects, but even then, some people still struggle.
Alternatives and Smarter Use
Dr. Caroline Robinson, a board-certified dermatologist, encourages starting slow and choosing the right product type. Washes and leave-on creams work differently; washes stay on the skin for less time, which might lessen side effects. Pairing benzoyl hydroperoxide with a gentle, fragrance-free moisturizer can help buffer its harsher impact.
People who keep reacting might look to alternatives. Ingredients like azelaic acid or niacinamide offer antibacterial and anti-inflammatory action without quite the punch of benzoyl. Salicylic acid, another over-the-counter option, sometimes feels gentler since it softens keratin and helps slough dead cells. No ingredient comes with zero risk, though, so trial and error often tells the true story of what your skin can handle.
Small Steps Toward Clearer Skin
Many people chasing clear skin feel tempted to throw the strongest stuff they can find at their breakouts. Sensitive skin demands patience and careful observation. Trusting the expertise of board-certified dermatologists and sticking to evidence-backed routines can protect your skin and your confidence. Ease in, pay attention, and don’t give up if benzoyl hydroperoxide isn’t your match—there are plenty of options for finding relief without all the fallout.
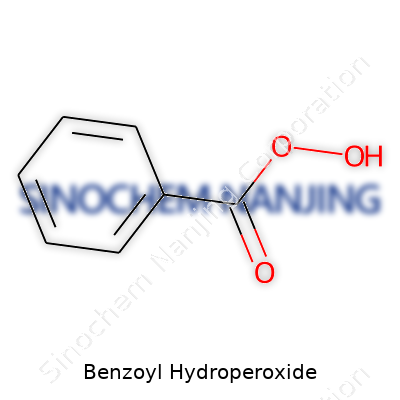
| Names | |
| Preferred IUPAC name | Benzenecarboperoxoic acid |
| Other names |
Benzoyl peroxide Peroxybenzoic acid Benzoperoxide Benzoyl superoxide Benzoic peroxide |
| Pronunciation | /ˈbɛn.zɔɪl haɪˌdrɒk.sɪˈpɜː.rɒksaɪd/ |
| Identifiers | |
| CAS Number | 94-36-0 |
| Beilstein Reference | 511873 |
| ChEBI | CHEBI:22902 |
| ChEMBL | CHEMBL1437 |
| ChemSpider | 7286 |
| DrugBank | DB14121 |
| ECHA InfoCard | 100.111.604 |
| EC Number | 202-327-6 |
| Gmelin Reference | Gmelin Reference: 132821 |
| KEGG | C14043 |
| MeSH | D001556 |
| PubChem CID | 7021 |
| RTECS number | DA8050000 |
| UNII | 5V5C8VB598 |
| UN number | UN3108 |
| Properties | |
| Chemical formula | C7H6O3 |
| Molar mass | 122.12 g/mol |
| Appearance | White, granular solid or crystalline powder |
| Odor | Acrid odor |
| Density | 1.2 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.27 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 11.3 |
| Basicity (pKb) | pKb = 9.71 |
| Magnetic susceptibility (χ) | -54.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.506 |
| Viscosity | Viscous liquid |
| Dipole moment | 5.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -323.3 kJ/mol |
| Pharmacology | |
| ATC code | D10AE01 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02, GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H241, H242, H302, H317, H319, H332, H335, H400 |
| Precautionary statements | P210, P220, P234, P240, P241, P261, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P332+P313, P337+P313, P370+P378, P403+P235, P410, P411, P501 |
| NFPA 704 (fire diamond) | 3-4-4-W |
| Flash point | 56 °C (133 °F) (decomposes) |
| Autoignition temperature | 50 °C |
| Explosive limits | Not explosive as per GHS classification |
| Lethal dose or concentration | LD50 (oral, rat): 7710 mg/kg |
| LD50 (median dose) | 162 mg/kg (rat, oral) |
| NIOSH | DDC35000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Benzoyl Hydroperoxide: "5 mg/m3 (OSHA TWA) |
| REL (Recommended) | 4 hours |
| IDLH (Immediate danger) | 200 mg/m³ |
| Related compounds | |
| Related compounds |
Benzoyl peroxide Peracetic acid Cumene hydroperoxide Tert-butyl hydroperoxide Acetyl peroxide |

