Benzoquinone: A Chemical with a Storied Past and a Complex Future
Historical Development
People have known about benzoquinone since the 19th century, well before anyone thought about industrial chemistry as a global force. Old lab notebooks tell stories of early chemists poking and tinkering, finding new smells and colors, and benzoquinone’s bright yellow hue left a mark both visually and in its legacy. Michael Faraday isolated related compounds, and later August Wilhelm Hofmann and Friedrich Wöhler dug deeper into the aromatic ring’s transformations. What started as academic curiosity grew into a foundation for modern knowledge of aromatic oxidation. Later generations of scientists expanded its use, weaving benzoquinone’s tale into countless innovations in organic synthesis, dyes, and pharmacology. These stories echo through research labs, showing how raw curiosity can ignite technological change.
Product Overview
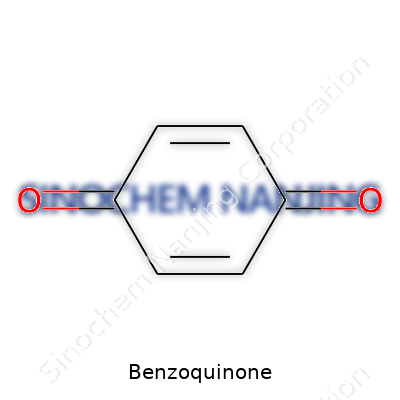
Benzoquinone describes a class of quinones, but most people in the lab usually mean 1,4-benzoquinone, a yellowish crystalline solid that catches the eye and challenges the nose. It isn’t some obscure or exotic chemical seen once in a blue moon—benzoquinone often pops up in college labs and large industrial plants alike. It acts as a mild oxidizing agent, working in everything from the synthesis of medicines to the production of chemicals and dyes. Its presence stretches even into polymer production and photochemistry. People who spend any time around synthetic organic reactions learn to respect the punch packed by such a small molecule.
Physical & Chemical Properties
Benzoquinone’s sharp, distinctive odor isn’t just a nuisance—it’s a reminder of volatility and sublimation. In solid form, the yellow crystals demand careful handling as they turn to vapor under normal pressure. Its melting point sits in a moderate range, making it easy to purify through sublimation in a fume hood. Solubility leans toward the organic: ether, alcohol, and benzene welcome benzoquinone, while water turns its back. Chemically, benzoquinone stands out for its two carbonyl groups on a six-membered benzene ring, both at para positions. This arrangement favors oxidizing behavior and scores of reactions, leaving the compound both predictable and endlessly useful. Such structural simplicity gives rise to surprising reactivity and lays a foundation for many modifications.
Technical Specifications & Labeling
The practicalities of working with benzoquinone keep labs vigilant. Packaging always screams caution: poisonous, corrosive, oxidizer, keep dry, avoid flames. Labels must inform of acute toxicity, strong irritant effects on skin and eyes, and long-term inhalation risks. Handling calls for sturdy gloves, full eye protection, and strict ventilation. GHS pictograms—dead fish and trees, exclamation marks—serve as stark reminders not to treat benzoquinone carelessly. Analytical standards demand clear labeling of purity and batch origin, especially as contamination or excess moisture can flip a reaction or risk safety. Every bottle tells a story, with regulatory oversight taking center stage to reduce hazards in the workplace and environment.
Preparation Method
Modern benzoquinone production brings together a range of chemical knowhow, historically scaling up from benzenediol oxidation. Common preparation involves oxidizing hydroquinone using agents like ferric chloride, silver oxide, or even manganese dioxide. This approach yields reliable results and fits within factory and research lab operations. Some manufacturers tweak conditions to suit environmental guidelines, choosing greener oxidants and solvent systems. Electrochemical methods also crop up in academic studies, emphasizing efficiency and reduced waste streams. These synthetic methods give access to high-purity material and set the tone for responsible chemistry in today’s world.
Chemical Reactions & Modifications
Benzoquinone acts as a workhorse in many organic reactions. It teams up with nucleophiles as a Michael acceptor, making it central to carbon-carbon bonding in pharmaceuticals and fine chemicals. Its ability to swap in for oxidants in dehydrogenations helps drive progress in the total synthesis of important compounds. Reductive modifications return benzoquinone to hydroquinone, offering a reversible redox pair that chemists exploit across redox cycling, spectroscopy, and even battery technology. Electrophilic substitution opens doors for chlorination or methyl group additions, expanding its versatility even further. If a researcher dreams up a novel catalyst or electron shuttle, chances are benzoquinone—or its relatives—deserve at least a cameo.
Synonyms & Product Names
In catalogs and technical literature, benzoquinone doesn’t hide behind one name. Most chemists refer to it as 1,4-benzoquinone, but names like para-benzoquinone or quinone often join the conversation. The term “p-benzoquinone” distinguishes it from its 1,2- counterpart (ortho-benzoquinone). Certain suppliers even use short codes or tradenames, though academic and industrial scientists prefer clarity over clever branding. Knowing the correct synonyms prevents costly errors and ensures safe handling.
Safety & Operational Standards
Working with benzoquinone demands real preparation—not just for its reactivity but for the health risks. Exposure to even modest quantities leads to skin blistering, eye injury, and serious irritation of the respiratory tract. Inhalation causes headache, dizziness, nausea, or worse. The compound’s toxicity profile pushes labs to enforce sealed containers, designated storage in cool and ventilated cabinets, and routine air monitoring. Training goes beyond telling people to ‘wear gloves’—it deals with the reality that a lapse can bring both acute and chronic health issues. Cleaning up spilled benzoquinone without full respirators and absorbent protocols would risk workplace safety. Waste disposal calls for strict adherence to regulatory standards to minimize leaching into soil or waterways, closing off one route of community exposure.
Application Area
Benzoquinone finds its way into pharmaceutical research, particularly for building complex molecules like antibiotics, vitamins, and anti-cancer agents. Its oxidizing power translates into crisp reactions in both academic and commercial settings. Polymer manufacturers value benzoquinone in making high-spec plastics, while the dye industry cherishes it for pigment production. Analytical labs recognize it as a standard reagent, harnessing its quick redox shifts for measuring reaction dynamics. The printing trade, photo industries, and even developers of organic semiconductors have a place reserved for benzoquinone or its analogues. Real-world applications depend not just on the parent molecule but on a family of modifications and derivatives tailored for each task.
Research & Development
New frontiers keep opening up for benzoquinone research—sometimes in unexpected places. Scientists working on rechargeable battery technologies harness the reversible redox nature of quinones to design new organic electrodes that could outperform traditional metal-based batteries. Medicinal chemists use benzoquinone scaffolds when hunting for fresh antibiotics or enzyme inhibitors, hoping that unique electronics will deliver new mechanisms of action. A growing body of environmental research considers benzoquinone’s fate in air and water, prompted by concerns over industrial discharges and accidental releases. The need for cleaner production methods spurs green chemistry labs, while analytical chemists refine methods to track trace levels in consumer goods and biological samples.
Toxicity Research
Benzoquinone’s health risks stretch beyond direct skin and eye contact. Medical papers document links between chronic exposure and long-term injury to liver, kidneys, and bone marrow. Animal studies have raised red flags about mutagenicity and reproductive effects, though results vary by species and exposure route. Occupational health specialists press for tighter permissible exposure limits and regular health checks for workers likely to encounter the compound. Toxicologists run newer assays to dissect how benzoquinone interacts with cellular macromolecules, aiming to spot the early signs of damage or set thresholds below which no harm appears. Calls persist for more in-depth studies, especially as production and usage rates continue to climb in emerging economies.
Future Prospects
The coming years will likely see benzoquinone branching into more sustainable roles, driven by tightening regulation and a push for cleaner chemistry. Green oxidant development balances effectiveness with waste reduction, thanks to steady advances in catalysis and photochemistry. New approaches to quinone-based battery electrodes could change how energy is stored at scale, with research groups jostling to translate lab results into commercial breakthroughs. Pharmaceutical innovation continues tapping benzoquinone scaffolds in the chase for rare disease treatments, while ongoing toxicity studies promise a clearer understanding of safe exposure levels and environmental fate. The future for benzoquinone looks complex—fraught with safety challenges, yet bursting with creative opportunities for those thinking beyond traditional boundaries.
What is benzoquinone used for?
A Closer Look at Benzoquinone
Benzoquinone stands out in chemistry for good reason. This yellow crystalline compound, with its sharp, penetrating odor, shows up in laboratories, pharmaceutical manufacturing, and even in the world of art restoration. I first came across benzoquinone in a college organic chemistry lab—the smell alone told us we were dealing with something important. It brings practical value that stretches from classrooms into the hands of major industries.
Benzoquinone in Chemical Synthesis
In the world of organic chemistry, benzoquinone helps chemists build more complicated molecules. Many pharmaceutical drugs begin their journey in a reaction where benzoquinone either gives up electrons or grabs them, acting as an oxidizing agent. This quality streamlines the production of antibiotics and cancer treatments. Factories that turn out bulk drugs, dyes, and pesticides rely on these reactions to keep their processes running smoothly.
Life Science and Research Applications
Lab researchers put benzoquinone to work in a host of ways. It serves as a core piece in certain biochemical assays and plays a critical role in electron transport studies, which look at how cells breathe and break down sugar. Every undergraduate learning about cellular respiration will bump into benzoquinone sooner or later. The molecule often pops up in textbooks—its structure helps students see how electrons flow from one chemical to another, letting cells produce energy.
Material Science and Industry
Manufacturers can use benzoquinone in making polymers, adhesives, and dyes. The pigment industry taps into this compound because it shapes the colors we see on fabrics and plastics. I visited a textiles factory last year and saw how benzoquinone derivatives played a part in creating stable, long-lasting dye compounds. Without these chemical steps, clothing and upholstery would fade fast.
Environmental Studies and Testing
Benzoquinone is more than a tool for building molecules; it serves as a marker for monitoring pollution. Scientists track benzoquinones in wastewater and even in smoke from wildfires. These studies help regulators track environmental damage and make smarter policies. Overexposure can harm humans and wildlife, pushing for careful handling and monitoring in any industry that uses it.
Risks and Handling
Handling benzoquinone calls for respect. This chemical proves toxic at higher doses, irritating the skin, eyes, and respiratory system. Workers need gloves, fume hoods, and training to stay safe, especially in large-scale operations. Oversight and proper disposal practices are not just red tape—they’re real protections proven to reduce risk. Even in a small lab, proper ventilation and safety gear make a clear difference. In the wider community, safer alternatives are under study, but nothing outpaces benzoquinone’s effectiveness in key applications—at least not yet.
What Lies Ahead
Chemistry, by nature, pushes towards innovation. Newer green chemistry routes are emerging, aiming to make benzoquinone use safer and less polluting. Promising research explores biodegradable analogues and sustainable ways to produce oxidants that could offer the same punch without the baggage. As regulations tighten and society pays more attention to environmental health, those in charge of production will need to adapt and innovate—just as benzoquinone has long driven innovation in science.
Is benzoquinone hazardous to health?
What Benzoquinone Is and Where You Might Encounter It
Benzoquinone, sometimes known to chemists as para-benzoquinone, pops up in a few surprising places. Factories use it for making dyes, fungicides, or as an intermediate in certain chemical processes. Cigarette smoke and car exhaust add it to the air you breathe in cities. If you work in labs, printing, or manufacturing, you may have come across its sharp, irritating odor.
Why Benzoquinone Poses a Health Risk
Breathing benzoquinone fumes irritates the nose and lungs pretty quickly. Folks report coughing, chest tightness, and sometimes headaches after exposure in crowded workrooms. Skin contact leaves red, itchy patches. Splash some on your eyes, and you’ll feel burning pain right away.
There’s more to worry about in the long run. Prolonged exposure affects the way your blood carries oxygen. Toxicology studies on animals point to changes in bone marrow and drops in red blood cell counts. Early-stage research links benzoquinone with disruptions in the immune system. Workers in plants using this chemical face far higher risks of chronic bronchitis and other respiratory trouble.
The United States Environmental Protection Agency marked benzoquinone as a hazardous air pollutant. Researchers from the American Conference of Governmental Industrial Hygienists recommend strict exposure limits: around one milligram per cubic meter as an eight-hour average. If you compare that to substances like benzene or formaldehyde, the dangers stack up quickly, especially with regular exposure.
Why Regular People Should Care
Not everyone walks into a lab or chemical plant. Still, benzoquinone sneaks into daily life through city air, cigarette smoke, or poorly ventilated homes. The risks get amplified for children, elderly folks, or anyone living with asthma. Even hobbyists should take care: photographers, woodworkers, or artists working with certain pigments or chemicals can get exposed without realizing it.
Normal routines sometimes don’t give warning. Workplaces cut corners, windows stay closed, and folks assume cleaning chemicals can’t hurt them. Recognizing these routine exposures can protect many people from unknowingly damaging their lungs or skin.
What Can Be Done for Protection?
Wearing gloves and masks makes a difference, but that alone never solves the problem. Stronger fume hoods and clean air systems in workplaces lower risks for everyone. Regular industrial safety checks can remind companies to keep airborne amounts far below the legal limits.
At home, common sense goes a long way: avoiding smoking inside, choosing safer cleaning products, or opening windows while doing DIY projects. Schools and workplaces with chemistry labs should invest in proper ventilation and up-to-date safety training. Local governments can chip in by enforcing air quality standards and cleaning up industrial sites.
Doctors and clinics need to ask about chemical exposure when patients struggle with unexplained coughs or skin rashes. Many early symptoms get chalked up to simple allergies or colds. Recognizing the role of environmental toxins can lead to better treatment and prevention.
Looking Forward
Tighter safety regulations and public awareness bring real change. Putting effort into cleaner production methods and monitoring air quality protects vulnerable groups. Teaching workers and students about the risks lets them make smarter choices. Personal responsibility, community standards, and policy shifts together build a much healthier future.
What are the storage conditions for benzoquinone?
Looking at Benzoquinone Up Close
Benzoquinone stands out in any chemical cabinet. Its sharp odor and bright yellow appearance make it hard to ignore, but there's more to it than meets the eye. Working in a lab, I learned the hard way about the importance of storing reactive substances like this safely. One cracked container can turn curiosity into catastrophe. Benzoquinone easily reacts with air and light, quickly degrading or even catching fire in the wrong scenario.
Risks and Hazards That Can’t Be Ignored
People who have handled benzoquinone know it doesn't play around. Inhaling its dust or vapors brings on headaches, coughing, and worse. Direct contact with skin or eyes burns and irritates. As a solid, it oxidizes quickly when exposed, especially under heat or direct sunlight. These are not imaginary risks—safety data sheets and dozens of workplace accidents back up their seriousness. I once saw an improperly closed bottle corrode from the inside out, warning me and everyone else in the lab.
Staying Ahead of Risks: Optimal Storage Practices
Benzoquinone needs a cool, dry, well-ventilated storage space. Locking it away from sunlight has to become second nature. Once, I opened a cabinet directly hit by afternoon sun. The heat had warped several bottles, threatening a mess that would require hazmat intervention. Nobody wants that on their shift. Acid-resistant shelves prevent leaks from spreading, and chemical-proof secondary containers add an extra layer of safety.
Humidity acts as an invisible enemy. Water triggers slow decomposition, with potential for unpleasant fumes or sticky residue. Every storage room I’ve worked in used desiccators or silica gel packs. This extra step pays off, saving you from unnecessary cleanup and exposure. Keeping storage spaces uncluttered also helps. If something spills, a clear area makes cleanup faster and safer.
Good Habits Make a World of Difference
Labs and warehouses handle these chemicals every day. Staff put limits on how much benzoquinone to keep open at once, and why put more people at risk? Smaller lots reduce danger and minimize loss if anything goes wrong. Clear labeling matters. My mentor always checked for outdated or damaged containers at the start of each shift. It’s easy to think you’ll remember, until you forget. Simple routine checks catch small problems before they grow.
Understanding Rules and Science
National chemical safety agencies set exposure limits for workers. The U.S. Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) both list benzoquinone as hazardous if inhaled over time. Their guidelines call for proper storage, not just for legal reasons, but employee health and peace of mind.
Research on benzoquinone’s environmental impact keeps growing. Accidental releases contaminate both air and water, so companies often invest in fireproof cabinets with self-closing doors. Spill control kits, personal protective equipment, and mandatory staff training reduce chances of injury or environmental accidents.
Practical Steps Forward
Any facility with benzoquinone can cut risks with simple steps—marking containers clearly, setting aside a storage room with good airflow and temperature control, using spill trays, locking cabinets, and having routine safety drills. Regular reviews of inventory and storage conditions catch problems early. As someone who’s seen both careful and careless handling, I can say that good storage keeps workers safe, saves money, and builds trust among everyone sharing the workspace.
What is the molecular formula of benzoquinone?
Why Benzoquinone Catches Attention in Chemistry
Benzoquinone pops up a lot during college chemistry classes and in industrial labs. The name gets thrown around, but not everyone outside chemistry circles knows what it’s made of. Looks simple enough: just a ring with some oxygen atoms double-bonded in. But what’s the actual count of carbons, hydrogens, and oxygens in that molecule?
There are different forms, but the one people usually mean—para-benzoquinone—holds a pretty special place. Its skeletal structure, which features a benzene ring with two double-bonded oxygens straight across from each other, forms the backbone for all sorts of reactions, pharmaceutical intermediates, and dye production.
Molecular Formula That Matters: C6H4O2
Here’s the formula right on the table: C6H4O2. That’s six carbons, four hydrogens, and two oxygens. For those folks who’ve spent afternoons wrestling with line drawings on chemistry test papers, it instantly brings back memories of aromatic rings and resonance structures.
Getting this thing right isn’t just about passing an organic chemistry quiz either. Plenty of labs rely on benzoquinone as an oxidizing agent—a way to give or take electrons from other molecules—because of the specific arrangement and number of atoms. Some undergraduate labs use it to teach electrochemistry. Its involvement in more advanced reactions -- like those creating vitamin K analogs or fungicides -- kicks the stakes up further. Mistake the formula and the whole plan falls apart.
Personal Encounters Behind the Formula
The first time I handled pure benzoquinone, I remember its sharp, almost acrid smell and how the powders stained gloves yellowish-brown. Reading a label, C6H4O2 seemed simple, but nobody in the room treated it lightly. I watched as an instructor explained the ring’s reactivity, and the way those two oxygens sitting opposite each other gave benzoquinone such versatility in redox chemistry. Students who didn’t double-check the structure made mistakes downstream, mixing up reagents, or misjudging hazards.
Chemical manufacturing crews also pay close attention to the exact formula. It determines how raw materials get measured and how reactions must be controlled. Sometimes a factory might pivot from para-benzoquinone to other quinone isomers, but the changes in the molecular formula require new handling procedures and safety checks. As a former lab tech working with chemical supply chains, I saw shipments refused over mismatches in documentation, usually because somebody confused compounded names or structures.
The Domino Effect of Misidentifying the Formula
Incorrectly identifying benzoquinone messes with more than just lab grades. Downstream industries risk faulty end products—think anti-malarials, agriculture fungicides, or dyes. Batch failures stack up, and safety issues grow if the actual toxicity and reactivity get ignored. Many industrial accidents trace back to paperwork errors as simple as a swapped hydrogen or extra oxygen.
Quality control teams jump into the fray here, double-checking chemical manifests against the C6H4O2 standard. Effective training means every chemist recognizes the pattern on sight—not just in a textbook, but on a shipment barcode or while prepping a flask on the bench.
Learning from Mistakes to Get it Right
Education rarely stops at the classroom wall. I’ve watched research teams adopt more visual aids, digital flashcards, and hands-on demonstrations to drive home what each chemical formula looks like in reality. Safety briefings drill the habit of double-reading chemical names and formulas before starting any prep work.
Benzoquinone’s formula may seem like a footnote once you’ve seen it enough times, but that atomic composition—C6H4O2—serves as a linchpin for smart, safe lab and plant work. Experienced chemists and careful newcomers both depend on getting it right, not just for clean experiments but for personal safety and product reliability.
How should benzoquinone be disposed of?
Looking Beyond the Trash Bin
Benzquinone doesn’t belong down the drain or tossed in the regular garbage. Anyone who has worked in a laboratory or dealt with chemical inventories knows the headache that comes from unwatched chemical waste building up. Even small spills of benzoquinone sting the nose, tingle the skin, and stain anything in its path with a distinct yellow. We learn right away—tiny amounts matter, both for safety and for the environment.
Human and Environmental Risks
Leaving benzoquinone unmanaged opens the door to health risks. This chemical irritates the eyes, causes respiratory trouble, and can trigger bigger problems for anyone with breathing sensitivities. Waterways take a blow too. Benzoquinone finds its way through pipes or landfill seepage, and aquatic life takes the hit. Fish and insects are a lot more vulnerable to toxins than we like to admit. The ecosystem doesn’t have a reset button. The crux here: responsible disposal keeps people and wildlife out of harm’s way, and the soil and water cleaner for the next person to use.
What Real Safe Disposal Looks Like
There’s a proper route for chemicals like benzoquinone, one that’s based on more than just checklists and compliance. Shelves and storerooms at universities and industries get full, and it’s tempting to pitch old bottles, but that shortcut comes at cost. The EPA and OSHA lay out rules for hazardous waste, and benzoquinone lands square in that category. A call to a licensed hazardous waste hauler is the first real step. These folks have training, containment vessels, and contacts at facilities that neutralize chemicals or break them down with incinerators at the right temperature.
At times, onsite neutralization makes sense, but only with the right gear, fume hoods, and chemical know-how. Sodium bisulfite reduces benzoquinone to less reactive hydroquinone, which is less toxic and easier to handle. Few outside of a chemistry lab are set up for that, so hauling it out with a professional makes the best sense for most situations. These waste haulers log every drum and provide a manifest, tracing waste from the origin to the final treatment. Seeing that paper trail shuts down worries about surprise audits or penalties.
Long-Term Mindset: Prevention Beats Disposal
Each year, schools, research labs, and manufacturing sites generate pounds of chemical waste. Thinking upstream helps curb the waste problem. Order smaller containers, share stock among departments, or dial in inventory spreadsheets. That way, fewer bottles expire and less benzoquinone ever needs disposal. Staff and students with a few years under their belts know the hidden cost in mismanaged chemicals—not just in cash, but in time spent worrying and paperwork later on.
On-the-Ground Solutions
Open conversations help too. New staff or students benefit from training on both storage and disposal. Dry runs with mock spills or waste labeling exercises get everyone on the same page and less likely to make mistakes. The simple move of labeling every bottle with purchase and opening dates helps keep track of what needs attention next. Asking local disposal teams for an annual review can turn up surprising gaps in your routine. Their feedback can spark process improvements that stick for years.
In the end, taking care of benzoquinone waste is about looking out for each other and the world outside our workspaces. The right mix of planning, teamwork, and proper hauling keeps everyone safer—and that’s worth every bit of extra effort.
| Names | |
| Preferred IUPAC name | cyclohexa-2,5-diene-1,4-dione |
| Pronunciation | /ˌbɛnzoʊkwɪˈnoʊn/ |
| Identifiers | |
| CAS Number | 106-51-4 |
| Beilstein Reference | 1203690 |
| ChEBI | CHEBI:35544 |
| ChEMBL | CHEMBL1400 |
| ChemSpider | 'GCJZKJUCBMHFED-UHFFFAOYSA-N' |
| DrugBank | DB04220 |
| ECHA InfoCard | 100.003.356 |
| EC Number | 1.6.5.6 |
| Gmelin Reference | 12938 |
| KEGG | C00183 |
| MeSH | D001584 |
| PubChem CID | 257 |
| RTECS number | DN3150000 |
| UNII | F639GH1D3S |
| UN number | 2587 |
| Properties | |
| Chemical formula | C6H4O2 |
| Molar mass | 108.094 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Strong, unpleasant odor |
| Density | 1.32 g/cm³ |
| Solubility in water | 6.4 g/L (20 °C) |
| log P | 1.01 |
| Vapor pressure | 1 mmHg (at 25 °C) |
| Acidity (pKa) | 9.88 |
| Basicity (pKb) | -3.78 |
| Magnetic susceptibility (χ) | −49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.538 |
| Viscosity | 0.964 mPa·s (25 °C) |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | '260.0 J·mol⁻¹·K⁻¹' |
| Std enthalpy of formation (ΔfH⦵298) | 35.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –2669 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D03AX04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H319, H332, H335, H341, H351, H372 |
| Precautionary statements | P210, P260, P280, P301+P310, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-2-0-A |
| Flash point | 79 °C (174 °F; 352 K) |
| Autoignition temperature | 285 °C |
| Explosive limits | Explosion limits: 2–17% (in air) |
| Lethal dose or concentration | LD50 oral rat 130 mg/kg |
| LD50 (median dose) | LD50 (median dose): 130 mg/kg (oral, rat) |
| NIOSH | TT3950000 |
| PEL (Permissible) | PEL: 0.1 ppm |
| IDLH (Immediate danger) | 50 ppm |

