The Journey and Future Outlook of 3-Aminobenzofuran-2-Carboxamide
Historical Development
After the early discoveries around benzofuran derivatives in the late nineteenth and early twentieth centuries, chemists realized these structures offered a door into new molecules with interesting biological activities. The 3-aminobenzofuran-2-carboxamide scaffold belongs to a family that saw a burst of interest once researchers linked benzofurans to potential uses in medicine and materials science. Synthetic routes picked up speed when chemists refined aromatic substitution and amide coupling reactions, making compounds like 3-aminobenzofuran-2-carboxamide easier to prepare with higher yields. Over time, this molecule has stepped into the spotlight, driven by the versatility that nitrogen, oxygen, and carboxamide groups lend to its chemistry, especially in developing enzyme inhibitors and advanced materials. Research groups across academic and industrial labs have integrated this compound into larger studies, searching for next-generation applications and building upon decades of chemical knowledge.
Product Overview
3-Aminobenzofuran-2-carboxamide stands out not only because of its distinctive aromatic ring, but also thanks to the presence of both an amine and a carboxamide functional group. The structure often serves as a versatile intermediate for more complex molecules, and it's found its way into medicinal chemistry libraries and combinatorial synthesis screens. Laboratories employ it as a backbone when exploring analogs or derivatives aimed at unraveling new structure-activity relationships. Pharmaceutical teams scour molecules like this one in the quest to uncover tools against challenging biological targets, while researchers in materials science take advantage of its conjugated system for potential electronic behaviors. This molecule means more than just a reagent on a shelf; for many, it’s a stepping stone to innovation.
Physical & Chemical Properties
The physical makeup of 3-aminobenzofuran-2-carboxamide reveals a crystalline solid, often pale or off-white, influenced by how it's purified and isolated. The molecule sports a balance between hydrophilicity and aromatic stability, a trait found in many functional benzofurans. Relatively moderate in molecular weight, it dissolves better in polar organic solvents, a plus for synthetic manipulation in the lab. The carboxamide and amino groups allow for hydrogen bonding, introducing both opportunities and complications during formulation or recrystallization. Thermal stability usually covers the range needed for most synthetic routes, but it pays to monitor conditions, since lab mistakes can result in decomposition or the formation of side products. The chemical resilience of the benzofuran core creates enough backbone strength for downstream modification, while leaving reactive points at the amino and carboxamide ends.
Technical Specifications & Labeling
Anyone handling this compound learns quickly to check for batch purity and uniformity, since side reactions and small impurities can skew reaction outcomes and toxicology readouts. Analytical tools like NMR, IR, and mass spectrometry play a role in confirming identity and quality. Labeling typically focuses on chemical identifiers, risk statements, and storage conditions, especially for those working in more regulated environments. Such rigor supports reproducible scientific work, which feels all the more urgent as researchers lean on specialty chemicals for advanced studies. Whether prepping a custom batch at a bench or scaling up for industrial runs, details matter. Accurate labeling and robust documentation protect both the scientist and the work at hand.
Preparation Method
Synthesis routes for 3-aminobenzofuran-2-carboxamide come from a mix of classical organic reactions and modern coupling methods. One of the more direct approaches involves constructing the benzofuran ring from a substituted phenol with an ortho-nitro or ortho-amino group, then cyclizing under acidic or dehydrating conditions. Converting the 2-position to a carboxamide often requires selective nitration or amination followed by amidation, such as reacting the corresponding acid chloride or carboxylic acid with ammonia or an amine. Purification steps—like recrystallization or chromatographic separation—often follow, shaped by the exact nature of the produced intermediates. Chemists gravitate toward routes that protect vulnerable groups, minimize hazardous reagents, and avoid excessive solvent use, reflecting a growing awareness of green chemistry.
Chemical Reactions & Modifications
Tinkering with 3-aminobenzofuran-2-carboxamide opens doors to diverse analogs. The amino group provides an anchor for acylation, sulfonylation, or even urea-forming reactions. The benzofuran core invites electrophilic aromatic substitutions such as halogenation or alkylation, with careful control needed to maintain regioselectivity. The carboxamide portion enables coupling with other functionalized amines, forming diverse amide bonds relevant to medicinal chemistry. Adding or manipulating substituents affects both solubility and biological activity, a fact that drug designers know well. Advances in palladium-catalyzed cross-couplings and photochemical transformations expand the options, while also raising questions about the safety of catalysts and reaction byproducts downstream.
Synonyms & Product Names
Across chemical catalogs and research literature, this compound takes different names: 3-aminobenzofuran-2-carboxamide, 2-carboxamido-3-aminobenzofuran, or by more systematic IUPAC identifiers. Such variance can trip up searches or inventory management, so researchers learn early to cross-check registry numbers and supplier product codes. Common names may surface in patent filings or research papers, especially in studies focused on enzyme inhibitors or candidate drug molecules. The challenge of nomenclature reflects a wider issue in chemical communication, highlighting the need for unambiguous naming whenever new derivatives make their debut.
Safety & Operational Standards
Safety in the lab often shapes the workflow more than the excitement of discovery. 3-Aminobenzofuran-2-carboxamide does not escape the watchful eye of institutional safety committees or regulatory bodies. Standard operating procedures advise the use of gloves, eye protection, and fume hoods—common sense steps that many young chemists learn early. Potential harmful effects, such as skin or respiratory irritation, push researchers to respect proper handling and waste disposal. When scaling up to pilot or manufacturing quantities, risk assessments take on extra weight, especially given the unpredictability of intermediate formation or thermal decomposition. Documentation around hazards supports ongoing vigilance and improves transparency for new workers or collaborators stepping into a project.
Application Area
Medicinal chemistry has shown major interest in benzofuran carboxamides, exploring them as antagonists or inhibitors against tough biological targets in oncology, infectious disease, or central nervous system research. The interplay of aromaticity and polarity in these molecules suggests potential for binding in enzyme pockets—attracted researchers looking for dual-action or selective drugs. Materials science teams investigate them for organic semiconductors, molecular sensors, and dyes, banking on their rigid structure and tailored electronic properties. Academic studies expand into environmental sciences, as nitrogen- and oxygen-rich compounds draw attention for their photostability and environmental fate. Each application brings its own set of challenges, but they share a reliance on building blocks like 3-aminobenzofuran-2-carboxamide to probe the frontiers of science.
Research & Development
The pace of research keeps accelerating. Collaborative teams pull data from hundreds of analogs, using machine learning to correlate structural changes with biological outcomes. High-throughput screening puts pressure on suppliers for ever-greater purity and documentation. I’ve watched friends in biotech push molecules like this through early-phase target validation, constantly refining the core scaffold for metabolic stability, selectivity, and physicochemical compatibility. Laboratory notebooks fill with iterations—one team modifying the carboxamide for greater solubility, another tweaking the aromatic ring to dodge off-target effects. Industrial chemists focus on process improvements, scaling up synthesis while addressing waste reduction, energy efficiency, and supply chain reliability. The push and pull between laboratory creativity and practical constraints drives continual reevaluation of how to best harness the chemistry of such promising compounds.
Toxicity Research
Toxicity always stands as a central consideration around new molecules, especially those intended for drug discovery or electronic applications with long-term contact risks. Early studies point to a need for comprehensive safety profiling—acute toxicity, mutagenicity, and the effect on environmental systems. I’ve seen entire projects stall when candidate molecules show unexpected persistence, bioaccumulation, or toxicity to model organisms. Regulatory bodies in the US and abroad expect clear data on health and environmental risks. Many research protocols now integrate in vitro and computational screens ahead of animal studies, looking for molecular signals linked to carcinogenicity or hormonal disruption. Such vigilance pays off by keeping both workers and end users better protected and forcing continual improvement in design and testing.
Future Prospects
Looking ahead, the story of 3-aminobenzofuran-2-carboxamide seems far from finished. Improved synthetic strategies promise broader access to diverse analogs, which fuels both the pipeline of modern drug discovery and innovations in organic electronics. Computational chemistry and artificial intelligence give researchers sharper tools to predict behavior and reduce dead-end experiments, making each iteration more informed. Increased scrutiny around safety and sustainability will likely shift priorities toward greener, less hazardous synthesis and comprehensive life-cycle analysis. As industry and academia keep mining the potential of the benzofuran scaffold, society stands to benefit from therapies and technologies that trace their origins back to thoughtful work at the lab bench. Armed with deeper data, smarter regulations, and open scientific exchange, the next chapters of this compound’s history could deliver practical answers to needs in health, technology, and sustainability.
What is 3-Aminobenzofuran-2-Carboxamide used for?
Looking Into 3-Aminobenzofuran-2-Carboxamide
Scientists and pharmaceutical researchers often come across chemical names that don’t seem to mean much to everyday folks. 3-Aminobenzofuran-2-Carboxamide might sound like something out of a dense textbook, but its story touches on health, treatment, and the drive to discover powerful medicines. In labs and journals around the world, this chemical has caught the attention of people hoping to design better drugs for complicated diseases.
The Connection to Medical Research
My own time spent poring over pharmacology reports taught me that most big breakthroughs start with small chemical tweaks. Adding or shifting a group on a molecule’s core can mean the difference between a dud and an active drug. 3-Aminobenzofuran-2-Carboxamide stands out for this exact reason. Researchers across Asia, Europe, and North America view it as a promising scaffold—a platform for building compounds that could tackle cancer, viral infections, or neurological issues. Publications have pointed to how its molecular framework interacts with certain enzymes involved in disease.
Fighting Cancer and Other Diseases
Cancer is quick to adapt, and that makes treatment tough. Over the past decade, specialists searched for agents that can block abnormal cell division without scorching healthy cells. Some groups developed derivatives of 3-Aminobenzofuran-2-Carboxamide for this, testing the new molecules against stubborn tumor lines. Results sometimes showed the compound stopping cell growth, or slowing down dangerous processes. Of course, these results usually came from pre-clinical data—cells in a petri dish or animal studies. Still, successes here spark bigger investigations.
Other teams have gone after viruses. With outbreaks and drug resistance growing, researchers chase options that can slip past a microbe’s defenses. A handful of studies used 3-Aminobenzofuran-2-Carboxamide variations to stop viral replication, aiming to halt a virus in its tracks. Many promising candidates never reach the pharmacy shelves, but the groundwork gets laid here.
The Roadblocks and Hard Questions
Those who have spent time in medicinal chemistry labs know that potential alone means little. Every hopeful compound faces a battery of tests for safety, toxicity, “druglikeness,” and cost. Sometimes a molecule looks solid at first but brings serious side effects or poor absorption. In past research around benzofuran derivatives, some caused unwanted reactions in healthy tissues. These risks demand careful investigation and transparency in sharing data. As patients and families wait for new therapies, any hint of progress brings hope, but responsible science refuses to cut corners.
What Matters For Patients and Policy
Every step in discovering new medicines starts with chemistry like 3-Aminobenzofuran-2-Carboxamide. This research doesn’t always grab headlines or lead to an instant cure, but each stage offers lessons. Regulators, academics, and industry leaders all play roles in deciding which paths get funding or approval. Open communication between labs, doctors, and the public helps weed out hype and keep everyone looking at real results. Adopting policies that encourage data sharing and patient safety gives the best shot at helping more people, sooner.
A Solution-Oriented Path Forward
If investment keeps flowing into creative chemistry, and if researchers stick to high standards, future derivatives from molecules like 3-Aminobenzofuran-2-Carboxamide could cross the finish line. Collaboration matters–across borders, universities, and companies. Listening to patient voices, sharing what works and what does not, and keeping the focus on safety make a difference. It all starts with paying attention to small molecules and big possibilities—in the lab and beyond.
What is the chemical structure of 3-Aminobenzofuran-2-Carboxamide?
Breaking Down the Basics
Chemistry feels intimidating to some, but anyone who has struggled with an organic chemistry textbook will recognize patterns in molecules like 3-Aminobenzofuran-2-carboxamide. Here’s what 3-Aminobenzofuran-2-carboxamide looks like by name alone: it starts with a benzofuran “core,” which combines a benzene ring with a fused furan—a five-membered oxygen-containing ring. This isn’t just textbook trivia. Structurally, each element in the name tells chemists what to expect, hinting at the molecule’s behavior and potential uses.
Dissecting the Structure
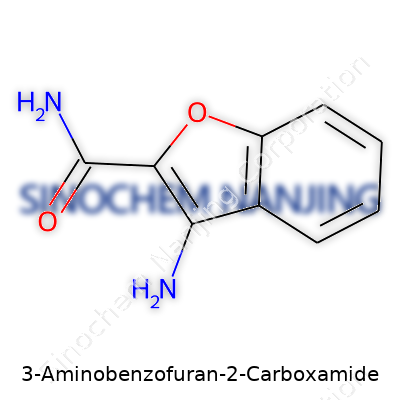
This compound holds a benzofuran backbone. On the second position of the furan ring, a carboxamide group (–CONH₂) attaches. On the third position of the benzene ring, an amino group (–NH₂) comes into play. For anyone who has mapped carbon atoms on notepaper, this is where chemical intuition starts to kick in—the molecule carries both nitrogen and oxygen, providing sites for hydrogen bonding. This matters, not just for solubility, but also for how the molecule might react with others.
Drawing Out the Formula
Chemists favor shorthand, so the simplified molecular formula for 3-Aminobenzofuran-2-carboxamide is C9H8N2O2. Its structure shows a benzofuran skeleton, with a carboxamide at position 2, and an amino group at position 3. For those visualizing it, the amino group sticks out from the benzene section, while the carboxamide hitches to the furan ring. This combination builds a versatile molecule—one that can participate in various kinds of chemical and biological interactions.
Why the Structure Matters
In research and medicine, structure steers behavior. The fusion of aromatic rings with functional groups means this molecule doesn’t just float around in a solution. The carboxamide group encourages stability, often increasing the compound’s lifetime in a biological environment. The amino group opens the door to further chemical modifications. Researchers count on these kinds of molecules for drug design, targeting enzymes, or binding to specific proteins.
Looking Toward Real-World Use
There’s practical impact, too. In the crowded fields of pharmaceuticals and material sciences, these structural features shape future applications. Medicinal chemists often test molecules like 3-Aminobenzofuran-2-carboxamide as starting points for new drugs—especially since many therapeutic compounds share aromatic and amide features. Having spent hours in the lab, I know how a simple shift, like swapping a hydrogen for an amino group, can change everything from water solubility to biological activity.
Pushing Boundaries and Solving Problems
Synthetic pathways for assembling these molecules pose challenges. Careful control of reaction temperature and selection of solvents often determine whether those sensitive functional groups survive the process. Green chemistry approaches get people talking these days, with researchers turning away from harsh reagents and toward milder, safer alternatives. This direction not only protects researchers but also takes aim at reducing environmental impact. In my own work, cleaner reactions bring fewer headaches during purification and waste disposal, which matters to every lab worker and environmental officer alike.
Bridging Basic Science and Application
Unlocking the secrets behind molecules like 3-Aminobenzofuran-2-carboxamide sets the scene for innovation. This isn’t just a static chemical drawing; it becomes a building block for future medicines, materials, and tools in chemical biology. As the scientific community digs deeper into these compounds, the blend of thoughtful design and practical synthesis gives real hope for useful new discoveries. This blend of elegance and hands-on challenge explains why the study of chemical structures keeps drawing researchers back.
What are the storage conditions for 3-Aminobenzofuran-2-Carboxamide?
Understanding the Nature of the Compound
3-Aminobenzofuran-2-carboxamide isn’t your average bench chemical. People in the lab or anyone handling specialty chemicals like this often realize its value—not just in terms of research output, but also in terms of lab safety and cost. Losing a batch due to poor storage practices hits both your budget and your timeline hard. Before considering fancy research, a few basics make a big difference in keeping this compound safe and potent.
Temperature Control Makes All the Difference
Don’t let temperature swings mess with your supply. Based on proven guidelines and firsthand experience, cool and consistent storage slows decomposition and helps this compound stay stable. Most research-grade benchtop chemicals benefit from storage in a dedicated fridge, kept well below room temperature but above freezing—often around 2 to 8 °C. Some labs use temperature loggers for a real-time alert if things go off track. At room temperature, chemical breakdown gets a chance to speed up, risking purity and consistency.
Keeping Moisture Out
Dry environments keep 3-aminobenzofuran-2-carboxamide in its best shape. Humidity can cause caking, hydrolysis, or other side reactions. For every bottle or vial, make sure the cap stays secure right after weighing or dispensing. Silica gel packs tucked in the chemical’s storage area can help keep local humidity low. Even in a busy lab, simple habits pay off—placing desiccators strategically or at least reminding coworkers why that extra twist of the cap matters.
Sheltering from Light
UV rays and even regular overhead lighting can start slow photochemical reactions in sensitive chemicals. A storage box or vial that blocks or limits light—amber glass, for example—reduces the risk of unwanted reactions. Close the cabinet door on the chemical shelf, and never leave it open for hours during set-up or cleaning. At times, forgetting these small steps ruins a compound faster than any temperature slip.
Labeling and Isolation
Cross-contamination makes even expensive chemicals useless. Storing 3-aminobenzofuran-2-carboxamide away from volatiles, acids, or bases in a well-labeled container prevents mix-ups. Labels need a clear purchase or expiry date, and signs explaining risks help everyone in the lab keep track. When trained properly, new team members spot issues before they turn into bigger problems.
Personal Experience: Shortcuts Lead to Waste
In every lab I’ve worked, someone eventually cuts corners on storage for the sake of convenience—the mindset that “just for today” won’t hurt. It does. Deteriorated samples mean wasted experiments, more time sourcing fresh material, and even safety concerns from unpredictable breakdown products. Nothing builds respect for chemistry like watching an unexpected reaction twist color right before your eyes—often traced back to poor storage.
Practical Solutions for Every Lab
Set up standard operating procedures everyone follows, not just senior scientists. Invest in a dedicated fridge, a small supply of desiccants, and plenty of amber bottles if you know you’ll handle photosensitive chemicals regularly. Do a regular inventory check. Tight schedules or tight budgets may tempt you to push things, but investing up-front saves time and resources in the long run.
Doing It Right Pays Off
Safe, consistent storage for 3-aminobenzofuran-2-carboxamide comes down to controlling temperature, moisture, and light while keeping the stock clearly labeled and separated from incompatible materials. Observing these habits preserves not just the compound, but also personal safety, research integrity, and cost-effectiveness. The groundwork matters as much as any breakthrough in the lab.
Is 3-Aminobenzofuran-2-Carboxamide hazardous or toxic?
Understanding What We’re Dealing With
3-Aminobenzofuran-2-carboxamide doesn’t exactly roll off the tongue, and its chemical name sounds harmless enough. You see similar structures in pharmaceutical research, but not much turns up in public safety databases. This gap doesn’t mean it should be taken lightly. Chemical safety depends just as much on what isn’t known as it does on long lists of proven effects. Without plenty of peer-reviewed toxicology data or common uses, reliable information ends up scattered and incomplete.
Looking at Chemical Relatives
Benzofuran rings pop up in many places, from plant-based compounds to some industrial products. Some derivatives serve as bases for drug development, others show up in dyes or pesticides. A few benzofuran analogues are notorious for posing risks. The mere presence of an amine group and a carboxamide unit in this molecule suggests some potential for both reactivity and bioactivity. Toxicologists would expect unpredictable properties, since tiny changes on a benzofuran framework can mean the difference between a dietary supplement and a hazardous molecule.
Health Risks: Known Unknowns
No established exposure limits exist for 3-aminobenzofuran-2-carboxamide. No widely published animal studies, no clear records of workplace injuries or poisonings have turned up. This doesn’t clear it. I’ve seen how new research chemicals often roll out with lots of hope and hush-hush testing, but sometimes slip through the cracks without a full picture on toxicity. Think of cases like benzofuran analogues used in synthetic drugs years ago: some proved much harsher than expected.
In practice, skin and eye contact, accidental inhalation, or ingestion could introduce unpredictable effects. Structurally similar benzofurans have caused irritation, headaches, and, in more severe cases, organ stress or even carcinogenicity. Until rigorous studies emerge, handling any such compound demands the same protocols as a known irritant or a possible toxin.
How Proper Handling Makes a Difference
When lab workers face a new or poorly studied compound, they rely on good habits: gloves, shields, fume hoods, respiratory protection as needed. Safe handling isn’t just about ticking boxes. It comes from a real understanding that uncertainty can be more dangerous than clear red flags, especially for people working in research or synthesis. Based on my own time in a busy chemistry lab, the worst incidents followed shortcuts—someone decided something “probably isn’t that bad.” These mistakes left colleagues with burns, allergic reactions, or worse.
Sources for Better Answers
The European Chemicals Agency (ECHA) and PubChem keep growing lists of research and incident reports, but sometimes it’s still a needle in a haystack for emerging chemicals. Toxicologists and environmental health researchers press for more transparency from chemical manufacturers, since gaps in knowledge put both workers and waste handlers at risk down the line. Education, up-to-date safety training, and quick access to the right protective gear keep harm to a minimum, even when the hazard picture isn’t ironclad.
Pushing for Solutions
We need manufacturers and research labs to publish more toxicology data on new chemicals like 3-aminobenzofuran-2-carboxamide. Funding for independent studies must climb, and chemical suppliers should attach clear safety information to rare or specialty compounds. Without these steps, users end up gambling, often without knowing the odds. Respect for what isn’t known can protect both workers and the public as chemical libraries keep growing and research picks up speed.
What is the purity and grade of 3-Aminobenzofuran-2-Carboxamide available?
Pursuing High Purity for Real Results
Anybody working in research or manufacturing chemistry knows that quality isn’t just a box to check. Sourcing 3-Aminobenzofuran-2-Carboxamide with a stated purity hits at the center of reliable results. Purity levels for this compound usually stand at 97% or higher. This figure comes from data provided by established chemical suppliers. Chemical synthesis and pharmaceutical development can demand that figure to go even higher—reaching up to 99%, especially for biologically sensitive assays.
Too many headache stories start with overlooked impurities. Contaminants running even a few percentage points above the spec can mess with spectral data, stall reactions, or trigger regulatory back-and-forth. Ask any synthetic chemist who’s run a column three times before realizing it never was their technique at fault—the problem sat in the bottle.
Grades Make a Difference
Lab-grade 3-Aminobenzofuran-2-Carboxamide deserves attention. These batches support basic research, method development, and teaching. Their specs handle a wide swath of flavors—sometimes ranging from 95% to 98% purity. They work fine in exploratory settings or for proof-of-concept work, but not so much for anything heading for patent portfolios or the clinic.
Moving to pharmaceutical or analytical grade, the story shifts. Purities stack up closer to 99% and above. These lots often carry full documentation—HPLC reports, NMR, and occasionally elemental analysis. That paperwork gets checked more carefully in heavily regulated environments. Failure to match grade to use case eats into time, budget, and even lab safety.
Supplier Verification—A Step You Can’t Skip
Looking for a high-grade sample starts at the Certificate of Analysis (CoA). Genuine suppliers list batch-specific test data, not just a catalog promise. Seeing a handful of brokers throw up vague “high quality” promises always set me on edge. I’ve seen teams burned by chasing bargain prices, only to spend the savings fixing avoidable contamination.
Several industry leaders in Europe, North America, and Asia supply this compound for both research and more demanding uses. Some back up their purity claims with validated testing, and occasionally third-party independent verification—especially for regulated applications. If you’re unsure about a supplier, checking recent publications for referenced materials gives a good sense of who delivers the real thing.
Improving Sample Consistency
Tracking down better grades gets a boost from tighter quality assurance routines at the site of manufacture. Rather than bank on spot tests, well-run operations monitor synthesis stages, waste management, and purification lines. They use GMP or ISO standards to spot-check and catch hidden variables that sneak through the process.
Feedback loops matter. Labs that track bad lots, report inconsistencies, and share impurities' snapshots help push the standard higher for the entire industry. As a chemist, I’d rather pay a premium once and avoid the pain of finding a mystery impurity halfway through a run—trading upfront cost for long-term savings.
What Works in Practice
Decisions around grade and purity of 3-Aminobenzofuran-2-Carboxamide have a direct line to lab performance. Spend the time on matching the product spec to your project. Look up batch-level certificates, ask for spectral data, and probe supplier reputation before opening a PO. Your notebook—and probably your manager—will thank you for it.
| Names | |
| Preferred IUPAC name | 3-Aminobenzo[f]furan-2-carboxamide |
| Other names |
3-Amino-1-benzofuran-2-carboxamide |
| Pronunciation | /ˈθriː-əˈmiːnoʊ-ˌbɛnzoʊˈfjʊərən-tuː-kɑːrˈbɒksaɪˌæmɪd/ |
| Identifiers | |
| CAS Number | 914349-41-6 |
| 3D model (JSmol) | `3D structure;JSmol;CC(=O)Nc1coc2ccccc12` |
| Beilstein Reference | 120793 |
| ChEBI | CHEBI:91209 |
| ChEMBL | CHEMBL3702049 |
| ChemSpider | 20216597 |
| DrugBank | DB08248 |
| ECHA InfoCard | 100.129.356 |
| EC Number | EC 620-107-7 |
| Gmelin Reference | 131268 |
| KEGG | C221499 |
| MeSH | D016426 |
| PubChem CID | 16314407 |
| RTECS number | GV7706000 |
| UNII | 9O2H547M4E |
| UN number | NA |
| Properties | |
| Chemical formula | C9H8N2O2 |
| Molar mass | 162.17 g/mol |
| Appearance | Off-white to pale brown solid |
| Odor | Odorless |
| Density | 1.32 g/cm3 |
| Solubility in water | Slightly soluble in water |
| log P | 0.01 |
| Acidity (pKa) | 13.79 |
| Basicity (pKb) | 9.23 |
| Magnetic susceptibility (χ) | -47.84×10^-6 cm^3/mol |
| Dipole moment | 4.53 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | The standard molar entropy (S⦵298) of 3-Aminobenzofuran-2-Carboxamide is "319.7 J·mol⁻¹·K⁻¹". |
| Std enthalpy of combustion (ΔcH⦵298) | ΔcH⦵298 = -4346 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07; GHS08; Warning; H302; H312; H332; H351; P261; P280; P301+P312; P308+P313 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332 |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P362+P364, P501 |
| Flash point | > 243.5 °C |
| NIOSH | NA |
| PEL (Permissible) | Not Listed |
| REL (Recommended) | 100 mg |
| IDLH (Immediate danger) | Not Listed |
| Related compounds | |
| Related compounds |
Benzofuran 2-Aminobenzofuran 3-Aminobenzofuran Benzofuran-2-carboxamide Indole-2-carboxamide 3-Aminobenzoic acid Furo[2,3-b]pyridine 2-Carboxamidoindole |

