Benzenesulfonyl Chloride: A Commentary on its Past, Present, and Future in the Chemical World
Tracing the Roots and Evolution
Benzenesulfonyl chloride started showing up in chemical labs long before modern organic chemistry took off. Early chemists noticed its reactivity and saw more in it than just a molecule for textbook reactions. Its first preparation, coming from treating benzene with sulfonating agents to create benzenesulfonic acid, then reacting this with phosphorus pentachloride or thionyl chloride, made organic synthesis more accessible. Over time, the process for making benzenesulfonyl chloride got less messy and less dangerous, but the basic approach—a two-step chemical transformation—hasn’t changed much in more than a century. Every lab that touched this chemical since its discovery has left a mark, from refining purification to boosting yields so it could be made in bulk for industrial and research needs.
What This Chemical Feels Like in the Real World
Anyone who’s spent time in a lab knows that benzenesulfonyl chloride stands out with its sharp, choking smell and fuming white colorless liquid look. Its formula (C6H5SO2Cl) doesn’t tell the full story, but regulars notice it reacts violently with water, sending off clouds of hydrochloric acid—a good reason for anyone in a lab coat to double-check gloves and eye protection whenever they crack open a bottle. Its melting point sits just above room temperature, and it boils well below the frying point of most oils, giving it a narrow window that keeps people alert in the lab, especially in summer. Few things make students more careful about storing chemicals dry and cool than a close call with benzenesulfonyl chloride escaping from a loose bottle cap.
Seeing Beyond the Label: Technical and Regulatory Realities
Labels on bottles of benzenesulfonyl chloride always shout danger, with hazard diamonds and notes about severe skin burns, eye damage, and lung harm. Over the years, regulators pressed for strict labeling, demanding clear warnings and handling instructions. Storage in tightly sealed containers and away from anything humid or reactive became the golden rule. Some packaging now comes with extra-strong seals and vented caps to let out pressure, which makes sense considering incidents in the past where bottles cracked or exploded from built-up fumes. These steps go beyond legal compliance—they’re lessons written in real-world experience from labs both big and small.
Reactions Driving Discovery and Use
Some people remember benzenesulfonyl chloride mostly from undergrad chemistry, where it shows the textbook idea of nucleophilic substitution. It’s more than a teaching tool, though. It powers the preparation of sulfonamides, dyes, pharmaceuticals, and other specialty chemicals. Scientists reach for it when they need to add the benzenesulfonyl group to molecules, offering a block for building more complex structures. The reaction with amines, producing sulfonamides, sits at the core of making antibiotics, antidiabetic drugs, and even herbicides. Even minor tweaks—swapping bases, shifting solvents, using microwave methods—can turn a slow, wasteful process into something clean, fast, and reliable.
Synonyms and the Landscape of Product Names
Names like Phenylsulfonyl chloride, Benzene sulfonic acid chloride, or PCI all point to the same stubborn, reactive liquid. In catalogs and chemical inventories, these synonyms reflect shifting preferences among countries and manufacturers. “BSC” turns up sometimes, but in most places, the direct chemical name still rules. In academic papers, the traditional benzenesulfonyl chloride appears, but anyone sourcing the material from suppliers stays alert to alternative synonyms to avoid mix-ups with sulfonic acids or their salts.
Practical Safety: Lessons from the Benchtop
Benzenesulfonyl chloride earns respect in the lab. Splash this on skin or breath in just a whiff, and the burning will linger for hours. Stories circulate about goggles fogging up, gloves melting or failing when this chemical spills. Safety standards keep evolving, often after close calls or near-misses. Fume hoods become home base for every transfer or reaction. Respirators and chemical-resistant suits aren’t excessive; they’re common sense. Labs now install continuous air monitors to catch leaks early, both for worker health and to head off environmental problems from accidental releases. These steps shape safe work, but a culture of caution—instilled by mentors and reinforced by experience—keeps both greenhorns and veterans on guard.
Carving Out a Niche: Where It Shows Up Daily
Benzenesulfonyl chloride’s reach stretches beyond synthetic labs. Its sulfonamide derivatives became the backbone of the first effective antibiotics, saving countless lives before modern drugs took over. Textile processing uses it to craft bright, stable dyes that bind tightly to fibers and resist fading. In agriculture, new crop protection agents wouldn’t exist without this molecule anchoring key parts of herbicide structures. Even today, cutting-edge research leans on old reactions, modifying benzenesulfonyl chloride to build novel molecules with fresh biological activity. Its presence in every sector, from medicine to agriculture to plastics, underscores its ongoing value.
Research on Toxicity and Exposure: Walking the Tightrope
Years of use led to piles of reports about what happens after exposure to benzenesulfonyl chloride. Skin contact brings intense burns, sometimes with permanent damage if not treated right away. Breathing its vapors means more than a cough—cumulative exposure can eat away at lung tissue. Animal studies put sharp limits on exposure, leading regulators to demand airtight standards. Waste disposal complicated matters, as any spill reacts with water, sparking acid clouds hazardous to both people and local ecosystems. Ongoing research digs deeper, looking at long-term, low-level effects for workers, and asking if breakdown products might harm water or soil. Hard-won wisdom emerged: only trained teams using strict precautions should handle this stuff, and facilities that ignore these warnings pay the price in worker health and regulatory fines.
Preparation: Keeping Up with Modern Needs
Modern preparation methods build on old routes but improve efficiency and safety. Early on, chemists used phosphorus pentachloride, but the toxic byproducts and tricky workup pushed industry toward thionyl chloride, which gives easier-to-purify products. Newer approaches focus on minimizing hazardous waste, using sealed reactors and better venting. Some labs now explore flow chemistry, letting them make benzenesulfonyl chloride exactly where it’s needed, reducing large-scale storage of a risky material. As technology marches on, greener and safer processes continue to emerge, though economic pressure weighs heavy since industrial customers watch cost per kilogram above all.
The Road Ahead: Imagining the Future
Chemists now look beyond making benzenesulfonyl chloride just for tried-and-true uses. Research chases derivatives that offer selective biological effects, especially as pathogens outsmart old drugs. Materials science explores new forms, hoping to use sulfonyl chloride chemistry to tweak polymer surfaces for electronics or medical implants. Regulatory scrutiny keeps tightening, pushing for safer manufacturing, real-time leak detection, and accountability for downstream waste. In my own experience, the best changes grow from small improvements tested by people on the ground—workers, students, and supervisors all giving feedback. Some see benzenesulfonyl chloride as a relic, but every year it serves as a stepping stone to new fields: personalized medicine, micro-manufacturing, and advanced diagnostics. The future hinges on balancing its remarkable reactivity with a sober respect for health and the environment—a lesson all chemicals ought to teach, but few do as well.
What is Benzenesulfonyl Chloride used for?
The Chemistry Scene in Everyday Life
Walk into a lab, even a classroom, and benzenesulfonyl chloride quickly pops up among the glassware and beakers. Sometimes it gets overlooked with all the talk about acids, bases, and more familiar chemicals. This compound plays a key role behind the scenes, helping create products that show up in medicine cabinets, garden sheds, and toolboxes.
Turning Simple Things Into Useful Tools
Think about the process where something plain becomes something special. Benzenesulfonyl chloride acts as a builder in organic chemistry, attaching itself to molecules and setting up what comes next. In practical terms, it’s used to make sulfonamides. These compounds helped turn the tide against bacterial infections before antibiotics became widespread. Today, they still pop up in some pharmaceuticals, quietly fighting infections and supporting other medicines.
Helping Chemists Make Better Products
Working with benzenesulfonyl chloride in the lab reminds me of laying down primer before painting a wall. It doesn’t look like much on its own, but everything after it goes smoother. This chemical turns weak alcohols and amines into better candidates for further reactions. In the classroom, it’s a go-to reagent for identifying or protecting certain groups (like amines) during synthesis. All those steps help keep reactions clean, efficient, and a little more reliable. The color changes and modest smell of benzenesulfonyl chloride often let you know the reaction is going as planned.
Industry Uses: Beyond the Test Tubes
Benzenesulfonyl chloride finds its way into industrial factories as well. Dyes and optical brighteners—the stuff that makes a white shirt look so fresh—often rely on this compound during production. Its use helps attach different groups to dye molecules, creating colors that last longer and look more vibrant.
Some companies count on benzenesulfonyl chloride for pesticides and specialty chemicals. Apply this compound the wrong way, and it can act as an irritant—to skin, eyes, and lungs—so proper handling belongs high on the safety checklist. Responsible use and good storage habits stay critical for those who work with it daily.
Pushing for Safer and Greener Chemistry
The story of benzenesulfonyl chloride reminds me of ongoing shifts in chemistry. Working with it brings a set of challenges. The fumes catch you off guard if you’re not careful, and safety glasses are a must. Over the years, lab managers and teachers stress the need for proper ventilation, sealed storage, and up-to-date safety training. Green chemistry approaches—like looking for less-hazardous reagents—start becoming more popular as people want safer labs and workplaces.
It’s worth talking about alternatives. Some researchers now develop processes that use milder sulfonylating agents or avoid certain hazardous steps altogether. They aim to keep the chemistry clean while reducing harm for workers and the environment, catching on as both industry regulations and public awareness move forward.
Why It All Matters
Benzenesulfonyl chloride might not grab headlines, but it leaves a mark on daily life, science, and safety. Learning about its uses keeps workers, students, and communities informed and encourages more responsible chemical practices. Real progress comes from asking tough questions, learning from experience, and always looking for ways to do things better.
What are the safety precautions when handling Benzenesulfonyl Chloride?
Understanding Benzenesulfonyl Chloride’s Hazards
Benzenesulfonyl chloride seems simple on paper—a colorless liquid, strong smell, used in chemical manufacturing and research labs worldwide. The trouble starts with how quickly it reacts with water or air. It gives off fumes that can sting the eyes, nose, and throat. Even a brief whiff can leave you coughing. Direct contact burns skin, leaves red welts or worse. These reactions aren’t rare mistakes—they hit even the careful, so old habits in the lab don’t measure up.
Personal Safety Steps
Gloves rule every interaction with this chemical. Standard latex won’t cut it. Nitrile or butyl gloves offer better protection because benzenesulfonyl chloride will eat straight through the thinner stuff. I’ve learned to double-check for small rips or pinholes before starting, as well as wearing safety goggles plus a face shield. Someone in my old lab learned the hard way: regular eyeglasses don’t block side splashes. He needed emergency rinsing and it rattled the whole team.
Lab coats help, but not just any shop-coat. I look for flame-resistant fabric and make sure sleeves are snapped tight. Respiratory gear comes out each time I handle it outside a fume hood. Simple paper masks offer nothing—only a full organic vapor respirator blocks the fumes that can fill your lungs in seconds.
Ventilation and Work Environment
Chemical fume hoods stand between lab workers and serious trouble. Benzenesulfonyl chloride doesn’t wait to release fumes. I always check the airflow gauge on the hood before bringing out the bottle. It’s tempting to skip if I’m only pouring a little—every shortcut raises the risk. Some have tried using these chemicals in quick jobs outdoors or by a window, but all that does is put everyone nearby at risk. I close the sash down as much as possible, keep containers tight, and never leave bottles open longer than it takes to measure.
Storage Smarts
Plastic breaks down and leaks, so glass containers with tight seals always store this chemical. I label every bottle with proper Hazard Communication Standard symbols, not just scrawling “benzenesulfonyl chloride” with a marker. That label helps everyone know exactly what’s inside, especially in emergencies. A cool, well-ventilated chemical cabinet does the job—never next to acids, bases, or water sources. Mishandled storage led to one incident at a friend’s workplace: someone put it closer to the rinse sink, a spill followed, and the cleanup required suits and respirators.
Dealing With Spills and Exposures
Spills demand attention, not panic. I keep spill kits stocked with absorbent pads, neutralizing solutions, and plenty of gloves. Water makes things worse; adding it causes more vapor. Using dry sand or sodium bicarbonate controls the spread. Rinsing skin immediately with running water for at least fifteen minutes keeps damage manageable. If anyone breathes in the fumes, fresh air and medical help come first, not assignments or notes.
Training and Team Awareness
Experience alone never replaces training. Newcomers hear real-life stories, not just rules, in labs that take safety seriously. Regular drills on what to do behind the hood, who to call for spills, and how to use the eyewash station make all the difference. Every team checks each other’s gear, and speaks up if something feels off. People often ignore the early signs—itchy skin, a funny taste in the air. Sharing those moments makes colleagues more likely to avoid them later.
What is the chemical formula and structure of Benzenesulfonyl Chloride?
Getting to Know Benzenesulfonyl Chloride
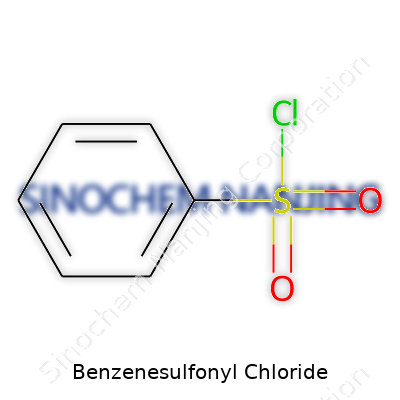
In chemistry class, the smell of certain chemicals leaves a lasting impression. Benzenesulfonyl chloride stands out, not just for its strong odor, but for the way it opens up doorways in organic synthesis. This compound has the formula C6H5SO2Cl. What this means in plain terms: you have a benzene ring, much like those hexagons you see in textbooks, holding tight to a sulfonyl chloride group.
Breaking Down the Structure
Let’s picture the molecule. The benzene ring shows up as six carbon atoms linked together by alternating double bonds, set up in a flat ring. Hanging off one of these carbons sits a sulfonyl chloride group, made up of a sulfur double-bonded to two oxygen atoms and single-bonded to a chlorine atom. This arrangement forms a classic functional group in organic chemistry—the sulfonyl chloride.
There isn’t a path through undergraduate labs without running into aromatic sulfonyl chlorides. The unique combination of the benzene ring and the reactive -SO2Cl group means chemists can build more complex molecules, especially for the pharmaceutical and dye sectors. You learn quickly that this compound, with that sharp, choking aroma, plays a central role in moving pieces around in reaction schemes.
Real-World Importance
On paper, a formula looks precise and distant. Working with benzenesulfonyl chloride, though, you see how it fits into the ebb and flow of the chemical industry. It crops up as a reagent for protecting the amino group in organic synthesis, something you can’t sidestep if you’re building molecules like sulfa drugs or prepping intermediates for agrochemicals. Its reactive nature means it can easily transfer sulfonyl groups, giving chemists more room to play with structure and function.
Handling benzenesulfonyl chloride in the lab made me respect both its usefulness and danger. Quickly, you learn not to open a bottle outside the fume hood. That pungency tells you something about its volatility—and its ability to irritate skin and breathing passages reminds us that chemical utility comes paired with responsibility.
Risks and Solutions
Exposure to benzenesulfonyl chloride causes more than a bad workday. It can lead to skin burns and respiratory issues. Data from workplace safety boards show that cases of chemical burns or accidental inhalation still happen, especially when personal protective equipment lapses or fume hoods fail.
Safety rules alone never feel like enough. We need regular reminders and hands-on demonstrations—not just warnings on a label. If institutions introduced safer, less volatile substitutes for key steps, or pushed for better ventilation equipment, fewer hazardous exposures would happen. Labs also benefit from clear spill response plans and easy access to emergency showers and eyewash stations. Eyewitness stories from seasoned chemists often stick better than generic safety modules, because experience shows just how quickly a careless moment with a reactive chemical can spiral into a health crisis.
The Chemical Community’s Responsibility
Benzenesulfonyl chloride sits at an intersection of usefulness and risk. As science keeps rushing forward, the chemical community can lead by example: strict labeling, careful training, and ongoing innovation for safer reagents. You don’t forget the tools that build the foundation of organic chemistry, but investing in people’s safety while using them matters just as much. Ultimately, staying alert and updating best practices benefits everyone in the lab—students and professionals alike—now and well into the future.
How should Benzenesulfonyl Chloride be stored?
Getting Storage Right Means Fewer Headaches Later
Storing benzenesulfonyl chloride isn’t something to gloss over if you spend time in a lab, warehouse, or even a college storeroom. Problems with this chemical can get out of hand quick—just a splash on the skin or inhaling its fumes ruins your day. I’ve run across sloppy storage conditions before, and believe me, fixing leaks or mopping up accidental spills leaves a lasting impression. A little care goes a long way.
Keep Moisture Far Away
Benzenesulfonyl chloride reacts with water, giving off dangerous fumes and creating a corrosive mess. Open bottles next to a sink, humid storerooms, or anywhere you get rain seeping in lay the groundwork for trouble. Instead, store the chemical in a dry place—an airtight, sealed container gets the job done well. Simple silica gel packets in the cabinet help soak up stray moisture, and making sure the container closes tightly becomes a habit for anyone doing the job right.
Glass or Plastic—But Not Just Any Kind
Containers matter almost as much as the room. I always choose thick glass (not the type that shatters if you look at it wrong) or certain plastics that don’t degrade. Polyethylene or Teflon containers stay in good shape. Thin plastic, old jam jars, or recycled water bottles don’t cut it. Investing in the correct vessel means lowering the risk of leaks or chemical reactions.
Ventilation Needs Attention
Fumes sneak up on you, and benzenesulfonyl chloride doesn’t stay silent. It gives off hydrogen chloride gas if it leaks, and nobody wants to smell that. Good airflow makes a world of difference, so cabinets with built-in ventilation or a storage room with exhaust fans bring down exposure risk. Never set containers near the main walkways, where a wayward hit by a cart and the next thing you know, everyone’s out coughing in the hallway.
Temperature Control Brings Peace of Mind
This chemical stays happiest at room temperature. Heat ramps up pressure and reactivity, and exposure to sunlight can break down the container or the product. Keeping benzenesulfonyl chloride away from windows, heaters, or steam pipes helps hold off disasters. Even just leaving it in the back of a hot car on the way from the supplier to the lab can lead to pressure build-up—something I’ve learned to avoid.
Store Apart From Incompatible Chemicals
Mix-ups often start with careless shelving. Organics like amines, strong bases, or alcohols trigger dangerous reactions with benzenesulfonyl chloride. Separate storage solves much of this—different shelves or cabinets, clearly labeled, make it almost impossible to grab two enemies at the same time. In research settings, color-coded labels save more than a few headaches.
Training Matters More Than a Sign on the Door
A locked cabinet won’t mean much if the person opening it doesn’t know what to expect. Training goes beyond reading a label or glancing at a safety sheet. Walking through the storage routine, pointing out what can go wrong, and showing where to find safety gear sticks better. Regular checks and honest conversations add another layer of confidence.
Final Tips from the Trenches
Clean up spills right away with gloves and proper cleanup kits, keep emergency contacts posted in the storage area, and toss damaged containers instead of patching them up. With everyday common sense and respect for the risks, storing benzenesulfonyl chloride won’t be something you worry about at night.
What are the potential health hazards of Benzenesulfonyl Chloride?
What Workers Face Every Day
Stepping into a chemical facility, people face invisible dangers, and benzenesulfonyl chloride joins a list of substances that demand respect. The sharp, acrid odor signals how quickly it irritates eyes and airways. One small splash in the eye or on the skin brings rapid burning pain, redness, and swelling. For many, contact brings stories of hospital trips and weeks or even months of lingering issues. I once visited a research lab after an accident with this chemical—colleagues talked about a mist that drifted in the air, turning a routine day into a race to wash out eyes and strip off clothes.
How Exposure Affects the Body
Most chemicals in this group behave as strong irritants. Benzenesulfonyl chloride acts fast: the liquid can cause chemical burns, blisters, and tissue damage. Vapors turn into hydrochloric acid in moist tissue, making inhalation a threat to your lungs and throat. Chronic exposure over weeks or months deepens the risks. Some researchers report asthma-like symptoms, chronic bronchitis, and skin sensitivity that triggers rashes with only light contact. Prolonged skin exposure may even lead to ulcerations that take weeks to heal.
Longer Term Concerns
Repeated exposure sometimes leads to a lifelong sensitivity. Medical journals have several case studies where continued workplace exposure caused lasting respiratory issues, with people developing asthma long after they stopped working in the lab. Some initial research suggests benzenesulfonyl chloride may carry carcinogenic properties too, though definitive proof remains elusive so far. Even short bursts of exposure leave people with headaches, dizziness, and a lingering metallic taste. It often takes only a small, airborne leak to make an entire room unfit for hours.
Ripple Effect in the Community
The risks do not end at the facility fence. Poor handling and storage sometimes send vapors wafting into nearby neighborhoods. In 2023, I spoke with workers in a chemical plant who described mornings with burning eyes, and local residents have filed complaints about unexplained nosebleeds and chest tightness. Accidental releases strain local hospitals and sometimes spark evacuations, especially if wind conditions push fumes toward residential streets.
What It Takes to Lower the Risk
Managing benzenesulfonyl chloride means vigilance every single day. Strong ventilation systems cut down airborne levels inside, and exhaust hoods become essential. Anyone working near the substance needs high-quality gloves, goggles, and protective suits—and a strict habit of double-checking for leaks. Quick access to emergency eyewash stations and showers shifts an accident from life-changing to merely inconvenient. Most experienced chemists keep detailed protocols posted in every lab, reminding newcomers not to cut corners.
Better monitoring and regular health screenings can flag problems early. Safety teams should track symptoms, not just spill numbers, because real recovery sometimes starts with a quiet conversation about irritated skin or a stubborn cough. Regulations play a part, but real safety happens face-to-face: training, reminders, and people willing to speak up before an accident happens.
Informed Decisions
Knowing the risks shaped how I moved around labs and factories. Anyone working with benzenesulfonyl chloride or living nearby deserves this same information—not just to protect health, but to demand improvements where gaps appear. Knowledge empowers workers to advocate for better equipment, cleaner air, and safer handling practices. Progress happens through awareness, vigilance, and the courage to ask tough questions about the chemicals that shape daily life.
| Names | |
| Preferred IUPAC name | Benzenesulfonyl chloride |
| Other names |
Benzenesulfonic acid chloride Benzenesulfonic chloride Phenylsulfonyl chloride Benzene sulfochloride |
| Pronunciation | /ˌbɛn.ziːnˌsʌl.fə.nɪl ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 98-09-9 |
| Beilstein Reference | 1209249 |
| ChEBI | CHEBI:52050 |
| ChEMBL | CHEMBL1235020 |
| ChemSpider | 2141 |
| DrugBank | DB08703 |
| ECHA InfoCard | 100.006.734 |
| EC Number | 211-698-6 |
| Gmelin Reference | Gm 824 |
| KEGG | C00692 |
| MeSH | D001568 |
| PubChem CID | 8715 |
| RTECS number | BO8925000 |
| UNII | FBL8X5565A |
| UN number | UN2612 |
| Properties | |
| Chemical formula | C6H5SO2Cl |
| Molar mass | Molar mass: 176.63 g/mol |
| Appearance | White to yellowish crystalline solid |
| Odor | Pungent odor |
| Density | 1.38 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.35 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | -6.5 |
| Basicity (pKb) | Basicity (pKb): 10.5 |
| Magnetic susceptibility (χ) | -59.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.587 |
| Viscosity | 2.14 mPa·s (20 °C) |
| Dipole moment | 3.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -259.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -858.6 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, causes burns, harmful by inhalation, toxic if swallowed, reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H314, H302, H317, H335 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P337+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 139°F (59°C) |
| Autoignition temperature | 205°C |
| Lethal dose or concentration | LD50 oral rat 960 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (rat, oral) |
| NIOSH | DS8050000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 20 ppm |
| Related compounds | |
| Related compounds |
Benzene Benzenesulfinic acid Benzenesulfonic acid Benzenesulfonamide |

