Benzenediazonium Nitrate: An In-depth Commentary
Historical Development
Back in the 19th century, European chemists wandered into unexplored territory while hunting for new reactions in organic chemistry. The birth of diazonium chemistry traces its origins to the mid-1850s—when Peter Griess, working on nitrous acid’s odd effects on aromatic amines, wrote his name into history books. Griess’s experiments started with simple aniline, but soon gave rise to diazonium salts like benzenediazonium nitrate, which introduced a new way of making azo dyes. The industrial surge of colored fabrics and synthetic dyes could not have arrived when it did without such discoveries. By working with diazonium salts and harnessing their reactivity, chemists across Europe designed better pigments, pharmaceuticals, and even the first photosensitive materials. The groundwork paved during the next century shows how one class of compounds can change the way researchers approach chemical synthesis.
Product Overview
Benzenediazonium nitrate isn’t something an average person has in their garage or under the sink. This compound, known for a short life and a big impact, sits in supply cabinets of research labs for good reason. It doesn’t travel alone; it’s usually kept under specific conditions because of its instability and potential hazards. Unlike stable household chemicals, benzenediazonium nitrate is valued for its ability to introduce new groups onto benzene rings. This makes it an important intermediate in preparing azo dyes, pharmaceuticals, and organic materials. Many in the research community have relied on its reactivity for decades. Despite risk, its value lies in the doors it opens for chemical modification.
Physical & Chemical Properties
This salt forms as a pale-yellow to colorless solid, usually as a crystalline powder. It dissolves readily in water and polar solvents. Its chemical behavior draws attention: it reacts explosively if subjected to friction, heat, or shock. Chemists know it cannot be stockpiled; even minor environmental triggers can set off a chain reaction, converting it into nitrogen gas and leaving behind phenol derivatives. Because of these risks, chemists prepare it on demand, then use it immediately or store it in ice-cold environments for short periods. The reactivity towards nucleophiles makes it a go-to for arylations or azo coupling reactions. Significant volatility, combined with the release of nitrogen, makes handling tricky and demands a steady hand.
Technical Specifications & Labeling
Strict technical specifications surround the shipment and use of benzenediazonium nitrate. Labels flag its explosive nature, urging careful handling and emphasizing the need for cold storage at temperatures just above freezing. The product must be pure—usually upwards of 98%—to avoid side reactions during experiments. Detailed safety data covers not only storage requirements but also instructions for neutralization in case of accidental spillage. Transportation follows regulations that treat it like other hazardous materials: packaging requirements, limited airflow, and protected containers are a must. Research suppliers clearly display the chemical’s UN number and hazard statements, making sure the risks aren’t buried in fine print.
Preparation Method
In professional laboratories, chemists build benzenediazonium nitrate through the diazotization reaction. They start by dissolving aniline in cold dilute nitric acid, then add sodium nitrite solution, also kept ice-cold. Temperature control remains critical; even a small jump above 5 °C can spell disaster, as the compound may decompose violently. The intermediate forms—nitrous acid and the diazonium cation—combine with nitrate ions in solution, crystallizing out as the desired salt. After formation, rapid filtration and cooling minimize decomposition and maximize safety. Every chemist in the lab pays attention here; the price for mistakes includes ruined experiments and potential injuries.
Chemical Reactions & Modifications
Chemists turn to benzenediazonium nitrate as a key player in substitution reactions. By replacing the diazonium group with halides, hydroxyls, or other functional groups, they forge molecules difficult to build through other routes. This reactivity underpins the creation of azo dyes, which throw vivid colors across textiles worldwide. Sometimes, the salt reacts with copper(I) salts—a process known as the Sandmeyer reaction—to provide chlorinated or brominated benzenes. The versatility doesn’t stop there. Coupling with electron-rich aromatics, it forms complex azo compounds, whose shades depend on ring substituents. These reactions illustrate the power and danger of this molecule: useful transformations hinge on the fragile stability of the diazonium group.
Synonyms & Product Names
Benzenediazonium nitrate pops up in the literature under several names. Chemists might call it "benzenediazonium nitrate", "phenyldiazonium nitrate", or sometimes just "diazonium nitrate" if context makes things clear. In catalogs, chemical suppliers sometimes use systematic names but always highlight its explosive properties in bold. Researchers recognize it quickly by its core structure: a benzene ring tied to a diazonium group, carrying nitrate as its counterion.
Safety & Operational Standards
Anyone handling benzenediazonium nitrate knows not to cut corners on safety. Laboratories restrict its use to qualified personnel, outfitted with glass shields, gloves, heavy coats, and safety eyewear. Cold rooms and ice baths remain on standby, reducing risk of runaway reactions. Waste must be neutralized with care—adding base in small portions and constant stirring to avoid local heating that would spark decomposition and gas evolution. Fire extinguishers and inert gas supplies should be within arm’s reach. Regular training refreshes everyone’s memory about stored volumes, allowed concentrations, and disposal procedures. Research organizations adopt protocols in line with global chemical safety boards, focusing not just on accidents but long-term health risks for staff.
Application Area
The legacy of benzenediazonium nitrate stretches into multiple industries. Textile dye companies once relied almost entirely on diazonium chemistry for a palette wider than nature ever offered. Researchers discovered new antibiotics and analgesics using the salt as a stepping stone to manipulate aromatic rings. Advanced materials science sees its value: it helps modify electrodes or build tailored sensory polymers. Even the photo industry, responsible for early blueprint paper and lithography, ran on reactions launched by diazonium salts. Newer developments center around organic electronics and catalysis, but the core challenge—controlling an extremely reactive molecule—always guides application choices.
Research & Development
R&D efforts focus on making benzenediazonium nitrate safer to use and opening new pathways for industrial chemistry. Chemists design reactions with greener solvents or switch to flow chemistry, using microreactors that lower risks by producing tiny amounts at a time. Computational models predict side reactions, helping teams optimize conditions before they step into the lab. Synthetic organic chemistry relies on these salts, whether searching for new agrochemicals or boosting the production yields of life-saving drugs. Efforts also address sustainability, replacing hazardous reagents and finding ways to reclaim or neutralize waste byproducts.
Toxicity Research
Diazonium compounds, including benzenediazonium nitrate, present toxicity risks—ranging from acute skin burns to systemic effects in cases of inhalation or accidental ingestion. Toxicology studies document oxidative stress and methemoglobinemia as potential concerns. Repeated exposure can damage liver and kidneys, and some breakdown products have links to mutagenicity. As a result, responsible labs invest both time and money checking ventilation systems, updating personal protective equipment, and monitoring work areas for residues. Researchers follow detailed incident reports and case studies to adapt protocols, often going beyond regulatory minimums. Because even brief lapses in concentration can have lasting health consequences, chemists pass down stories and reminders that stick with everyone who works with diazonium salts.
Future Prospects
As chemistry continues to evolve, the story of benzenediazonium nitrate is far from finished. There’s a growing push toward safer derivatives, ones delivering the same reactivity but with lower risks. Automation, better engineering controls, and remote reaction monitoring aim to cut lab accidents. Our digital age will likely see more computational work before any hands-on handling, reducing waste and introducing new classes of functional materials quickly. In emerging fields such as targeted drug design or molecular electronics, chemists still use diazonium salts as key tools, reimagining old reactions for new challenges. With stricter regulations on hazardous compounds, manufacturers must rethink supply and packaging to support the next wave of chemical discovery without repeating the safety mistakes of the past.
What is Benzenediazonium Nitrate used for?
The Science Behind the Compound
Benzenediazonium nitrate isn’t common in daily conversation, but its impact stretches wide in advanced chemistry labs and specialized industries. Made from benzene and treated with nitrous acid, this chemical forms a diazonium salt. The “nitrate” part signals that it’s paired with a nitrate ion, making the substance not just chemically interesting, but also notably reactive.
Main Uses: More Than Just a Lab Curiosity
In my years around people working in organic chemistry, benzenediazonium nitrate pops up where precise molecular changes matter. It’s mostly used as an intermediate—a building block that chemists push to other, more complex compounds. For example, educators use it in advanced chemistry classes to show how benzene’s structure can be changed or reorganized. These transformations are possible because this compound makes it easy to swap out the nitrogen group for something else, like a halogen atom or a hydroxyl group.
Dye manufacturing still leans on this compound because it helps chemists create ‘azo’ dyes. These bright, lasting colors show up in textiles, food coloring, and inks. Walk through most fabric stores, and you’ll see shades that owe their brilliance to a process starting with chemicals like benzenediazonium nitrate. The reaction that links two aromatic rings (a process called coupling) stands at the heart of what makes these dyes so stable and colorful. Without satisfactory intermediates, many industries would struggle to deliver the range of colors people take for granted every day.
Challenging Safety Concerns
No amount of practical use changes the reality that benzenediazonium nitrate brings serious safety risks. Chemists talk about its instability even at room temperature; it can explode if roughly handled or too much builds up. That’s not some abstract danger. Inattention means people get hurt. The chemical’s hazardous behavior means only experts should ever handle it, and then only with controlled temperatures and equipment designed to prevent shock or friction. In the day-to-day work of an experienced chemist, a moment’s lapse around diazonium salts could turn dangerous in a heartbeat.
Alternative compounds exist, but they don’t always offer the same effectiveness, especially for making certain dyes or pharmaceuticals. Local storage rules, specialized glassware, and clear communication protocols all help reduce risk. Factories and university labs train strictly for this kind of chemical. Nevertheless, accidents remind us just how stubborn and unpredictable some chemical substances can be.
Looking for Safer Solutions
Synthetic chemistry keeps evolving. Some researchers look for replacements for unstable diazonium compounds, hoping to find more stable, less hazardous options that can pull off the same transformations. Experts also test new digital modeling to predict instability before scaling up, and work on faster, safer methods to generate the same effects in a controlled way. Green chemistry initiatives ask inventors to design production that reduces dangerous byproducts as much as possible. That challenge hasn’t been fully solved, but every year, new techniques push the field further.
Benzenediazonium nitrate’s story highlights how basic science can shape technical progress—while always reminding us to respect what powerful chemistry can do, both for better and for harm.
Is Benzenediazonium Nitrate stable at room temperature?
Why Stability Matters with Benzenediazonium Nitrate
Benzenediazonium nitrate doesn’t usually show up on home chemistry sets, and there’s a reason for that. The compound draws attention among chemists for its notorious instability, especially at room temperature. Stability, sometimes taken for granted in other molecules, becomes more than a technical issue with diazonium salts—it shapes every aspect of how, where, and if they get used outside the lab.
Let’s try to picture this chemical as more than a formula. It has two reputation-shaping features: a diazonium group and a nitrate group, both known for contributing to explosive properties. Put them together, and you’ve got a molecule primed to break apart fast and violently. Research and practical experience confirm this. Take the classic work by chemical safety experts at universities like Cambridge and MIT—these teams report that benzenediazonium nitrate can detonate if even a small sample dries out or gets slightly warmed. Handling even milligram quantities can cause an accident with disastrous results.
Real World Evidence: Danger Comes Easily
Anyone tempted to handle this compound outside a professional lab should know about its track record. Laboratories worldwide keep diazonium nitrates behind reinforced barriers, and they have good reason. The United States Chemical Safety Board, along with national chemical safety organizations in Germany and Japan, have documented injuries—sometimes fatal ones—linked to mishandling. Benzenediazonium nitrate sometimes makes the news for lab explosions at room temperature to slightly elevated temperatures, catching scientists off-guard.
A closer look at its chemistry helps explain the fear. The bond holding the diazonium group together has an urge to break, releasing nitrogen gas. If the stress builds up inside the crystalline solid, the release turns violent. Relatively mild nudges—friction from a spatula, slight heat, or even static discharge—can cause the whole thing to go off. Organic chemistry classes teach these facts not to scare, but to save lives.
Safer Alternatives and Sensible Practices
So what are scientists doing to avoid another accident? Many labs shift towards safer substitutes—tosylate and tetrafluoroborate salts, for example, show more stability, letting chemists carry out their reactions without risking an explosion. Developed protocols call for making diazonium nitrates right before using them, never storing or transporting the dry compound at room temperature. Stories circulate in safety seminars about the dangers, keeping new researchers alert.
Institutions enforce strict controls. Staff enforce temperature limits and humidity controls. Training programs run through what can happen with just a few careless minutes—like that time a graduate student learned the hard way, a clean-up turned deadly because someone didn’t pay the warning labels enough respect. For anybody working in a lab, or overseeing one, learning these lessons seems non-negotiable.
Putting Respect Back Into Chemistry
Benzenediazonium nitrate doesn’t make headlines the same way other dangerous substances do, but it carries a risk that sits in the background of chemical laboratories across the globe. Good safety practices protect the people driven by curiosity and necessity to keep experimenting. The solution never comes down to just crossing your fingers—or treating warnings like red tape. Chemical respect saves lives.
How should Benzenediazonium Nitrate be stored?
What Makes Benzenediazonium Nitrate Stand Out?
Benzenediazonium nitrate stands apart from typical lab reagents. This dry compound may look unremarkable, but beneath the surface sits a real hazard. A small nudge from friction, heat, or even static can trigger it. Chemists know all too well that a single lapse in judgment may invite disaster. The nitrate ion only adds to the risk, making accidental ignition even more likely compared to other diazonium salts.
Storing with Safety Top of Mind
Experience teaches that a locked, segregated chemical cabinet goes a long way. Placing benzenediazonium nitrate beside ordinary reagents in a general storage cabinet can spell trouble. Humidity and light creep in more easily than you think. Drier, cooler spaces help to slow decomposition and keep this compound from acting up. Flammable storage rooms or cabinets near direct sunlight create more risk, not less. Fire detection and suppression equipment should stand ready nearby.
Most chemists recommend glass or PTFE containers, never metal or plastics that build static charges. Thin plastic makes an inviting home for static buildup, which sparks shatter lives more often than most care to admit. Labels deserve clear language, so colleagues know exactly which container holds potential peril. Every time I stepped into a lab, the first glance went to labels and lids, checking nothing sat open by mistake.
Keeping benzenediazonium nitrate apart from reducing agents, strong bases, or combustibles makes sense. Experiences with minor shelf fires or spills remind us that just a stray grain near the wrong solvent may spark a serious sequence.
Small Quantities, Better Odds
In practice, no one stores pounds of this compound unless they want a headline for the wrong reason. Synthesis teams work with fresh batches, making just enough for each project. Storing more than a few grams at a time only increases the chance something goes wrong. Even the smallest quantity in a sealed vial can react uncontrollably under certain conditions.
Old samples become unpredictable. Chemists and safety officers stress routine reviews of storage records. Unlabeled or outdated stock should go to waste disposal without delay, never waiting for someone “brave enough” to test if it’s still stable.
Backup Systems and Emergency Planning
A single layer of defense won’t always hold. That’s where backup planning takes top priority. Good labs keep spill control kits and sand buckets close. Evacuation routes ought to stay unobstructed, and staff should run drills that don’t just stay on paper. Posters and quick-reference guides help everyone recall the steps in a rush.
Stories float through the scientific community about labs that failed to respect the volatility of diazonium salts. Explosions carve out lessons that get passed down from one technician to the next. Many learned that simple habits—checking humidity, double-sealing vials, only opening them on grounded, antistatic work surfaces—can keep the odds in your favor.
Training: The Human Factor
No policy replaces personal responsibility. One coworker with little training can undo the care of a dozen experts. That’s why recurring safety workshops carry so much weight, especially for visitors or new students in academic environments. Senior staff who share their real-world stories bridge the gap between textbook procedures and daily vigilance.
Putting safety first starts with respecting the risks. Everyone benefits when the most dangerous compounds are stored with intention—not out of fear, but out of earned experience and respect for what can go wrong. Benzenediazonium nitrate deserves nothing less.
What are the hazards associated with Benzenediazonium Nitrate?
This Chemical Packs a Punch
Anyone who has spent time in a chemistry lab knows that not all liquids, powders, or crystals carry the same risks. Some news stories only mention the hazards after a major spill or accident. Benzenediazonium nitrate, with its intimidating name, sits far from the fleeting risks of spilled vinegar or table salt. Safety discussions about this compound aren't just academic. They can save lives.
Explosion Is Not a Remote Possibility
This chemical falls into the class of diazonium salts—compounds famous among chemists for their instability. Mix benzene’s aromatic ring with a diazonium group, throw in nitrate as a counterion, and the product doesn’t like sitting still. It can detonate from friction, bumps, or just being left in a warm room. Laboratory stories passed down in university corridors sometimes mention shattered glass and ruined benches after someone underestimated this white crystalline powder.
Breathing Is Not Safe Around It
It’s more than just an explosion risk. Even in small quantities, exposure risks run high. Benzenediazonium nitrate doesn’t just look innocent; dust in the air can irritate lungs and eyes. Skin burns are real. My own gloved hands tingled after an accidental brush during graduate school, and friends reported headaches after fume hood leaks. Symptoms like dizziness, coughing, or eye redness signal real danger, not just minor inconvenience.
No Firefighter Wants to Face This Flame
Firefighters hate dealing with chemicals that explode or erupt with toxic fumes. Benzenediazonium nitrate decomposes, shooting out gases like nitrogen oxides when ignition occurs. These gases poison air, set off alarms in emergency rooms, and complicate evacuations. Fire blankets and water won’t solve the problem. The heat or even static sparks can set off a chain reaction. I heard from a hazmat specialist that even small bottles require full protective suits during cleanup.
It Doesn’t Like Ordinary Containers
Storage brings a new list of headaches. Some plastics react with it. Sunlight, heat, even shaking from moving boxes can trigger decomposition. Most research labs store it in tightly controlled environments, isolated from incompatible chemicals such as metals and reducing agents. Glass vials, double-sealed, in a cool dark cabinet, show up on safety data sheets. After an incident in a student lab, I watched our chemical inventory shrink, with this compound quickly pulled off shelves and flagged for immediate disposal.
Handling Calls for Skill and Respect
Lab training doesn’t gloss over this one. Protocols call for face shields, thick gloves, and blasting the fume hood fan at full power. Waste disposal takes certified containers, not ordinary trash bins. Any spill means evacuations and special response teams. Regulations exist for a reason: the risk isn’t just for the person making a batch, but neighbors and entire buildings. I have never seen a research supervisor relax around this compound, not even for a minute.
What Should Change?
Information alone isn’t enough. Safety drills must stay up to date, with all lab members familiar with the evacuation routes and spill procedures. Chemists should favor alternative reagents if possible. Waste collection should happen more often, reducing storage time. Institutions can invest in better containment and sensor systems. Supply limits should stay strict, with no unnecessary stockpiling. Keeping the community safe begins with real respect—and real caution—around compounds like benzenediazonium nitrate.
How is Benzenediazonium Nitrate synthesized?
The Chemical Steps Matter
In the corner of a well-ventilated lab, the route to making benzenediazonium nitrate looks simple on paper. In practice, each step brings a story, both fascinating and risky. This compound builds off aniline, a clear liquid with a fishy odor that’s always reminded me more of undercooked beans than anything chemical. The goal is to coax aniline into a reactive form without letting the reaction run wild. Liquid aniline mixes with nitric acid and hydrochloric acid, and sodium nitrite acts as the star chemical, generating nitrous acid right in the mixing flask. The temperature sits just above freezing, often under ten degrees Celsius. Go warmer, and the product breaks down, sometimes violently, which keeps even seasoned chemists on their toes.
Watching this process, a mix of tension and focus grows—no two syntheses play out exactly the same. The pale solution turns almost electric with yellow tints. The smell, combining sharp acids and an earthy base note, is tough to forget. With steady swirling and careful control, the mixture creates benzenediazonium chloride first. Nitrate then joins the dance. Dripping in a bit of sodium nitrate or potassium nitrate slowly swaps out the chloride for nitrate, giving benzenediazonium nitrate—used in coupling reactions and organic syntheses around the world. Each step draws on fundamental concepts from organic chemistry, but the practice feels more like careful cooking with explosive ingredients than textbook chemistry.
Why This Matters Outside the Lab
Synthesizing compounds like benzenediazonium nitrate changed how dyes, medicines, and polymers get manufactured. These diazonium ions, once made stable enough to survive long enough to react with something else, open new pathways to aromatic compounds that can’t be built any other way. Without diazonium chemistry, the color on your favorite shirt, the working ingredient in antiviral drugs, or the simple rubber in tires wouldn’t be nearly as easy to produce or as affordable. Skilled lab work, not just theory, keeps this field thriving. I’ve seen talented researchers work wonders by adjusting drop rates and using purified glassware, getting higher yields than anyone could from guessing or rushing the steps.
The real trick, though, isn’t only about making the nitrate salt. Safety always pulls focus—diazonium nitrates have a reputation for surprise decomposition. Even a small spill or shock can kick off an explosive reaction. Labs that forget to keep temperatures down or mishandle their reagents learn that lesson fast. Over the years, strict rules and up-to-date procedures have saved both chemistry students and industry veterans. The difference between a successful synthesis and a dangerous accident often comes down to respect for the tools and a knack for spotting trouble early.
Moving Forward: Safer and Cleaner Chemistry
Green chemistry principles push modern researchers to rethink classic syntheses like this one. Generating less acidic waste, using milder reagents, and designing better containment gear have all made their way into standard operating procedures. Schools and startups now invest in closed systems that contain fumes, recycle acids, and keep heat under control. Based on first-hand experience, open discussion among chemists and a culture that encourages reporting near-misses makes a world of difference collecting reliable data—especially on how to avoid trouble.
In the end, the road from aniline to benzenediazonium nitrate serves as a perfect example of why chemistry still depends on sharp observation, constant learning, and respect for the smallest detail. Every step adds to a tradition of hands-on experience backing up scientific progress, not just theory or hope.
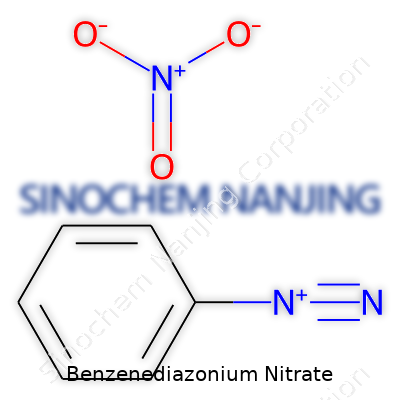
| Names | |
| Preferred IUPAC name | Benzenediazonium nitrate |
| Other names |
Benzenediazonium nitrate Diazonium, benzenediazo-, nitrate Phenyldiazonium nitrate |
| Pronunciation | /ˌbɛn.ziːn.daɪˈæz.oʊ.ni.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | [578-61-2] |
| Beilstein Reference | 1209275 |
| ChEBI | CHEBI:75956 |
| ChEMBL | CHEMBL2340688 |
| ChemSpider | 12694177 |
| DrugBank | DB11438 |
| ECHA InfoCard | 100.023.706 |
| EC Number | 208-888-1 |
| Gmelin Reference | Gmelin Reference: 10804 |
| KEGG | C00971 |
| MeSH | D013578 |
| PubChem CID | 12858573 |
| RTECS number | GE9100000 |
| UNII | 7Z0B72CKF9 |
| UN number | UN3384 |
| CompTox Dashboard (EPA) | DTXSID2020857 |
| Properties | |
| Chemical formula | C6H5N2NO3 |
| Molar mass | 155.12 g/mol |
| Appearance | White to yellowish crystalline powder |
| Odor | Odorless |
| Density | 1.69 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.5 |
| Vapor pressure | Vapor pressure: 0.0253 mmHg (25°C) |
| Acidity (pKa) | -2.9 |
| Basicity (pKb) | 7.0 |
| Magnetic susceptibility (χ) | -82.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.641 |
| Dipole moment | 1.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 122.6 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | +137.5 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | Benzenediazonium Nitrate: ΔcH⦵298 = -1267 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V04CX27 |
| Hazards | |
| Main hazards | Explosive, Toxic, Harmful if inhaled, Causes skin and eye irritation |
| GHS labelling | GHS02, GHS03, GHS06 |
| Pictograms | GHS01,GHS02,GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | H271, H300, H301, H314, H330, H410 |
| Precautionary statements | P261, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 3-3-1-OX |
| Autoignition temperature | Autoignition temperature: 110°C |
| Lethal dose or concentration | LD50 (oral, rat): 50 mg/kg |
| LD50 (median dose) | LD50 (median dose): 50 mg/kg (intravenous, mouse) |
| NIOSH | B0144 |
| PEL (Permissible) | No PEL established |
| REL (Recommended) | Recommended: 2C |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Benzenediazonium chloride Benzenediazonium tetrafluoroborate Phenyldiazene Aniline Nitrobenzene |

