Barium Selenite: An In-Depth Look
Historical Development
Barium selenite didn’t grab the spotlight overnight. It took root in the landscape of inorganic chemistry in the late nineteenth century, back when researchers scoured for minerals that could drive advances in glassmaking, pigments, and analytical reagents. Old school chemists noticed the strange combination of barium’s weight and selenite’s secluded presence in rare mineral ores. Building on the work of Berzelius, who isolated selenium in 1817, later generations learned to handle and purify compounds like barium selenite with confidence, using careful precipitation work. That small leap opened doors for industrial researchers decades later, helping them hint at its role in making specialty glasses and refining selenium recovery from ores. Every step, from basic isolation to small-scale synthesis, showed how methodical advances in laboratory chemistry could bring another obscure compound into practical circulation.
Product Overview
This compound, known to chemists as barium selenite or barium oxide selenate, shows itself as a colorless, crystalline solid. Industrial suppliers market it to labs hunting for a reliable selenium source or seeking non-chlorinated barium salts. Even in small volumes, reputable distributors list it specifically for analytical chemistry, catalyst production, and a growing group of niche manufacturing requests. The solid’s firmness and lack of strong color fade into the background, but that’s the hook for glassworkers and researchers who crave consistent formulae without surprise impurities. Bulk buyers often turn to quality inspection reports and certificate of analysis documentation to keep their compliance teams happy and supply chains consistent.
Physical & Chemical Properties
Barium selenite doesn’t sparkle like a fancy crystal, but its stubborn insolubility in water sets it apart. An off-white to colorless crystalline powder, it feels heavy for its size—thanks to barium’s density. Its chemical formula, BaSeO3, marks out a one-to-one pairing of barium and selenite units. It won’t dissolve freely in cold water, but douses in acids split it up and liberate selenium. It won’t melt easily, holding out until it’s heated past 500°C, and even then it can decompose rather than melt the way common salts do. Mock up a beaker of barium selenite and you’ll notice little smell, no strong taste, and a gritty feel. The crystal lattice produces a solid that doesn’t react frantically with air or mild reagents, which has made it popular for those looking to store or ship small but precise quantities of selenium.
Technical Specifications & Labeling
Suppliers often provide a detailed technical data sheet. The purity—frequently above 99%—dominates the front of the label, since trace contaminants like lead or arsenic complicate precise lab work. Labels usually justify the batch by listing its water content, noticeable color, and the mesh size of the ground powder. That’s not only for compliance; regulatory bodies such as the European Chemicals Agency and US EPA routinely review and update limits on heavy metal content. Whether the product serves as a research chemical or an industrial intermediate, containers carry UN and hazard statements referencing its toxic and irritant nature. Seeing both GHS pictograms and specific lot numbers helps busy labs with ordering, traceability, and audit requirements.
Preparation Method
True to its roots, creating barium selenite relies on solid and solution chemistry. Mix an aqueous solution of a soluble barium salt—like barium chloride or barium nitrate—with a solution of sodium selenite. The job happens at room temperature and in open vessels, which makes the process straightforward for both lab-scale syntheses and small manufacturing runs. The barium selenite drops out of solution as a fine precipitate, leaving nitrate or chloride ions behind. After filtering and a thorough wash with distilled water, the solid dries at moderate heat. Consistent yields count on precise control of stoichiometry, temperature, and mixing rate, especially since over-washing can carry off tiny grains and mess up batch counts.
Chemical Reactions & Modifications
Barium selenite doesn’t behave as a particularly reactive salt under mild lab conditions, staying put as a solid in neutral to mildly alkaline environments. In strong acids, it dissolves to release selenium dioxide and barium ions. Treatment with reducing agents can shift selenite (SeO32–) into lower oxidation states, sometimes leading to elemental selenium or even more complex barium selenides under the right conditions. The ability to pivot its selenium state has made it a handy intermediate for those hunting for specialty selenium alloys and catalysts. Direct interaction with organic acids rarely produces much action, but strong bases can dissolve it, creating soluble barium selenite complexes.
Synonyms & Product Names
Across catalogs and regulatory inventories, barium selenite sometimes goes by the name selenious acid, barium salt, or barium salt of selenious acid. Its registry numbers bring up synonyms in multiple languages, such as bario selenito in Spanish and Selensaures Barium in German. These aliases help track its reputation and distribution across global supply chains, ensuring buyers and safety officers trace the substance correctly through shipment, research, and disposal paperwork.
Safety & Operational Standards
Every container of barium selenite comes backed by concrete safety standards. Handling it means gloves, eye protection, and well-ventilated workspaces. Exposure to the dust or ingestion risks classic barium toxicity symptoms: nausea, vomiting, and even muscle paralysis in large doses. Regulatory bodies like OSHA set exposure limits, while the GHS tags it as both an acute toxin and an environmental hazard due to selenium’s delicate environmental profile. Storage needs to stay dry, cool, and away from acids that could release toxic vapors. Regular safety reviews motivate organizations to institute annual audits, chemical inventory checks, and spill response drills—often going well beyond the minimum compliance paperwork.
Application Area
Barium selenite carves out small but critical roles for itself. In analytical chemistry, it acts as a selenium standard or a gentle selenium donor for colorimetric detection. Some rare glass formulations benefit from its oxygen-stable selenium content, especially in infrared and specialty optical glass. Small amounts enter the world of ceramics, pigments, and catalyst research. Metallurgists sometimes look twice at it for refining selenium impurities in copper ore blends. Given selenium’s tightrope walk between biological need and toxic risk, health-related applications remain strictly non-therapeutic, but environmental testing labs find barium selenite reliable for selenium quantification.
Research & Development
Chemists and materials scientists stay interested in barium selenite’s potential as both a raw material and a testbed for new selenium compounds. Nanotechnology labs investigate its potential in producing nanoscale selenium clusters, while analytical chemists continue to refine detection and titration methods that can distinguish selenite from selenate at ever-lower concentrations. There’s also persistent work exploring its catalytic behaviors under high-temperature, reducing environments—especially in converting industrial by-products into more stable selenium forms. Environmental labs dive into its solubility properties to improve soil and water selenium remediation strategies, closing the gap between laboratory knowledge and practical fieldwork.
Toxicity Research
Toxicologists keep a steady eye on both animal and cellular studies involving barium selenite. The main concern stems from both barium’s potent effects on muscular and nerve signaling, and the oxidative stress selenium imparts in high doses. Research catalogs dozens of acute toxicity studies, with LD50 values in rodents providing lower bounds for safe human exposure. Long-term exposure studies show disrupted cellular respiration, highlighting the need for careful dosing and exclusion from food and consumer product streams. Regulatory agencies regularly update permissible exposure limits based on these animal studies, erring on the safe side due to variations in absorption and sensitivity across species and even between individuals. Cleanup and disposal teams receive specific training on improper discharge, since both barium and selenium threaten aquatic ecosystems at parts-per-million levels.
Future Prospects
Looking ahead, barium selenite likely won’t flood the mass market, but it’s earning a reputation in precision manufacturing and analytical chemistry. As the electronics sector grows hungry for selenium-based semiconductors, niche suppliers keep fielding more requests for tailored, high-purity intermediates like this one. Tightening environmental regulations continue to shape how the substance gets produced, shipped, and recycled; research into greener synthesis routes and less toxic selenium analogs may eventually reduce reliance on heavy barium. Meanwhile, improvements in nanoscale analytics and material science could unlock new applications that haven’t left the lab bench yet, especially if researchers address persistent toxicity and waste management issues. Seeing its trajectory in the specialty chemicals market, the compound’s main role stays anchored in accuracy, reliability, and that fine balance between hazard and utility that marks so many niche industrial salts.
What is Barium Selenite used for?
Peeking Into Barium Selenite’s World
Barium selenite tends to show up in places most folks never hear about. Its main home sits in chemical laboratories and factories, not in daily life. A pale yellow-white powder, this compound quietly feeds several niche uses, with its most notable appearance as a source of selenium in glass, ceramics, and pigment production.
Selenium Source—Why It Matters
Selenium traces in our world punch above their weight. In the right hands, this element colors, strengthens, and amplifies other materials. Mixing selenium into glass makes it a shield against damaging light. Glassmakers often blend in barium selenite to get the process moving—like loading the right cartridge in a printer. Adding selenium shifts glass to soft pinks, reds, or clears the greenish tint from iron. This subtle tweak helps eyeglasses, solar panels, and decorative bottles do more than just look pretty.
Not all colorants get chosen. Some leave toxic by-products, fade too fast, or play badly in the kiln. Barium selenite offers more stable coloring than many metal oxides, and glassworkers notice how easily it disperses compared to raw selenium powder. These subtle advantages ripple outward, making jobs smoother in factories where every hour counts.
Role In Ceramics and Pigments
Ceramicists crave vibrant hues and long-lived results. Selenium’s punchy color palette transforms tiles and pottery. Barium selenite lets manufacturers harness the best of selenium without confusing side reactions spoiling the mix. This matters for big tile plants and small pottery studios alike.
Pigment creation is also a big deal. Mixing in barium selenite helps pigment specialists create paints and plastics with sharp, precise colors. Most important is controlling dust and waste—barium selenite’s controlled reactivity helps reduce mess and cuts risk for workers.
Concerns and Hazards
Using barium selenite isn’t all upside. Both barium and selenium compounds have known toxic effects. Workers exposed to fine powder can inhale or absorb unsafe amounts through the skin. Water runoff from factories risks contaminating soil and aquatic life if not carefully treated.
This isn’t a theoretical worry. The United States Environmental Protection Agency highlights barium and selenium as chemicals of concern. Even small spills can create problems since both elements persist and build up. Long-term exposure to either can stunt growth in plants and harm ecosystems.
Making Use Safer
Reducing risks means paying attention well before mistakes happen. Ventilation systems, sealed processes, and regular air checks keep factory floors safer. Teams learn proper handling through hands-on training—nobody throws caution to the wind near these compounds.
Recycling and disposal rules continue to tighten. Chemical makers track every gram from delivery to waste, logging and scrubbing waste streams for stray toxins. Some companies are investing in greener alternatives for applications like glass coloring, pushing for bio-based pigments that don’t lean on heavy metals. It's a tougher challenge, but with profits on the line and tighter rules ahead, more research points toward greener chemistry.
Why Attention Matters
Understanding barium selenite’s role and risks reminds us all that industrial chemistry shapes far more than lab textbooks. Most folks use products with hidden journeys—untold colors, treatments, and additives layered silently into our world. For everyone invested in safety, paying close attention to how chemicals get used and managed turns small changes on the factory floor into brighter outcomes for workers and the world outside.
Is Barium Selenite toxic or hazardous?
Understanding the Real Risks
Barium selenite might sound like one of those chemicals that few people run into, but its risks deserve attention. This compound brings the dangers of both selenium and barium, two elements not known for being gentle on the human body. Plenty of researchers and workers in specialized fields face these risks, not just chemists in white coats. Anyone handling this material benefits from real perspective about just how toxic it gets, not only for peace of mind but for safety.
Health Hazards That Demand Respect
You don’t want barium selenite dust drifting anywhere near your face or skin. Inhalation or exposure can lead straight to trouble, thanks to how the compound interacts with our organs. Selenium compounds can build up quickly: even a small dose above dietary needs already tilts toward toxic levels. Short-term exposure during an accident can bring burning eyes, sore throat, or upset stomach. Over time, selenium harms the liver and nervous system. Barium, on the other hand, takes a different path. It disrupts the body’s ability to control muscle function and can even mess with your heart rhythm.
Fact is, both elements on their own have pages of medical literature describing their dangers. Combining them in one compound doesn’t make things any safer—actually, it creates new headaches for anyone in charge of safety protocols. During my early days working around raw minerals, we always took extra steps with selenium and barium, treating both like respected bullies in different corners of the lab. Mix them, and the effort doubles.
Where Accidents Happen Most
Lack of training spells trouble. Folks with limited experience can easily underestimate how quickly a powder can contaminate hands, clothes, or surfaces. Eating or drinking near a barium selenite spill means adding risk, especially if someone licks a contaminated finger or forgets to wash up. Even small oversights, like a loose-fitting mask or cut glove, mean dangerous exposure. Historically, most serious cases of poisoning come from labs, mining, or improperly labeled waste, but school labs and hobbyists sometimes hit headlines too.
Safeguarding Lives—Immediate Steps Matter
Simple steps usually cut risk right down. For starters, solid gloves and fitted masks provide the basic line of defense. Anyone around this chemical stays safer working under ventilation hoods and using sealed containers. I’ve always urged those new to the scene to double-check all labels and never rush through clean-up: one small shortcut can set up a major health scare.
Firms also have a duty here, beyond the lab. Regular audits, fresh training for new staff, and spill drills keep everyone on their toes. Local authorities and regulators should enforce tough inspection schedules, since oversight tends to slip over time. Supply chains matter too: fake or poorly labeled chemical shipments have caused disasters before, so traceability helps.
The Environment Pays a Price Too
Letting barium selenite slip into water or soil triggers environmental damage. Both barium and selenium accumulate in wildlife and can wreck local ecosystems. Fish kill incidents in selenium-rich zones stand as recurring reminders that mishandling just a few grams produces big ripple effects. So responsible waste disposal—never just down the drain—can’t be treated as an afterthought.
Every day, more materials enter the market with hidden risks. Barium selenite stands as a clear example: smart handling and open transparency protect both those working with it and the broader community.
What is the chemical formula of Barium Selenite?
Digging Into the Science
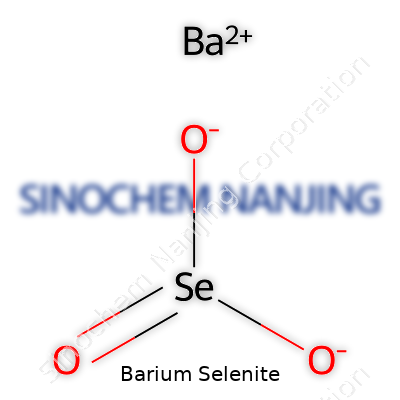
Barium selenite isn’t something you stumble on at the hardware store. It pops up in the world of chemistry labs, where folks explore how different chemicals interact. The chemical formula for barium selenite is BaSeO3. You’ve got a barium ion, a selenium atom stuck in the element’s +4 oxidation state, and three oxygens rounding out the group. That formula tells more than just structure—it marks the signature of barium selenite’s unique behavior.
Why Barium Selenite Catches Attention
The element barium stands out with its bulky, heavy characteristics, while selenium brings a hint of drama thanks to its reactivity. This mix creates a compound with real solutions for the industrial world. For anyone handling glass or specialty ceramics, barium selenite can help adjust the way those materials absorb light or heat. Science companies and certain manufacturers have noticed the specific spectral properties brought by selenite ions—useful for tuning lasers or specialty optical coatings.
Mixing things up can get risky, though. Both barium and selenium offer some potent risks if handled without care. Barium compounds are toxic; swallowing small amounts can turn serious fast. Selenium in the wrong dose isn’t friendly either—too much can trigger a range of health problems. Chemistry teachers regularly bolt warnings about these dangers right on the cabinet doors, and strict disposal rules aim to keep lab techs and the environment safe. If you’ve ever been in a college lab with a stern instructor, you know their frustration over mishandled chemicals isn’t just for show.
Facts on Safety and Community Impact
Most folks assume a specialty compound like barium selenite won’t touch their lives. Indirectly, it probably does. If you depend on electronics, medical imaging, or even renewable energy tech, you might benefit from rare and complex chemical processes that involve compounds like this one. That said, any real value comes with risk if storage tanks burst, or if factory workers breathe dust over time. International safety rules address these concerns for a reason. According to the CDC, barium-based substances have found their way into some accidental poisonings, and the Environmental Protection Agency pushes for careful waste tracking. For selenium, improper disposal can mess with aquatic ecosystems, disrupting life downstream of a major plant.
Potential Roads to Safer Use
Instead of scrambling to ban every risky compound, people could focus on developing better handling methods and tighter workplace rules. Few folks realize the shift toward closed-system manufacturing—machinery sealed to keep dust or vapor locked down—has cut spills and accidents by huge margins in recent decades. Ongoing training and safer packaging make a long-term difference. Seeking alternatives helps too; sometimes there’s a similar compound with less toxicity or environmental impact that can step in without changing production quality.
On the education side, students reading up on BaSeO3 dive into real chemistry, not just memorizing. They learn the why behind the formula—how structure reveals risk, how lab protocols protect people, how every glass slide relies on reactions traced back to simple equations. Knowledge matters most when it’s lived out with respect for what even the toughest-sounding compounds can do.
How should Barium Selenite be stored and handled?
Why Barium Selenite Demands Respect
Barium Selenite isn’t a chemical you leave sitting out on a shelf in a dusty storeroom. This salt brings some real hazards, both to workers and to the environment. The dangers come mostly from two sources: its soluble barium component and the selenium it contains. Exposure can cause harm to people, and Barium Selenite doesn’t shy away from contaminating soil or water. That’s reason enough for folks working around this compound to keep their eyes open and their hands steady.
Storage: More Than Just Lock and Key
I remember walking into a university lab where somebody had stashed all sorts of chemicals together under a single bench. One cracked lid, one leaky bottle—that’s all it takes for things to go wrong fast. Barium Selenite calls for an extra layer of respect. Forget storing it in a flimsy cabinet. This stuff fits best in sealed, clearly labeled containers that don’t react with it. Smart labs space these containers apart from acids, bases, and anything that might spark trouble.
Temperature matters too. Drastic temperature swings lead containers to crack. A cool, dry spot with good airflow helps keep Barium Selenite stable. Humidity shouldn't be allowed, since this compound shifts into nasty forms with moisture present. I’ve seen a whole stash wasted because someone parked it near a steam pipe.
Handling: Think Protection
Gloves, goggles, and a good lab coat create a barrier between your skin and this compound. Even small spills can cause irritation. Breathing in dust or fumes pushes the danger up a notch. So, chemical users who respect their health never handle this powder outside a fume hood. Once, in a hurry, I skipped gloves for just a second—skin irritation reminded me why those safety steps matter.
A written log tracks who handles Barium Selenite and how much moves in and out. This method doesn’t just help with compliance—it keeps everyone accountable. I’ve seen groups catch errors and mishaps before accidents by keeping tight records. Labeling every container and educating newcomers keeps people sharp. No one wants a slip-up because someone grabbed the wrong container in a rush.
Dealing With Waste
Throwing leftovers in a trash can was common decades ago—but those days are long gone. Unused Barium Selenite joins the ranks of hazardous waste. Tossing it down the sink lets it sneak into water supplies. Responsible users collect every crumb and gear up to send it off with licensed hazardous waste handlers. Disposal records keep inspectors and community members reassured.
Learning from Real Mistakes
Sometimes, the headlines tell stories of labs or factories hit with fines after someone mishandled a substance like Barium Selenite. Usually, it comes down to ignoring known steps, such as skipping the right storage or failing to educate new employees. Solutions often lie in regular training, honest discussions about mistakes, and clear working guidelines. The whole team needs to buy in, from the greenest intern to the most seasoned chemist. Everyone’s safety, and the safety of the planet, depends on treating materials with the right amount of caution and care.
Where can I buy Barium Selenite?
Real-World Roadblocks
Barium selenite isn’t something people see sitting next to table salt or baking soda at the supermarket. I once worked in a research lab where we needed rare inorganic compounds for very particular tests. Sourcing materials like this was never simple. Most chemical suppliers list what they carry, but some inventory doesn’t stay up to date, or what’s in stock only ships to universities and established companies. Barium selenite sits squarely in this gray zone.
Why This Compound Is Different
This is not some everyday commodity. Barium brings toxicity concerns. Selenium compounds pose their own health risks. Anyone working with barium selenite must read the safety data sheet and wear serious protective gear. If you’re outside academic or industrial settings, you will hit more walls trying to buy it.
Supplier Expectations
Reliable chemical suppliers care about credentials and proper use. The best-known names—Sigma-Aldrich, Alfa Aesar, Fisher Scientific—carry compounds like barium selenite. Their online catalogs require creating an account, verifying your business or research needs, and usually sharing a tax ID, shipping address, and sometimes a project description. This is not about red tape for its own sake. It helps cut off sales to people without legitimate use, lowering risks of poisoning, environmental spills, or diversion into unsafe experiments.
Online Sellers Versus Trustworthy Brands
You might find claims to sell barium selenite on independent marketplace websites. Deals here usually bring trouble. Quality control remains uneven, labeling gets sloppy, and buyers deal with uncertain origins. Some of my former colleagues used auctions and forums to track down rare compounds, but they always worried about purity, legality, and sometimes even scams. With regulated materials, this gamble goes beyond annoyance—health and legal safety count more.
Ethics and Responsible Sourcing
There’s talk these days about ethical sourcing in everything from coffee to cobalt. Chemical buyers need to pay attention too. Responsible suppliers don’t just handle shipping—they track chain of custody, safe packaging, and disposal guidance. They report suspicious orders and follow up on export controls. People outside the field often overlook these steps, but they exist for public safety and for protecting the environment from improper disposal or accidental release.
Alternatives and Solutions
For educators or hobbyists, approaching a university lab might open doors. Professors with proper lab licenses sometimes order on behalf of student projects, but strict recording stays in place. Local hazardous waste programs or public science labs sometimes loan rare chemicals under supervision. Buying on the open market without documentation won’t work for most.
For companies, setting up an account with a well-known supplier and assigning trained staff to handle procurement changes the game. This creates a paper trail and lets suppliers offer disposal advice, replacement parts, or alternate compounds with fewer risks.
I’ve learned over years in science that cutting corners on chemical sourcing isn’t worth it—even for an oddball experiment. There’s no substitute for doing things by the book, working through trusted channels, and keeping safety up front. For barium selenite and compounds like it, sticking with legitimate suppliers and documented processes protects people, communities, and future research.
| Names | |
| Preferred IUPAC name | Barium selenite |
| Other names |
Barium selenate Barium(2+) selenite |
| Pronunciation | /ˈbeə.ri.əm ˈsɛl.ɪ.naɪt/ |
| Identifiers | |
| CAS Number | [10138-15-9] |
| Beilstein Reference | 14628 |
| ChEBI | CHEBI:86467 |
| ChEMBL | CHEMBL510876 |
| ChemSpider | 53609910 |
| DrugBank | DB14636 |
| ECHA InfoCard | 03b973d1-37c1-4178-a593-53e2c2c6fd40 |
| EC Number | 240-914-1 |
| Gmelin Reference | 140775 |
| KEGG | C18636 |
| MeSH | D001471 |
| PubChem CID | 24974 |
| RTECS number | CQ9625000 |
| UNII | U6Y9H85KLT |
| UN number | UN1564 |
| CompTox Dashboard (EPA) | DJ46641E5S |
| Properties | |
| Chemical formula | BaSeO3 |
| Molar mass | MS = "232.39 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.89 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -2.36 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.28 |
| Basicity (pKb) | 7.79 |
| Magnetic susceptibility (χ) | +30.0e-6 cm³/mol |
| Refractive index (nD) | 1.972 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 107.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -746.1 kJ/mol |
| Pharmacology | |
| ATC code | A12CE03 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause damage to organs; harmful to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled |
| Precautionary statements | P261, P264, P270, P271, P272, P301+P310, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Lethal dose or concentration | LDLo oral rat 4 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5.2 mg/kg (oral, rat) |
| NIOSH | WW3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Barium Selenite: "0.2 mg/m3 (as Ba) |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Barium sulfate Barium selenate Barium oxide Barium carbonate |

