Barium Permanganate: A Substance Shaped by Curiosity and Chemical Progress
Understanding the Roots: Historical Development
Barium permanganate hasn’t enjoyed the same public spotlight as other reagents, but its road stretches deep into the 19th century. Chemists set their sights on permanganates soon after manganese(IV) oxide lit up the scientific world, and blending this potent oxidizer with barium salts proved irresistible. The vivid colors and strong oxidizing nature tugged at researchers’ imaginations, driving curiosity-driven synthesis in cramped university labs and larger industrial ventures. Without the big-name fame of potassium permanganate, barium permanganate attracted attention in smaller circles where niche requirements favored its unique characteristics.
A Rugged Profile: Product Overview and Physical & Chemical Properties
Sitting in the world of inorganic salts, barium permanganate stands out for its deep purple hue. Granular at room temperature, this compound throws off a metallic sheen that hints at its charged internal makeup. It dissolves sparingly in cold water, offering a striking violet color, and as temperatures rise, solubility creeps up. Although not volatile, barium permanganate needs gentle handling—the oxidizing punch makes it unfriendly to organic contaminants and flammable materials. As a barium salt, toxicology always hangs in the background, causing many to proceed carefully. Mixing this compound with the wrong reducers or combustibles can turn a typical experiment into a lesson in chemical fire hazards.
Technical Details: Specifications & Labeling
Any lab-stamped bottle will tell you barium permanganate is more than a simple oxidizer. In my own time spent with similar oxidizing salts, a strong warning label isn’t seen as overkill; it’s earned from experience. The product shows up as crystalline, deep-violet, and sometimes with a greenish tint if impurities sneak in. Purity for research calls for above 97 percent, particularly for kinetic studies. Handling and shipping bring regulatory stickers warning of both the oxidizer and toxic nature.
Chemistry in Action: Preparation Methods
Synthesizing barium permanganate takes deliberate steps. Many chemists start with potassium permanganate and a soluble barium salt, such as barium chloride, using double displacement in aqueous solution. The barium ion swaps places with potassium, yielding a purple precipitate. Careful filtration and controlled crystallization help tease out the product. The process may sound straightforward on paper, but anyone who has slogged through precipitation reactions knows they rarely run without a hitch. Slow stirring, managing temperature, and fighting off contaminants shape each batch.
A Dynamic Chemical: Reactions & Modifications
The real action with barium permanganate shows up in its chemical behavior. As an oxidizer, it jumps at the chance to strip electrons from a long list of substances, tossing out manganese dioxide and other products in the process. Reactivity ramps up when heat enters the picture. I’ve watched permanganates darken and break down in the presence of acids, spitting out oxygen and ushering in colorful shifts. Reducing agents can push the permanganate ion down the oxidation ladder, shifting from deep purple to pale brown. This dynamic chemistry unlocks uses in organic synthesis and analytical work, especially where its high reactivity can be an asset.
What’s in a Name? Synonyms and Product Labels
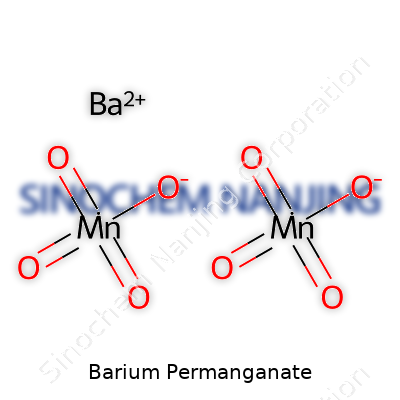
In the wild, chemists might spot this compound going under different monikers. You’ll hear “permanganic acid, barium salt,” “barium manganate(VII),” or more plainly, “barium permanganate.” The chemical formula Ba(MnO4)2 crops up in catalogs and scientific papers alike. Juggling these names can feel like code-switching, especially across international borders or in older literature, but it underscores the compound’s deep roots in a chemist’s toolkit.
Staying Safe: Operational Standards
Any technician who has worked with strong oxidizers respects what they can do in the wrong hands. Barium permanganate stands up alongside other permanganates in terms of risk—it burns skin, poisons tissues, and sets flammable surroundings alight if not treated right. Good laboratories put careful rules in place, from gloves and goggles to fume hoods and secure storage. I’ve worked in places where using permanganates only after a full team discussion wasn’t a luxury, it was standard practice. Proper waste procedures get drilled into anyone handling these salts, as water sources and landfill sites do not forgive mistakes with heavy metals or oxidizers.
Where It Fits: Application Area
Barium permanganate has a story to tell in various fields, though its limelight is limited. It stakes a claim in organic synthesis, where stubborn substrates demand a strong oxidant. Environmental laboratories occasionally turn to it for specialty oxidation tasks or for decontaminating persistent organic pollutants. In pyrotechnics, the compound’s strong oxidizing power and colorful emission can attract interest, but safety issues and regulatory pressure mean it rarely displaces more popular choices. Academic research keeps the compound in rotation because its reactivity helps unpack mechanistic pathways in redox chemistry.
Building Tomorrow’s Chemistry: Research & Development
Barium permanganate doesn’t draw the R&D budgets of more famous reagents, but it still occupies valuable ground in specialty chemistry. Researchers explore its role in catalytic cycles, searching for new transformation routes in organic chemistry. The search for greener processes could open the door for alternative oxidizing methods, where barium permanganate’s reactivity serves as a benchmark. Functional material scientists eye its potential for making high-performance ceramics or generating surfaces with catalytic properties, always forced to weigh toxicity and handling hazards against utility.
Health Matters: Toxicity Research
Toxicologists give barium salts the side-eye for solid reasons. Ingesting even small doses leads to disturbances in muscle function and heart rhythm, while the permanganate ion brings its own baggage with the potential for tissue damage and oxidative stress. Animal studies show clear signs of harm at relatively low exposures, which has kept regulatory bodies on high alert. Occupational safety campaigns in chemistry curricula teach the importance of keeping these risks front and center, both for the person on the bench and for anyone downwind of a possible spill or improper disposal.
A Glimpse Ahead: Future Prospects
Looking towards tomorrow, the story of barium permanganate is shaped by both opportunity and constraint. Research interest in selective oxidation could provide an avenue for more extensive application, especially if engineers learn how to tame its toxicity or develop robust containment systems. Advances in solid-state chemistry and surface functionalization hint at roles this salt could play in creating durable, efficient catalytic systems. At the same time, rising environmental standards and workplace safety expectations will limit its use. The future of barium permanganate likely hinges on how chemists and engineers solve the duel problems of performance and safety, and whether greener alternatives can be made that compete without the same health hazards.
What is Barium Permanganate used for?
What People Really Do with Barium Permanganate
Barium permanganate doesn’t land in the spotlight very often, but it holds a strange spot in industrial chemistry and a few specialty areas. It grabs attention mostly for being a strong oxidizer with a purple pop—something that’s rare and often powerful. Growing up around labs and picking up odd summer jobs at paint and metal shops, it was clear how these crystalline powders play behind the scenes. People treat these chemicals almost like magic ingredients, pulling off things you can’t just do with table salt or vinegar.
Take pyrotechnics. This is where barium permanganate really cuts loose. Firework makers hunt for colors that pop out vivid and crisp. Barium compounds have this knack for shooting up green flames, the kind that stick in memory during a July night. Permanganates bring oxygen to the mix, driving reactions harder and pushing color even brighter. Sockets of air greenhouse fireworks, but the oxidizer’s job is to keep the fire burning hot enough for minerals to show their shade. That purple-pink hint in a rare show sometimes starts with permanganate. Not every fireworks compound can claim that level of excitement or risk—factories don’t let beginners tinker with barium permanganate, since it’s touchy and floods air with oxygen if mishandled.
Learning firsthand about oxidizers teaches caution as much as creativity. Offering up pure oxygen to the wrong situation—an oily rag or a bit of stray powder—turns an experiment into a hospital trip. Safety means everything in these settings, and everyone gets the message hammered home before a gram of material ever hits the table. Scientists and technicians wear dust masks, gloves, and lab coats for a reason. Even outside of big factories, school demonstrations and chemistry clubs steer clear of barium permanganate unless a qualified instructor is nearby. It doesn’t forgive mistakes, and guidelines from agency manuals make that clear. It pays to read up on the Material Safety Data Sheet, since ingestion or skin contact leads to all kinds of trouble, from nerve issues to outright poisoning.
Barium permanganate also turns up in niche water treatment. In rare cases, municipal plants and labs use it to break down troublesome organic matter or iron and manganese stains. Its ability to rip electrons away from tough pollutants has a practical side, but this compound’s price and toxicity keep it from filling every filter. Managers usually prefer safer, cheaper options unless some tricky contamination pops up that nothing else can handle. My neighbor once worked on a water project in an old mining town, and he told stories about tiny amounts of specialty chemicals pulling color out of rusty pipes like nothing else, but even then, the team measured everything ten times and wore double gloves.
In the classroom, teachers might show off permanganates to illustrate the basics of oxidation and reduction—watching a deep purple solution fade as it attacks iron or sugar is a memory that sticks with students. These demonstrations leave a mark that long lectures never manage. Seeing chemistry in motion has always made the risk feel real, and the curiosity behind it feels worth it. Still, none of these lessons happen without careful prep and solid training.
Barium permanganate grabs the chemist’s imagination and stirs a fair bit of caution. It has its place in pyrotechnics and rare treatment plants, but it reminds us that real science doesn’t shy away from risk. Respect fuels progress as much as curiosity does. For those handling the powder—or admiring the green flash in the sky—paying attention keeps everyone in the game for another round.
Is Barium Permanganate toxic or hazardous?
What Is Barium Permanganate?
Barium permanganate isn’t something most people keep in a kitchen cabinet, but it’s pretty important in certain scientific settings. This chemical, recognized by its deep purple hue, plays a role in oxidation reactions. Folks in labs and some industries turn to it thanks to these powerful properties. Its mix of barium and permanganate puts it in a category of compounds that can create real concerns for health and safety.
Hazards and Risks: What the Evidence Shows
Holding a bottle labeled “barium permanganate” actually means holding two hazards in one. Both barium and permanganate can cause trouble, each in their way. Barium, even outside the permanganate salt, can disrupt potassium channels in muscle and nerve cells. Swallowing enough can cause muscle weakness, irregular heartbeat, or even paralysis. The Environmental Protection Agency and the Centers for Disease Control both flag barium salts as toxic, and for good reason.
Permanganate, on its own, causes severe burns if it comes into contact with skin or eyes. Ingesting permanganate doesn’t just burn the mouth — it can deliver toxic effects to organs like the liver and kidneys. Combine barium with permanganate and the recipe turns risky for anyone not using proper safety gear or working in a well-controlled setting.
Cases of barium poisoning often come from contaminated water or accidentally swallowing the wrong chemical at work. Stomach pain, vomiting, and muscle spasms all can hit quickly. Long-term exposure? Medical literature links it with nerve damage and changes in blood pressure. Using barium permanganate by mistake could pose those same risks, only with a punch from the permanganate’s corrosive nature.
Why Regulation and Training Matter
Strict rules exist for a reason. The Occupational Safety and Health Administration has clear standards when it comes to barium, mandating limits on exposure and calling for protective gloves, goggles, and fume hoods. The National Institute for Occupational Safety and Health set exposure limits at 0.5 mg per cubic meter for soluble barium compounds. The logic: nobody wants workers developing health issues just to make reagents or coatings.
Proper training goes a long way. When I worked alongside research teams at a university chemistry lab, we'd spend as much time setting up our safety protocols as we did running experiments. Fume hoods stayed down, gloves stayed on, and we always made sure all waste bottles were labeled. No one wants to deal with an emergency spill when a little caution can keep everyone safe.
Reducing Harm: Practical Steps
Skipping shortcuts saves lives. Folks handling barium permanganate need to keep food and drinks away from their workbench. Skin contact? Always wash up with lots of water after use, and treat any spills with caution. If anything splashes in the eyes, running water and quick medical help matter. Fresh air and good ventilation help keep vapors in check. Spills need a plan — dedicated disposal containers, not the nearest trash bin.
Substitution sometimes works best. In teaching labs, using less hazardous oxidizers cuts down on risks. For industry settings, strict engineering controls — sealed systems, local exhaust, precise inventory management — mean fewer dangerous interactions.
What to Watch For
No chemical is entirely without risk, and barium permanganate’s record shows that extra respect is non-negotiable. Taking shortcuts rarely ends well. Following the rulebook isn’t about making extra work — it keeps emergencies rare and lets science move ahead without adding new health risks.
What is the chemical formula and appearance of Barium Permanganate?
What Is Barium Permanganate?
Barium permanganate comes with a rather eye-catching purple color. Its chemical formula stands as Ba(MnO4)2, pairing a heavy alkaline earth metal with a potent oxidizer. Anyone who has spent lab time with oxidizing salts knows this is not a compound to keep on the kitchen shelf. It forms purple to dark-violet crystals, often shimmering and almost hypnotic—if one forgets the dangers for even a moment.
Where Science Meets Safety
The interesting thing with barium permanganate is how it blends two elements known well in science education—barium and the permanganate ion. Both have their quirks and, frankly, both raise red flags for most chemists. Take barium: in small doses, barium ions can be toxic. Ancient alchemists couldn’t know swallowing barium compounds could lead to muscle paralysis or even death, but today we have good data to back up those warnings. On the manganese end of things, the permanganate ion serves as a powerful oxidizer—one spark away from giving off a breath of oxygen and burning through organics.
Anyone who’s mixed potassium permanganate with glycerin in high school chemistry has seen what these purple ions can do. Barium permanganate doesn’t behave very differently, just throws in the added heaviness of barium’s own reactive touch. It decomposes with heat, releasing oxygen and leaving behind barium manganate (BaMnO4) and manganese dioxide (MnO2) among the ashes. For pyrotechnicians, the vibrant green flame of barium makes it a tempting ingredient—though strict bans exist for safety reasons, and green fire isn’t worth the ER visit.
The Industry and Real-World Value
There aren’t a lot of home uses for barium permanganate. Industrial labs, research institutions, and specialty chemical companies are its main environments. Water treatment sometimes looks at permanganates thanks to their ability to break down organic contaminants, but potassium permanganate takes the prize there for being easier and safer. Some niche syntheses and analytical chemistry routines use barium permanganate, though getting one's hands on it involves red tape and legitimate credentials.
Understanding the Risks—and Solutions
People working with barium permanganate need solid training. That’s not just regulation talking—my own time handling oxidizers taught me to respect both their fire risk and their toxicity. Purple color lures the curious, but mixing with organic matter or acids can spell disaster. Sometimes, the best safety tip involves simply not taking shortcuts. Using proper gloves, goggles, a fume hood, and having neutralizing agents on hand makes the difference between running an experiment and triggering a hazardous response.
Disposal brings its own headaches. Because both barium and manganese remain toxic to aquatic life, pouring leftover material down the drain can contaminate groundwater. Best practices mean working with certified waste services, neutralizing residues, and never treating laboratory reagents like household trash. In my experience, labs that cut corners on disposal often wind up fielding emergency calls from environmental authorities.
Bringing It Back to Science
Barium permanganate may never become a household name, but it serves as a reminder that every chemical reveals both promise and peril. Training, respect for data, and patience keep those crystals beautiful but safely distant from the dangers that lie beneath their shimmering purple surface.
How should Barium Permanganate be stored and handled safely?
Why Safe Storage Matters
Barium permanganate’s bright purple looks can be misleading. It’s an oxidizer with a serious kick, and that spells trouble if ignored. I remember working once in a lab where an old jar of the stuff leaked near a shelf with cardboard. Things could have gone sideways fast. Nobody wants to deal with that kind of scare—or worse, a preventable fire or exposure.
Understanding Its Real Hazards
These crystals store lots of energy. When they hit organic material or bump up against heat, a reaction often follows. That reaction throws off oxygen and heat. Fires start easier and burn hotter. Inhaling its powder isn’t good, either—lungs don’t fare well with it. Swallowing some doesn’t do a body any favors, either. Toxicity concerns make respect for this compound more than a luxury—science backs this up, with emergency room cases pointing straight to oxidizer mishandling as the root.
Simple Rules That Work
Clear, well-marked glass or plastic containers cut down on confusion. No confusing white powder sitting right next to sugar or washing soda. Labels should actually mean something to people who don’t spend days reading MSDS sheets. A good habit I picked up: Print the full chemical name, highlight that it’s hazardous, and toss in a couple of sentences about risks.
Dry shelves in a cool space keep trouble at bay. Moisture launches reactions, so don’t store it anywhere leaks can find it. Metal shelves should be avoided. Rust or loose scraps just increase the risk.
Combining storage is asking for trouble. Mixing oxidizers with fuels or acids creates a tossup. A shelf crash could make an ordinary day turn ugly quick. There’s a reason most regulatory guides say to keep oxidizers separate—and not out of bureaucratic habit, either. Flammable items like solvents, paper, and rags should get their own locked cupboard or, better yet, be kept elsewhere.
Personal Protection Matters
I never step near this chemical without goggles and gloves. Skin contact is hard to shake off, and eye splashes can go from irritation to permanent harm in minutes. Disposable nitrile gloves do the trick. Eye protection stays on as long as the bottle stays open. People often forget respirators, but in powder form, a half-mask respirator with good filters keeps fine particles out of lungs.
In Case of Accident
Spills do happen, no matter how careful anyone is. Keeping neutralizing solutions nearby helps. Absorbent material—never sawdust or rags—soaks up the mess. Scooping it up with plastic tools is safer than using metal shovels.
Disposal isn’t for the household trash. Professional chemical disposal services handle it best. There’s not much charm in seeing headlines about water supply disasters, and barium carries real health risks if it enters the system.
Why All This Effort Makes Sense
Every time a story hits about a lab explosion or environmental mess, it’s clear that cutting corners brings big consequences. Good habits, clear labeling, and the right gear keep real threats at arm’s length. There are no shortcuts. If you work with oxidizers, treat each step in the process as personal, not just professional. Other folks and the environment stand to benefit from a bit of extra care—science, history, and common sense all back that up.
Where can I purchase Barium Permanganate and what are the shipping restrictions?
Finding a Supplier Isn't as Simple as It Sounds
Barium permanganate pops up on the radar for chemists and researchers who want a potent oxidizer. It’s not something you just pick up on Amazon or order through local hardware outlets. Most legitimate purchases happen through chemical supply companies—think Sigma-Aldrich, Fisher Scientific, or Alfa Aesar. There, you get guarantees on purity, full documentation, and info sheets that detail every risk and recommended handling procedure.
It’s tempting to Google around for smaller distributors. In practice, credible suppliers never ship barium permanganate to casual shoppers. You’ll almost always face background checks and need to verify your identity, business type, and intended use. You may need to show professional credentials. That helps block unsafe or illegal uses and shows respect for the danger this compound carries.
Shipping Restrictions—No Shortcuts Allowed
Barium permanganate can’t take a ride in regular mail. Its oxidizing power means it’s flagged by shipping companies and chemical regulators. Air shipping is often off-limits due to the risk of fire or explosion from shocks or leaks. UPS, FedEx, DHL—these guys follow strict rules and most refuse to handle this substance unless you’re certified, trained, and authorized.
The U.S. Department of Transportation and agencies in Europe, Canada, and Asia classify this chemical under “hazardous materials.” If you’re ordering from abroad, import paperwork multiplies. Customs will want proof you’re permitted, sometimes even for research labs. Any attempt to skirt these hurdles leads to shipment seizures or investigation.
Couriers need the chemical packed inside special containers. Double bagged, sealed, and cradled in shock-absorbing material, each parcel sports warning labels and documentation. Any shortcut risks danger to handlers and the public, so the extra steps make sense.
Safety Isn't Just a Buzzword
I’ve spent years in lab environments where strong oxidizers show up. One careless moment with compounds like barium permanganate could cause toxic fumes, fires, or even explosions. Even a pinch on open skin, or a whiff, can send you to the hospital. That drives home why reputable distributors keep such a tight grip on who gets to buy.
No matter how tempting it sounds, skipping these protections for a faster or cheaper purchase isn’t worth it. Every properly-run lab sets up special storage (cool, dry, away from organic matter), clear staff training, and written emergency procedures. In my experience, safety culture—more than any label—is what keeps everyone healthy and productive.
Responsible Sourcing and Alternatives
Sometimes you hunt for a chemical like barium permanganate only to realize a safer or more available substitute meets your needs. Alternative oxidizers such as potassium permanganate are much easier and legal to buy for most experiments and industrial needs. Before placing any order, talk with qualified staff about whether you can replace barium permanganate with something less hazardous.
For research groups, setting up a relationship with a major chemical supplier beats combing through hobbyist forums or black market sites. The paperwork takes time, but you get peace of mind and real support if anything goes wrong. Education remains the best tool: teachers and professors can foster a culture where safety, not shortcuts, becomes second nature.
| Names | |
| Preferred IUPAC name | Barium manganate(VII) |
| Other names |
Permanganic acid, barium salt Barium manganate(VII) Barium permanganate(VII) |
| Pronunciation | /ˈbɛəriəm pərˈmæŋɡəˌneɪt/ |
| Identifiers | |
| CAS Number | 7787-36-2 |
| Beilstein Reference | 14656 |
| ChEBI | CHEBI:137742 |
| ChEMBL | CHEMBL1229770 |
| ChemSpider | 8770878 |
| DrugBank | DB13765 |
| ECHA InfoCard | 03d104c8-b473-418c-b32e-1e46fd6b4852 |
| EC Number | 233-587-9 |
| Gmelin Reference | 80704 |
| KEGG | C18781 |
| MeSH | D001471 |
| PubChem CID | 24643 |
| RTECS number | CM8600000 |
| UNII | BB8R8E6SP1 |
| UN number | UN1476 |
| Properties | |
| Chemical formula | Ba(MnO4)2 |
| Molar mass | 437.24 g/mol |
| Appearance | Dark green crystalline solid |
| Odor | Odorless |
| Density | 3.74 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -2.06 |
| Basicity (pKb) | 7.68 |
| Magnetic susceptibility (χ) | +3030.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1255 kJ/mol |
| Pharmacology | |
| ATC code | V09CA03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS03, GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 20 mg/kg |
| LD50 (median dose) | LD50: 15 mg/kg (oral, rat) |
| NIOSH | SD2650000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended): 0.5 mg(Mn)/m3 |
| Related compounds | |
| Related compounds |
Calcium permanganate Potassium permanganate Sodium permanganate Barium manganate |

