Barium Periodate: Progress, Promise, and Problems
Historical Development
Barium periodate stands as one of those inorganic compounds chemists stumbled upon while digging through the rabbit hole of oxidative chemistry in the late nineteenth century. Early work, especially in European laboratories, showed a special interest in periodates due to their ability to yield oxygen-rich anions. Barium wasn’t an obvious pick—the heavy metal’s intimidating toxicity had been demonstrated with its chloride and carbonate forms long before anyone dared introduce it to periodate. Over time, researchers settled on barium periodate for niche oxidizing needs that surpassed common agents like potassium permanganate. Chemical literature showed peaks of interest, often after world conflicts spurred munitions research or in periods when mainstream oxidizers failed to deliver on selectivity. In my own undergraduate project years ago, our lab shelves featured only the dustiest bottle of periodate under lock and key, echoing its historical arc: present, powerful, a little forgotten, but never irrelevant.
Product Overview
Unlike the household chemicals stirring under your kitchen sink, barium periodate claims a reputation few compounds can match. Chemists eye it for its strong oxidative punch, knowing that its periodic structure offers multiple oxygen atoms ready for release. The formula, typically displayed as Ba(IO4)2, signals how dense, reactive, and potentially hazardous it is right from the get-go. Commercial interest fluctuates: some years, demand jumps as niche synthesis or academic curiosity spikes; other years, only high-level labs and a handful of industries give it a second look. Its uses crop up mainly in advanced synthesis and certain analytical techniques, but the product rarely spills into consumer markets. Barium periodate always carries an aura of potential, complicated by the real-world limits of cost, safety, and regulations.
Physical & Chemical Properties
The white, powdery crystals of barium periodate look deceptively unremarkable at first glance. What sets them apart is the dense barium ion paired with the periodate anion, creating a compound that resists easy dissolution in water. This limited solubility shapes its behavior in the lab, often forcing chemists to rely on vigorous stirring or heating if they want full reaction. With a melting point climbing over 500°C, it sits stubbornly inert to moderate heat until, sometimes explosively, the compound starts breaking down and giving off oxygen and iodine. The high oxidation state of iodine within periodate delivers its signature reactivity, and the dense atomic make-up of barium adds heft, making handling a matter of deliberate attention. During one internship, I remember learning to keep hands dry and steady, as this was not a material you let drift about near your skin or mucous membranes.
Technical Specifications & Labeling
Handling and packaging barium periodate requires organized labeling, with hazard codes and oxidation warnings. Usually, the compound appears in solid form with varying grain sizes, from fine powder to granular product. Purity often hovers above 98% for most research-grade material, with some suppliers guaranteeing low moisture content and limited contamination by other alkaline earth metals or halides. Strict rules and prominent warning symbols land on the bottles, not just for barium toxicity but also for the oxidizing power. This isn’t a substance to store near organics, acids, or open flames. In my academic experience, the label itself commands respect—unlike more benign salts, the combination of barium and periodate drives home the potential for harm, so proper storage and meticulous record-keeping always become second nature.
Preparation Method
Synthesis of barium periodate follows a dance with solubility and oxidation state. Most commonly, preparation starts by reacting barium hydroxide or barium chloride with a soluble periodate, such as sodium or potassium periodate. The insoluble nature of barium periodate helps, as it falls out of solution to be collected, washed, and dried. Any contamination, especially from other ions or remaining alkali, affects not just quality but safety. Each batch demands attention: incomplete washing leaves salts behind, improper drying raises the risk of hazardous dust. At various points in senior lab coursework, we followed methodical steps under close supervision—to limit risk, a glass of distilled water always within reach to douse any unexpected spill, a reminder that synthesis here sits less on the side of curiosity and more on the side of respect.
Chemical Reactions & Modifications
The chemical curiosity in barium periodate springs from its coveted ability to deliver oxygen in demanding environments. It oxidizes alcohols to carbonyl compounds and can cleave certain organic bonds other oxidizers won’t touch. Reacting barium periodate with acids or reducing agents often leads to the release of iodine, occasionally in violent fashion, and evolution of oxygen gas, turning a simple misstep into a safety incident. Attempts to modify the molecule, by pairing the periodate with other heavy metals or seeking composite materials, rarely outperform its standard crystalline form, though ongoing research sometimes finds ways to control dissolution rate or boost selectivity. For most chemists, the real draw lies in its role as a “cleanup hitter,” stepping up for tough oxidation needed at critical moments, despite its limitations and risks.
Synonyms & Product Names
Across laboratories and chemical catalogues, barium periodate hides behind several aliases. Internationally, it goes by barium tetraoxoiodate(VII) or barium metaperiodate. Occasionally, suppliers market it as simply “barium salt of periodic acid.” Each synonym reinforces the important detail: this isn’t any everyday barium salt, but one unified by its periodate anion. Despite the interchangeable names, the underlying risks don’t change, a fact hammered home in every introduction to the product’s documentation.
Safety & Operational Standards
No one with actual hands-on experience takes safety protocols around barium periodate lightly. The barium ion stands well known for cardiac and gastrointestinal toxicity, while the oxidizing periodate increases risks of acute irritation, tissue burns, and spontaneous combustion when exposed to organic materials. Standard lab practices demand fume hoods, goggles, full gloves, and frequent hand-washing after handling. Spillage responses mean rigorous containment and neutralization, never casual mop-ups. Regulatory guidance gets strict here: most institutions require restricted access, special dispensations, and disposal through licensed hazardous waste operators. This level of scrutiny pushes users to internalize safety as part of any daily workflow, never skipping steps, always assuming the worst could happen if procedure falters even once.
Application Area
Barium periodate remains a bit-player in the wider chemical industry, but it claims legitimate fame in analytic chemistry and rare synthetic transformations. Researchers use it to oxidize challenging organic structures, especially in carbohydrate analysis, where selective bond cleavage helps determine sugar motifs and branching. The compound stirs interest in advanced electronics research, especially as material scientists push toward higher-performance ceramics and specialized coatings. Small-scale usage in dye manufacture, forensic chemistry, and even some explosives testing highlights its functional range. Only experienced personnel, surrounded by proper equipment, ever touch it outside of the most controlled industrial projects or research settings.
Research & Development
Every step toward new applications for barium periodate brings renewed focus on balancing benefit and burden. Modern research fixates on ways to harness its oxidizing power while limiting exposure, improving selectivity, and finding greener synthesis routes. In some university projects I’ve followed, scientists explore ways to immobilize periodate anions within polymer matrices or nanostructured supports, seeking safer handling and tailored release for advanced chemical synthesis. Collaborations between academia and industry have achieved only modest success here, but the promise remains: safer, targeted, more accessible use could open whole new corners for innovation in analytical chemistry and advanced material science. Investment and regulatory support often lag, but the steady stream of peer-reviewed studies hints at momentum. Periodically, new modifications make headlines, though very few find reliable scale-up beyond the lab bench.
Toxicity Research
Toxicology research on barium periodate never gives reassuring answers. Exposure to soluble barium compounds brings risks of muscle weakness, cardiac irregularities, and even death at surprisingly low doses. Iodine in the periodate form, especially when freed by reaction, pushes the hazard even further: inhalation of dust causes acute respiratory irritation, long-term handling may introduce risk of chronic poisoning or environmental contamination. Animal studies and limited clinical incident reports reinforce the same message—engineering controls, procedural rigor, and proper training cannot be optional. Modern toxicological studies focus on ways to monitor exposure, limit uptake in biological systems, and improve emergency response. Alternate compounds with similar reactivity but lower toxicity haven’t yet displaced barium periodate, keeping its handling within the domain of well-equipped professionals.
Future Prospects
Demand for powerful yet selective oxidizers in pharmaceutical manufacturing, advanced chemical analytics, and next-generation materials keeps barium periodate’s future unpredictable. Advances in containment, handling, and environmental remediation could open doors to safer, more widespread use, but regulatory burdens and public scrutiny sit heavy. Research targeting modified forms—whether supported on inert matrices, integrated into reusable composites, or manufactured under greener conditions—may push the compound’s utility further. Still, unless clear advances make its handling and disposal less onerous, barium periodate will likely remain a valuable yet specialized tool, championed by those willing to accept its considerable risks in pursuit of chemical innovation.
What is Barium Periodate used for?
What Sets Barium Periodate Apart
Barium periodate catches attention mainly because of its strong oxidizing ability. In labs, people lean on it for reactions that need serious muscle to break certain bonds. Take organic chemistry — creating or modifying molecules sometimes calls for a bit of force, and this is where barium periodate steps into the mix. I recall a discussion with a colleague who used it to oxidize alcohols without pulling in moisture from the air, something that made other oxidizers misbehave. Barium periodate brought them clean, crisp results, which made a real difference in their research and, sometimes, in scaling reactions for pilot projects.
Making Dyes and Pigments Work
The dye industry often explores new ways to get colors to last longer or set better on fabric. Barium periodate has seen use here, especially with pigments that interact strongly with oxidizers. Its stiffness as an oxidant brings consistency when manufacturers push for brighter shades or sharper contrasts. I once read about small batch fabric dyers tapping into its power for tough palettes that lesser oxidants ruined or dulled out. Of course, with any chemical like this, folks need to respect the safety rules, but for certain specialty colors, it’s earned its keep.
The Dig Into Analytical Chemistry
When researchers check for traces of chemicals in water, air, or food, they sometimes look to barium periodate. Its strong reactivity lets analysts detect sugars or other compounds at levels that other reagents miss. Back in my grad school days, a friend wrestled with a stubborn sugar contamination problem. They turned to a periodate-based approach and watched as tiny traces lit up under analysis, finally getting numbers they could trust. That reliable detection can keep pollution in check and food safer.
Electronics: Not Just a Minor Role
In electronics, rare chemicals occasionally come out from behind the scenes. Barium periodate plays a low-key role in making some specialty batteries and circuits. Its stability helps avoid breakdowns at high voltages, especially in devices that go through frequent charging and discharging. I’ve seen reports from niche capacitor makers in Europe citing this compound as a reason for performance gains in harsh environments. Products like these often run in infrastructure where downtime stings, so reliability matters more than almost anything else.
What About Safety and Environmental Impact?
Barium compounds build up in soil and water. I’ve heard experienced chemists talk about the challenge of disposal — it’s not something to pour down the drain. We should always handle it with tight protocols and respect for regulations. Communities near production sites deserve open conversations about risks and management. Innovation in recycling and neutralization could bring solutions that let scientists and engineers keep the benefits without the headaches of contamination.
The Search for Alternatives
Researchers keep looking for greener chemicals that do what barium periodate can, but for some processes, the alternatives just don’t cut it yet. Funding pushes for safer substitutes do help. I’ve watched younger labs swap in hydrogen peroxide for some jobs, though that comes with limits. Being able to balance progress with responsibility pushes us to keep learning, questioning, and improving how we use these powerful but tricky substances.
What is the chemical formula of Barium Periodate?
Why Barium Periodate Matters
Barium periodate, a less common compound, gets attention in certain circles of chemistry and materials science. This compound falls under the category of periodates, which hold promise for their oxidizing abilities. Getting the chemical formula right matters, especially in labs and industries where a simple tag error on a bottle leads to faulty experiments or unsafe practices. From my own time in a university laboratory, I remember how often a misplaced prefix or wrong subscript sent students scrambling. In chemistry, clarity and accuracy go hand in hand.
Breaking Down the Chemical Formula
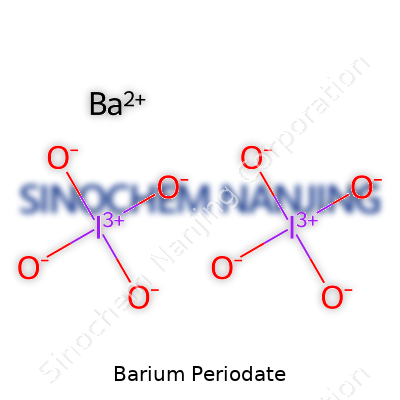
Looking at its structure, barium (Ba) teams up with the periodate ion. The periodate anion appears in two forms—metaperiodate (IO4-) and orthoperiodate (IO65-). For most practical purposes and in common literature, the metaperiodate grabs the spotlight. The barium ion carries a charge of +2, and one periodate anion brings a -1 charge. To even out the charges, two periodate ions combine with one barium ion, giving the formula Ba(IO4)2.
Applications and Concerns
In some rare settings, barium periodate finds use as an oxidizer in reagents or experimental work. Unlike potassium periodate or sodium periodate, it does not show up often in textbooks, but some researchers have explored it for analytical chemistry and synthesis. Barium’s presence raises safety flags: it is not a friendly metal for humans. Soluble barium compounds are toxic, and all work with them calls for gloves, eye protection, and fume hood ventilation. Inexperienced hands may overlook this risk—my old professor repeated warnings anytime barium or periodates appeared on a worksheet.
Periodates, in general, can break down organic material and serve as strong oxidizers. These features make them both useful and dangerous. Spills or improper storage could result in unwanted chemical reactions, possibly releasing iodine or causing combustion if mixed with reducing agents. Institutions responsible for chemical education and industrial safety carry the responsibility to communicate these risks clearly, following Environmental Protection Agency (EPA) and Occupational Safety and Health Administration (OSHA) regulations. Reports suggest that clearly labeled bottles and up-to-date safety data sheets cut down on mishandling and emergency room visits.
Getting the Facts Straight
Barium periodate may not headline many discussions, but its correct chemical formula—Ba(IO4)2—serves as a reminder of how important details become in real-world chemistry. Students, researchers, and lab supervisors rely on accuracy for safety and results. Official chemical registries, like the PubChem database, confirm this formula, showing that rigorous sources align when mistakes are not an option. Anyone handling periodates will want to use trustworthy resources and double-check every label.
Steps Toward Safer Practice
Mistakes with formulas have real consequences. Educational programs help bridge the gap by teaching students the habits of checking sources, reviewing chemical properties, and following protocols. Updating reference materials in laboratories, investing in regular training, and keeping open lines of communication about risks help prevent accidents. Focusing on these clear actions turns chemistry from a potential hazard into a tool for innovation and discovery.
Formula: Ba(IO4)2Is Barium Periodate hazardous or toxic?
The Hazards Lurking in Barium Periodate
Barium periodate isn’t found on everyday shopping lists, but it lands in scientific settings and industries that deal with specialty compounds. Still, just because something was made in a lab doesn’t mean ordinary folks or workers can ignore what it might do to the body or the environment.
From early college chemistry courses, my classmates never shrugged off the barium family. Teachers repeated stories about barium compounds and their potent health effects. It only takes a few mistakes—maybe some powder on your fingers or dust in the air—to learn not everything white and powdery is flour or sugar. Periodate versions of barium combine two sources of risk: toxic barium and a strong oxidizer. Even experienced workers pay attention when weighing or mixing this stuff in an open lab.
What Makes Barium Periodate Risky
Inhaling or touching barium compounds kicks off problems fast. Barium messes with muscles, including the heart. Some common symptoms after accidental exposure include nausea, breathing trouble, and muscle weakness. Cases from places with barium-tainted water tell the story pretty clearly: this element doesn’t belong inside the body.
Periodates offer another layer of concern. They’re oxidizers, which means under the wrong conditions, they can start fires or help other things burn. That’s trouble if storage gets sloppy, or somebody stacks the wrong chemicals together. If spilled, periodate dust can irritate the eyes, skin, and throat—a sharp reminder that laboratory safety needs respect, not shortcuts.
Potential for Real-World Harm
Some may think “specialty chemical” means safety rules don’t spill into daily life, but supply chains, transportation, and waste all carry risk. A truck carrying barium periodate in a crash or someone disposing of it down a regular drain could cause harm that lingers—especially where water contamination is involved. Drinking water with barium in it increases blood pressure, and chronic exposure links to more serious diseases.
Lab accidents crop up in news headlines every year. The common root? Somebody skipped a safety procedure, used the wrong container, or overlooked training. I’ve seen experienced techs hurried by deadlines—gloves coming off early or fume hoods left open. The cost comes later: health checks at the doctor, cleanup crews in hazmat suits, or even lawsuits.
Building a Safer Culture Around Chemicals
A strict routine doesn’t just protect the person mixing up barium periodate; it shields everyone working nearby and people outside the facility. Rules help, but they only work if people understand why they’re in place. Labeling every chemical, providing personal protective gear, and storing reactive compounds in separate rooms each matter. Those steps sound routine, but they keep disaster out of the headlines.
Alternatives exist in some cases, but sometimes only barium periodate will do. So the best that can be done is demand respect for the hazards. Training programs should focus on real-world mistakes, drills for spill response, and easy reporting if gear or storage looks off. The companies I trust most share their incident reports—not because they’re proud something went wrong, but because learning from mistakes keeps the rest of us safe. Honest communication builds better habits.
Handling barium periodate safely means accepting it isn’t an ordinary material. Proper care starts with education, continues with clear protocols, and never stops watching for blind spots. That attitude protects both workers and everyone downstream. Ignoring its toxicity or treating it like another bag of salt is where trouble usually begins.
How should Barium Periodate be stored?
Taking Risks Seriously
Bringing barium periodate into a workspace or lab means stepping up precautions. Many folks might think to treat it as just another inorganic compound, but skipping safety could bring severe consequences. I’ve seen labs that squeezed dangerous chemicals onto makeshift shelves or into old plastic bins, figuring the stuff would “probably be fine.” That mentality invites tragedy. Barium periodate stands out: it holds strong oxidizing power and, mixed with the wrong materials, can ignite or cause toxic exposure.
Understanding the Compound
Barium periodate contains both barium and periodate ions. Both pose their own hazards. Barium on its own can be toxic if inhaled or ingested. The periodate side brings a serious oxidizing punch, which means even a bit of stray organic material—a rag, a stray cardboard box, wood—could kick off a risky reaction. Mixing or even storing such a compound near fuel, flammable solvents, or dust means rolling the dice with unpredictable chemical reactions. I’ve read accident reports that started with a chemical like this sitting in a damp storeroom corner, forgotten about until “something smelled funny” or worse.
Practical Measures Matter
Never store barium periodate near organic chemicals, acids, or materials that burn. Secure it in a tightly sealed, non-reactive container. In my experience, glass storage jars—with proper labeling—work much better than plastic. Store the compounds in a cool, dry place to keep moisture away. Humidity triggers decomposition or, in the worst-case scenario, aggressive chemical reactions.
Storing this compound in a locked chemical cabinet—ideally dedicated to oxidizers—sets a real-world standard. Someone at a community lab I knew once repurposed a metal medicine locker for oxidizers, double-checking that each shelf carried only one class of chemicals. That way, if a spill happened, incompatible substances wouldn’t blend and cause an issue. Cabinet labels in bold colors kept anyone from “grabbing the wrong jar.” Extra padding, such as lined shelf trays, helped to catch messes and reduce the chance of glass cracking. Having a spill kit and fire extinguisher nearby, with staff trained on how to use them, added extra protection.
Documentation and Responsibility
Every time a new member joined our team, we sat down and reviewed the storage plan—not just rules on paper, but the real-life examples of what can go wrong. Safety data sheets for barium periodate belong in an easy-to-find binder. Clarity and transparency serve everyone—nobody should guess how to store or handle a reactive compound.
Cleaning Up the Mess
Even with vigilance, spills sometimes happen. That’s where preparation makes the difference. Keeping a dedicated area for oxidizer storage limits the size of a potential mess. Having absorbent materials, chemical neutralizers, protective goggles, and gloves in arm’s reach doesn’t come from paranoia. It comes from listening to those with scars and stories.
Building a Culture of Safety
The point isn’t to scare people away from scientific work; it’s about building habits that keep people healthy. I’ve watched labs with tight safety routines breeze through inspections and stay accident-free for years, while labs that “wing it” run into bad luck faster than most imagine. No rule matters more than direct respect for both the chemical and the human lives nearby. Barium periodate, respected, can sit on a shelf for years without causing trouble. Take shortcuts, and the risks show up fast.
What are the physical properties of Barium Periodate?
The Substance at a Glance
Barium periodate doesn’t show up on many shopping lists, but in labs, it often pops up when chemists need a strong oxidizing agent. This compound, made of barium and periodate ions, pulls a fair bit of interest due to its striking properties. Let’s look at how it behaves in a real-world context.
Looks and Feel
Barium periodate stands out as a colorless or white crystalline solid. If you ever run into it in a lab, don’t expect it to fade away—its crystals tend to catch the light, producing a bit of a sparkle. The solid form is robust and feels much like fine sand or table salt if you press it between two pieces of glass. Its density tells a key part of the story: hovering around 4.17 grams per cubic centimeter. Compare that to table salt, and you realize you’re handling something much heavier for its size.
Brittleness and Stability
Drop a small sample, and it shatters. Barium periodate breaks easily. This brittleness comes down to the structure at the atomic level. While the compound might look tough, those tightly arranged ions don’t hold each other with the flexibility seen in metals. It crumbles rather than bends. Heat takes its toll too. Barium periodate handles moderate temperatures without fuss, but push it past around 200°C, and it starts to decompose. Oxygen and other byproducts get released—bad news for stability, especially in storage.
Solubility with a Twist
If you’ve got water handy and think you’ll just dissolve some barium periodate, think again. It barely budges in water. A pinch in a glass beaker leaves most of itself behind. Compare that to table salt, which vanishes fast in water, and you see why its water-resistance matters for safety—spills don’t spread quickly. For chemists, this property forces them to handle it with even more care during reactions because undissolved clumps could lead to uneven results or surprises in the lab.
Oxidizing Muscle and Risks
Barium periodate owes much of its attention to oxidizing power. Drop some into the wrong substance, and it sets off strong chemical changes, handing out oxygen atoms to whatever’s nearby. This makes it valuable for some chemical synthesis, analytical chemistry, or even pyrotechnic applications. Yet the same power means accidents can turn serious in a hurry. A spill near combustible material chills experienced lab workers. The compound may not ignite by itself, but it boosts burning with the oxygen it can provide.
Toxicity in Everyday Handling
A huge risk lies in its barium, not just the periodate part. Barium compounds, when soluble—even a little—pose health problems. Scientists understand that accidental ingestion or inhalation can lead to muscle weakness, stomach distress, or much worse. I remember a mentor who stressed gloves, goggles, even a full-face shield, because a single careless moment could hurt for years—especially if small particles go airborne. Labs that care about health keep clear safety protocols: good ventilation, spill kits nearby, and strict waste disposal rules.
Safe Storage and Moving Forward
To get around the hazards, facilities store barium periodate in sealed containers, far from anything that burns easily. Dry, cool cabinets with proper labels help keep accidents from ever starting. Tech isn’t standing still, either. Research teams look at less risky oxidizers and new packaging to avoid dust or contact. Some hope digital inventory tracking can tell workers exactly what sits where, cutting human error.
Understanding these hands-on physical properties of barium periodate—texture, density, fragility, solubility, strength as an oxidizer, and toxic potential—helps everyone from students to pros make safer, smarter choices, whether in research, industry, or regulatory fields.
| Names | |
| Preferred IUPAC name | Barium periodate |
| Other names |
Barium metaperiodate Barium periodate(VII) |
| Pronunciation | /ˈbeəriəm pəˈrɪəʊdeɪt/ |
| Identifiers | |
| CAS Number | 13718-50-8 |
| Beilstein Reference | 358126 |
| ChEBI | CHEBI:31114 |
| ChEMBL | CHEMBL1201646 |
| ChemSpider | 24237 |
| DrugBank | DB11145 |
| ECHA InfoCard | 100.032.415 |
| EC Number | 234-374-9 |
| Gmelin Reference | 39200 |
| KEGG | C18763 |
| MeSH | D001477 |
| PubChem CID | 24891768 |
| RTECS number | CQ9625000 |
| UNII | 13S278A93H |
| UN number | UN1472 |
| Properties | |
| Chemical formula | Ba(IO4)2 |
| Molar mass | 487.136 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 4.28 g/cm3 |
| Solubility in water | Insoluble |
| log P | -1.32 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 12.2 |
| Magnetic susceptibility (χ) | +65.0e-6 cgs |
| Refractive index (nD) | 2.65 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 220.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1286.71 kJ/mol |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes eye and skin irritation |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H332 |
| Precautionary statements | P220, P221, P280, P370+P378 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 3, Special: OX |
| LD50 (median dose) | LD50 (median dose): Oral rat 118 mg/kg |
| REL (Recommended) | REL: 0.1 mg/m³ |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Barium permanganate Barium chlorate Barium perchlorate Barium iodate Barium nitrate |

