Barium Perchlorate: More Than a Simple Chemical
Tracing the Path: A Short History of Barium Perchlorate
Barium perchlorate isn’t just another overlooked salt. It has roots stretching back to early studies in inorganic chemistry, when scientists chased the mysterious properties hidden in rare alkaline earth metals paired with powerful oxidizers. The mix of barium—a heavy metal known for its density and vivid flame test—with the highly reactive perchlorate ion has kept labs experimenting for more than a century. Way back, researchers exploring oxidative energetics discovered its unique ability to break apart stubborn compounds, which quickly caught the attention of those working with explosives and rocket propulsion. Since its early synthesis and documentation, barium perchlorate has found a stubborn spot in the toolkit of both the analytical chemist and the eager engineer.
A Closer Look: What Sets Barium Perchlorate Apart
Most people walk past barium perchlorate every day (at least metaphorically), but those who have handled it know its distinct personality: a white, crystalline solid, easily soluble in water, with a bitter taste that fortunately stays locked away under strict lab controls. Its chemical formula, Ba(ClO4)2, hints at trouble if misused. Unlike its relatives barium chloride or potassium perchlorate, it poses more risk due to both the toxicity of barium ions and the instability of perchlorate when near organic material or heat.
What Science Says: Physical and Chemical Traits
This compound shows a straightforward crystalline structure, sometimes forming as a monohydrate. Its high solubility stands out compared to similar salts; dump it into water, and it disappears fast, which is both a blessing and a curse. Barium’s weight dominates its physical characteristics. At the same time, perchlorate’s eager oxygen atoms give it strong oxidizing power—a feature sought after but dangerous in untrained hands. The pure salt manages to keep long-term stability when stored dry, but startle it with sparks or friction mixed with flammable materials and it turns from benign to explosive.
Details Matter: Technical Specs and Lab Labels
Anyone who has spilled some on the lab bench can attest: barium perchlorate deserves respect. The labels tend to shout warnings: “Strong oxidizer. Toxic. Handle with gloves.” Standard to see CAS No. 13465-94-6 on the jar, along with hazard codes that remind one of grade-school chemistry gone serious. Professionals working with it check purity above 98 percent, prefer tightly sealed moisture-resistant packaging, and pay close attention to the unavoidable contaminant profile, since stray impurities can tip the compound from inert to reactive.
Making the Stuff: Preparation on the Bench
The classic method involves reacting barium chloride or barium carbonate with a soluble perchlorate, usually sodium or potassium perchlorate, in water. Precipitation and simple filtration drop out sodium or potassium ions, leaving barium perchlorate to crystallize from the solution. This seemingly easy process runs into snags, though, due to perchlorate’s tendency to form stubborn hydrates and its risk of contaminating water supplies. Industrial production scales up the process, focusing on reducing residual contaminants and capturing wastes, especially since both barium and perchlorate face environmental scrutiny.
Getting Reactive: Chemical Conversion and Tweaks
Barium perchlorate jumps into action in a bunch of reactions. It gives up its perchlorate ion willingly, making it a favorite oxidizer in analytical and synthetic chemistry. Bomb calorimetry relies on its ability to provide oxygen in a controlled, measurable way. Given the right conditions, it plays a role shifting perchlorates between different metal cations. The reactivity makes it versatile but also demands tight storage and sensible handling, especially anywhere organic material or reducing agents might be found. Those who ignore the warnings about mixing perchlorates with sulfur or phosphorus often become stories told in safety briefings.
Other Names: Synonyms on the Shelf
Open a chemical catalog, and you might see it offered not just as barium perchlorate, but as barium diperchlorate or perchloric acid, barium salt. Sometimes labels mention its monohydrate form. No matter the spelling or the language, the risks and the benefits remain unvarnished.
Treading Carefully: Safety and Handling
Barium perchlorate doesn’t belong in the hands of the unwary. Both barium and perchlorate bring toxic baggage. Inhalation of dust invites trouble, and accidental ingestion opens the door to the well-documented dangers of barium poisoning: muscle weakness, heart irregularities, even death. The perchlorate half disrupts thyroid hormones, throwing metabolism out of balance. So, responsible labs invest in fume hoods, gloves, and locked chemical cabinets. Waste management plans matter just as much as accident response drills. The compound’s strong oxidizing ability means it can't share storage with flammable liquids, organic powders, or even some metals. Outdoor exposure carries regulatory limits, driven by groundwater protection and long-term health studies.
Where It Shows Up: Real-World Applications
Despite its risks, barium perchlorate still makes a name for itself in specialized corners. Analytical chemists use it as a powerful oxidizing agent in bomb calorimetry—a precise method for measuring the energy content of fuels. Military and space research tap its oxygen-delivering punch to boost propellants and specialty explosives, enabling controlled power release where nothing else quite fits the bill. Even in civilian labs, some water testing kits call on its properties, although environmental regulators frown on its use in open systems. Electrochemists and materials scientists sometimes reach for it when working with unusual batteries or electrolyte systems, chasing properties that mainstream salts just can’t copy.
Pushing Boundaries: A Tool for Research and Experimentation
Researchers never seem to exhaust their questions about barium perchlorate. Some keep teasing apart its electrochemical behavior, hunting for ways to store and release energy efficiently. Others chase its crystallization path and complex forming habits, using X-ray diffraction to map structures and predict new reactions. Environmental chemists monitor its release and degradation, hoping to understand just how it migrates through soil and water. Medical and toxicology labs test its impact on health, tracking subtle shifts in metabolism and hormone pathways to spot long-term risks that go unnoticed outside the lab.
Facing the Risks: Ongoing Toxicity Studies
It doesn’t take a PhD to read the warning on a bottle of barium perchlorate, but ongoing research only broadens the picture of its risks. Studies focus on two fronts: immediate dangers if handled unsafely, and chronic effects from tiny exposures over years. Barium ions slip across membranes, disrupting muscle and nerve function in ways both spectacular and subtle. Perchlorate, stubbornly persistent in water, blocks iodine uptake in the thyroid. Communities living near industrial plants producing perchlorates often show higher rates of thyroid issues. Regulatory agencies keep tightening limits, motivated by animal studies and a slow trickle of human epidemiology that refuses to declare the compound safe outside carefully controlled environments.
On the Horizon: The Road Ahead for Barium Perchlorate
Looking toward the future, the road for barium perchlorate winds between advancing science and rising caution. Industry leaders and chemists alike search for safer and greener oxidizers, driven both by regulation and a growing sense of social responsibility. Alternatives do appear on the horizon, but for highly specialized uses—where performance and predictability matter as much as price—barium perchlorate’s tough-to-replace features keep it relevant. The challenge will be finding smart ways to contain, recycle, or neutralize its toxic legacy while letting its formidable chemical power serve research, industry, and even space exploration. For now, it remains both a tool and a warning, teaching risk management alongside reactivity.
What is Barium Perchlorate used for?
A Closer Look at Its Uses
Barium perchlorate often rings a bell in conversations about pyrotechnics or laboratory reagents. Scientists reach for it when strong oxidizers are needed, especially in chemical synthesis and analytical chemistry. Its high solubility in water gives it a unique spot among perchlorates, making it handy for experiments that depend on accurate and predictable behavior in solutions. In my college chemistry days, I watched a lab group handle this compound with serious care—its reactivity with organic matter demands nothing less. This isn’t just a matter of lab safety; cities and manufacturers have learned expensive lessons about what happens when reactive chemicals aren’t managed with respect.
Outside academic settings, engineers sometimes turn to barium perchlorate for creating solid rocket propellants. Its properties help engines burn hotter, faster, and cleaner—a necessity in aerospace where every ounce of thrust counts. Fireworks artists also rely on it for brilliant displays, thanks to the intense flames produced during combustion. Some industrial water treatment operations involve small amounts, exploiting its power to remove impurities that other agents leave behind. There’s no shortage of creativity in how people have applied the compound across fields.
Health and Environmental Concerns
People working with barium perchlorate need to watch their step. Barium is toxic, and perchlorate ions disrupt thyroid function by blocking iodine uptake—a threat that grows with repeated exposure. My first experience handling such chemicals drilled the importance of gloves and fume hoods into me, long before any incident happened. Years ago, a minor spill at a friend’s workplace led to a full evacuation and days of soil remediation. Even small mishaps can ripple out, closing down workspaces and causing headaches for local authorities. Long-term exposure to perchlorates, whether at work or through contaminated drinking water, poses health risks that communities struggle to manage. Studies from the CDC point out thyroid disorders can show up in areas with high perchlorate contamination.
Environmental impact stretches beyond safety goggles and lab coats. Improper disposal or accidental release into waterways makes perchlorates tough to clean up. Some towns in California have already grappled with groundwater perchlorate plumes, forcing millions into expensive water treatment. Regulators like the EPA have pushed for tighter controls, but enforcement can lag behind industrial innovation. Barium perchlorate’s strong oxidizing nature means it accelerates decomposition in organic matter, complicating cleanup efforts whenever an accident occurs.
Safer Handling and Future Solutions
People’s lives change when accidents happen, which pushes professionals to chase safer processes. More and more research shifts toward greener alternatives or more stable oxidizing agents. At some rocket labs, teams have swapped out barium perchlorate for less hazardous substances, aiming for similar performance with fewer risks. Companies investing in better spill containment and real-time monitoring avoid some of the costly disasters seen elsewhere. My own fieldwork taught me to double-check protocols, especially with compounds that can threaten both people and drinking water supplies. Training, regular audits, and community transparency do more than simply tick off boxes—they help prevent the next big crisis.
It all boils down to staying informed, proactive, and honest about the ingredients that drive science, technology, and art forward. Barium perchlorate remains useful but comes with demands that shouldn’t get brushed aside. That respect saves jobs, protects health, and shields entire neighborhoods from trouble.
Is Barium Perchlorate hazardous or toxic?
The Realities Behind a Powerful Chemical
Science classes often teach that chemistry isn’t just about mixing harmless ingredients. Some substances bring excitement, others come with a fair share of risks. Barium perchlorate falls into that tricky zone. Used mostly in the lab or industry—not household cabinets—it packs energy and carries real dangers for those handling it.
Why Precautions Matter
When someone mentions barium perchlorate, the phrase “explosive compound” can grab attention quickly. This chemical contains both barium and perchlorate ions, each carrying distinct hazards. Let’s break it down. Barium itself is well-known for being toxic to humans and animals. Swallow even a small amount, and serious health effects show up—ranging from muscle weakness to dangerous shifts in heart rhythm.
The perchlorate ion isn’t innocent either. It can disrupt the thyroid’s natural ability to absorb iodine. That leads to problems with hormone production. Most research on this link points to high levels or chronic exposure, but it reminds us not to take shortcuts with safety.
Beyond the Lab Bench: Environmental Impact
My years around chemistry labs taught me that spills or careless disposal haunt communities for a long time. Perchlorates travel through soil and water pretty easily. That persistence lets them slip into drinking water, where even tiny doses matter. Several states in the U.S. flag perchlorate as a water contaminant. Regulatory limits exist for a reason. Nobody wants to take chances with kids’ health or a municipality’s public supply.
Another headache comes with the barium ion. Plants and animals don’t handle barium exposure well, so runoff or improper dumping causes ripple effects through an ecosystem. It doesn’t just go away on its own—modern clean-up efforts get expensive and take time.
Workplace Safety: Experience Counts
Anyone asked to handle barium perchlorate in a job setting gets warned about its twofold risk: it’s highly oxidizing and hazardous to health. Even simple friction or a stray spark could trigger violent reactions with organic materials, metals, or reducing agents. That’s a recipe for a lab fire. I’ve seen colleagues gear up in full protection, double-check protocols, and keep neutralizing agents within arm’s reach. An emergency shower and eyewash station shouldn’t feel like an afterthought.
One slip in safety training means exposure by skin, inhalation, or accidental ingestion. Acute poisoning cases need quick attention—respiratory issues and cardiac problems can follow.
Building a Safer Future
A single accident teaches people the value of engineering controls and training over shortcuts. Plenty of organizations tackle these dangers by storing barium perchlorate away from anything flammable, using strict temperature and humidity controls, and conducting regular safety drills. Workers check their equipment before every use, not because a checklist says so, but because someone they know already got hurt.
For community safety, more investment should go toward water testing, transparent reporting of perchlorate levels, and safe disposal technology. Government oversight matters, and so does honest communication with the public—especially near sites where this compound turns up in large amounts.
Barium perchlorate might solve technical challenges in industry, but ignoring its hazards brings problems nobody wants. My own experience says prevention, not reaction, leads to safer work and healthier neighborhoods. Facing up to the risks head-on, with reliable information and commonsense action, makes the difference.
How should Barium Perchlorate be stored?
Respecting the Hazards of Barium Perchlorate
Anyone who has worked in a chemistry lab remembers that some chemicals seem ordinary at a glance, but they hold big risks behind that white powder or crystal appearance. Barium perchlorate fits right into that list. This compound stands out because its contact with flammable materials can cause fires, and its ability to absorb water means that storing it anywhere humid spells trouble.
Choosing the Right Storage Location
I have seen several near-misses born simply from a poor storage spot. Using advice from safety resources and OSHA standards, the dry cabinet in a chemical storeroom wins as a proper place to keep barium perchlorate. Any cabinet holding it should keep out water vapor, stop light from breaking down the crystals, and remain far from any door or air vent to cut down on accidental mixing with other chemicals.
Compatible Storage Materials
Experienced chemists already know glass or high-quality plastics resist corrosion and keep oxidizers from reacting with storage containers. Metal isn’t the friend you want in this case; even tiny corrosion or an unnoticed nick can start a reaction. I keep barium perchlorate only in containers with screw-tight lids that seal out air and minimize the risk of moisture slipping in. Labels don’t seem like a big deal day to day, but clear writing prevents anyone from grabbing the wrong jar in a rush. Skipping regular label checks can lead to mistakes nobody wants to clean up.
Limiting Quantity and Isolation
No one needs an oversized stash in regular labs, so bottles only hold what’s needed for short-term use. Extra stock stays in central chemical storage, behind locked doors, out of sight. Never store this compound with organics, reducing agents, acids, or anything combustible. An old coworker once kept perchlorates near alcohols; cleanup crews had to vent the building before anyone could work, all because of improper storage. Pro tip: never let perchlorates share a shelf with anything they could react with.
Environmental Controls
Air conditioning and dehumidifiers shouldn’t be overlooked. Lab managers who ignore humidity let crystals clump, making them unpredictable to handle. I use hygrometers to keep the space under 50% relative humidity, no exceptions. If there’s a spill or a leaky jar, the dry space keeps reactions from starting until cleanup teams can step in.
Fire Safety and Spill Preparedness
Barium perchlorate’s role as a strong oxidizer means standard fire extinguishers don’t cut it. Only Class D extinguishers work against metals and their compounds. A lab that keeps this compound must have a plan: fire blankets, sand buckets, and metal fire extinguishers within reach. Training everyone who steps into the room beats reading about hazards on a safety sheet. I’ve run more drills than I can count, and they’ve always made a difference in tense moments.
Respect for the Risk, Respect for Each Other
Long experience in the lab teaches that short cuts with storage run out fast. Barium perchlorate must be kept with careful respect for its instability, always separated by physical distance and with solid barriers. People’s safety depends on more than theory; strong habits save lives and livelihoods. It’s not just the right chemical practices—it’s the right culture.
Practical Solutions for Better Storage
Strong habits start with regular checks on containers, maintaining humidity systems, and real training on spill response. Digital inventory logs prevent over-ordering. In tough budget times, group purchasing to cut down on unnecessary stocks can spare resources and risk. Ignoring proper storage for barium perchlorate isn’t an option. The payoff is a lab that runs safely, people who can trust each other, and a healthy respect for what powerful chemicals can do if left unchecked.
What is the chemical formula of Barium Perchlorate?
Understanding the Chemistry
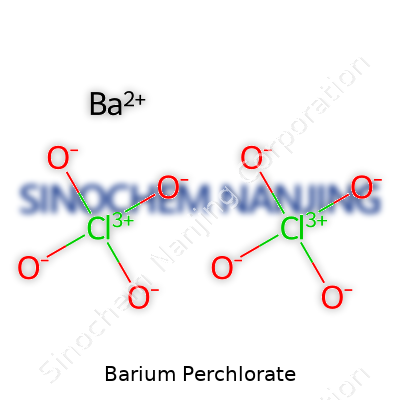
Barium perchlorate stands out in chemistry labs and industrial plants thanks to its unique make-up. This compound carries the formula Ba(ClO4)2. That’s not just some random arrangement of elements. On a basic level, it comes down to a single barium atom bound with two perchlorate groups. Each perchlorate group contains a chlorine atom surrounded by four oxygen atoms. The barium atom gives up its electrons, and the resulting ion pairs with the highly reactive perchlorate groups.
To someone working in chemical manufacturing or environmental testing, these details aren’t just textbook trivia. A misstep in stoichiometry leads to costly mistakes. If you grow up around farms or local water treatment facilities, you probably notice safety measures plastered all over any container that mentions perchlorates or barium. Both carry risks to plants, animals, and people if handled carelessly.
Why Does Ba(ClO4)2 Get Used?
Beyond lab curiosity, barium perchlorate plays a real role in fireworks, explosives, and rocket propellants. Those bright green colors on the Fourth of July? That often comes from barium-based salts. Perchlorates, with their high oxygen content, feed combustion reactions and release energy fast. That’s great for pyrotechnics, but bad news if you worry about environmental fallout.
Some water treatment companies run across perchlorate contamination. Perchlorates don’t break down quickly in the environment. Out in the real world, regions near defense plants and fireworks factories track groundwater for perchlorate runoff and barium buildup. My work with a small-town water utility drove home how tough it is filtering these ions out. You end up relying on advanced techniques like ion exchange, raising costs for communities already struggling to upgrade old pipes and equipment.
Risks and Regulations
Barium itself isn’t the neighbor you want leaching into your drinking supply. Exposure brings health risks, especially for people with hypertension or heart problems. The Environmental Protection Agency set limits for how much barium contaminates public water. Perchlorates, while present in everyday items like airbags and matches, come under special scrutiny. A single spill at a fertilizer warehouse nearly shut down our local river for weeks during my college years, and the community pushed for tougher inspection rules after that.
Industrial sites handling barium perchlorate operate under strict safety codes. Accidents draw heavy penalties, not just for environmental clean-up, but for employee safety. OSHA rules kicked in during several projects I've watched up close, adding frequent inspections and new containment equipment any time this compound made it onto the supply list.
Addressing the Challenges
Tackling the double edge of barium perchlorate starts with better handling and disposal. At my job, I watched seasoned technicians double-check containers for leaks and review spill kits before even touching a drum. New research has focused on less-reactive alternatives in fireworks and fertilizers to cut down perchlorate migration. Some teams now pilot test advanced filtration membranes, aiming to capture both barium and perchlorate ions before they enter wastewater streams.
Public awareness works wonders, too. Our town set up annual “Chemical Safety Days” at local schools, pairing hands-on demos with info about how substances like barium perchlorate show up in everyday life. Investing in education, paired with smart regulation and new technology, gives communities their best shot at balancing the perks of chemistry with the safety net everyone deserves.
What are the safety precautions when handling Barium Perchlorate?
Understanding the Risks
Barium perchlorate stands out in any lab for its crisp white crystals, but there’s nothing harmless about it. The stuff kicks off heavy reactions with organic material, and it doesn’t play around with moisture or heat either. A friend once worked in a facility where a careless spill meant the safety team lost a week scrubbing and testing everything for residue—no one wants that kind of panic. It’s oxidizing and highly toxic to humans; even a bit of dust in the air means trouble for the lungs and nervous system. Research tells us that even ingesting a small amount causes muscle spasms, irregular heartbeat, and in the worst cases, respiratory failure.
Personal Protection Comes First
Simple rules make the difference. I never skip goggles or a face shield when I’m near this compound. A thick lab coat—preferably flame-resistant—serves as a barrier. Decent chemical-resistant gloves are a must, and not just nitrile; for this job, go for gloves engineered for oxidizing agents. Stepping into a good pair of closed work shoes (no sneakers allowed) finishes out the basics.
Here’s a real lesson—after handling barium perchlorate, ditch those gloves, scrub up, and keep hands away from the face. It’s easy to forget, especially after a long day, but the consequences just aren’t worth the risk. I keep skin creams off before a shift too, since lotions can trap chemicals close to the skin.
Ventilation and Workspace Controls
Ventilation saves lives. Always work with barium perchlorate in a fume hood or a well-ventilated area. Leaving dust or vapors to build up in a closed room almost guarantees a visit to the doctor—or worse. I remember a session where our aging exhaust system failed mid-way through a process, setting off alarms. No one got hurt, but only because we made it a habit to wear respiratory protection, even when things seemed normal.
Keep your work zone clean, dry, and free of clutter. Static discharge easily sets off this compound, so grounding equipment is not a suggestion, it’s an absolute. Forget paper towels—grab spill control pads rated for oxidizers. Store barium perchlorate far from organic materials, flammable solvents, and acids. If storage space runs short, prioritize clearing flammables and acids to another part of the building—never try to fudge this one.
Emergency Response Plan
Everyone on the team needs to know where eyewash stations and safety showers are. It’s not enough to just point them out—run practice drills for chemical spills with the real equipment, so panic never becomes the main event. Quick access to an emergency spill kit is essential. Know the clean-up procedure cold: douse minor spills with soda ash to neutralize, then scoop up carefully for hazardous waste disposal.
Barium perchlorate often means hazardous waste, both liquid and solid. Label waste containers before use, and always track disposal in line with local and federal rules. I once saw an unlabeled bottle stored under a bench for weeks before it was found—no one could remember who left it there, and it forced a costly shutdown for hazmat removal.
Supporting Safe Work Every Day
Training matters more than fancy equipment. Supervisors keep sessions up-to-date and make time for refresher courses on chemical hazards. Sharing near-misses during meetings isn’t about blame; it builds a culture where no mistake gets swept under the rug. Speak up if something feels unsafe—it’s not overreacting, it’s protecting everyone in the lab.
Handling barium perchlorate safely depends on everyday habits, honest communication, and respect for the power of the material. Taking one shortcut erases all those careful steps, and that risk is never worth it.
| Names | |
| Preferred IUPAC name | Barium diperchlorate |
| Other names |
Perchloric acid, barium salt Barium diperchlorate |
| Pronunciation | /ˈbeəriəm pərˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 10099-19-9 |
| Beilstein Reference | 358691 |
| ChEBI | CHEBI:63043 |
| ChEMBL | CHEMBL3750616 |
| ChemSpider | 12266 |
| DrugBank | DB11104 |
| ECHA InfoCard | 0260-377-9 |
| EC Number | 231-868-0 |
| Gmelin Reference | 84511 |
| KEGG | C18650 |
| MeSH | D001467 |
| PubChem CID | 24636 |
| RTECS number | CU6120000 |
| UNII | 2YDH18Z5RN |
| UN number | UN1476 |
| Properties | |
| Chemical formula | Ba(ClO4)2 |
| Molar mass | 336.23 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.74 g/cm³ |
| Solubility in water | Very soluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -2.0 |
| Basicity (pKb) | -6.8 |
| Magnetic susceptibility (χ) | -72.0e-6 cm³/mol |
| Refractive index (nD) | 1.684 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 202.5 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1408 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | Barium Perchlorate: –2540 kJ/mol |
| Pharmacology | |
| ATC code | V09CA02 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H301, H314 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3 0 2 OX |
| Lethal dose or concentration | LD₅₀ orally in rat: 9.5 mg/kg |
| LD50 (median dose) | Barium Perchlorate LD50 (oral, rat): 117 mg/kg |
| NIOSH | NL3675000 |
| PEL (Permissible) | PEL: 0.5 mg/m3 |
| REL (Recommended) | PEL-TWA 0.5 mg/m3 |
| IDLH (Immediate danger) | 150 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium perchlorate Magnesium perchlorate Sodium perchlorate |

