Barium Nitrite: Unfolding Its Importance in Science and Industry
Historical Development
Barium nitrite grew out of the early days of chemical synthesis, when researchers explored nitrite chemistry for military and agricultural uses. Chemists in the 1800s recognized its utility because of its reactivity and distinctive yellowish hue. Early methods relied on converting available barium compounds, often casting a wide net for ways to use barium salts, as nitrite salts caught the curiosity of academics and explosives manufacturers alike. Work in laboratories during the Industrial Revolution led to better control over purity and safer handling, which allowed its wider use. Barium nitrite earned a place in textbooks not only for its unique colorimetric applications but also for acting as a stepping stone to more complex compounds that fueled further discoveries in inorganic chemistry.
Product Overview
Barium nitrite stands out as a pale yellow, water-soluble powder. Its chemical formula, Ba(NO2)2, places it squarely within the realm of heavy-metal nitrites. Its commercial value emerges from its ability to serve as a precursor or reagent in several synthesis processes, including organic transformations and specialized compound preparations. Producers and users value its predictable reactivity, but also respect the toxic edge carried by the barium ion.
Physical & Chemical Properties
Barium nitrite appears as a crystalline, light-yellow solid. It dissolves easily in water, forming a solution that remains clear as long as contaminants are controlled. Its density and melting point, at about 2.99 g/cm³ and 120°C respectively, give insight into its robustness and handling requirements. The compound remains stable under ordinary storage, but exposure to acidic conditions may release toxic nitrogen oxides. This reactivity with acids renders it a useful but risky participant in nitrite-related chemical reactions. Its solubility, over 50 grams per 100 mL at room temperature, is high enough to ensure broad practical application without the need for excessive solvents.
Technical Specifications & Labeling
Suppliers ship barium nitrite under strict labeling protocols, marking containers with proper hazard symbols relating to both toxicity and environmental release. Labels specify purity–commonly above 98%–and lot number, aiding traceability. Transport regulations demand use of UN identification code 1564, with instructions clear on separation from food or feedstuffs and away from flammable material. Material Safety Data Sheets offer additional instructions for personal protective equipment, ventilation, and first aid measures. Clarity here reduces accidents and ensures professionals respect the risks.
Preparation Method
Industrial-scale production relies on double displacement. Typically, manufacturers react barium chloride with sodium nitrite, forming barium nitrite as a precipitate and sodium chloride as the byproduct. The equation looks simple but delivering high purity demands washing, filtration, and often multiple recrystallization steps. Homegrown chemistry may fall short without lab-grade reactants and careful control over temperature and mixing speed. The process doesn’t forgive shortcuts—failure brings unreacted starting materials or dangerous contamination, especially with unwanted azides or nitrates sometimes forming under the wrong conditions.
Chemical Reactions & Modifications
Barium nitrite’s role as a nitrosating agent attracts both educators and industrial chemists. In the presence of acids, it liberates nitrous acid (HNO2), which in turn opens up the synthesis of diazonium salts, vital for the dye and pharmaceutical industries. Strong reducing agents will break it down to barium oxide or elemental barium, an exercise better saved for controlled settings. Its reaction with ammonium salts generates barium ammonium nitrate, used as a starting material for certain explosives. Each modification brings a new hazard profile, so protocols anchor themselves around careful reagent addition and strict observation.
Synonyms & Product Names
Barium nitrite goes by a handful of names, including nitrous acid barium salt. Some catalogues list it as barium dinitrite. In European regulation documents, it appears under EINECS number 233-653-7. Specialty chemical suppliers sometimes adopt proprietary branding, but the molecular formula tends to clarify confusion quickly.
Safety & Operational Standards
Few materials demand respect like barium nitrite. Inhalation, ingestion, or prolonged skin exposure brings significant health risks because both barium and nitrite ions disrupt biological processes. Acute exposure may result in vomiting, muscle cramps, and cardiac arrhythmias. As a result, laboratories and production environments require gloves, eye protection, and mechanical ventilation. Waste barium nitrite is never left for landfill; hazardous waste contractors collect and treat it through precipitation or stabilization. Facilities hold regular training, emphasizing spill containment, emergency eyewash stations, and strict inventory management. Regulatory compliance guides its every move from shelf to disposal, tying together personal responsibility and institutional oversight.
Application Area
In the real world, barium nitrite plays a role in fabric dyeing, certain pyrotechnic compositions, and as a laboratory reagent. Dyers use it for color fixation on fabrics, capitalizing on its controlled release of nitrogen oxides. Explosives manufacturers appreciate its role in formulations where reliable ignition and burn rates make all the difference. Specialized batteries research sometimes points to its electrochemical properties, though toxicity curtails widespread use. In analytical settings, chemists employ it to detect certain cations, including cobalt and manganese, via spot tests in educational or research labs.
Research & Development
Modern research addresses two broad goals: increasing safety and expanding green chemistry alternatives. Teams push methods to capture barium and nitrite waste, developing protocols for recovering or neutralizing spent materials. Others chase organic synthesis pathways that swap barium nitrite for less hazardous nitrite donors. Interest continues around its colorimetric responses, with new sensor technologies leveraging its reactivity. Pharmaceutical development sometimes revisits classic diazonium chemistry, with barium nitrite as the nitrosating agent of choice despite health risks—pursued only under the tightest controls.
Toxicity Research
Research on barium nitrite’s toxicity digs into both acute and chronic effects. Studies link ingestion to muscle paralysis and sudden drops in blood pressure; historic accidental exposures led to strict storage rules in schools and research centers. Chronic exposure research still lags because long-term cases remain rare outside of manufacturing environments, but animal studies hint at possible kidney stress and nervous system effects. Regulatory bodies, including OSHA and REACH, maintain low exposure limits and demand immediate cleanup following any spill or suspected contact.
Future Prospects
Looking ahead, future prospects lie in pairing better containment technologies with greener substitutes where feasible. Pressure builds for closed-loop chemical processes, especially in dye and explosives industries, where barium nitrite’s hazards force a choice between efficiency and corporate responsibility. Researchers continue to test organometallic nitrite compounds, aiming for the same reactivity without the downsides. Interest in next-generation sensors and analytical kits still values barium nitrite for reproducibility, so niche applications likely remain, though expectations grow for improved safety and real-time exposure monitoring in every setting where it appears.
What is Barium Nitrite used for?
Why Barium Nitrite Matters in Today’s Labs
Most folks don’t talk about barium nitrite down at the coffee shop. Still, if you look into industrial chemistry or research labs, this yellow-green salt pops up more than you might guess. As a person who has spent a fair share of time in college chemistry labs, I’ve witnessed its use up close. It’s not headline news, but its role doesn't get the attention it deserves outside expert circles.
How Barium Nitrite Ends Up in Industry
Barium nitrite mostly finds its place in scientific research and specific manufacturing settings. In practice, it acts as a strong oxidizer in the synthesis of other chemicals. Chemists reach for it when they need something to jumpstart complex reactions — especially for creating other nitrites or preparing barium azide. They pick it because it gets the job done fast and doesn’t slow down production lines.
Its main home sits with specialty glassmaking. Adding barium nitrite to glass can boost certain optical properties, making the material a bit more useful in things like precision lenses or specialty lab equipment. It gives the glass a refractive index bump and can help filter out some unwanted wavelengths. That’s not something people think about when they snap a photo or peek through a microscope, but it makes a difference for companies trying to stand out in a crowded market.
Challenges with Safety and Handling
Chemists think twice before touching barium nitrite without proper gear. This stuff can cause serious health problems if it gets into the body, either through breathing, eating, or skin contact. Once inside, it messes with the body’s ability to carry oxygen. In my early days at the lab, our supervisor repeated one rule above all: never let your guard down around toxic salts like barium nitrite. The stories I heard about careless handling and hospital trips left an impression.
Industry workers use heavy-duty gloves, masks, and tightly regulated handling and storage procedures. Strict rules protect both people and the environment, since letting any barium compound slip into the water supply spells trouble for ecosystems and public health. Agencies like OSHA and the EPA keep a close eye on how companies store and discard this material, with hefty fines for slipping up.
Environmental and Ethical Considerations
Even with good safety rules, accidents still happen. Back in the 1990s, several companies faced hard questions about heavy metal leaks near manufacturing sites. Some of those leaks involved barium compounds, with harmful effects on drinking water and local wildlife. Damage control teams spent years cleaning up the mess. Since then, modern plants have brought in upgraded containment systems — real investments in better storage and emergency response plans. Responsible manufacturers test storage tanks and drainage lines on a regular basis, not just once a year.
Smarter Alternatives and the Road Ahead
New tech and fresh policies open the door for stronger protections and even new substitutes. Green chemistry initiatives encourage scientists to search for safer oxidizing agents and alternatives in glass production. While barium nitrite hasn’t vanished, more labs experiment with less toxic compounds. The push for sustainability is only getting louder, and this could mean less demand for hazardous salts in the years ahead. I keep rooting for that shift — progress in science should never come at the cost of human or environmental health.
Is Barium Nitrite hazardous to health?
What I’ve Learned About Barium Nitrite
Before I ever worked around chemicals, I’d never heard of barium nitrite. I only ran into it because my first job out of college put me near a small lab, and, as the new guy, I had to handle inventory. That’s where I got a crash course in chemical safety from a patient old chemist named Mr. Huang. He made a point to look me in the eye and say, “You need to respect what you’re handling.” Barium nitrite was on his “don’t mess with this” list.
What Makes Barium Nitrite Risky?
Barium nitrite carries serious health concerns. Its formula, Ba(NO2)2, masks real dangers. Many people don’t realize tiny exposures to soluble barium compounds matter, not just the big, obvious spills. The nitrite part gives this salt its bright color, but also brings toxicity. Breathing the dust or getting it on your hands can introduce it straight into your system. Once inside, barium ions interfere with muscle control. Instead of just a cough or rash, you’re looking at muscle weakness, irregular heart rhythms, and, in heavy doses, organ shutdown.
The Science and the Symptoms
Barium interrupts the normal flow of potassium in and out of cells. That can spark everything from tingling fingers to violent muscle spasms, depending on your exposure. Nitrites turn the blood’s hemoglobin into methemoglobin, which doesn't carry oxygen well. Someone exposed might look blue or get short of breath. It’s not rare—industrial accidents have shown these symptoms can show up fast. Several case studies, some from the US National Library of Medicine, show that routine mistakes (like not wearing gloves or working in a poorly ventilated space) put workers in the ER after a spill.
Real-World Precautions
My early days in the lab taught me that personal protection isn’t just lab-coat theater. Mr. Huang drilled it into new hires. Gloves, goggles, and strict cleaning routines mattered. Barium nitrite never left the chemical fume hood. Respirators came out if anything needed mixing or measuring. In my experience, someone ignoring this routine believed nothing bad would ever happen to them. That belief could get someone sent home in an ambulance.
Better Choices—Training and Accountability
Fixing chemical hazards doesn’t rest on warning labels. Regular training—talking through real cases, not just handing out a sheet with hazard codes—keeps people alert. Every lab manager I respected had a “no shortcuts” mindset about safety. Emergency eyewash stations, immediate spill cleanup policies, even color-coded aprons for toxic substances—they all provided extra insurance against mistakes.
On the policy side, it’s about transparency. Workplaces should publish records of chemical incidents and near misses. That helps set realistic expectations—nobody ever benefits from hiding close calls. I look at it this way: knowing what’s gone wrong before gives everyone a shot at preventing it next time.
Alternative Approaches
Education—starting as early as high school chemistry—helps future workers recognize chemical hazards quickly. Modern labs can swap in less-toxic compounds for classroom demos. Online safety simulators let people practice responding to spills without real risk. In industry, routine medical screening for those handling toxic salts can pick up subtle symptoms before they become emergencies.
Why This Matters to Me
People with no reason to know about barium nitrite often face it on accident—tinkering at home, working a new job, cleaning up a spill. My experience tells me one careless mistake can snowball. Treating every hazardous compound with skepticism changes outcomes. If you work near barium nitrite—even just for a few hours—every small step you take for safety counts. That attitude, not just protocols, keeps people healthy and out of the hospital.
What is the chemical formula of Barium Nitrite?
Digging Into the Basics
Barium nitrite lives between high school chemistry textbooks and industrial supply catalogs. Its formula, Ba(NO2)2, tells a bigger story than just a string of letters and numbers. That little formula links lab work with large-scale applications, stretching from pyrotechnics to specialized reagents.
Understanding the Building Blocks
Barium belongs to the alkaline earth metals. Anyone who dropped a chunk in water during a science lab knows about the fizz and energy these metals release. Nitrite, on the other hand, brings a twist from nitrogen’s many forms. Mix barium ions (Ba2+) and nitrite ions (NO2-), and you get a white to yellowish powder that dissolves well in water—and can be hazardous in the wrong hands.
Real-World Relevance
What makes Ba(NO2)2 more than a footnote? For starters, it acts as a reagent in analytical chemistry. Labs use it to test for various metals and to understand nitrite interactions. That leads to bigger questions in environmental and health research. Nitrites play a role in water quality and food safety, and understanding how barium nitrite behaves helps address contamination risks.
Personal Connection to Chemistry’s Hidden Helpers
I’ve watched students freeze during experiments because they saw a chemical name that looked intimidating. When you take apart a formula like Ba(NO2)2, suddenly it isn’t so mysterious. You start to see each element’s part in the story. Plenty of folks outside a lab feel disconnected from this kind of information, but these “scary” formulas often shape safety protocols in treatment plants, hospitals, or construction sites.
Hazards Hide in Plain Sight
Safety demands that we pay attention to how compounds like barium nitrite interact with our bodies and the environment. Ingestion or inhalation can damage organs. Accidental release into waterways spells trouble for aquatic life. Industrial settings provide the kind of real-world stress test that highlights why knowledge beats ignorance; a label with Ba(NO2)2 signals danger and urgency to anyone trained to recognize it.
Paths Toward Safer Chemistry
Industry experts already set safety strategies, but not every workplace takes proper care with barium nitrite. It’s possible to cut injuries and spills by requiring more regular training and encouraging clear labeling. Using substitutes where possible, or tightly controlling processes that use barium nitrite, supports a safer environment. Where that’s not possible, strict routines for storage and disposal become essential.
Moving Forward With Knowledge
Knowing the formula Ba(NO2)2 matters, not just as trivia but as part of a toolbox that keeps people safe and operations running smoothly. Responsible handling comes down to actual understanding, beyond what’s printed on a container. That’s what bridges the gap from textbook to toolkit, from risk to responsibility.
References and Fact Checks
Barium nitrite appears on lists from safety watchdogs like the Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH). Chemists will find its information in the CRC Handbook of Chemistry and Physics. These resources, and sound training, keep workers safe and provide the practical context that anyone who works with chemicals deserves.
How should Barium Nitrite be stored?
Why Safe Storage Deserves Attention
Barium nitrite often turns up in labs and factories thanks to its use in chemical synthesis and the textile industry. Many might not realize just how hazardous this compound can get when ignored or left sitting in poor conditions. My own years in research labs taught me one thing above all else—complacency with strong chemicals always brings trouble. Barium nitrite, with its toxic and oxidizing nature, deserves respect every step of the way, especially once it arrives at the facility.
Toxic by Nature, Demanding in Practice
Let’s get real: ingesting or inhaling even small amounts of barium nitrite has a high chance of health consequences like muscle weakness, heart rhythm changes, or worse. Its oxidizing ability also means it stokes fires rather than dampening them. Reports from industrial accidents point directly at mislabeled containers, ruptured bags, and humidity creeping in where it was never supposed to. Taking shortcuts doesn't just put product quality on the line—it risks lives. That’s not an exaggeration; OSHA cites barium nitrite on its list of substances demanding appropriate controls precisely because of its history in workplace injuries.
Key Steps for Responsible Storage
Keeping barium nitrite contained—and everyone safe—starts at the receiving dock. Forget leaky barrels or mystery bins—only use tightly sealed containers made of compatible, corrosion-resistant material. Polyethylene or polypropylene often gets picked, with glass in smaller quantities also working if clearly labeled. Letting dampness sneak in leads to clumping, dangerous reactions, and faster breakdown. A dry, cool indoor room with strong ventilation stays the gold standard, away from flammable materials and acids that could trigger a violent reaction.
Once, I helped clean out a supply room after a minor flood. Water had seeped into cardboard drums of lab chemicals. Luckily, barium nitrite sat up on shelving, sealed airtight, which kept us from cleaning up a much bigger problem. Water doesn’t just create a mess: it can turn an oxidizer into a chemical time bomb.
Storage space always needs clear hazard signage. Every label on a barium nitrite drum must name the chemical, show hazard symbols, and include advice on handling and emergency steps. Careless labeling is an invitation for disaster, especially during shift changes or emergencies. Lockable storage cabinets with spill trays underneath bring extra peace of mind for anyone sharing space with these materials. I only ever felt secure knowing extra safeguards stood between me and unforgiven chemicals like this.
Training and Oversight Make the Difference
Every technician, not just safety managers, ought to know what barium nitrite does and how to handle an emergency. Drills, clear written SOPs, and PPE requirements—like gloves, aprons, and goggles—should come standard. Many incidents reported in academic and industrial settings trace back to skipped steps, missing gloves, or outdated instructions left on the shelf.
Periodic inspections—checking for leaks, broken seals, or outdated stock—keep surprises to a minimum. During my time auditing a university stockroom, we found expired barium nitrite tucked behind newer stock, bottles sticky from old spills. That’s how you lose track of what belongs where, and end up risking contamination or mixing the wrong compounds. Old stock should move out first (FIFO methods really matter here), and any container showing damage heads straight for safe disposal.
What Works Goes Beyond Rules—It’s Culture
Storing barium nitrite safely isn't a one-time checklist. It relies on a workplace culture where everyone values safety over speed. Real stories and hard experiences keep that lesson alive—one missed step can echo for years. Anyone who has seen a close call with hazardous chemicals knows: you never want to see it again. That lesson sticks, long after the bottles have moved on.
What precautions should be taken when handling Barium Nitrite?
The Risks Lurking in the Lab
Barium nitrite draws attention because of what it can do—and not always in a good way. It doesn’t spare your lungs or skin. Get it in your system, and you’ll soon understand why most chemists carry a deep respect for this chemical. The stuff releases toxic gases and can damage your organs. Touching or inhaling it can lead to nausea, muscle problems, and even serious heart issues.
Gloves, Goggles, and the Gear Non-negotiables
Barium nitrite belongs nowhere near bare hands. From my own days working in teaching labs, students always looked for shortcuts—trust me, this habit led to more accidents than carelessness. The right gloves (nitrile, not cheap latex), a good lab coat, and chemical-splash goggles block almost every point of entry. Wearing those layers might feel clumsy at first, but no experiment is worth the risk of chronic illness or a trip to the hospital.
Clean Benches, Clean Habits
The cluttered workbench looks familiar: open containers, ring stands crusted with unidentifiable residue, expired bottles stuck at the back. It doesn’t help anyone stay safe. Spills get missed, and accidental mixing happens far too often. For anyone handling dangerous chemicals like barium nitrite, keeping work areas tidy means fewer surprises and fewer exposures. Most incidents I saw in the lab stemmed from poor housekeeping. Wipe down surfaces. Label bottles every time. Dispose of wipes, gloves, and waste material as soon as you’re done. It’s impossible to overstate how much basic tidiness prevents disaster.
Good Ventilation Isn’t Optional
Open windows or desk fans don’t cut it. Barium nitrite needs a functioning fume hood. No excuses. It only takes a minor spill to send invisible fumes drifting across the room. I remember a case where someone left a petri dish uncovered outside the hood “just for a second.” The result: headaches, eye-watering, and a warning from the building manager. Fume hoods pull away not just obvious smoke, but also the subtle toxic gases that linger. If there isn’t a working hood, that’s the time to demand better facilities before opening a single jar.
Safe Storage—Don’t Store Next to Food
Barium nitrite doesn’t mix well with food, acids, or organic stuff. Cabinets meant for chemicals exist for a reason. Separate shelves, tight containers, and a clear label keep hazardous chemicals from accidental cross-contamination. It is tempting to cut corners, especially in shared or smaller spaces. I’ve witnessed the aftermath of barium compounds being stored above break-room coffee supplies. That sort of mistake brings consequences. Proper storage saves lives and jobs.
Know What to Do in an Emergency
People forget to check their eyewash stations and safety showers—then find out the hard way they aren’t working. Have the right spill kits close by, and don’t improvise with paper towels or basic cleaning sprays. Make sure the first-aid kit isn’t empty. Memorize the emergency contacts. These steps take minutes but make all the difference in a crisis. Regular drills help everyone stay sharp. Experience proves that panic sets in during a real emergency, and only well-practiced routines prevent small mistakes from turning big.
Learning and Passing on the Lesson
Every chemistry student or worker should receive real-world training about barium nitrite. Stories carry weight. The more we talk about mishaps and best practices, the fewer repeat accidents show up in reports. At the heart, responsible handling comes down to respect—respect for your own health, the well-being of everyone around you, and the power of the chemical itself. That respect never stops being relevant, no matter how long you’ve worked with dangerous compounds.
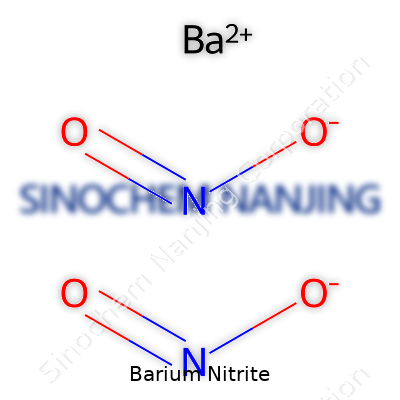
| Names | |
| Preferred IUPAC name | barium dioxidoazaniumylazanide |
| Other names |
Nitrous acid, barium salt Barium dinitrite |
| Pronunciation | /ˈbeə.ri.əm ˈnaɪ.traɪt/ |
| Identifiers | |
| CAS Number | 10022-31-8 |
| Beilstein Reference | 13607 |
| ChEBI | CHEBI:81045 |
| ChEMBL | CHEMBL1201533 |
| ChemSpider | 12222 |
| DrugBank | DB14536 |
| ECHA InfoCard | ECHA InfoCard: 03b713e7-c6e0-40a3-b1e5-42f227f32ae2 |
| EC Number | 233-322-0 |
| Gmelin Reference | 83341 |
| KEGG | C18615 |
| MeSH | D001459 |
| PubChem CID | 24964 |
| RTECS number | CQ9625000 |
| UNII | N9L58Q64QQ |
| UN number | 1466 |
| Properties | |
| Chemical formula | Ba(NO2)2 |
| Molar mass | 197.34 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 3.6 g/cm³ |
| Solubility in water | 56 g/100 mL (20 °C) |
| log P | -2.14 |
| Vapor pressure | Vapor pressure: Negligible |
| Acidity (pKa) | 11.6 |
| Basicity (pKb) | 10.09 |
| Magnetic susceptibility (χ) | +62.0e-6 cm³/mol |
| Refractive index (nD) | 1.67 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 138.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -486.6 kJ/mol |
| Pharmacology | |
| ATC code | V09CA04 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes eye, skin, and respiratory irritation; may cause methemoglobinemia; strong oxidizer, may intensify fire. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H272, H301, H332, H410 |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P391, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Lethal dose or concentration | LD50 oral rat 250 mg/kg |
| LD50 (median dose) | 185 mg/kg (rat, oral) |
| NIOSH | NA8490 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Barium nitrate Barium oxide Nitrous acid Sodium nitrite |

