Barium Chlorate: A Chemical with Complex Roots and Modern Relevance
Looking Back: How Barium Chlorate Became Part of the Chemical Toolbox
Long before complex lab instrumentation or digital controls came on the scene, chemists looked for reliable oxidizers to power everything from early fireworks to experiments in the nature of flame and color. Barium chlorate was born out of that era, its discovery tracing back to a time where practical chemistry met human curiosity. Potassium chlorate had become standard, but researchers craved sharper greens for pyrotechnics, leading them to barium-based compounds. I remember reading how nineteenth-century chemists would describe the “apple green” flame of barium salts, almost reverentially, as if they had found a piece of magic. Barium chlorate’s commercial synthesis improved over time, making it more available for industry and research alike. Its history teaches us how necessity, and sometimes showmanship, drives chemical innovation.
What Sets Barium Chlorate Apart
Barium chlorate exists as a colorless, crystalline solid with a serious knack for releasing oxygen. It stands out because of its role as an oxidizing agent: it can break down, usually with a good push from heat, and throw off oxygen in impressive quantities. This property makes it valuable in work where oxygen delivery needs to be fast and predictable. Handling barium chlorate comes with distinct sights and smells—sharp, bitter, and chemical—reminding me why certain labs insisted on double gloves and careful disposal practices. With a molecular formula of Ba(ClO3)2, barium chlorate’s two chlorate groups pack a punch, fueling reactions that leave a mark, both literally and figuratively, in any demonstration.
Not Just Another Chemical: Physical and Chemical Characteristics
Barium chlorate’s physical look might not set off alarms—its crystals dissolve well in water, and they’re usually stable under dry storage. But its real value comes out in chemical behavior. This compound heats up, decomposes, and—unlike many common solids—spills out both oxygen and barium chloride. Its capacity to vigorously fuel combustion means safety sits front-and-center in labs and workshop storerooms alike. I’ve seen seasoned professionals insist on glass containers and tightly-sealed caps to keep stray sparks or moisture at bay. Melt it, and it’s not just a hot liquid; it’s a powerhouse looking for the next oxidizable partner.
Labeling: More Than Just Ink on Paper
Labels on barium chlorate offer sharp warnings and don’t mince words. Hazard diamonds, oxidizer symbols, and skull-and-crossbones stand out for good reason. The technical details go beyond regulatory niceties—packing groups and handling categories reflect real dangers. Too often, people get lulled into a false sense of security by everyday lab routines, but the wrong move with this chemical could bring injury or contamination. Clear, legible information supports safe practice, especially for students and new technicians who might be handling such compounds for the first time.
The Craft: Preparation and Synthesis
Manufacturing barium chlorate usually involves reacting barium carbonate with sodium chlorate. It’s a process that’s both art and science—careful mixing, strict temperature controls, and enough time to let crystals form right. Larger batches, intended for industry, follow similar paths but juggled with higher volumes and necessary purity checks. Each synthesis cycle produces a highly reactive product, sometimes requiring additional purification steps like recrystallization or filtration to get rid of unwanted barium contaminants. Even minor lapses, say in drying or washing, can leave residues or trigger dangerous reactions down the line. The care at this stage carries through the chemical’s whole lifecycle.
Chemical Reactions: Friend and Foe
Barium chlorate’s thirst for redox action makes it a favorite scenario for educators with a flair for dramatic experiments. Drop a reducing sugar or a bit of sulfur into its path, and heat brings about rapid transformation: oxygen bursts free, often in a flash of light and smoke. The compound’s instability in the face of heat and combustible materials means that experienced hands stay on high alert. Too many stories circulate about accidents in poorly ventilated spaces or with hurried measurements. On the flip side, the same reactivity can help researchers modify it into specialty barium or chlorate derivatives, opening windows to new chemical exploration.
Name Games: Synonyms and Street Names
Chemistry loves its lists of synonyms, and barium chlorate is no exception. Old textbooks and newer safety data sheets toss around names like “chloric acid, barium salt" or “barium(II) chlorate” interchangeably. Each label reflects a slice of its history—some, etched from Latin roots, and others, leftovers from early scientific arguments. The confusion might seem trivial, but even a mix-up in a supplier’s database can cause headaches. Ensuring that everyone agrees on what’s actually in the bottle connects directly to safe, reliable results, especially for those outside the chemical industry who might encounter the substance under a different name.
Taking Safety Seriously
There’s no shortcut to safe handling of barium chlorate. Its toxic nature—due to both the barium ions and the chlorate—demands more than just gloves and goggles. Workspaces investing in proper ventilation and dust controls go a long way toward protecting people. Any hint of contamination in water supplies or improper disposal can turn a minor incident into a community health scare. Regulations dictate storage away from organics, acids, and combustibles. Waste handling protocols demand neutralization and confirmed disposal routes. I’ve seen companies fine-tuned to the point where even the floor sweepings are handled like hazardous waste. These standards matter, not only for plant workers but for the neighborhoods around them.
Industrial Reach and Experimental Playgrounds
Barium chlorate has lit up the night sky through fireworks and signal flares, where its role in creating green hues holds up against modern colorants. In the classroom, small samples power vivid demonstrations of combustion chemistry, showing students the speed and energy waiting in a jar of oxidizer. It’s also done time in agriculture, once pitched as a defoliant, although this use has dropped off. Environmental regulation and safety worries have shaped its role, nudging it out of widespread industrial use towards more restricted, specialized applications. Familiar uses revolve around scientific research, where the mix of reactivity and toxicity offers both a challenge and a lesson.
Pushing the Boundaries: Research and Development
Chemists and engineers search for better, safer oxidizers, yet barium chlorate still finds its way into new research every decade. Innovations in synthesis and purification keep cropping up, driven by the goal of higher yields or lower unwanted byproducts. I remember reading about efforts aimed at reducing barium’s environmental footprint, such as trapping or transforming waste products into safer forms. Academic groups continue dissecting its combustion traits, searching for clues that could improve other energetic materials. Every improvement in handling, disposal, or detection technology feeds back into both educational and industrial settings, keeping safety and efficiency front of mind.
Toxicity Research: Not Taking Any Chances
Anyone who has seen the litany of symptoms attached to barium poisoning—muscle weakness, irregular heartbeat, even death—understands why toxicology studies rack up year after year. Animal studies expose the dangers at low doses, raising alarms about water contamination or accidental ingestion. Chlorate ions add their own category of trouble, especially for anyone with compromised red blood cells. Medical case reports offer stern reminders about what can happen with improper use or unintentional exposure, which keeps pressure on industry to design and enforce strict limits. The focus on improvement in both detection and treatment remains strong, as public health agencies stay wary of these hazards.
Looking Ahead: The Next Chapter for Barium Chlorate
The future for barium chlorate looks complicated. On one side, its signature green flame and strong oxidizing power leave a legacy that isn’t so easy to replace in specialty arenas. On the other, rising safety and environmental standards have cut down its mainstream roles. Research continues to search for less toxic, more sustainable alternatives, but few match its combination of properties. Whether it survives in niche applications or is completely replaced by safer chemicals will depend on advances in synthetic methods, waste treatment, and perhaps breakthroughs in green chemistry. For educators, regulators, and chemists alike, the story of barium chlorate stands as a case study in the balance between utility and risk, coloring both our past and how we shape the future of chemical handling and discovery.
What is Barium Chlorate used for?
Not Just Another Chemical Compound
Barium chlorate doesn’t often make headlines, but it plays a role in things that catch our eyes and sometimes even affect our environment. This white, powdery salt carries a punch when it comes to its uses—especially in fireworks and pyrotechnics. From my years living around small towns where fireworks shows are more than just July entertainment, I’ve seen how barium chlorate’s green color sets the sky apart during festivities. Without it, those brilliant green bursts would fade into dullness.
Lighting Up the Night—But There’s a Cost
People have leaned on barium chlorate for its power to create bright, vibrant displays. Take campfire colorant packets, for example, or signal flares—many owe their sharp hues to this compound. Folks rarely pause mid-celebration to think about what’s behind the scenes of this magic. The truth is, each colorful explosion comes with a side note. Barium compounds, including barium chlorate, are toxic if they get into groundwater or the air in a big enough dose. It reminds me of growing up near rivers that sometimes had strange colors during local festivals. In hindsight, those odd shades probably meant more barium than I’d like.
Industrial and Lab Uses—Not Just for Shows
Industry taps barium chlorate in the manufacturing world, mainly for dyes, explosives, and matches. Scientists use it for basic experiments because it breaks down easily, offering a clean source of oxygen. I worked a summer in a lab where the focus turned to chemical reactions fueled by oxidizers like this. Seeing its effect firsthand made clear the energy packed in each granule. That’s also why accident risk runs high if storage and handling skip safe practices.
In agriculture, barium chlorate once appeared in weed killers. Farmers around my home switched away from it, feeling uneasy about potential soil contamination and toxicity. Regulations eventually tightened, reflecting a broader change toward safer herbicides.
Concerns for Health and Safety
Barium chlorate is strong stuff. Ingesting it or breathing its dust can harm nerves, kidneys, and muscles. I remember a friend’s father—who worked in a fireworks factory—telling stories about coworkers getting sick after too much airborne dust. He always wore heavy-duty masks and gloves, swearing by them. Strict rules in modern workplaces help, but the risk hasn’t vanished. Parents in communities near firework shows sometimes wonder if the bright colors are worth possible downstream health effects.
What Can We Do?
There’s no getting around the wow factor of those green bursts in the sky. Some companies now test alternative, less toxic compounds to achieve similar effects. Research grows year by year in crafting safer chemical blends for fireworks and industrial uses. We can’t ban every useful material, but we can push for responsible handling, thorough cleanup, and transparent labeling and reporting. Supporting local policies that limit unnecessary chemical use keeps both people and ecosystems in mind. Drawing lessons from the past, a little caution and care can stretch a long way.
Is Barium Chlorate toxic or hazardous?
Understanding the Risks
Barium chlorate lands on the list of chemicals that don’t belong in the kitchen or around curious hands. On paper, it seems bland—just another white, crystalline powder. In practice, this chemical brings a punch of risk with it. Many industrial labs use it because barium chlorate helps create fireworks and explosives. That alone can turn heads. But health concerns stretch beyond what happens during a misfired celebration.
Health Effects and Real-Life Examples
Contact with barium chlorate can cause real harm. It can damage skin, eyes, or even lungs if someone breathes in its dust. Ingesting even a small amount leads to poisoning. Symptoms hit hard: vomiting, diarrhea, muscle weakness, and life-threatening changes to heart rhythm. I’ve seen emergency reports from rural hospitals that struggled after a call came in about a child eating a mysterious white powder—barium poisoning isn’t rare in regions where chemical storage is less secure.
The core problem lies in barium ions blocking potassium channels in the human body. That leads to muscle paralysis, including the one muscle everyone relies on: the heart. Agencies like the CDC classify barium compounds as acute toxins. That’s not lab exaggeration—emergencies have shown the damage firsthand, especially where chemical literacy isn’t high.
Fire Hazards Around Barium Chlorate
There’s another layer of risk that often gets missed. Barium chlorate is a powerful oxidizer. If it comes into contact with organic material, fire can break out with little warning. In places where storage sheds get hot or dusty, a small leak can turn into a big problem. Fire departments have documented incidents where sections of storage facilities were lost, entirely because of an unchecked spill or poor labeling.
Many folks working around fireworks know this risk by heart. Still, recordkeeping in small shops or backyard operations doesn’t always stay top-notch. All it takes is an unlabeled drum or a missing warning sticker, and suddenly a stack of sawdust or some spilled sugar can kick off a hazardous blaze or explosion. The environmental fallout is real too—local streams pick up the runoff, putting wildlife and water supplies at risk.
Safer Handling in the Real World
After seeing what mistakes can lead to, safer storage feels urgent. Locking up barium chlorate in weather-proof containers changes outcomes. Setting up areas with clear labels, strong ventilation, and no sources of ignition goes a long way. I’ve worked with teams who added secondary containment and simple ventilation fans—those changes helped them avoid all-nighters cleaning up spills or rushing to the ER.
Some facilities switched to safer alternatives where possible, cutting down on barium’s footprint in regular operations. Training helps too. Walking through the handling steps—gloves, goggles, dust masks—shouldn’t just happen in a classroom. Practice drills build habits, and I’ve watched small changes keep people healthy and shops open.
Taking Hazards Seriously
Barium chlorate doesn’t make headlines often, but those who come across it, whether at work or through a simple mistake at home, can’t afford to take chances. Chemical safety depends on information. Labels, proper gear, and community awareness might sound simple, but they have stopped disasters in places that used to trust luck over planning. If those steps start sounding repetitive, it just shows how vital they are for keeping people out of harm’s way.
What is the chemical formula of Barium Chlorate?
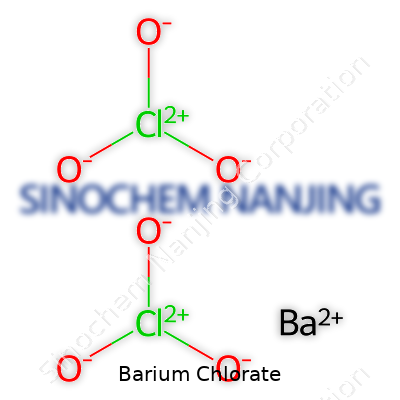
What Is the Chemical Formula of Barium Chlorate?
Barium chlorate holds the formula Ba(ClO3)2. Simple as it appears, this combination of barium, chlorine, and oxygen turns up in real-world situations that reach well beyond textbooks or lab benches. If high school chemistry was your thing, you might remember that barium always carries a 2+ charge, chlorate clocks in at -1. Two chlorate ions balance a single barium ion, so Ba(ClO3)2 forms a neutral compound.
The Role of Barium Chlorate in Everyday Life
Barium chlorate mostly grabs attention in fireworks and explosives. Talking about firework displays, that riveting green is tough to get. An experienced pyrotechnician knows that barium compounds, especially barium chlorate, bring out some of the brightest, richest greens possible. Potassium chlorate does the job for other colors, but it can't match the brilliance of barium chlorate's green flash.
Firework fans rarely consider what this means for manufacturing safety. Barium chlorate stands as a powerful oxidizer, so safety demands more than gloves and goggles. Without solid safety practices, the dangers stack up quickly. Chemical burns, inhalation risks, or unintended detonations have happened, especially where regulations get ignored. From my time in a university research lab, nobody cut corners with chlorate salts—folks remembered stories where mistakes led to ruined equipment or far worse.
Understanding the Risks: Health and Environment
Knowledge about barium chlorate's formula also brings up concerns about health. The barium ion turns toxic in the body. Workers crafting fireworks or handling this chemical in factories need well-ventilated spaces, dust control, and steady habits around food and drink. Not everyone realizes that even a small dose could disrupt how nerves send signals in the body, with muscle weakness, cramps, or dangerous heart rhythms. The Centers for Disease Control and Prevention list barium poisoning as a medical emergency—people who know about the chemical formula stay mindful of these dangers in their workspace.
Barium chlorate doesn't stay put after use. Wastewater problems pop up when disposal rules get bent or ignored. Plants and aquatic life can take a hit if the compound slips into surface water, since chlorate ions put stress on how organisms break down nutrients or carry out photosynthesis. Environmental agencies point to proper chemical handling and neutralization before disposal. During my undergraduate studies, lab supervisors stressed neutralization as non-negotiable lab cleanup. They bought test kits by the dozen for monitoring waste before pouring anything down the drain.
Finding Safer Solutions and Better Practices
Knowing formulas goes farther than passing exams. It creates a foundation for safer work and smarter decisions. Some fireworks companies now look for alternative oxidizers that hold less risk for users and bystanders. Researchers keep developing techniques for waste treatment that neutralize barium and chlorate ions on an industrial scale. Chemistry can’t exist apart from its real-world consequences. The formula Ba(ClO3)2 comes with a duty to handle, store, and dispose of this compound with precision.
Barium chlorate’s bright green might light up the night sky, but it only does so safely through respect for science, regulations, and the people who put their hands on it. Anyone working with chemicals like these shouldn’t disconnect equations from experience, not in the lab, factory, or one July evening at the local fairground.
How should Barium Chlorate be stored safely?
Understanding the Risks
Barium chlorate brings more risk than many folks realize. This compound isn’t just toxic if swallowed; it reacts violently with organic material and can cause fires or even explosions if ignored. Plenty of chemistry classrooms keep supplies like this in the back closet, but experience shows that one slip—an accidental spill, mixing with the wrong substance, or even just humidity creeping into a jar—can cause havoc. The stories aren’t rare. Just a few years back, a warehouse in Eastern Europe lost an entire shipment room to a single contaminated batch.
Staying Dry: Moisture Is an Enemy Here
Moist conditions make barium chlorate even more unstable. If water vapor or any sort of liquid gets in the container, the risk of decomposition goes up sharply, and the product can become less predictable. Experts working in chemical management recommend tightly sealed, airtight containers for this reason. Glass works well, as does heavy plastic, provided there’s no chance the seal will fail. Even one splash can turn what should be a stable salt into a dangerous, oxidizing powder.
Fire Hazards: Store Away from Fuel Sources
Barium chlorate stands out as a powerful oxidizer. In practical terms, that means it feeds fire rather than starves it. Keeping it near paper, wood, solvents, or any flammable stockpile is a big mistake. One overlooked can of paint thinner or a forgotten cardboard box creates the perfect kindling. For that reason, fire-resistant cabinets are the industry norm. Years of following safety audits made me see that anything less than steel, with a self-closing door, only invites disaster. Some facilities also put up clear signage so that firefighters know the danger if something goes wrong.
Temperature Control Isn’t Optional
Hot rooms push barium chlorate closer to its decomposing point, and if that happens, there’s a real risk of oxygen release. Sudden pressure builds up. Chemical storage rooms should stay cool and avoid direct sunlight. Temperature swings destroy more than just shelf life; they change how substances behave. From my own work in laboratories, digital temperature logs gave peace of mind. Regular checks catch broken air conditioning systems before any product suffers.
Segregation by Compatibility
Combining incompatible chemicals creates the worst accidents. At a university I partnered with, a mix-up left barium chlorate stored with acids. Even the slightest contact puts staff at risk for toxic gas releases. Barium chlorate belongs on its own shelf, away from acids, organics, combustibles, and reducing agents. Color-coded shelves and inventory logs aren’t bureaucratic overkill—they prevent mistakes, especially for new employees or visiting contractors unfamiliar with the space.
Transport and Labeling: Never Take Shortcuts
Moving chemicals brings new risks. Use containers designed for hazardous material, with hard-to-miss hazard diamonds. Too often, small operations skip these steps, thinking small amounts don’t warrant fuss. After seeing someone try to move barium chlorate in an old jam jar, I saw just how fast things can go wrong. Proper labels, insurance paperwork, and rigid protocols help catch mistakes early.
Training and Emergency Preparedness
No storage method saves lives if no one knows what to do in an emergency. Staff need real-world drills and regular training. Simple measures—like fresh spill kits and eyewash stations nearby—are investments that pay off. Sharing firsthand stories about close calls goes further than any printed manual. People remember what almost went wrong, not just the dry rules.
What is the appearance and solubility of Barium Chlorate?
Crystal Clarity: The Appearance of Barium Chlorate
Barium chlorate stands out on a lab bench. The compound shows up as colorless crystals—clear and sharp if you look closely. Run your fingers near the edges and they reflect light a bit, sort of like grains of salt but larger, sometimes in flat plates or chunky clusters. The surface feels hard, not powdery, and it crunches if pushed between slides. In the right light, the crystals almost glimmer, which feels odd considering their chemical punch.
Water and Barium Chlorate: Solubility You Can Rely On
Drop a scoop into a beaker of water and the process starts fast. Barium chlorate dissolves easily, leaving virtually no sediment, even when the water’s not warm. Room temperature works, no need for extra heat or agitation. That’s handy for labs and industries handling this compound, whether for producing oxygen in chemical reactions or testing combustion effects.
The science backs up this observation. At 20°C, you can dissolve about 27 grams in just 100 milliliters of water. Shift the temperature up, even a little, and the solubility keeps rising. No distinct smell, no fizz, just clear liquid, unless you hit over-saturation. At that point, some crystals linger at the bottom, slow to fade away, a gentle reminder that even soluble salts set their limits.
Why Barium Chlorate’s Solubility Matters
High solubility grabs attention for a reason. Handling chemicals that dissolve completely in water changes the way cleanup or accidental release works. With barium chlorate, the risk to water supplies jumps up. Barium ions, once in solution, travel easily and won’t get stuck to soil as much. I remember working on a team investigating well water near an old industrial site—the legacy of soluble barium compounds left persistent contamination that traveled downstream, tough to trap and harder to clean out.
For manufacturing, high solubility brings benefits. Solutions mix evenly for use in pyrotechnic and oxidizing formulas. It acts as a reliable oxygen source in specialty glass production and certain chemical syntheses. The flip side—if crystals spill, they’ll dissolve into moisture quickly, raising hazard potential at the worksite or in streams. Solubility increases the risk profile, especially where spills can reach wet surfaces.
Managing the Hazards and Finding Safer Alternatives
Using barium chlorate safely requires strategy, not just gear. Spill trays, absorbent pads, and keeping lab water drains isolated can block some accidental spread. Regular training and extra storage precautions, like humidity control and sealed containers, make a real impact. Risk grows in settings with poor oversight or lax discipline.
Switching to chemicals with less environmental mobility takes real effort, but makes sense where water quality matters. In some cases, less soluble oxidizers, such as potassium perchlorate, serve similar roles with fewer risks if they escape. It takes more upfront research and sometimes a cost increase, but preventing barium from entering groundwater pays off long-term—less hassle with remediation and fewer community worries.
What the Facts Give Us
Barium chlorate does not blend into its surroundings quietly. The see-through, hard-edged crystals grab attention, and their fast dissolution means one spill or misstep can change water chemistry nearby. Chemists, plant operators, and site managers should all pay attention to the details—both shimmering in a beaker and disappearing in water. The facts highlight the need for respect, right containment, and eyes on possible alternatives for the sake of workers, neighbors, and the environment.
| Names | |
| Preferred IUPAC name | Barium dioxido(dioxo)chlorate |
| Other names |
Barium chlorate(VII) Barium dichlorate |
| Pronunciation | /ˈbeəriəm ˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 13477-00-4 |
| Beilstein Reference | 2318730 |
| ChEBI | CHEBI:61449 |
| ChEMBL | CHEMBL509031 |
| ChemSpider | 21137 |
| DrugBank | DB11090 |
| ECHA InfoCard | 100.037.273 |
| EC Number | 233-189-7 |
| Gmelin Reference | Gm. 798 |
| KEGG | C18625 |
| MeSH | D001468 |
| PubChem CID | 24589 |
| RTECS number | CQ9625000 |
| UNII | F7V02P6LQ1 |
| UN number | UN1564 |
| CompTox Dashboard (EPA) | DTXSID1022912 |
| Properties | |
| Chemical formula | Ba(ClO3)2 |
| Molar mass | 304.23 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 3.18 g/cm³ |
| Solubility in water | 52.5 g/100 mL (20 °C) |
| log P | -2.13 |
| Vapor pressure | Negligible |
| Basicity (pKb) | -1.02 |
| Magnetic susceptibility (χ) | +36.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.995 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -858.87 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −801 kJ/mol |
| Pharmacology | |
| ATC code | V09CA03 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H301, H373 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P301+P312, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Lethal dose or concentration | LD50 oral rat 74 mg/kg |
| LD50 (median dose) | LC50 (oral, rat): 100 mg/kg |
| NIOSH | NA0450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Barium Chlorate: "0.5 mg/m3 (as Ba) |
| REL (Recommended) | REL: Ca (0.5 mg/m³) |
| IDLH (Immediate danger) | 25 mg/m3 |
| Related compounds | |
| Related compounds |
Barium nitrate Barium perchlorate Calcium chlorate Sodium chlorate |

