Barium Arsenite: History, Science, and Today’s Realities
Tracing the Path: Historical Development
People started paying attention to barium arsenite in the 19th century, drawn in by the arsenic compounds already popular in glassmaking and pigments. Early chemists mixed barium and arsenic, chasing new colors and formulations. The industrial revolution brought interest in unique compounds that blended earth metals with toxic elements. Pioneering scientists, with limited safety measures, explored barium arsenite for glass tinting and ceramics. The story of barium arsenite runs alongside that of its cousins like barium carbonate, bringing hopes of better material properties. By the early 20th century, awareness of arsenic-related toxicity started making headlines, forcing industries to change their approach, and research shifted from curiosity to risk management.
The Product Up Close: Overview
Barium arsenite is recognized as a chemical compound combining barium and arsenite ions, usually forming a pale yellow solid. Its structural framework appeals to inorganic chemists due to its distinct ionic arrangement—one barium atom surrounded by arsenite groups. Commercially, it gets stored in sealed glass or plastic containers, marked clearly to warn about toxicity. Unlike some other barium or arsenic compounds, barium arsenite rarely finds its way into everyday products due to regulatory restrictions and alarming toxicity levels.
Understanding Physical & Chemical Properties
The solid form appears pale, sometimes off-white when pure, but can turn light yellow if impurities get in. Solubility stays low in water, though it isn’t entirely water-resistant. It melts at a temperature higher than most home ovens can reach, and its density exceeds most routine minerals found in home collections. Barium arsenite reacts in acidic environments, releasing both barium and arsenic ions, posing a real risk for environmental contamination if not carefully controlled. On paper, the formula reads Ba(AsO2)2, with the barium ion doing the heavy lifting in binding to two arsenite units.
What the Label Says: Technical Specifications & Labeling
Strict standards dictate how barium arsenite gets shipped and handled due to its toxicity. Chemical labels carry clear symbols for acute toxicity and environmental hazards. Details on purity—usually above 98% for research-grade batches—show up on the technical datasheet, along with storage instructions such as “keep in a cool, dry place away from acids.” Shipment uses UN numbers for hazardous substances and all containers sport tamper-evident seals. Spec sheets list bulk density, melting point, and main uses mainly for lab use, not for consumer products. Any lab technician working with this substance keeps material safety data sheets close by.
Making It: Preparation Method
Labs make barium arsenite through controlled double decomposition. Barium chloride reacts with sodium arsenite in an aqueous solution, forming barium arsenite as a precipitate. That reaction quickly brings out a yellowish solid, which goes through filtration and washing to remove any chloride or sodium ions stuck to the surface. Drying happens under vacuum or over a gentle heat source, depending on quantity and risk measures. Any leftover solution and solid residue require careful disposal as hazardous waste, under strict supervision from experienced chemists.
Chemical Reactions & Modifications
Barium arsenite interacts easily with acids, breaking down and releasing soluble arsenious acid and barium ions—this poses big risks for water supplies and ecosystem health. Thermal decomposition can produce arsenic oxides and barium oxide, both classified as health hazards by agencies such as the EPA and OSHA. Researchers often study how small structural modifications—like swapping out ions or tweaking crystal conditions—might affect the toxicity and solubility. Most experiments focus on “what if” scenarios rather than practical applications due to the dangers involved.
Name Game: Synonyms & Product Names
This compound goes by several tags depending on the lab or historical context. Expect to see “barium(II) arsenite,” “arsenous acid, barium salt,” or “barium meta-arsenite” on safety documentation. Synonyms change with language and trade regulations, but all point to the combination of barium and arsenite, flagged for poison control and tracked on inventory lists in academic and industrial settings.
Staying Safe: Operational Standards
Working with barium arsenite requires full compliance with hazardous substance standards from organizations such as OSHA and the International Labour Organization. Lab coats, goggles, and gloves count as non-negotiable. Fume hoods must run before opening any container, and emergency showers need to be close by. Facilities log every gram of barium arsenite, and disposal heads directly to approved hazardous waste plants. Anyone training in a lab where barium arsenite appears gets drilled on spill response and decontamination. Data tracking keeps records on exposures, with periodic health checks for personnel handling this compound over long periods.
Where It’s Used: Application Area
Current uses rarely show up in consumer life. Most demand comes from specialty research in solid-state chemistry and some limited historical applications in pigment studies. A few industries investigate barium arsenite for reference materials or legacy glass-formulation analysis, but safer alternatives dominate the field. For decades, regulations and toxicity awareness have kept its use tightly locked under institutional review—very little industrial-scale processing survives outside academic curiosity.
Pushing Knowledge: Research & Development
Most R&D on barium arsenite happens in the context of environmental chemistry, toxicology, and material science. Teams investigate how certain compounds behave under harsh conditions, especially longevity and breakdown products for waste management. Analytical chemists test detection techniques in groundwater using barium arsenite as a reference for arsenic contamination, since the ion mix pushes the limits of modern sensor technology. Any push for applications in energy storage, catalysis, or materials engineering gets weighed carefully against real-world safety concerns and regulatory red tape.
Toxicity Research: Why It Matters
Toxicity sits front and center in every discussion about barium arsenite. Both barium and arsenic carry reputations for poisoning humans and animals. Even low exposure damages internal organs, affects nervous and circulatory systems, and disrupts cell function. Chronic exposure can trigger cancer, birth defects, and long-term ecological effects. Researchers test thresholds in mice, rodents, and insects for environmental risk assessments, all aiming to understand transmission, buildup, and acute effects. Regulatory bodies record barium arsenite near the top of hazardous substances due to how quickly small doses turn dangerous. Cleanup efforts for sites contaminated with barium or arsenic treat any discovery of barium arsenite as a red flag for immediate action.
Future Prospects: Looking Ahead
Bans and tighter regulations block most mainstream uses of barium arsenite. Many labs use it only as a benchmark for toxicity and detection method development. Cutting-edge work centers on alternatives with similar properties but much lower hazard risk. Some research delves into ways to neutralize or recycle existing waste containing barium arsenite. Advances in sensor technology, closed-system handling, and real-time environmental monitoring draw on the lessons learned from handling such risky compounds. Ultimately, barium arsenite prompts a larger discussion in science and society about balancing curiosity, benefit, and the responsibility that comes with handling toxic materials.
What is Barium Arsenite used for?
Understanding Barium Arsenite’s Place in Industry
Barium arsenite doesn’t show up on store shelves or in household products. This chemical mainly turns up in certain industrial traditions, particularly in the past, thanks to its properties as a pigment and insecticide. In everyday life, you’re unlikely to cross its path, but that doesn't mean the story ends there.
Tracing Its Use in Pigments and Pesticides
Mixing bright greens for paints or wallpapers once drew on barium arsenite. The brilliant shade it produced came with a dark side—the substance contains arsenic, an element that’s rightly earned a reputation for toxicity. During the Victorian era, homes looked pretty but the cost was steep: people inhaled arsenic dust flaking from green wallpaper, risking nervous problems, and sometimes deaths. Art historians and chemists now recognize how these pigments shaped not just interiors but also public health policies.
On farmland, barium arsenite pulled duty as a pesticide. Its strong effect on insect populations led to its use where other options fell flat. Farmers and agricultural workers wanted crops that would feed families and supply cities, but often little thought landed on long-term health impacts or environmental fallout. Soil, rivers, and living things soaked in these chemicals. Decades later, scientists still track lingering contamination in old agricultural zones.
Health and Environmental Hazards
Both barium and arsenic bring health concerns. Arsenic ranks among nature’s top toxins, muting immune responses, upsetting stomachs, and in severe cases, turning deadly. Barium, depending on its form and exposure, can disturb heart rhythms and hit muscles hard. When paired in compounds like barium arsenite, risks don’t vanish—they stack up instead.
Separating technical talk from daily reality, the risk isn’t just about direct exposure on the job. Forgotten dumpsites and historic buildings let small traces slip into groundwater or dust, threatening people who moved in long after factory whistles fell silent. The CDC and WHO regularly warn about heavy metals in drinking water, and many local health departments urge property tests, especially near former industrial lands.
Why We Need to Pay Attention
I’ve walked past old rail yards and towns with boarded-up factories, wondering what sits beneath the weeds. Public records sometimes hide scary stories—a pigment plant dumping toxic barrels, a pesticide mixer blowing dust across fields. Families now play on what used to be loading docks or run along creeks bordering past farmlands. The legacy of compounds like barium arsenite lingers, often invisible except to experts with soil probes and test kits.
Regulations now bar barium arsenite in most developed countries. New pigments and safer farming methods crowd out the old standbys. But a quick Google search shows online sellers pushing vintage pigments or chemical supplies. People experiment with old recipes for paints or powders, sometimes missing the toxic baggage that comes with nostalgia.
Pushing for Solutions
Big changes take teamwork. Cities and counties need to dig through historical records, test soil, and make cleanup plans. Companies must open their books and share what chemicals linger on their old sites. Schools and community groups benefit from lessons in environmental history—not fearmongering, but straight-up education about risks and safe handling. Artists using historic techniques can source or substitute safer modern materials. Family doctors should stay alert for unusual exposure symptoms, especially in regions with industrial legacies.
Barium arsenite paints a cautionary tale. Chemistry unlocks bright colors and powerful tools, but past oversights set traps for the future. Talking openly about old hazards and acting to clean them up matters to everyone who wants safe water, safe play, and real peace of mind.
Is Barium Arsenite toxic or hazardous?
Understanding What We’re Dealing With
Barium arsenite is no friendly neighbor on the chemical block. This compound packs a punch with two heavy hitters—barium and arsenic. Those who spent any time in a chemistry lab remember that both these elements hold plenty of bad news for health. As I learned scrubbing glassware in college, even a smudge of the wrong chemical can stir up trouble. Barium arsenite falls into that camp.
Why Toxicity Matters Here
Barium by itself causes muscle spasms, high blood pressure, and irregular heart rhythms if swallowed or absorbed in enough quantity. You won’t see barium salts on menus for a reason. Arsenic is a legend in the poison books—linked to skin, lung, and bladder cancers, and infamous for chronic low-dose effects. Anyone living in an area with arsenic in well water knows just how tricky it is to dodge contamination.
Mixing these two in a salt—like barium arsenite—means compounding the risks, not canceling them out. Even small exposures stack up over a lifetime. The Environmental Protection Agency spells out clear limits on arsenic in drinking water, and I’ve watched friends and neighbors dig new wells or retrofit their filtration to duck under the danger zone. Barium concentrations have their limits, too, thanks to its impacts on the cardiovascular system.
Hazards Beyond Toxicity
It’s not just about swallowing or breathing in these chemicals directly. Powdered barium arsenite disperses into the air without much effort. One uninterested gust of wind can send dust where nobody wants it—on skin, clothing, or even into a nearby cup of coffee. Cleaning up spills in a workplace or research lab means suiting up, taping off doors, and bagging all wipes and tools as hazardous waste. Waste disposal rules treat this kind of compound with plenty of caution for a reason—nobody wants a leaky landfill or poorly sealed trash bag turning into a neighborhood worry.
Preventing Problems Before They Start
If you work around barium arsenite—lab staff, cleanup crews, scientists—training is just the start. Spills lose their drama if everyone wears gloves, goggles, and avoids stirring up dust. Good ventilation and sealed containers belong on anyone’s checklist. I remember the relief of finishing a shift without a breach or incident, just because the team stuck to the process every time.
Community exposure rarely crops up outside of mishandling or factory accidents—but that’s not much comfort if it happens. Regulators don’t just pull safety rules out of a hat; these come from hard-won lessons over time. There’s plenty of history showing towns paying the price for shortcuts, from contaminated water supplies to lingering cleanup zones. Barium arsenite’s story isn’t unique in this respect, but the risks stay real.
Better Protection—For All of Us
Manufacturers, schools, and waste handlers all play their part in safety. Locking up supplies behind keycard doors, running regular training, and investing in proper disposal don’t just tick boxes. They prevent someone’s poisoning or a clinic visit. For the average person, odds of running into barium arsenite at home land close to zero. Still, supporting chemical safety rules—backed by solid science—keeps that risk where it belongs: far away from kitchens, playgrounds, and backyards.
What are the safety precautions for handling Barium Arsenite?
Getting to Know the Risks
Barium arsenite stands out on a lab shelf for a reason. Barium and arsenic are both toxic, so mixing them isn’t doing safety any favors. In my early days working with chemicals, the experienced techs always warned about respecting this compound. They told stories of headaches, nausea, and lab evacuations that started with the careless opening of a container. Barium arsenite attacks the nervous system and can damage organs over time. It doesn’t take much to cause irritation, especially if you inhale or get it on your skin.
Personal Protection Isn’t Optional
Personal protective gear keeps the worst risks at bay. In my chemistry training, gloves and safety goggles were the first layers. There’s no shortcut; double gloves make sense, especially if the risk of spill or splash exists. I learned the value of splash-proof goggles after seeing a colleague get a single fleck of powder in his eye. He spent the afternoon rinsing—with lucky results. Add on a lab coat and, if you’re moving any quantity of powder, a face mask or proper respirator. Even the briefest exposure can backfire, so don’t “just grab something” with bare hands.
Work Under a Fume Hood
Most of the trouble starts when dust or fine particles become airborne. Labs that handle barium arsenite never work in open air—fume hoods make a real difference. Turning on that noisy hood made sense after breathing in a few too many chemical odors in school. These hoods suck up airborne particles before anyone asks for trouble. Every spill I’ve ever seen was a reminder that accidents come fast. Never work with this material without a real ventilation system—home experiments have no place for this.
Separate and Clearly Label Everything
Clear labeling saves lives. I walk through the stockroom and see stickers and bold warnings. Containers holding barium arsenite need clear hazard symbols and a warning about poison—no room for faded tape. Store it on a shelf lower than eye level and never above acids or anything reactive. Keep incompatible materials and strong acids far apart, since unpredictable reactions only end one way.
Keep a Cleanup Plan Ready
The best labs I worked in ran regular drills for spills and exposure. It might feel silly pouring fake powder and rushing to find the spill kits, but those minutes count in a real emergency. Every handling area I’ve seen used lined, spill-proof trays. If powder gets out, a proper HEPA vacuum and wet cleaning methods collect the mess—never sweep or blow it around. Bag up everything with the right hazardous waste label, and follow disposal rules down to the letter.
Health and Training Matter
Routine health checks mean more than just ticking boxes. Places that handle barium arsenite watch for headaches, rashes, and breathing troubles among their staff. I remember health officers pulling workers out of the shift at the faintest sign of trouble, all in the name of safety. Training isn’t a one-time PowerPoint—every person working anywhere near this material practices safe steps and knows exactly where to turn for help.
Better Substitution and Changing Practices
In my experience, the safest labs find alternatives whenever possible. Engineers, researchers, and managers talk together to find less toxic substitutions or alter reactions. If a safer chemical can do the job, there’s no reason to keep barium arsenite around except where it’s absolutely necessary. The push for safer labs comes from people at the bench as much as from policy.
Why It Still Matters
People make mistakes, but strong habits prevent the worst outcomes. No lab is too experienced to skip safety. Following real-world stories and learning from colleagues set a foundation that keeps everyone going home healthy. Barium arsenite remains a stark reminder—respect for the material is non-negotiable.
How should Barium Arsenite be stored?
Handling Something This Dangerous Demands Respect
Barium arsenite brings trouble from both the barium and arsenic corners. Each brings toxicity to the table, and together they form a real headache for anyone in a lab or warehouse. Exposure to this compound, through skin, breathing, or accidental swallowing, causes lasting damage. Stories of people suffering tremors, vomiting, and even long-term nerve problems never leave my mind from safety trainings I've attended. Anyone working near this chemical should remember that accidents can happen in the blink of an eye, and sloppy storage shortens that fuse.
Choosing the Right Storage Container
Glass jars with airtight lids often set the gold standard for storage because barium arsenite doesn’t chew through high-quality glass. Strong polyethylene bottles also do the job if glass is off the table. Cheap plastic, on the other hand, can react with barium salts and end up getting brittle or warped, offering one more path to leaks. No matter the container, it must seal tight to keep dust inside and humidity outside. Water shouldn’t get even a sniff, or suddenly you have a much more dangerous substance leaking gases or dissolving compounds that might find their way onto a worker’s skin.
Don’t Rely on Just One Layer of Safety
Secondary containment kicks in as insurance. I learned early, from a grizzled supervisor, never to stack poisons directly on a shelf with no spill tray or plastic tub underneath. Containers sometimes fail. A slip, drop, earthquake, or chemical reaction inside that jar cracks open, and without a catch system, powder goes everywhere. With secondary containment, at least the mess stays in one box, not an entire storeroom. That extra barrier also keeps dust from spreading through air vents, settling on clothing, or ending up in a lunch area by mistake.
Labeling is Not Optional
Bold hazard symbols do more than tick a compliance box; they instantly warn everyone nearby what lives inside the jar. In case of a spill, emergency responders know what they are facing, and anyone grabbing the bottle—workers new or seasoned—thinks twice before moving or handling it without full protective gear. Labels must name the compound and spell out toxic, carcinogenic, and environmental hazard warnings. In one of my jobs, we ran unannounced audits; the most frequent slip-up was faded or handwritten, nearly illegible labels on toxic substances. People get hurt that way.
Safe Location: Far from Heat, Food, and People
No chemical this dangerous gets stored next to water pipes, food, or personal protective equipment. Even trace contamination does real harm. Lock cabinets limit who can reach the compound. It should sit in a cool, dry place, away from sunlight and far from acids, bases, or reactive metals. Unauthorized access leads to theft, sabotage, or careless sampling; situations everyone would like to avoid. My own practice always included keeping a signed logbook for access, so no one could “borrow” a little for an experiment and forget to return it. Accountability saves lives.
Solutions: Training, Checklists, and Regular Inventory
Throwing funds at fancy cabinets or new containers won’t help if workers shrug off real risks. I’ve seen labs forget about outdated training or let chemical stocks grow dusty until nobody remembers what’s inside. Regular checks, annual retraining, and a hard-nosed approach to storage rules all keep people safe. A missed safety meeting or skipped monthly inventory sometimes leads to expired chemicals or misplaced containers, increasing risk. Checklists reinforce best practices and catch carelessness before disaster strikes. In the end, a culture of respect around hazardous material management saves time, money, and health.
What is the chemical formula of Barium Arsenite?
Understanding Barium Arsenite
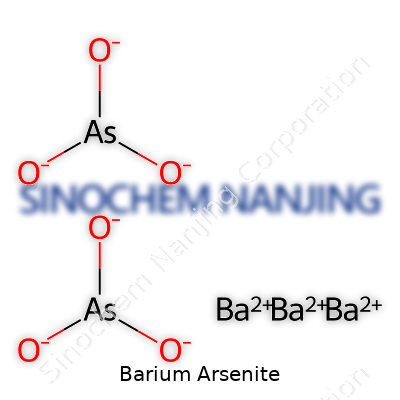
Barium arsenite brings together two elements: barium, a naturally occurring metal, and arsenite, the anion derived from arsenic acid. The chemical formula for barium arsenite is Ba(AsO2)2. That means each molecule contains one atom of barium and two arsenite groups.
The Relevance of Chemical Formulas in Real Life
Digging into formulas like Ba(AsO2)2 reveals more than just how atoms line up. The way chemical components bind together shapes a compound’s behavior, including solubility, toxicity, and how it interacts with the environment. In my experience as a science enthusiast, understanding the structure of a compound changes the way we handle it. With barium arsenite, the risks aren’t just theoretical. Both barium and arsenic come with health warnings. You can look at their place on the Periodic Table and see danger flags right away—barium can affect nerves and heart rhythms, arsenic’s reputation for toxicity is notorious, and their combination calls for some serious handling guidelines.
Safety and Environmental Rough Edges
The reality of barium arsenite’s risks goes beyond the lab. Cases of soil and groundwater contamination have led to health concerns. Arsenic compounds enter the water table in some communities, and the effects can linger for decades. Regulatory efforts recognize this, especially in drinking water limits. The U.S. Environmental Protection Agency watches arsenic closely, setting a maximum contaminant level of 10 parts per billion in water. That guideline comes from decades of research linking arsenic with cancers and other chronic health issues. Barium gets its own scrutiny, too, because of its effect on the human body. When found together, as in barium arsenite, their impacts can overlap and amplify.
Using Chemistry to Reduce Risks
Addressing contamination takes more than awareness. Community groups, researchers, and regulators focus on testing and remediation. There’s energy behind efforts to map old industrial sites, clean up spills, and test private wells. I’ve seen how community science brings together neighbors, students, and scientists to trace these compounds. Simple test kits make a difference, especially in rural parts of the country where access to high-end labs isn’t possible.
Sustainable chemistry offers another path. Green chemistry principles push for less toxic alternatives. Industries get challenged to substitute safer raw materials and invent new processes. Some universities have set up labs where students test chemical replacements and look at the whole life cycle. In a world that’s getting smaller, one spill in a remote place can echo through the water supply and food chain. Every improvement in chemistry education has a ripple effect.
Why Details Matter
Getting the formula right gives us a foundation. With Ba(AsO2)2, the specifics shape everything from industrial application to basic safety procedures. Reliable details mean fewer mistakes. I’ve learned that chemistry is only as strong as its careful measurements. From school experiments to full-scale site cleanups, knowing exactly what you’re working with shapes every outcome. That’s why a clear chemical formula matters: it sets the stage for science, safety, and smart choices.
| Names | |
| Preferred IUPAC name | Barium trioxidodiarsenate |
| Other names |
Barium arsenate |
| Pronunciation | /ˈbeə.ri.əm ˈɑː.sɪ.naɪt/ |
| Identifiers | |
| CAS Number | 1303-14-0 |
| Beilstein Reference | 363827 |
| ChEBI | CHEBI:86457 |
| ChEMBL | CHEMBL510867 |
| ChemSpider | 21569368 |
| DrugBank | DB11572 |
| ECHA InfoCard | 100.013.955 |
| EC Number | 235-028-6 |
| Gmelin Reference | 73138 |
| KEGG | C18763 |
| MeSH | D001477 |
| PubChem CID | 166859 |
| RTECS number | CY8575000 |
| UNII | 4V6YKW85H4 |
| UN number | UN1566 |
| CompTox Dashboard (EPA) | `DTXSID7035272` |
| Properties | |
| Chemical formula | BaAsO₂ |
| Molar mass | 315.23 g/mol |
| Appearance | White powder |
| Odor | odorless |
| Density | 4.68 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.19 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.3 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.89 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 114.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1161.7 kJ/mol |
| Pharmacology | |
| ATC code | V09BA02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes damage to organs; may cause cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P310, P302+P352, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 48 mg/kg |
| LD50 (median dose) | TDLo-hamster-oral: 115 mg/kg |
| NIOSH | WA8900000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Barium Arsenite: "0.5 mg/m3 (as Ba) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 1 mg/m³ |
| Related compounds | |
| Related compounds |
Barium arsenate Calcium arsenite Strontium arsenite |

