Barium Arsenate: An Editorial Look at an Unassuming Compound
Tracing Roots: A Short Walk Through the History of Barium Arsenate
Chemical discovery often unfolds in response to need, and barium arsenate entered the world of science in a time when poisons both fascinated and alarmed people. In the late 1800s, researchers focused on compounds that carried promise and peril at the same time. Barium arsenate surfaced on the back of this curiosity, shaped by advances in mineralogy and inorganic chemistry. Early on, it drew attention for its potential as a pigment and insecticide, but this interest soon mixed with caution. By the late 19th and early 20th centuries, scientific journals reported on its uses, but also on its attendant dangers. I often think about how much of chemistry’s story springs from that balance between pursuit of utility and recognition of risk.
Getting Acquainted: Product Overview
In the simplest language, barium arsenate is a white to off-white powder, usually found in crystalline or amorphous form. It’s not a household name among compounds, yet it’s shaped practices in areas like ceramics and pest control. The thing that sets it apart is the direct melding of two elements—barium and arsenic—each with a notorious reputation in its own right. The barium lends heft and density; the arsenic delivers the biological punch people have leaned on, at a devastating cost, for centuries. My early lab days taught me that this type of material is never just about formulas but about context: its legacy, consequences, and what we manage to learn from handling it.
What We See and What We Measure: Physical & Chemical Properties
Barium arsenate stands out in the lab for its appearance and its attitude toward the elements around it. Visually bland, it’s typically an odorless, white powder with a hefty density (in the ballpark of 5.2–5.6 g/cm³). It resists dissolving in water, much as other arsenates do, but will part ways with its ions in strong acid. Shift it to high temperatures and decomposition sets in, spinning out toxic fumes of arsenic oxides and barium compounds. Speaking as someone who’s cleaned up a fume hood after an arsenate run, I can say there isn’t a whiff of romanticism about those fumes. You brace for the worst before even opening the sash. Chemically, it keeps to itself unless provoked by acids or certain reagents, a trait that’s both a blessing and a curse, especially if spills or environmental escapes come into play.
Looking at the Numbers: Technical Specs & Labeling
Classification falls under the GHS (Globally Harmonized System), marked clearly for acute toxicity and environmental danger. In practice, any shipment or lab use wraps in hazard pictograms and stern signal words. Its molecular formula, Ba3(AsO4)2, bears repeating in safety circles. One part barium, two parts arsenate—each portion brings hazards as well as use potential. Even students in introductory chemistry courses notice that the labeling isn’t just bureaucratic—it’s a loud warning sign to approach with know-how, not bravado.
Making It Happen: Preparation Method
Crafting barium arsenate relies on pretty straightforward chemistry: a precipitation reaction. In typical practice, one mixes a water-soluble barium salt—like barium chloride—with a solution of a soluble arsenate compound, often sodium arsenate. As the two meet, a white solid steps out of solution: barium arsenate, pretty as a picture but no safer for it. The mixture gets filtered, washed, and dried. From memory, this is not a “set it and forget it” synthesis. Meticulous handling, control of pH, and temperature management are all crucial because one misstep means toxic fumes, corrosive residues, and an end product no longer fit for use. Laboratories spend real time and effort ensuring every synthesis batch stays both pure and contained.
Whipping Up Change: Chemical Reactions & Modifications
Barium arsenate wears several hats in reactions. Acidic conditions break it down, liberating arsenic-bearing ions that take part in new compounds—sometimes as intermediates, sometimes as waste. It doesn’t dissolve in neutral water, but acids like hydrochloric or nitric cut right through, slicing up the structure. In my own research days, I saw barium arsenate pressed into service as a precursor for more exotic arsenic compounds, especially for analytical standards in environmental work. Chemists tweak conditions to pull out different arsenates or even to recover the barium fraction. Every experiment gets planned with heavy attention to waste, since residues cross over into hazardous waste categories—no shortcuts.
Names It Goes By: Synonyms & Product Names
In scientific circles, one runs into synonyms like tribarium diarsenate or barium orthoarsenate. It can also appear as barium(II) arsenate, just to keep things clear about oxidation state. These alternate names pop up in regulatory documents, research protocols, and supply catalogs. Whether digging through a dusty chemical registry from decades past or scrolling a modern database, the aliases might obscure the fact that the substance in question is one and the same—hazardous, useful, and demanding careful stewardship.
Working Safely: Operational Standards
No other topic deserves such attention as workplace safety for substances like barium arsenate. Anyone who’s ever taped up a glovebox or double-checked the integrity of a respirator cartridge gets it: mistakes leave a mark, often permanently. Regulations set limits for airborne exposure, mandate secondary containment, and require training before use. Spill protocols go well beyond a spritz of bleach and a paper towel. Disposal, too, follows specific hazardous waste rules, since both barium and arsenic present ongoing environmental threats. Proper PPE (personal protective equipment), fume hoods, and rigorous documentation make the difference between safe use and disaster. Anyone handling material this toxic absorbs a certain humility—there’s no winging it, only disciplined care.
Where It Ends Up: Application Area
Barium arsenate may have started as a curiosity, but its niche uses shaped several industries. Historically, it played a part in insecticide formulations, taking advantage of arsenic’s biocidal force. Ceramics and glassmaking also drew on its properties, although this has declined as safer alternatives appeared. In pigment work, the compound showed a unique white with particular staying power. Each sector that touched barium arsenate, though, learned the hard way that even small releases could cause harm. Now, as environmental limits tighten, barium arsenate bows out in many places, leaving behind environmental considerations and health-related questions that remain relevant.
Digging Deeper: Research & Development
The research world treats barium arsenate quite differently today than it did in the early days. Work now focuses not just on synthesis, but on containment, detection in the environment, and remediation. Analytical chemists put in long hours developing better ways to find traces in soils, waters, or industrial residues. Materials scientists chase after encapsulation options that lock the toxic potential inside stable matrices, cutting the risks of leaching or dust formation. Regulatory science keeps a sharp eye on the thresholds for acceptable exposure. The thrust in modern R&D isn’t making more of the compound, but controlling it, understanding its pathways in nature, and overcoming its long-term impacts—especially in contaminated land.
Risk and Reality: Toxicity Research
Walk into any toxicology lecture, and you’ll see why barium arsenate doesn’t inspire much nostalgia. Both barium and arsenic have notorious records—the one disrupting potassium channels in muscle, the other interfering with cellular energy metabolism and DNA repair. Studies confirm significant risk on inhalation, ingestion, or skin contact. Chronic exposure links directly to neurologic, cardiovascular, and carcinogenic outcomes. Animal research, though uneven, points to multi-organ damage even at doses lower than with some organoarsenic compounds. Regulatory bodies classify barium arsenate as a Category 1 acute toxin and as a carcinogen—this drives its phase-out from common use. As someone who’s tracked biochemical markers from heavy metal exposure, I view every warning label as hard-won knowledge rather than bureaucratic red tape.
Looking Down the Road: Future Prospects
With its hazards laid bare, barium arsenate faces a future shaped by restrictions, innovation in remediation, and deeper investigation into legacy contamination. Its use in manufacturing or agriculture now stands at the margins and continues to shrink. Environmental chemists spend their careers tracing plumes of leached arsenic and barium, looking for ways to neutralize or contain old waste sites. New governance from agencies tightens standards, and the market answers with substitutions that promise the same function but less long-term harm. In the lab, barium arsenate serves as a reminder about the price of utility—just because a compound solves one problem doesn’t mean it won’t spawn new ones down the line. The search for safe alternatives, improved cleanup methods, and real-world exposure data keeps barium arsenate a subject of ongoing, cautious study rather than enthusiasm.
What is Barium Arsenate used for?
Where Barium Arsenate Shows Up
Barium arsenate isn’t something you find on a grocery shelf. This compound, built from barium and arsenic, found a place in the world through specialized use in industry and science. The white powder holds a story that says a lot about the push and pull between innovation and responsibility.
One-Time Role in Ceramics and Glass
Years back, barium arsenate added value in ceramics and glass making. Manufacturers favored it for certain glazes because it helped produce a smooth, bright finish on tableware and art pieces. It wasn’t about looks alone. Glazes needed chemical stability through firing, and the right additives mattered. But evidence mounted about the risks tied to arsenic compounds. Paint chips or dust raised safety questions for both workers and people using these products at home. Countries tightened their rules as researchers sounded the alarm on chronic exposure—which can hurt everything from skin to major organs.
Use in Pigments
Barium arsenate also played a part in producing pigments for specialized paints and inks. These pigments created long-lasting, vibrant colors, especially for industrial coatings. Color alone didn’t justify the risks linked to arsenic’s toxicity. Alternatives grew more common, and most pigment production in the West moved away from recipes that require heavy metals or arsenic-based compounds. Heavy metal pigments might outlast others against weather, but safer modern chemicals do the job without endangering workers or the environment.
Insecticides and Rodenticides: A Dark History
Looking back, public health records still show trails of barium arsenate in commercial pesticides. For decades, farms used arsenic compounds to fight insects and rodents. At first, these poisons worked well. Damage to crops dropped, yields rose, but hidden costs were steep. Poisoned water, tainted food, and harm to those working the fields changed the story. More scientists connected the dots linking cancers and other illnesses with long-term, low-dose exposure. Today, most countries ban arsenic-based pesticides, recognizing that safety comes first and sticking with less hazardous options wherever possible.
Lab Work and Specialized Research
Modern use sticks closely to research settings. Barium arsenate helps in analytical chemistry and mineralogy—fields where rare compounds still matter under strict control. Labs might turn to it for experiments studying material behaviors or for validating chemical detection techniques. Here, personal protective gear and closed systems keep risks in check. Dangerous as it is, barium arsenate still plays a part when nothing else will do, but only in places with processes shaped by decades of scientific learning.
Health, Safety, and Better Answers
No one should overlook the danger that barium arsenate brings. Both barium and arsenic rank high on the toxicity scale. Inhaling dust, ingesting powder, or getting residue on skin can all lead to acute poisoning or long-term harm. That’s why regulators demand strict labeling, locked storage, and thorough training for anyone dealing with this compound. Everyone from chemical suppliers to university labs stays on their toes, checking guidelines, updating safety data sheets, and pushing for alternatives. In my own work, nothing clears a lab faster than the word “arsenic”—the risks echo from older chemist stories, and modern teams won’t gamble their safety for tradition.
What’s Next?
Science has moved far from barium arsenate’s heyday. Industries phase out hazardous substances for newer, safer materials. Reaching for less toxic substitutes doesn’t just tick a compliance box; it keeps people and waterways clean. There’s no need to roll the dice with health where better answers exist. The future favors solutions that work for everyone, from workers to local communities. The story of barium arsenate isn’t just about what it does, but why we’re ready to leave it behind for good.
Is Barium Arsenate toxic or hazardous?
A Look at Barium Arsenate’s Risks
Barium arsenate shows up on lists of seriously hazardous chemicals for good reason. Touch it, inhale its dust, or have it mixed with water sources—none of these are safe. It combines two elements with notorious reputations in toxicology. My time working with lab chemicals and reviewing OSHA documentation made it clear: both barium and arsenic push nearly any substance they bond with straight into the high-risk category.
Human Health Concerns
Barium causes muscle weakness, heart irregularities, and digestion problems. Arsenic sits among the worst offenders for cancer risk, especially through long-term exposure. Put them together, and you face a material dangerous through skin contact, inhalation, or accidental consumption. The CDC highlights acute symptoms of arsenic poisoning: nausea, vomiting, abdominal pain, and in severe cases, cardiac arrest or nerve damage. Add in barium’s tendency to disrupt potassium channels in the body, leading to abnormal heart rhythms, and you quickly grasp the joint hazard posed by this compound.
Environmental Impact
Barium arsenate doesn’t stay where it’s put. Any careless disposal—say, dumping in a landfill, flushing small quantities down the drain, or mishandling lab glassware—invites trouble. Runoff can introduce arsenate into soil and water, exposing plants, animals, and people to slow and unseen poisoning. Arsenic likes to accumulate in food webs, so a contaminated stream or patch of farmland doesn’t just affect today’s crops but also keeps harming local ecology for years. Barium compounds hang around in sediments and don’t break down easily.
The Workplace Reality
Anyone handling barium arsenate needs high-level safety protocols. No shortcuts. I remember working in labs where even opening a container required gloves, goggles, and a ventilated hood. Safety Data Sheets demand immediate action for any exposure: emergency washing, medical attention, and incident reporting. There’s no room for casual handling. Workers in industries like ceramics or glassmaking, where these compounds sometimes appear, often take additional training to prevent dust clouds or spills. The risk becomes shared—not just to the worker, but to their families if dust is carried home on clothing.
Prevention and Solutions
Prevention always costs less than cleaning up after a spill, and certainly less than treating an illness. Facilities can switch to safer alternatives in most applications where barium arsenate once featured. For storage and disposal, secure containers and registered hazardous waste services remain the only responsible method. Community education about the risks of arsenic-bearing substances has curbed accidental poisonings in schools and small businesses in cities I’ve lived in, but the danger isn’t gone. Local and national regulations lay out tight controls, though enforcement relies on clear protocols and regular inspections. Regular workplace monitoring, proper signage, and lockable storage cabinets reduce risk for everyone on site. Proper training for all staff—not just chemists—brings down the odds of a preventable mistake.
Why It Matters
Barium arsenate sits in a league of its own among chemical hazards. Real people pay the price for lapses—be it a contaminated well in a rural area, or a technician suffering after a splash. Chemical safety isn’t drama; it’s a day-in, day-out commitment. The facts and the science say: vigilance and respect for toxic compounds save lives.
What are the safety precautions for handling Barium Arsenate?
Why Barium Arsenate Sets Off Alarm Bells
Barium arsenate does not show up in most people’s daily lives, but in chemistry labs and a handful of industrial settings, it’s a real concern. From what I’ve seen, any compound with arsenic demands full attention. Cases of accidental exposure stick with you, even years later. It is no secret: barium arsenate can be deadly. The combination of barium and arsenic means you’re dealing with a toxic punch that harms organs, nerves, and can cause long-term health problems. A slip-up does not just send you home early—sometimes, it sends you to the hospital or worse.
What Has Worked Best for Safety
Experience has taught me never to trust that any surface is uncontaminated. Gloves come on before you even open the bottle. My preference is for nitrile gloves because latex does not stand up to all chemicals. If a glove gets torn or something spills, no hesitation—change them. Lab coats and eye protection aren’t suggestions; they protect against all those micro-splashes most people never notice. I’ve seen colleagues turn away for a second and end up with powder on their sleeves or worse, in their eyes.
Working in a fume hood brings real peace of mind. Those exhaust systems trap powders and vapors that could otherwise end up in your lungs. At my university’s lab, the only exposures I ever heard about happened when someone tried to work on an open bench. It sounds convenient until someone drops a beaker and the dust clouds up.
Protecting Your Workspace
One habit I picked up from a safety officer was to treat every tool and container used for barium arsenate like it carries a threat. I keep separate spatulas, brushes, and glassware—then mark everything clearly. Cleaning is not just a wash with water; it's a careful rinse, collecting all wastes in special containers. Water alone does not neutralize barium arsenate, so disposal has to go through hazardous waste protocols. Dumping it in the sink or regular trash creates a risk for everyone who comes into contact later.
After working with barium arsenate, washing hands twice feels necessary. I never eat or drink in the same room, and I encourage others to call out anyone they catch breaking that rule. It just is not worth the risk.
Storing and Transporting the Compound
I keep barium arsenate locked away in a dedicated cabinet, clearly labeled and far from acids or other incompatibles. The label carries more weight than most people realize. If someone new comes in, there is no mistaking what’s inside. Transporting barium arsenate, say, between sites, requires sealed containers and all paperwork in order. If it spills in transit, the cleanup is far more complex than mopping up a water spill. Quick action with the right spill kit helps, but any major spill means activating emergency response. I learned this the hard way during a transfer that got messy—a sealed container, PPE, and quick reporting protected everyone involved.
Training and Constant Vigilance
In every lab session, I remind students and colleagues to review safety data sheets before touching anything new. No one is too experienced for a refresher. I’m not above running drills, either. Quick reactions make a difference if something goes wrong. Anyone who works with barium arsenate should know emergency numbers—and emergency eyewash stations and showers should always be within arm’s reach, not buried behind clutter.
If something does happen—symptoms like difficulty breathing, nausea, or weakness—do not wait. Get to medical help right away and bring the safety sheet. It’s saved lives before.
Better Regulations, Safer Workplaces
Regulators and employers can do more by enforcing strict storage rules, regular training, and quick access to spill kits and cleanup materials. Routine air and surface tests spot contamination early. Insisting on strong safety cultures creates workplaces where accidents stay rare. Education plays a bigger role than fancy equipment; confident, trained workers prevent most accidents.
How should Barium Arsenate be stored and disposed of?
Understanding the Risks
Barium arsenate isn’t a substance you want lingering around. With its reputation as both a toxic and carcinogenic compound, it has landed itself on serious watchlists for good reason. Once in the body, barium arsenate starts wreaking havoc with the nervous system, kidneys, and other organs. On top of that, both arsenic and barium compounds have clear links to cancer. Speaking from my background in environmental reporting, I’ve seen incidents where a moment’s carelessness led to dangerous exposures. No one wins in that scenario—worker health suffers, communities worry, and companies bear the brunt of regulatory action.
Safe Storage: Don’t Leave Room for Mistakes
Barium arsenate belongs in a specific type of container. Polyethylene or glass containers keep it in check, no corrosion or leaching. One thing I always tell people who handle chemicals: keep your labels clear, simple, and permanent. No hastily scribbled warnings or faded tags. The storage spot gets just as much attention—dedicated shelves, locked cabinets, or chemical vaults away from anything food-related or flammable. Keep water and acids away, since mixing those with barium arsenate can start a nasty, hazardous reaction.
Ventilation in the storage room proves crucial. I’ve visited labs that cut corners on airflow—bad move. Dust or fumes collect, raising the odds of accidental breathing exposure. Best practice calls for regular inspections, with a checklist that doesn’t skip any step. Gloves and goggles should hang nearby, and spill kits stay stocked—nobody scrambles during an emergency, because the tools are right there.
Handling and Personal Safety
People sometimes underestimate the value of personal protective equipment. I’ve heard of workers dismissing gloves or masks for the sake of speed. Those shortcuts create health risks no one wants to live with years down the road. Thick nitrile gloves, fitted goggles, and well-sealed respirators guard against dust or accidental splashes. Work over spill trays, not unprotected tables, and make sure there are clear processes for cleaning up even the smallest mess.
Training goes a long way here. Teams that practice regular safety drills respond faster and smarter when accidents happen. Written protocols nearby help in moments of panic, keeping folks away from improvising with potential life-or-death decisions.
Disposal: Doing Right by Health and the Environment
Nobody tosses barium arsenate in the regular trash. Hazardous waste facilities exist for a reason. My own experience with waste programs shows a big difference between facilities that respect regulations and those that cut costs. Choose a transporter with strong safety credentials—ask about their processes, licenses, and past violations.
Barium arsenate disposal follows strict hazardous waste laws. Each shipment leaves a paper trail, from cradle to grave. Environmental agencies run spot checks, so accuracy in records matters. Laboratories or industrial sites should connect with certified hazardous waste contractors, never relying on random hauling outfits.
Pre-treatment helps in some cases, breaking the compound down or binding the arsenic and barium, making leaks less likely if a barrel cracks. Still, professional waste handlers take the lead. Home chemists or amateur collectors need to contact local hazardous waste drop-off days—resisting the urge to “dilute and dump,” a mistake that could poison groundwater for years.
Better Solutions and Moving Forward
We push for safer substitutes and tighter controls, but barium arsenate remains in some supply chains. Spreading awareness means fewer accidents down the line. Community outreach programs make a difference, especially where hazardous sites sit close to schools or homes. Regulators and industry both share responsibility—cut corners and we all end up paying the price.
Safer handling, better disposal, smarter storage—these steps add up to a safer world. It’s not about ticking boxes; it’s about taking real steps to protect people and ecosystems we all depend on.
What is the chemical formula and structure of Barium Arsenate?
Understanding the Formula
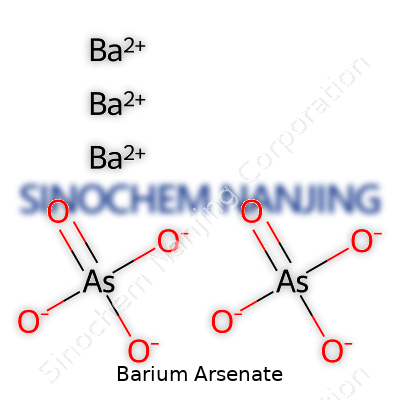
Barium arsenate comes down to a straightforward chemical formula: Ba3(AsO4)2. The formula tells you it holds three barium atoms bonded with two arsenate groups. Now, each arsenate group (AsO43−) looks a lot like a phosphate group—a central arsenic atom surrounded by four oxygens. The compound forms through ionic interactions between the positively charged barium ions and the negatively charged arsenate ions.
Getting a Picture of Its Structure
Chemists often talk about Ba3(AsO4)2 as a white, crystalline solid. The mineral world gives us an example called hamiltonite, which shows how these atoms line up in nature. Barium ions space themselves out among arsenate tetrahedra, forming a three-dimensional network. The structure dictates the physical properties—low solubility counts as one of its defining traits. This means dumping this substance in water hardly makes a dent, unlike more soluble salts. Working in a lab years back, I remember needing strong acids just to dissolve samples; tap water didn’t stand a chance.
Why It Matters in the Real World
The science might sound a bit technical, but barium arsenate landed on my radar for another reason. Both barium and arsenic land on many health hazard lists. The Environmental Protection Agency (EPA) points out that arsenate compounds pose risks to human health and the environment. Both elements can cause harm: barium affects the nervous system, arsenic links to all sorts of toxicity.
Even though industrial use of this compound dropped over the decades, you still find traces in some ceramics, glass, and the odd research lab. Most folks never encounter it directly, but I’ve seen environmental teams check old disposal sites for arsenic leaching. Soil and groundwater tests keep an eye on these contaminants, turning up compounds like barium arsenate if old industrial waste heads south. Safe handling and proper disposal call for more than gloves and goggles—you need air systems and secure storage.
Potential Solutions and Safer Approaches
For folks working with or living near industrial sites, risk comes down to controlling exposure. Strong regulation around safe disposal limits the spread into the environment. Companies and universities substitute safer materials wherever possible. In my own lab work, we shifted away from arsenate chemistry for most experiments, sticking with phosphates or silicates. Cleanup means containment: testing waste streams with chemical sensors, using precipitation methods to remove barium and arsenate ions from liquids, and storing residues in well-designed landfills.
For long-term safety, public education about arsenic’s risks and careful tracking of waste help keep both workers and communities safe. Research continues on newer materials to replace legacy compounds in industrial processes. The story of barium arsenate holds a straightforward lesson: understand the chemistry, respect the risks, and move toward safer alternatives. Knowledge, in this case, really does protect both people and the planet.
| Names | |
| Preferred IUPAC name | Barium arsenate |
| Other names |
Barium orthoarsenate |
| Pronunciation | /ˈbeəriəm ɑːr.sə.neɪt/ |
| Identifiers | |
| CAS Number | 13464-38-5 |
| Beilstein Reference | 3552097 |
| ChEBI | CHEBI:88275 |
| ChEMBL | CHEMBL3986611 |
| ChemSpider | 21171272 |
| DrugBank | DB11103 |
| ECHA InfoCard | 100.006.604 |
| EC Number | 215-609-9 |
| Gmelin Reference | Gm.2.164 |
| KEGG | C18679 |
| MeSH | D001459 |
| PubChem CID | 24866193 |
| RTECS number | CX9310000 |
| UNII | 3F88B85N3E |
| UN number | UN1564 |
| Properties | |
| Chemical formula | Ba3(AsO4)2 |
| Molar mass | 439.07 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 4.8 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.43 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.1 |
| Basicity (pKb) | 12.32 |
| Magnetic susceptibility (χ) | +14.0e-6 cm³/mol |
| Refractive index (nD) | 1.92 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 128.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1660 kJ/mol |
| Pharmacology | |
| ATC code | V03BA03 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs through prolonged or repeated exposure; very toxic to aquatic life. |
| GHS labelling | **"GHS07, GHS08, GHS09, GHS06"** |
| Pictograms | GHS06, GHS08, GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P264, P270, P273, P280, P301+P310, P301+P330+P331, P304+P340, P311, P305+P351+P338, P308+P313, P314, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-4-OX |
| Explosive limits | Explosive limits: Not explosive |
| Lethal dose or concentration | LDLo oral rat 30 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 20 mg/kg |
| NIOSH | GR8335000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Barium Arsenate: "0.01 mg/m3 (as As) |
| REL (Recommended) | 0.01 mg/m3 |
| IDLH (Immediate danger) | **75 mg/m3** |
| Related compounds | |
| Related compounds |
Barium phosphate Barium sulfate Barium carbonate Calcium arsenate Lead arsenate |

