Barium Acetate: Old Roots, Modern Impact
Historical Development
Barium acetate didn’t pop up out of nowhere. As early as the 1800s, chemists found ways to react barium carbonate with acetic acid. They worked through candlelight and handled strong-smelling acids, showing early curiosity about the basic building blocks of matter. In the nineteenth century, applications centered on laboratory studies and curiosity about the periodic table. Later, growth in industrial chemistry shaped the way barium acetate entered manufacturing, especially as new dyes and pigments emerged. Generations of chemists picked up those early recipes and refined them, showing that sometimes, the building blocks of innovation depend on someone else's groundwork.
Product Overview
Walking through any chemical stockroom, you’ll find barium acetate as a white, crystalline substance with a faint vinegar smell. The chemical formula is Ba(C2H3O2)2, and in the world of reagents, it lands between common salts and specialty chemicals. People often look past it until they notice its place in glass production, chemical analysis, and even fireworks. In water, it dissolves easily, which helps in certain chemical syntheses. Most suppliers mark it as suitable for laboratory or industrial use, depending on the purity level and packaging.
Physical & Chemical Properties
Barium acetate’s sharp, needle-like crystals give it a subtle sparkle under light, almost like frost on a cold morning. Its molecular weight is around 255.42 g/mol. It melts at roughly 450 °C, and above that, decomposes rather than boiling. When dropped into water, it dissolves quickly and leaves no color. It doesn’t pack much scent, though a faint vinegar-like tang sometimes greets the nose. Chemically, it reacts with acids and strong bases, releasing acetic acid or forming other barium salts. Its reactivity profile suits those working in analytical labs and preparative chemistry.
Technical Specifications & Labeling
Barium acetate usually comes listed with purity above 99%, but grades may run lower for non-lab settings. Manufacturers print batch numbers, expiry dates, and hazard warnings on containers. Labels signal the material’s toxicity and proper storage—dry, cool places away from acids or ammonium salts. Globally Harmonized System (GHS) icons warn of risk if swallowed or inhaled. Weight, batch, and supplier info all appear on the drum or bottle, so every step of usage—from warehouse to benchtop—gets tracked.
Preparation Method
The most direct approach mixes barium carbonate with acetic acid, yielding carbon dioxide gas and barium acetate in solution. Chemists heat the mix, filter away leftovers, and evaporate water so the barium acetate crystals can form. For better purity, repeated recrystallization does the trick. Some dig for efficiency in industrial methods, searching for ways to cut energy steps by tweaking concentration, temperature, and input chemicals. These tweaks often save money and reduce environmental impact.
Chemical Reactions & Modifications
Barium acetate plays well with a series of chemical reactions due to its ionic nature and solubility. It swaps acetate ions for chloride in double decomposition reactions, letting manufacturers prepare barium chloride. People might use it to introduce acetate groups into other compounds, open the door for organic transformations, or act as a source of barium ions in inorganic syntheses. Barium acetate’s ability to form mixed salts or react further with sulfuric acid or chromates has kept it relevant in both research and teaching laboratories. Not every reaction leads to something glamorous; sometimes, it’s about reliability and repeatability.
Synonyms & Product Names
In catalogues and shipment manifests, people refer to barium diacetate, acetic acid barium salt, or simply Ba(CH3COO)2. Some packaging bears trade names from regional suppliers. Whether as barium ethanoate or under brand labels, it moves through global supply chains with different tags, but the science stays the same.
Safety & Operational Standards
Handling barium acetate takes care and training. Barium compounds pose risk if ingested, inhaled, or absorbed through skin. Proper gloves, goggles, and lab coats help protect workers, and well-ventilated storage keeps dust levels down. Many operations lock it in chemical stockrooms with restricted access, logging every release. Disposal heads to approved chemical waste streams, never down the drain. Emergency guidelines suggest calcium or magnesium solutions in case of poisoning, since barium blocks muscle activity and can disrupt heart and nerve function. Local and international agencies provide strict workplace rules so that accidents don’t lurk in the shadows of the supply closet.
Application Area
Barium acetate has found surprising staying power in the arts and sciences. In the lab, analysts use it as a reagent for preparing other barium salts, testing sulfates, and separating chemical mixtures. The glass and ceramics industries value it for shaping color and properties in finished goods. Firework producers lean on its contribution to green hues that arc across the night sky. Textile dye production taps into its reactivity for mordant work, helping dyes bind to fabric better. Water treatment, pigment manufacture, and feedstock applications tap its versatility, much like a trusty tool that never leaves the bench.
Research & Development
Across universities and private labs, ongoing research eyes barium acetate for cleaner synthesis routes, better efficiency, and reduced waste streams. Modifying the crystalline structure, tweaking reaction pathways, and digging into nanoparticle systems all widen the field. Some researchers use it as a source for novel organic and coordination compounds, while others track its fate in the environment. Digital monitoring and advanced sensors make R&D safer, reducing exposure and marking hazardous steps more clearly.
Toxicity Research
Toxicity studies put spotlight on barium’s effects on biological systems, with most attention on ingestion and inhalation pathways. Animal tests show that even moderate doses cause muscle paralysis, hypokalemia, and sometimes fatal heart rhythm changes. Chronic exposure links to kidney and liver effects, so industrial hygiene protocols keep strict boundaries. Many research projects pursue less hazardous substitutes, or aim to lock barium ions into matrices that cut bioavailability. Scientists also track environmental fate, since soluble forms easily enter waterways. Monitoring keeps the focus on user safety and broader ecosystem impacts.
Future Prospects
Looking ahead, demand for barium acetate shows no sign of vanishing, but trends point toward higher sustainability and safety. Research on greener production aims to shrink the carbon footprint, tapping recycled acids or renewable power. Digital inventory management will sharpen oversight and cut accidental misuse. Innovations in packaging may further reduce risks in transport and storage, with QR codes linking every drum back to its origins and test records. Cross-disciplinary teams see potential in fields ranging from advanced ceramics to energy materials, with an eye toward harnessing barium’s properties while meeting tighter safety and environmental standards. The story of barium acetate isn’t just about a chemical compound; it reflects ongoing work to match technological progress with stewardship for worker, user, and planet alike.
What is Barium Acetate used for?
Digging Into the Daily Impact
Some fine chemicals drift so far off the radar that most folks walk through life never hearing their names. Barium acetate fits this category, yet it lands in more corners of our lives than most realize. Many times, the things we don’t see carry a much heavier weight than what’s obvious. In my own circle, friends working in labs crack jokes about obscure salts, but behind the humor sits a chemical playing a real role in shaping other products that actually make life easier.
Crafting Glass and Ceramics
Barium acetate stretches far past lab benches. It supports glassmakers in the hunt for clarity and strength. What sets glass apart is its ability to transmit light and stand up to everyday knocks. One dose of the compound tightens up these qualities, pulling out bubbles and smoothing out flaws. A technician from a local ceramics shop told me they rely on this to keep color consistent and to fire at higher temperatures without risks of cracking. Through trial and error, they find that it can mean the difference between a batch destined for the trash heap and one that goes straight to market.
Chemical Research and Analysis
Many of my classmates, early in our chemistry days, got their first real exposure to barium acetate when preparing for qualitative and quantitative analysis. It dissolves in water much more smoothly than some other barium compounds. This property makes it a go-to choice for researchers running tests in solution. Tackling a synthesis or running routine tests, you’ll find that switching to acetate gets results faster and with less crud clogging up the works.
Textile Finishing
Textiles soak up plenty of specialty chemicals to reach the final fit and feel. Once, at a textile mill in the South, I watched technicians steady the finish on batches of cotton using barium acetate. The goal: step up water resistance and soften sheets. That factory chose it after running into issues with cheaper alternatives that didn’t hold up under scrutiny. It’s this side of the process that gives customers the feeling of smoothness or firmness that ends up making or breaking a sale.
Role in Pigment and Ink Production
Brighter, truer prints often lean on a handful of unexpected helpers. In several pigment plants I visited, barium acetate popped up on ingredient lists, especially for specialty inks used in industrial labeling and fine art materials. As inks mature and dry, the right chemical background ensures wood, cotton, and synthetic sheets won’t blur. My own attempts at printmaking taught me that skipping this step can send even the sharpest lines straight into muddy chaos.
Environmental and Safety Considerations
Handling of barium acetate tends to raise eyebrows among safety professionals. Exposure risks push everyone, from university students to factory veterans, to keep protective gear handy and double-check storage rules. Poisoning cases have been reported, though mostly tied to flagrant neglect of safety basics. Just down the road, a lab tech landed in urgent care after ignoring a torn glove. His story gets rolled out at every safety meeting now. These lessons add up: a big part of responsible use circles back to training and good habits.
The Path Forward
Safer practice means mixing the right training with better alternatives where they exist. Some manufacturers target substitutions, swapping out barium acetate for safer salts—even at added cost—because no production gain covers for a major health scare. Regulators encourage transparency and recycling programs. By sticking to clear communication, a shop can guard both its team and its reputation, and maybe teach others to step up too.
Is Barium Acetate toxic or hazardous?
What Barium Acetate Does and Where People Encounter It
Barium acetate shows up in labs, chemical processes, some electronics, and even specialized glassmaking. The name might look harmless on a label, but its effects can turn dangerous if ignored. Barium compounds often catch folks off guard, especially outside of industries with regular chemical training. Research and accident reports point to problems from careless storage or handling, and not many people read up on barium chemistry before getting started.
The Core Danger: Toxicity on Contact and Ingestion
Exposing skin to barium acetate brings itching, burning, or rashes for some people. Most of the concern focuses on swallowing or inhaling dust, which gets risky fast. Once barium enters the body, it messes with muscles and nerves. Weakness, breathing problems, heart rhythm issues, and even death can happen if a large enough dose sneaks in. Science journals and OSHA guidance stress that barium blocks potassium movement in cells, and that throws nerve signals out of whack.
Environmental Risks and Waste Problems
Dumping this salt into drains or soil leaches toxic barium ions that stick around and seep into groundwater. Even low doses poison fish and aquatic life, and it lingers in food chains. The U.S. Environmental Protection Agency includes barium in its contaminant lists for a reason. Cleanup turns expensive when spills go unchecked, so careful containment matters even outside the workplace.
Living with the Material: Personal Experience and Safety Steps
I spent several years working in a university chemistry stockroom. Bottles of barium acetate collected dust on back shelves. We carried it in sealed plastic drums with large warning labels, wore gloves and masks, and kept it away from acids to avoid releasing barium acetate fumes. New students often underestimated its hazards at first, thinking of acetate as a common ingredient in vinegar. The reality required clear instructions and training before anyone worked with it independently.
Handwashing, using dedicated scoops, and never tossing barium salts in regular trash bins set the baseline for safety. Chemical fume hoods, spilled material kits, and proper storage—far from food preparation areas—kept mistakes from turning into emergencies. Resisting shortcuts made all the difference, because the cost of one slipup reaches far beyond an individual’s health.
Finding Solutions: Smart Alternatives and Better Training
Some labs started shifting to less toxic alternatives wherever possible. If a reaction or process doesn't truly need barium, switching saves hassle and reduces risk. When teaching or training new staff, hands-on demonstrations go much further than just reading a safety sheet. Some organizations use detailed safety data sheets, videos, and real accident stories during onboarding. No substitute exists for face-to-face discussion, where questions bring out hidden gaps in understanding.
On the industrial side, engineers build safeguards like air handling systems, secure packaging, and clear labeling throughout the supply chain. Industry standards develop alongside technology, not as afterthoughts. Clear policies against improper disposal and regular waste audits make up another layer of protection.
Wrap-Up: Why Diligence Matters
The story of barium acetate serves as a reminder—no one can ignore risks just because a chemical seems obscure or looks ordinary. With honest, ongoing education, proper equipment, and enforcement of safety habits, real harm can be avoided. People’s lives depend on respect for what science tells us about toxic materials, and shortcuts have no place in the equation.
What is the chemical formula of Barium Acetate?
The Essentials: Formula and Structure
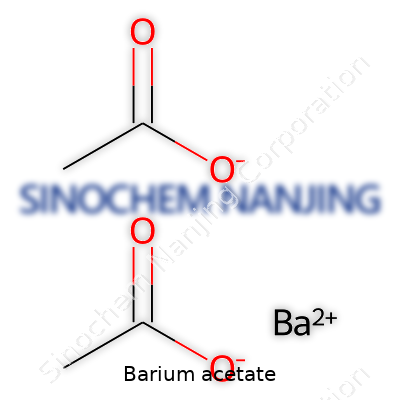
Barium acetate carries the chemical formula Ba(C2H3O2)2. Breaking it down, each molecule holds one barium atom and two acetate groups. The acetate bit comes from acetic acid, the same acid found in vinegar, minus a hydrogen ion. Each acetate delivers two carbon atoms, three hydrogens, and two oxygens, so you build up a molecule that’s both simple and surprisingly useful once those pieces join with a barium atom. Chemists often write it out as Ba(CH3COO)2, but either version points to the same thing—a rather straightforward salt that’s ready to react in just the right hands.
Why Barium Acetate Matters
Don’t let the plain-looking structure fool you. Barium acetate has its fingerprints on several key industrial and lab processes. For anyone in a chemistry lab, dissolving it brings up familiar sights—white, odorless crystals, ready to mix into solutions for research or manufacturing. It plays a role in organic synthesis and can act as a precursor for making other barium-based compounds. Some folks run across it in applications like printing textiles or making certain pigments. Even pyrotechnics experts find value here; barium acetate can color flames green for fireworks and displays, making those vivid sky shows possible.
Safety First: Handling Barium Acetate
I remember back in school, the first warning about barium compounds got drilled in fast. Soluble barium salts like this one carry health risks. Ingesting barium acetate can disrupt the nervous system, muscles, and even cause dangerous drops in potassium levels. You learn to double up on gloves, keep the goggles snug, and always clean up spills—habits that stick with most chemists for good reason. Local water sources have to stay barium-free; contamination can threaten wildlife and public health. This is why safe storage, correct disposal methods, and environmental monitoring can’t get skipped, especially in schools and factories.
Barium Acetate in Everyday Science
Barium acetate shows up in classic chemistry demonstrations. It dissolves in water and reacts easily, letting students see with their own eyes how ions swap and new compounds fall out of solution. Want to make barium carbonate? Toss in carbon dioxide and watch it turn. I’ve seen beginners light up when a simple salt like this turns into evidence that the world changes through chemical reaction, not just words in a book. This hands-on experience helps keep science approachable.
Looking Forward: Sustainable Practices and Alternatives
Industries continue using barium acetate for all the reasons above, but growing attention falls on keeping its use safe and sustainable. Many companies move toward tighter regulation and waste management. Others search for less risky alternatives where possible. The European Chemicals Agency and the US Environmental Protection Agency keep tabs on its environmental impacts. Future progress could mean using barium acetate only where no safer option exists, or developing ways to recycle it. Chemists sharing what works, and what doesn’t, will move the field forward while protecting health and the planet.
How should Barium Acetate be stored?
The Why Behind Storage Choices
A lot of folks hear “barium acetate” and picture a chemistry textbook. The truth is, this compound pops up in research labs, chemical manufacturing, glassmaking, and even some lessons on ionic reactions. Barium acetate brings certain risks. Swallowing it can lead to real trouble — we’re talking severe symptoms or worse. So putting it wherever there’s space under a lab bench won’t cut it. Fact sheets from the CDC and OSHA make that clear. Storing it right helps avoid health emergencies and stops chemical messes that spread farther than one room.
Don’t Trust Luck: Control What You Can
Barium acetate looks like a benign white powder, but it doesn’t belong near food, drinks, or common workspaces. I remember working in a classroom where the janitors stacked cleaning supplies beside jars of metallic salts. Not only was that risky, but the shelf was full of open boxes. Just because barium acetate won’t explode on contact with paper towels, mixing those materials invites mistakes. Cross-contamination or an accidental sprinkle near an acidic cleaner can start a chain reaction. All it takes is a moment of distraction.
Right Place, Right Package
Glass or high-density polyethylene bottles do the job. Both resist corrosion and can handle the powder’s moisture-drawing habits. I always pick a tight lid, something that doesn’t loosen over weeks or months. Don’t reach for metal — some grades of steel or aluminum react with barium salts over time, and that creates more problems. If you forget one day and leave the container open, humidity changes the chemical’s character and shortens shelf life.
Temperature swings ruin storage too. Barium acetate prefers a steady, cool climate, away from direct sunlight and hot water pipes. Cabinets built for chemicals offer real peace of mind. I’ve watched cabinets with lockable doors stop curious hands from poking around. They also bring order to what often looks like organized chaos in a busy science building. Label the bottle with big lettering — hazard signs, date received, expiry — so nobody guesses what’s inside.
Manage Spills and Keep Clean
Even if the bottle seems sealed, double up: set everything inside a chemical-resistant tray. If a leak occurs during an earthquake, cleanup takes minutes instead of hours. Store barium acetate apart from acids and organic solvents. Mixing those by accident makes hazardous fumes. In my old college lab, a careless shelf shuffle placed barium acetate next to sulfuric acid. Quick thinking averted a dangerous incident, but it showed how overlooked storage details can pile up.
Safety supplies deserve their own shelf nearby. Gloves, goggles, and dust masks — don’t stash them across the building. Keep sodium sulfate or magnesium sulfate on hand in case barium splashes. Both work as neutralizing agents. Training everyone in the room is worth the time; it creates habits. Simple rules: never scoop with a bare hand, always sweep up any granules, and wipe counters after using.
Mind the Rules
Hazard communication standards push us to respect these chemicals. Barium acetate isn’t just another white powder. It’s a substance that can harm — or help — depending on the care behind each action. Using trusted resources, as OSHA and the CDC recommend, backs up good lab culture. Each step — tight containers, cool storage, clear labels, organized shelves — makes it far less likely for a simple mistake to turn into a disaster.
What are the safety precautions when handling Barium Acetate?
Understanding the Risks
Barium acetate’s crisp white powder looks harmless, but it brings real risks to health if handled carelessly. In my time around lab chemicals, I’ve seen how easy it is for folks to dismiss powders and salts because they aren’t bubbling or giving off strong fumes. Yet, barium compounds have a notorious track record—skin contact, inhalation, or accidental swallowing cause serious problems, including muscle weakness, irregular heartbeat, and even paralysis. This isn’t just speculation; the Centers for Disease Control confirms the dangers, citing symptoms even from small exposures.
Personal Protective Equipment Isn’t Optional
No one should approach barium acetate without safety glasses. A friend of mine thought regular prescription lenses offered enough coverage. They don’t. Fine dust travels quickly, and eyes are too valuable to take that sort of risk. Gloves, preferably nitrile, earn their place for a reason; bare skin can absorb toxins, and even a small spill leads to trouble. A standard lab coat shields your arms and regular clothing from splashes and spills.
Unlike some lab chemicals, barium acetate powder is just as hazardous dry as it is in solution. A simple breeze can send dust into the air, so using a chemical fume hood becomes standard practice, not overkill. Those who rely on disposable masks instead of a fitted respirator are playing with luck. Particulate respirators (look for N95 or higher) guard the lungs against fine powders that never advertise their presence.
Clear Labeling and Storage Practice
Storing chemicals sounds simple; the reality is mistakes pile up if people cut corners. I once visited a small lab where barium acetate sat with food containers. This makes cross-contamination far too easy. Locking up barium acetate in a well-labeled, sealed container—not a reused plastic tub—prevents accidental mix-ups. Chemical safety experts recommend keeping it away from acids and combustible materials. It’s not just over-cautious; improper storage has led to emergencies, like toxic gas releases and fires.
Preparation and Spills
Mixing solutions with this chemical produces heat, sometimes enough to splatter. Slow, careful addition becomes second nature. After, all surfaces need a thorough clean, using water and a non-reactive cloth. Spilled barium acetate doesn’t “disappear” if brushed to the floor, and dry sweeping adds danger by making dust airborne. A trained response calls for damp cloths and sealed waste bags. I’ve seen labs rush cleanup without following protocols; only by treating every spill as a serious threat do you protect everyone nearby.
Training and Emergency Plans
Time and again, emergencies show training matters more than fancy equipment. Every worker needs to know signs of barium toxicity—tingling hands, dizziness, stomach discomfort. The CDC lists these as early warnings, and knowing what to watch for saves time in calling medical help. Clean water for rinsing eyes and showers for skin exposures put safety within reach. Local poison control numbers should stay posted where all can see, not buried in a folder.
Why Following Protocols Matters
Big chemical incidents rarely start with an explosion. Often, they begin with overconfidence. I trust proven systems: keep inventory, track who uses what, and never rely on memory alone. Following standards isn’t red tape; it’s respect for my own health and that of coworkers. The few extra seconds for gloves and goggles or the discipline to store chemicals right keeps labs open and people safe. Those moments are worth it every time.
| Names | |
| Preferred IUPAC name | Barium diacetate |
| Other names |
Acetic acid, barium salt Barium(II) acetate |
| Pronunciation | /ˈbeəriəm ˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 543-80-6 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Barium Acetate**: ``` CC(=O)[O-].CC(=O)[O-].[Ba+2] ``` |
| Beilstein Reference | 319835 |
| ChEBI | CHEBI:133876 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 14218 |
| DrugBank | DB11134 |
| ECHA InfoCard | 100.024.325 |
| EC Number | EC 213-026-2 |
| Gmelin Reference | 13611 |
| KEGG | C18636 |
| MeSH | D001474 |
| PubChem CID | 24636 |
| RTECS number | AG8756000 |
| UNII | Z80Y3I6QEC |
| UN number | UN1564 |
| Properties | |
| Chemical formula | Ba(C2H3O2)2 |
| Molar mass | 255.42 g/mol |
| Appearance | White powder or crystals |
| Odor | Odorless |
| Density | 2.46 g/cm³ |
| Solubility in water | 39.8 g/100 mL (20 °C) |
| log P | -2.85 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.76 |
| Basicity (pKb) | 3.98 |
| Magnetic susceptibility (χ) | −52.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.513 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 205.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1016.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1627.0 kJ/mol |
| Pharmacology | |
| ATC code | V09BA03 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. Toxic to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H332, H373 |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 921 mg/kg |
| LD50 (median dose) | 920 mg/kg (Rat, oral) |
| NIOSH | BA1750 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Barium Acetate: 0.5 mg/m3 |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Barium carbonate Barium chloride Acetic acid Calcium acetate |

