Phenylarsine Dichloride: A Closer Look at Its Role, Hazards, and Future
Historical Development
Chemistry doesn’t sit still. Researchers in the nineteenth and early twentieth centuries chased after new elements and their combinations, often at great personal risk. Phenylarsine dichloride sits in the shadow of that era—discovered as part of broader studies into organoarsenic compounds. Back when organometallics were taking the world by storm, some believed these arsenic-containing chemicals would solve pest problems and maybe even lead to medical advances. Wartime use of similar arsenicals left a dark mark, with these compounds showing up in chemical warfare agents—this is real chemical history, not just lab trivia. Exposure studies, decades of accidental poisonings, and environmental concerns shaped the legacy. Scientists learned the hard way what can happen when innovation outpaces awareness of risk.
Product Overview
Phenylarsine dichloride brings together arsenic, chlorine, and a phenyl group, creating a compound that’s both highly reactive and hazardous. Laboratories value it for its ability to serve as a reagent, reacting with sulfhydryl groups and other sensitive targets. The compound isn’t something you’ll find in a hardware store or garden shed, and there’s a good reason: handling it requires skill, serious training, and the right infrastructure. Even trace exposure means real, sometimes irreversible harm. This is not a chemical for the casual tinkerer or even most hobbyists—specialized uses and research are where this molecule gets to work.
Physical & Chemical Properties
If you’ve worked with phenylarsine dichloride, its pungent odor and clear to pale yellow appearance stand out. This material is volatile, giving off dangerous vapors at room temperature. Chemically, it bridges organic and inorganic chemistry—arsenic at the center, two chlorines clinging, and a phenyl ring. Reactivity with moisture and nucleophiles describes its behavior better than any theoretical property table. Pull the top off a freshly prepared container and there’s no mistaking the threat—protective gear isn’t optional.
Technical Specifications & Labeling
Labels on bottles and in storage areas need to shout out the risk, not whisper. Phenylarsine dichloride often comes with hazard pictograms matched to its acute toxicity and environmental persistence. Regulations demand clear indication of toxic, irritant, and ecological danger. By law and for good reason, the amounts supplied remain small, and only registered institutions with serious chemical hygiene programs have access. In my experience, regulators look for bulletproof documentation on what’s inside each bottle and how it’s tracked from cradle to grave—this isn’t red tape, it’s real protection.
Preparation Method
Synthesis calls for deep respect and finely tuned technique. Most methods ask for reaction between phenylarsine oxide and thionyl chloride or phosphorus pentachloride, using careful temperature control. The result, distilled under dry and inert conditions, leaves no room for shortcuts. Those who cut corners with arsenic chemistry often pay with long-term health consequences, not to mention regulatory penalties. I’ve seen firsthand how even minor sloppiness in handling precursors or managing waste can unleash trouble beyond a single experiment.
Chemical Reactions & Modifications
This chemical’s action with thiols stands out. Phenylarsine dichloride will grab onto sulfur atoms, chewing through protein structures and disrupting enzyme functions. In organic synthesis, it’s a sharp tool for modifying peptides and proteins. Once, working on sulfhydryl labeling, I found out that speed and precise stoichiometry really matter—overdo it and the whole experiment turns toxic in more ways than one. The compound’s readiness to swap its chlorine groups for other ligands makes it versatile, but also unpredictable if misused.
Synonyms & Product Names
Names for this chemical stretch across languages and catalogues: phenylarsine dichloride, dichloro(phenyl)arsine, and for those who follow historical echoes, SA-forming agents. That range of names points to mixed legacies, with utility on one hand and hazardous applications on the other. Keeping track of synonyms in documents and supply chains matters, since confusion can lead to wrong shipments or mishandling. Anyone ordering, transporting, or using chemicals with a profile like this knows to triple-check every reference.
Safety & Operational Standards
Everything about phenylarsine dichloride screams for respect and rigid controls. Proper fume hoods, closed-handling systems, and substantial personal protective equipment aren’t optional extras. I remember one lab safety day where the instructor poured time into our protocols for arsenic chemicals—no shortcuts. Waste disposal presents another sinkhole for mistakes: residues can’t just hit the drain. Years ago, a nearby university paid heavy fines after arsenic traces turned up in routine water tests—a sobering consequence for incomplete handling. Regular blood and urine monitoring for workers using arsenicals speaks to the depth of the risk. It all comes down to a single truth: mistakes can linger far beyond the lab.
Application Area
In research, phenylarsine dichloride finds a purpose mostly as a highly specific probe or modification reagent. It’s used to bind thiol-containing proteins, map out enzyme function, or build models for toxicology. Outside strict research, very few processes justify its risk. Historical applications in warfare put a lasting stigma on anything with arsenic’s fingerprint. Most current legitimate use appears behind locked doors in universities and advanced research labs, with strict oversight. Before assigning it to any project, a scientist needs a concrete rationale—something that can’t be achieved with safer chemicals.
Research & Development
Inventive chemists and toxicologists look at phenylarsine dichloride as a window into sulfur-arsenic chemistry and protein structure. Anyone who’s prepared arsenical reagents in the lab knows the precision these studies demand. For instance, advances in labeling techniques for proteins still sometimes reach for this compound because alternatives fail to capture its specificity. Unfortunately, most R&D involving phenylarsine dichloride needs triple the paperwork and quadruple the vigilance compared to less toxic reagents. Graduate students and principal investigators navigate a tough balancing act between scientific curiosity and rigorous caution.
Toxicity Research
Every major study of organoarsenic compounds circles back to toxicity. Acute symptoms of exposure range from severe skin irritation to life-threatening systemic poisoning. Chronic exposure? That means neurological damage and increased cancer risk. Data on breakdown, residue, and long-term environmental fate put the compound in the crosshairs of environmental scientists. Animal testing shows a brutal path from exposure to organ failure. Even with modern ventilation and PPE, accidental exposures leak through—one slip, one forgotten mask, one moment of inattention can end up in a publication’s footnote or a journal’s retraction. Talking with colleagues, I hear stories where fast action after spills or splashes made all the difference: a chemical shower, a medical response, a report filed in full detail. Sharing these lessons keeps old mistakes from cycling through the next generation.
Future Prospects
Changing regulations and increasing ethical scrutiny limit the reach of phenylarsine dichloride, but chemists never stop searching for sharper, safer alternatives. With each new project, institutions question whether the specific reactivity justifies the risks. The trend in research moves toward molecules that do similar jobs with drastically less hazard. Automation and microfluidics might one day limit human contact with this and related compounds, shrinking exposure to near zero. Environmentally, the pressure stays on for total elimination from regular lab inventories. Looking back at the evolution of chemical safety, the arc bends toward replacing hazardous reagents with benign ones wherever possible. People still working with phenylarsine dichloride know the stakes, and each lab protocol reflects that: a handshake agreement between scientific ambition and a moral obligation to protect workers, the public, and the environment.
What is Phenylarsine Dichloride used for?
The Hidden Side of an Industrial Chemical
Phenylarsine dichloride sounds intimidating. Most people never encounter it outside of a chemistry textbook, yet its influence stretches into labs, manufacturing spaces, and beyond. This compound isn’t a household word, but anyone walking through the corridors of a modern research lab would catch a whiff of its significance.
How Scientists Put Phenylarsine Dichloride to Work
Chemists chasing complicated molecules appreciate the unpredictability of organic arsenic compounds. Phenylarsine dichloride fills that spot. Its knack for reacting with thiol groups offers up a rare tool for probing biochemical mysteries. Researchers dig into proteins and enzymes and use phenylarsine dichloride to “tag” the sulfhydryl groups. That way, they tell which cysteines in a protein are important. Honestly, this saves endless hours and offers pretty crisp results.
My old supervisor at a university lab used to describe these reagents as his “molecular magnifying glass.” He’d wave around a glass bottle, and though most undergrads flinched at the skull-and-crossbones warning, he kept reminding everyone that understanding proteins sometimes demanded risky tools. In fields where biochemistry crosses paths with pharmacology and medicine, this kind of reagent shortens years of searching for active sites to a matter of weeks. That transforms how quickly we can develop new medications or tackle enzyme malfunction in disease.
Manufacturing Utility: More Than a Lab Tool
Industrial chemists don’t just use phenylarsine dichloride as a research probe. They rely on its power as a chemical intermediate, especially when the end goal involves other organoarsenic compounds. I remember a chemical manufacturer in Texas using it to build more complex structures—a kind of village blacksmith, but one with goggles and gloves handling dangerous reactions instead of steel. Its reactivity opens the door for new materials, specialized catalysts, and even advances in electronics, though always with tight safeguards against exposure.
Health Risks No One Should Ignore
Just saying the word "arsenic" gets everyone’s guard up, and for good reason. Government agencies have strict rules about using and disposing of phenylarsine dichloride. Direct contact and inhalation bring a real risk of poisoning. Chronic exposure messes with the nervous system and even smaller spills trigger emergency cleanups. Meeting these challenges takes more than one-off training videos. Anyone working with phenylarsine dichloride needs ongoing education, proper gear, and fast emergency response. I once worked with a safety coordinator who insisted on daily checks—no shortcut, no negotiation. The culture of caution stuck, making everyone safer in the long run.
Staying Responsible with Chemistry
Looking at the broad picture, phenylarsine dichloride teaches an important lesson. Powerful chemicals transform the way science works, but each benefit comes with heavy responsibilities. Companies and research organizations do better when they double down on transparent procedures and respect for safety. Switching to less toxic alternatives or greener processes should stay top of mind. Policymakers set the ground rules, but respect for human life starts right among those who pour, weigh, and react these substances every day.
What safety precautions should be taken when handling Phenylarsine Dichloride?
Understanding the Risks
Phenylarsine dichloride brings both danger and opportunity to the bench. It acts as a powerful reagent, useful in organic synthesis, but its toxicity compares to some of the worst substances found in old-school chemistry. Even trace exposure causes harm—skin blisters, nasty burns, and over time, irreversible damage to organs. Arsenic compounds also raise cancer risk after frequent contact. Stories from chemists show what happens after letting basic safety slip: coughing, headaches, rashes that last for days, cases where protective gloves broke down within minutes and gave folks unforgettable lessons.
Protection Starts With Planning
Preparation always matters more than bravado. Before opening a bottle, most labs review the Safety Data Sheet and run through emergency steps out loud. Spills can’t always be avoided, so knowing where to reach an eyewash station or how to call hazmat makes a difference if trouble strikes. Some use the buddy system, never working alone, so someone will help if things get out of control.
The right space helps too. Fume hoods do more than keep annoying smells down—they carry dangerous vapors far away from your lungs. Labs with good exhaust systems also limit the spread of invisible dust that could settle on skin and clothes.
Picking the Best Gear for the Job
Exam gloves alone don’t cut it with phenylarsine dichloride. Thicker butyl rubber or layered gloves will keep hands safe. Throw-away lab coats stop splashes from soaking shirts. Always swap out clothes after finishing, because small amounts cling and travel home with you if you’re not careful. Simple glasses don’t protect enough. With this compound, chemists wear splash goggles and even face shields for heavy-duty tasks.
Handling these chemicals demands extra care with storage. Tightly sealed bottles go into vented cabinets, far from acids, bases, or anything that reacts with moisture. Ask anyone storing these for a while—they keep them dry, upright, and labeled in big letters, not tucked away in busy parts of the lab.
Reducing Exposure and Managing Waste
Once some phenylarsine dichloride is in use, even the air can turn hazardous. Always having the right masks on hand matters. Respirators with filters for organic vapors should always be ready, especially if accidents could send chemical clouds into the room.
Waste needs careful handling. Pour-off and soaked materials need sealed, labeled drums, collected by professionals no matter the cost or hassle. A friend in chemical disposal once told stories about folks who risked fines, or worse, by dumping small amounts down drains. That’s a shortcut with huge consequences.
The Importance of Good Habits and Training
The best labs hold training more than once. Relying on memory from college isn’t enough. Sharing real stories about close calls turns the rules into living habits. Labs who track incidents and talk mistakes provide lessons you can’t get from a printed poster.
Using phenylarsine dichloride safely is all about respect—respect for the compound, for coworkers, and for your own future health. Trust in good routines, not shortcuts, and watch out for each other. Every step counts, because nobody wins from a half-safe lab.
What are the storage requirements for Phenylarsine Dichloride?
Getting Real with Chemical Storage
Phenylarsine dichloride deserves respect. This chemical’s dangers don’t end at the lab bench. It’s used in research and industrial work, but it doesn’t reward carelessness. It’s toxic, corrosive, reacts with water, and turns nasty when things go wrong. Labs that keep it on the shelf can’t treat it like any other bottle.
Keep It Sealed, Keep It Dry
Water and humidity spell trouble. Get a bit of moisture in the bottle, and you start releasing hydrochloric acid fumes and other hazardous gases. If you’ve worked in a lab, you know fumes don’t just stay put—they move fast. So, always seal containers tightly and use materials that don’t react, like glass with Teflon-lined caps. Every chemist learns early that shortcuts with dangerous chemicals leave scars or worse.
Environment Makes All the Difference
Don’t just toss this on any shelf. Cool, dry, and dark storage keeps risks in check. Light sometimes triggers reactions, especially with sensitive arsenic compounds like this. A flammable cabinet or a dedicated corrosives cabinet works, just make sure it’s not near water sources, sinks, or materials that soak up moisture from the air.
Separation Isn’t Optional
This isn’t something you store next to acids, bases, or other reactive chemicals. If phenylarsine dichloride sits too close to incompatible materials—like oxidizers or strong reducing agents—even small leaks or spills can result in violent reactions or toxic releases. Keep it apart, and use labeling that warns staff what’s in there. Memories of mixed-up chemicals in high school chemistry might raise a smile, but mistakes now cost more than a detention.
Ventilation Matters More Than You Think
Every experienced lab worker recognizes the telltale sting or smell of released gases, even when the bottle looks fine. Ensuring good airflow—like ventilated cabinets and working fume hoods—means that if something leaks, the danger doesn’t linger and threaten people nearby. It just takes one overlooked vent to turn a small problem into an emergency. I've seen what happens when custodial staff open a storage room without warning—the smell chased everyone out for hours. Signs and procedures are part of the protection.
Don’t Forget The Emergency Gear
Eye wash stations, showers, and spill kits belong close to wherever toxic chemicals get stored. Phenylarsine dichloride exposure is a medical emergency, not just something you shrug off with a sink rinse. Supervisors and staff need to make sure that gloves and goggles always come on before opening the cabinet, and that proper containers are on hand for cleanup.
Regulations Aren’t Just Paperwork
International and local rules exist for good reasons. The CDC, OSHA, and EPA all set limits for how much of this stuff you can store, how you must label it, and what’s required for ventilation and containment. Fines for violations are steep, but consequences for people are worse. Every researcher and facility manager should review these rules before new stock arrives, not after someone gets exposed.
Safe Storage Is Teamwork
Phenylarsine dichloride highlights why training never stops. Turnover happens, and newcomers sometimes miss details. Taking the time to explain why these rules exist—beyond "because it’s the procedure"—builds habits that last. Nobody forgets the lesson from a near-miss, so the aim should be to avoid those moments.
There’s Always Room for Better Solutions
Labs with strong safety records look for ways to swap out highly toxic materials where possible. For those that can’t, checklists, regular audits, and transparent reporting make a difference. If you find a leaky lid or a mislabeled shelf, bring it up. One person noticing a risk stops a problem before it grows.
What are the possible health effects of exposure to Phenylarsine Dichloride?
What Exactly Is Phenylarsine Dichloride?
Phenylarsine dichloride shows up in history books about chemical warfare. It popped up during World War I as a “vomiting agent.” This chemical falls under the family of arsenic compounds, and arsenic’s reputation for causing trouble spans centuries—from being suspiciously present in some cases of poisoning to being flagged as a carcinogen.
Breathing It In: What Happens in the Body
Anyone coming into contact with phenylarsine dichloride quickly notices the nasty side effects. The compound targets the respiratory tract, starting with a burning sensation in the nose and throat. Exposure brings on coughing, sneezing, and nasty headaches. A person can start vomiting or feel tightness in the chest within minutes. Eyes go watery, skin gets irritated, and someone exposed long enough can collapse or even stop breathing. The Centers for Disease Control and Prevention warns that inhaling it doesn’t just sting—it shuts down normal breathing and can cause permanent lung damage.
Touching or Swallowing: More Than a Nuisance
It doesn’t stop with breathing problems. Skin contact leaves people with blisters and rashes. Eyes exposed to the vapors become bloodshot, swollen, and painful. Ingesting phenylarsine dichloride brings arsenic’s toxic traits into play. People can suffer abdominal pain, diarrhea, and eventually damage to the liver and kidneys. Poisonings involving arsenic compounds often bring chronic health issues. The body, when faced with continued exposure, may even start showing neurological symptoms—confusion, numbness, and tremors.
Risks That Don’t Just Go Away
Long-term health effects creep up long after the first encounter. The World Health Organization and the U.S. Environmental Protection Agency both label arsenic compounds as carcinogens. The risk of certain cancers climbs higher with repeated exposure. Beyond cancer, people can wind up with chronic skin lesions, heart issues, low red blood cell counts, and decreased resistance to infections. Even if the symptoms at first seem minor, the bigger risks pile up over time.
Protecting People: Real Solutions Matter
Many believe chemical threats like phenylarsine dichloride linger mostly in war stories and dusty history books. Lab accidents and illegal uses still pop up. Thousands of scientists, emergency responders, and workers in hazardous waste clean-up zones remain at risk.
Personal experience in chemical safety work makes one fact clear: lack of access to proper protective equipment and training leads to tragedy. Good ventilation, full protective gear, and real-time chemical detection don’t just check off boxes during inspections—they save lives. Regular training sessions keep people sharp and aware. Educators stress the need for fast-acting medical responses. Washing off the chemical and delivering oxygen early can cut down lasting damage.
Communities living near old industrial sites worry about stored chemicals. For peace of mind, people push for stronger laws on chemical storage and disposal. Emergency preparedness—local warning systems, evacuation drills, and clear communication—protects more than the workers; it protects neighborhoods.
Science points to the dangers. Real-world stories of accidental exposure prove no one is immune. Arsenic compounds aren’t relics. They’re an ongoing concern for public health, and it falls on all of us to act with what we know.
How should Phenylarsine Dichloride spills or leaks be managed?
The Heavy Risk Behind a Colorless Liquid
Phenylarsine dichloride doesn’t make the headlines every day, but it brings real hazards. This chemical, known for its sharp and biting odor, can turn an ordinary workday upside down. A drop on the floor, a leaky container, a fumbled transfer—each carries the potential to harm skin, lungs, and even the water table. I’ve seen firsthand how a single oversight can send a team scrambling for safety showers and panic kits. The danger doesn’t just evaporate with time or dilute with wishful thinking; it calls for discipline, know-how, and real courage to speak up and act fast.
Protecting People Comes First
Folks dealing with phenylarsine dichloride should never gamble with safety. Chemical-resistant gloves, long sleeves, goggles, and good ventilation are tools that keep coworkers out of the ER. Respirators safeguard lungs if fumes escape. Quick thinking in the workplace can be the difference between a minor incident and a bad tragedy. Stories circulate through labs about rushed spills and the heroes who stopped them in their tracks. People remember the sound advice of a mentor who taught respect for the baggy suit and face shield, not just for state inspectors or insurance claims, but because every headcount after a shift matters.
Containment: Act Before It Spreads
Phenylarsine dichloride attacks quickly once loose. Absorbent pads, spill berms, gritty sand—all should be sitting close by, ready to go, not locked in a distant closet. That way, anyone can jump into action. A small spill turns massive if it seeps under doors or soaks into cracks. Sometimes, simple clay or vermiculite grabs the liquid and holds it in place while the cleanup team gets suited up. Spraying water offers zero help; it only cascades the poison into drains. Practice runs matter here. Folks who’ve run drills don’t freeze up, and they remember to block the exit with barriers, not just their bodies.
Cleanup Isn’t Glamorous—It’s Essential
After containment, shovels and heavy-duty bags do the dirty work. Crews trained in hazardous material response handle the final mop-up. I’ve stood outside scenes like these, watching wide-eyed, knowing the right call keeps those inside from lifelong scars or worse. Waste disposal requires total transparency with haul-away companies. No one wants to open a barrel of nasty secrets months down the road. I’ve seen places where clear labeling and accurate logs saved time and avoided drawn-out investigations. Regulators and honest reporting keep shortcuts from creeping in.
The Link to Health and Community Safety
Arsenic doesn’t vanish after a chemical takes a spill. Groundwater, local wildlife, workers, and kids biking home after school—each can feel the aftershock. A single accident in a poorly prepared shop can poison entire blocks. Genuine outreach with neighbors, first responders, and public health teams gives everyone a fighting chance if things go south. Open training and sharing lessons learned build trust. Good relationships between companies and emergency teams, plus a real plan for notification, keep confusion low and response rates high.
Solutions Rely on Experience and Training
Effective management comes from treating phenylarsine dichloride with the respect it deserves. That’s not just about rules on paper—the right tools, awareness, updated training, honest oversight, and team spirit keep workplaces and communities safe. Trust grows with every near-miss reported and every worker who chooses the right protection, even if no one watches. Protecting each other from a spill isn’t just about compliance; it’s a promise that every shift ends safely.
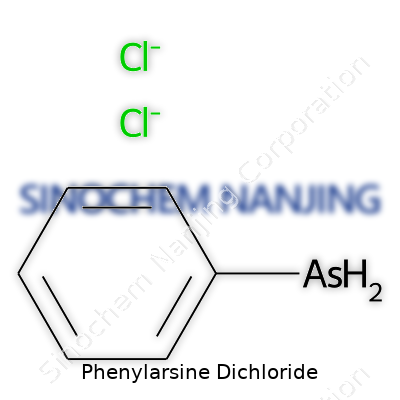
| Names | |
| Preferred IUPAC name | dichloro(phenyl)arsane |
| Other names |
Dichlorophenylarsine Phenylarsenic dichloride Fenylarsindichlorid Phenylchloroarsine Chlorophenylarsine |
| Pronunciation | /ˌfiːnɪlˈɑːrsiːn ˈdaɪklaɪd/ |
| Identifiers | |
| CAS Number | [696-28-6] |
| Beilstein Reference | 1366056 |
| ChEBI | CHEBI:52262 |
| ChEMBL | CHEMBL510532 |
| ChemSpider | 15310 |
| DrugBank | DB13916 |
| ECHA InfoCard | 100.004.529 |
| EC Number | 207-139-9 |
| Gmelin Reference | 163383 |
| KEGG | C18730 |
| MeSH | D017729 |
| PubChem CID | 66201 |
| RTECS number | CN9625000 |
| UNII | W76P9M7A3P |
| UN number | 1607 |
| Properties | |
| Chemical formula | C6H5AsCl2 |
| Molar mass | 272.93 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | garlic-like |
| Density | 2.2 g/cm3 |
| Solubility in water | Decomposes in water |
| log P | 1.94 |
| Vapor pressure | 0.05 mmHg (25°C) |
| Acidity (pKa) | 10.8 |
| Basicity (pKb) | 6.54 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.713 |
| Viscosity | 1.47 cP (20 °C) |
| Dipole moment | 1.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 225.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | ΔfH⦵298 = -31.6 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause cancer; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: "Fatal if swallowed. Fatal in contact with skin. Fatal if inhaled. Causes severe skin burns and eye damage. Causes damage to organs. |
| Precautionary statements | P261, P264, P271, P280, P301+P310, P304+P340, P308+P311, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | Flash point: 87°C |
| Lethal dose or concentration | LD50 oral rat 7 mg/kg |
| LD50 (median dose) | LD50: 3.1 mg/kg (oral, rat) |
| NIOSH | VL8575000 |
| PEL (Permissible) | PEL: 0.05 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit) of Phenylarsine Dichloride: "0.002 mg/m3 |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Phenylarsine oxide Triphenylarsine Diphenylchloroarsine |

