Arsenic Trifluoride: Caution, Curiosity, and the March of Chemistry
Historical Development
Arsenic trifluoride carries a long, layered story, winding through the annals of chemical discovery. Early chemists were driven by both necessity and curiosity, poking at the dark edges of the periodic table’s infamous elements. In the late 1700s, as arsenic-based pigments and compounds found wider use, researchers started digging into halide chemistry. Arsenic’s reputation as a poison didn’t scare off early experimenters; instead, its dual nature sparked interest. As more scientists navigated the risks, arsenic trifluoride began finding roles in both laboratory research and industrial processes. The journey to understanding this simple, yet hazardous, molecule illuminated just how hard it can be to strike a balance between scientific progress and public safety.
Product Overview
Arsenic trifluoride rests in the shadowy intersection of utility and danger. As a potent fluorinating agent, it holds sway over reactions that need a strong—but not explosive—fluoride source. Chemists value it for its ability to introduce fluorine atoms where others fail. Most folks won’t ever spot this material in a consumer setting; it mainly roams specialized industries and research labs, where experience and deep knowledge shape its handling. Its reputation precedes it, partly because of the broader legacy of arsenic and partly because of its own nasty traits.
Physical & Chemical Properties
Fresh arsenic trifluoride falls as a colorless, volatile liquid that manages to escape most containers as a sharp, acrid vapor. It doesn’t blend well with water―instead, it reacts to liberate toxic fumes and plenty of heat. With a boiling point not far above room temperature and a density greater than water, it behaves more like a heavier organic solvent than a common salt. This stuff fills the air with a choking sense of danger at the first sign of a leak—its thumbprint is unmistakable to anyone who has encountered it. Highly corrosive, eager to chew through tissue and metal alike, it commands both respect and careful storage.
Technical Specifications & Labeling
Every bottle of arsenic trifluoride comes stamped with hazard warnings that speak for themselves. Black-and-white toxic symbols cover the label, and precise storage conditions get priority placement. Packaging rarely strays far from airtight, corrosion-resistant containers, sometimes lined with materials that resist even aggressive fluorides. It doesn’t take much for a small misstep to escalate, so labels emphasize not just content, but conditions: keep cool, keep dry, keep sealed. In professional circles, labeling isn’t just a regulatory box to tick—it marks the razor’s edge between safe handling and disaster.
Preparation Method
Making arsenic trifluoride isn’t kitchen chemistry. Most synthesis happens in controlled industrial or laboratory setups. Chemists usually start with arsenic trioxide or metallic arsenic and treat it with hydrofluoric acid. Folks familiar with industrial procedures know that both starting materials and end products demand armored equipment, fume hoods, and technicians who’ve learned the hard way about the risks. Generating white clouds of fluoride-laced vapor pushes everyone’s safety muscle to the limit. Improper technique can feed into dangerous runaway reactions, which is why scaling up production generally happens behind multiple barriers and under watchful eyes.
Chemical Reactions & Modifications
Arsenic trifluoride draws in chemists with its ability to hand over fluoride ions. It transforms oxides into fluorides—a trick with value in both research and specialty production. Some intricate organic synthesis pathways also rely on this compound to swap out functional groups, giving it niche appeal. On the flip side, arsenic trifluoride can react violently with water or alcohols, sometimes breaking down with a hiss and puff that warns everyone to step back. Mixing it with strong acids or bases rarely ends well and usually finds mention only as a cautionary tale in chemical safety lectures.
Synonyms & Product Names
Through history, arsenic trifluoride has gone under many monikers. Sometimes it’s called simply “arsenous fluoride,” “trifluorarsine,” or, on old labels, “fluoroarsine(III).” These names echo its structure: three fluorines attached to an arsenic core. Sometimes product listings specify the hydrate form, but nearly all common references point back to the same volatile liquid. The chemical world has little forgiveness for confusion, so clarity in labeling matters even more where a misstep means real risk.
Safety & Operational Standards
Working with arsenic trifluoride leaves no room for shortcuts or bravado. Personal protective equipment goes beyond the usual lab coat and goggles; full-face respirators, acid-resistant gloves, and heavy-duty fume handling infrastructure form the baseline. Ventilation and emergency protocols receive constant review. Even a minor spill can trash expensive instruments or force a facility stand-down. Medical surveillance programs sometimes monitor people exposed to arsenic compounds, reflecting a sobering awareness of long-term toxicity. A single exposure can leave a mark—skin burns, respiratory inflammation, and, in the worst cases, systemic toxicity that lingers long after the vapor clears.
Application Area
In today’s laboratories, arsenic trifluoride finds its biggest role in fine-tuned chemical synthesis, especially when fluoride chemistry demands subtle, controlled manipulation. Some routes to specialty fluorinated materials lean on it for efficiency. Electronics manufacturing and advanced material science once looked at arsenic trifluoride as a tool, but increasing pressure for safety and environmental stewardship has squeezed its space. Academic interest still drums up new tricks, but tighter rules around environmental emissions and workplace exposure shrink its playground. That’s not to say it has disappeared—specialists in organic and inorganic synthesis still reach for this classic reagent in their most challenging projects.
Research & Development
Long after chemists first bottled arsenic trifluoride, questions about its chemistry keep cropping up. Researchers probe its reactivity, chase after ways to tame its hazards, and look for greener, safer fluorinating alternatives. Computational chemists model its electron cloud to forecast reactivity. Process engineers eye waste stream treatment, aiming to limit its environmental impact. Progress happens, but never fast enough to sweep away hard-earned caution. Some R&D labs try to replace arsenic trifluoride with organofluorides or more complex, buffered systems that offer similar function with reduced risk. Funding leans toward methods that lower exposure, recycle byproducts, and shrink the environmental footprint.
Toxicity Research
The dangers of arsenic compounds have shaped both regulation and research culture for decades. Arsenic trifluoride, with its high volatility and potency, intensifies that focus. Animal studies and epidemiological surveys underline how exposure can lead to multi-system toxicity—liver, kidneys, nerves, and skin all stand at risk. Acute contact can scar lungs or even prove fatal, while chronic low-level exposure links up with cancers and neurological issues. Toxicologists keep peeling back new layers, studying not just the parent compound but also the breakdown products that linger in workplace air or wastewater. Regulatory agencies track air and water levels, enforcing strict cleanup and reporting. These rules stem from grim evidence: overexposed workers in the past suffered everything from peripheral neuropathy to respiratory collapse.
Future Prospects
Environmental regulation and safety demands put arsenic trifluoride in a tough spot. There’s a tug-of-war between its undeniable value in deep-fluorination chemistry and the heavy baggage of toxicity. The search for safer substitutes runs parallel to efforts at miniaturization—using precisely tuned, micro-scale reactions to do more with less material, cutting spill risks and waste alike. Emerging research sometimes points toward encapsulated reagents or automation, keeping human hands as far as possible from the hazard. As green chemistry gathers steam, pressure to avoid volatile arsenic-fueled fluorination grows stronger. Despite shrinking mainstream relevance, arsenic trifluoride won’t vanish soon. Its value in odd corners of research lingers on, serving as both a warning and an inspiration to chemists who know the high price of progress.
What is Arsenic Trifluoride used for?
Understanding Why Arsenic Trifluoride Matters
Arsenic trifluoride isn’t a household name, but for folks working in chemical plants or labs, it’s something they encounter more often than many would expect. With a reputation for being toxic, it often gets overshadowed by that big red warning sign. Still, this sharp, colorless gas plays a role in science and business most don’t realize.
Turning Raw Materials into Useful Chemicals
One of the main jobs of arsenic trifluoride is to help chemists make other chemicals. In factories, workers turn it into organoarsenic compounds, which support everything from pesticides to pharmaceuticals. For a while, certain pesticides and wood preservatives relied on arsenic chemistry. These compounds helped keep crops healthy and wood safe from bugs and decay. Concerns about toxicity pushed many companies toward alternatives, but in some places, these chemicals haven’t fully gone away.
Another big use comes during chemical reactions that need to swap out one atom for another. In my own grad school days, I remember a chemistry professor explaining that fluorides are tricky, but incredibly useful when making more advanced materials. Arsenic trifluoride acts as a fluorinating agent, meaning it helps add a fluorine atom to other substances. That might sound simple, but fluorinated molecules end up in pharmaceuticals, specialty polymers, and even some refrigerants. A single reaction using arsenic trifluoride can make the difference in whether a new drug works better, or a plastic product holds up longer in harsh conditions.
A Cautionary Tale from the Lab
I once spent part of a summer internship shadowing a chemist in an industrial plant. He explained how handling arsenic trifluoride called for serious training. Without strict controls, accidents could leave people or the environment exposed to something truly dangerous. Even a small spill needed immediate attention. Many chemical companies build entire safety plans around storage, transport, and proper ventilation, so the gas doesn’t make its way into the air. Some plants install special scrubbers and containment systems to catch leaks or accidents before anyone gets hurt.
Challenges and Making Things Safer
Toxic chemicals don’t go away on their own, and history is full of stories about companies that once dumped byproducts in nearby rivers or fields. Regulators today put legal pressure on manufacturers to control arsenic emissions. Some countries track every gram, and require regular inspections. Workers often need special licenses or certifications to even approach storage tanks filled with this chemical. Tools like digital leak detection and closed-system processing help cut risks even more. I’ve seen how regular training sessions make a huge difference—when everyone understands the risks, they keep each other alert and careful, pushing for a culture where incidents stay rare.
Moving Toward Cleaner Solutions
The push for greener chemistry has created a search for substitutes that offer the same chemical punch as arsenic trifluoride, without the baggage. Some labs now use other fluorinating agents with lower toxicity, and the industry keeps moving away from arsenic wherever possible. As technology advances, safer and just-as-effective alternatives may eventually replace it, but for now, arsenic trifluoride hangs on in some corners of industry. It’s a reminder that the materials pushing science forward demand respect, serious care, and a willingness to keep learning how to handle them better.
What are the safety precautions when handling Arsenic Trifluoride?
Understanding the Risks
Arsenic trifluoride can fool you. You look at it—colorless and no strong smell—and it doesn’t scream danger. Don’t let appearances trick you. Years ago, I got tasked with preparing a small sample for a reaction in an undergrad lab. Nobody had a casual attitude about it, and for good reason. This stuff packs a punch. The fumes corrode metal and rip into your lungs, burning tissue on contact. No glove would save your skin if it got through.
Suit Up: It Matters More Than You Think
Your average safety goggles and nitrile gloves just don’t cut it here. My old supervisor used to double up on gloves: a thick nitrile layer on the outside, backed by neoprene or butyl underneath. A full face shield—not just goggles—keeps splash danger off your eyes and skin. The right lab coat makes a difference, too. Go for a chemical-resistant material. I always chose one that went past my knees.
Ventilation Rules the Day
If the lab you’re working in doesn’t have a high-quality fume hood, stop right there. There’s no safe shortcut. Years back, someone in our department skipped using a hood late one night, thinking they'd be quick. Two minutes later, they started coughing and felt dizzy. Emergency showers and a trip to the hospital followed. When the hood gets turned on, check the airflow with a simple strip of tissue or flow gauge—never assume it works. Arsenic trifluoride fumes will attack airways and can linger at dangerous levels, even after you’ve cleaned up.
Spills: Fast Reactions Save Health
Spills create a real mess, not just a “grab paper towels” situation. Soda ash or lime neutralizes small amounts on hard surfaces, but these powders need to sit until bubbles stop. Use heavy gloves and a scoop for the cleanup. Don’t ever let liquid go down the drain; arsenic compounds can contaminate water supplies and won’t break down on their own. Every lab session where we handled arsenic, we posted a reminder at the door: “Spill = Evacuate. Call Hazmat”. Unpleasant, but practical.
Storage: Isolation Beats Convenience
Storing arsenic trifluoride with acids or water-based chemicals builds a recipe for disaster. Moisture sets off a violent reaction, so dedicated cabinets kept the bottle surrounded by dry materials like silica gel packs. Labels faced out and containers stayed tightly sealed. We logged every use in a notebook—not because anyone wanted busywork, but because losing track could bring a hidden hazard months down the road.
Waste: Respect Every Drop
Consistent waste management—one of those boring but lifesaving routines. Labeled “Arsenic waste only” containers with sealed lids cut down exposure. Local disposal rules get strict for a reason; arsenic has no business in regular trash or sinks, ever. Years working chemistry taught me that logging out every waste container created accountability. Every ounce went through proper channels, with no exceptions.
Don’t Work Alone, Don’t Skip Training
Chemical safety works better as a team sport. Every time arsenic trifluoride came out, nobody worked solo. Backups make a difference in emergencies. Regular training sessions go beyond just ticking a box. Sharing stories about scrapes and close calls helps make the risks real for anyone handling hazardous materials. A real respect for chemicals like arsenic trifluoride grows not from fear, but from understanding how quickly things can go wrong.
What is the chemical formula and structure of Arsenic Trifluoride?
The Chemical Makeup
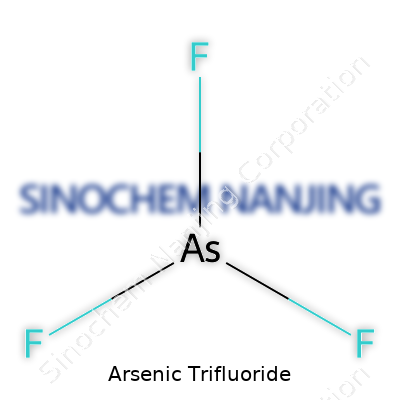
Arsenic trifluoride carries the formula AsF3. The letters look simple, but the way these atoms fit together changes how chemists approach this compound. You get one arsenic atom and three fluorine atoms forming each molecule. This isn't just a string of letters tossed together to fill space on a label—each part matters.
Visualizing the Structure
Picture a flat pyramid shape if you want to imagine arsenic trifluoride at the atomic level. The arsenic atom anchors the base, and three fluorine atoms reach out, forming a trigonal pyramidal geometry. That shape isn't random. It comes from the way electrons push against each other—something called VSEPR theory back in chemistry class. Fluorine, being highly electronegative, pulls hard on shared electrons, distorting the electron cloud around the central arsenic atom.
Why Structure Matters in Daily Practice
If you work in a lab or ever need to handle industrial chemicals, structure means more than a textbook answer. The specific shape of arsenic trifluoride affects how it interacts with water, glass, and even the air you breathe. Exposure to moisture makes it react fiercely, producing toxic hydrogen fluoride gas. I remember a fume hood demonstration going wrong in university—nothing drives home the risks like watching a glass beaker etch and fog up from chemical vapors. Companies dealing with this compound set strict rules and require proper ventilation, not just for compliance, but because structure determines hazard.
Applications and Hazards in the Real World
Arsenic trifluoride rarely shows up in everyday life, but it’s central in some specialized chemical synthesis and organic reactions. People making organoarsenic compounds and fluorinating agents depend on it to transfer fluoride ions. Since the molecule is sharply reactive, workers bring serious respect to the job. The danger isn’t only from arsenic’s toxic reputation—fluorine’s aggression also turns this compound into a chemical hazard.
Mistakes around arsenic trifluoride can lead to burns, poisonings, or environmental contamination. The U.S. National Institute for Occupational Safety and Health (NIOSH) highlights the importance of protective gear. Gloves, full-face masks, and chemical fume hoods aren’t optional. Handling spills or leaks calls for quick, practiced action and solid emergency procedures, because there’s little margin for error with both arsenic and fluoride on the table.
Reducing the Risk: Smarter Practices
An ounce of prevention beats scrambling during a spill. Training new staff on chemical structure isn’t an academic exercise—it’s life or death. Labs put up clear signage and color-coded containers because knowing a compound’s formula translates to respecting its dangers. Waste handling takes extra care to avoid environmental harm. Environmental Protection Agency (EPA) rules forbid casual disposal, pushing for sealed containers and certified waste collectors, which limits runoff and airborne risk.
Better alternatives exist for some applications, and research pushes for less hazardous reagents where possible. While arsenic trifluoride still earns a niche spot in chemistry, modern labs always look twice before bringing it in the door—everybody from the janitor to the PhD needs to know what those three letters, AsF3, actually mean beyond just a formula.
How should Arsenic Trifluoride be stored and transported?
A Look at the Risks Involved
Arsenic trifluoride doesn’t belong anywhere near where people work without precautions. This chemical releases toxic fumes that burn the lungs and corrode skin. Its vapor can inflict lasting health issues with the slightest exposure. Companies and researchers that use arsenic trifluoride dance with a serious hazard every day. A single unsafe container or accidental spill could put entire teams, neighborhoods, or cities at risk.
Trustworthy Containers Matter
Chemists and engineers rarely take risks with something as reactive as arsenic trifluoride. Reliable storage starts with nickel or Monel containers – not glass, which this compound eats straight through, and certainly not plastic, which doesn’t last against its fumes. Steel sounds tough until you see how quickly arsenic trifluoride chews holes through it, so stainless-steel containers with specialized inner linings become the only safe bet.
It’s not enough to use just any nickel drum or cylinder. Containers must form tight seals, because even a pinprick leak in a gasket brings instant danger. Workers double and triple-check for any weakness or rust before a shipment goes anywhere. History comes littered with stories of disastrous leaks and ruined equipment, all because someone looked the other way for a moment or thought they could get by with subpar gear.
Climate and Handling
Dry, cool, well-ventilated storage rooms save lives when it comes to this chemical. Moisture in the air reacts with arsenic trifluoride, generating hydrofluoric acid and releasing arsenic gas. Air conditioning and humidity control aren’t luxuries; they’re strict requirements. Operators keep them in locked cabinets, with only a handful of trained staff holding the keys. Spill containment equipment sits nearby in case anything goes wrong, including neutralizing agents and breathing gear.
No Replacement for Good Training
Hazmat teams rattle off the risks of arsenic trifluoride without missing a beat, because their own training drilled it into them. Every new technician learns about the cloud-white vapor that can blind and choke. Regular drills and refreshers keep everyone sharp. Employees grow into the rituals of donning full personal protective equipment (PPE). Suits, gloves, goggles, and respirators form the daily armor. No one skips steps, even on rushed days.
Training covers more than just handling. Emergency plans play out like clockwork, with every worker knowing escape routes and first aid for chemical burns. Response plans grow out of real incidents, not just textbook formulas. The importance of this hands-on training becomes painfully clear with each near-miss that could have turned tragic if someone hesitated.
Transport Demands Vigilance
Few chemicals demand more attention during transit. Any company moving arsenic trifluoride needs explicit approvals, clear labeling, and paperwork that documents every mile from source to destination. Vehicles must carry up-to-date hazmat placards and proper containment gear, with drivers who carry certification for handling extremely hazardous substances. Forget shortcuts—every law, industry regulation, and good practice gets followed, since any mistake hits hard and leaves little room for recovery.
I’ve seen the industry’s respect for this substance. Regulations may look strict, but the stories behind them tell of burned hands, hospitalized workers, and towns evacuated under a ghostly white cloud. Ordinary people put trust in those professionals to shield everyone from such incidents. Arsenic trifluoride reminds everyone why serious chemicals need serious respect, every single day.
What are the potential health hazards of exposure to Arsenic Trifluoride?
Understanding the Risks
Arsenic Trifluoride isn’t a household name, but that doesn’t let anyone off the hook for ignoring its risks. With a sharp chemical bite, it sends a clear message: handle with care. Exposure to this compound has sent plenty scrambling for better workplace protections, and there’s a good reason for that.
Health Hazards Aren’t Subtle
Breathe it in or let it slip through the skin, the body pays a heavy price. Right after exposure, lungs might feel irritated; coughing, tightness, and shortness of breath follow. Eyes and skin sting. The real trouble starts as the chemical starts to eat away at tissue. It can trigger blisters or burns, and the risk of blindness rises quickly if it splashes into the eyes.
Once inside, arsenic compounds build up in the body. Over time, arsenic links to all sorts of chronic health issues—cancer, nerve damage, and heart disease. The nervous system starts to fail, muscles weaken, and judgment turns fuzzy. These aren’t distant problems lining up for retired chemists; they’re clear and present dangers for anyone cutting corners with safety on the job.
Science Backs Up the Warnings
Facts back up these fears. Agencies like the Occupational Safety and Health Administration (OSHA) and the World Health Organization (WHO) wave big red flags over arsenic exposure, with limits that barely allow for breathing room in industrial settings. Research loads up the data with stories of cancer clusters in communities with contaminated water, and workers facing lung and skin troubles in manufacturing plants handling arsenic-fluoride compounds.
Looking over poison control reports, it becomes clear: symptoms often punch in fast. Nausea strikes, coordination drops, and breathing gets shallow. At its worst, arsenic trifluoride doesn’t just threaten long-term health—it can be lethal if medical help lags behind. I’ve never worked hands-on with it, but talking to a cousin from the chemical industry, she described strict handling procedures that leave little room for mistakes, and for good reason.
Facing the Problem Head-On
Ignoring the hazards isn’t an option for anyone hoping to make it home from work in one piece. Proper personal protective equipment forms the first line of defense: gloves, goggles, full-face shields, and chemical-proof suits. Even the best respirators matter, as just a whiff could send someone straight to the hospital. Well-ventilated areas and strict hygiene rules—clean clothes, handwashing, and sharp separation of work and break spaces—stop this chemical from following people home.
Companies play a part here, with clear protocols and training that actually sinks in. It’s not enough to slap on some warning labels and hope for the best. Workers need to spot symptoms, know who to call, and understand how to clean up without risking their health. For anyone outside a factory’s gated community, clean water supplies and strict industrial oversight shield the public from fallout.
Room for Change
Regulation and education about arsenic trifluoride save lives, plain and simple. Ready access to safety data sheets, hands-on emergency drills, and tough inspection schedules all pile on the protection. The lesson here sounds simple: don’t underestimate what a single chemical can do to a person who isn’t ready to fight back. Science, experience, and some sobering stories have shown over and over—the cost of carelessness with arsenic trifluoride runs high.
| Names | |
| Preferred IUPAC name | Trifluoroarsane |
| Other names |
Arsenic(III) fluoride Arsenous fluoride |
| Pronunciation | /ɑːrˈsiː.nɪk traɪˈfluːəˌraɪd/ |
| Identifiers | |
| CAS Number | 7784-12-3 |
| Beilstein Reference | 358715 |
| ChEBI | CHEBI:30169 |
| ChEMBL | CHEMBL1231102 |
| ChemSpider | 12142 |
| DrugBank | DB11599 |
| ECHA InfoCard | 100.029.668 |
| EC Number | 232-066-3 |
| Gmelin Reference | 837 |
| KEGG | C07369 |
| MeSH | D001151 |
| PubChem CID | 24597 |
| RTECS number | CG0875000 |
| UNII | 9U6A2R3D2G |
| UN number | UN1557 |
| Properties | |
| Chemical formula | AsF3 |
| Molar mass | 139.9204 g/mol |
| Appearance | Colorless fuming liquid |
| Odor | Pungent |
| Density | 3.9 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.78 |
| Vapor pressure | 128 mmHg (at 25 °C) |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | 1.94 |
| Magnetic susceptibility (χ) | −49.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.344 |
| Viscosity | 1.68 mPa·s (at 20 °C) |
| Dipole moment | 1.84 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 256.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1012 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1371 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB54 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, or skin absorption; causes severe burns; reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H311: Toxic in contact with skin. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P234, P260, P262, P264, P270, P271, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P314, P363, P370+P378, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-Ac |
| Autoignition temperature | 620°C |
| Lethal dose or concentration | LD50 oral rat 20 mg/kg |
| LD50 (median dose) | 65 mg/kg (oral, rat) |
| NIOSH | RN0082 |
| PEL (Permissible) | PEL = "0.05 mg/m3 |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Arsenic trifluoride oxide Arsenic pentafluoride Antimony trifluoride Arsenic trichloride |

