Arsenic Trichloride: Old Roots, Modern Dilemmas
Historical Development: A Long Shadow in Chemistry
Arsenic trichloride has a history tangled in both innovation and infamy. Chemists started experimenting with various arsenic compounds back before modern laboratories existed, searching for useful properties without always knowing what risks lay ahead. In the 19th century, arsenic trichloride started showing up in industrial processes as chemists leveraged its reactivity, especially for synthesizing organoarsenic chemicals. Through both war and peace, the world kept rediscovering arsenic’s double-edged sword: a potent tool for science, a hidden menace for health. Military use crept into the story during the First World War, when arsenic compounds found their way into chemical weapons. These darker chapters remind us that progress often walks hand-in-hand with ethical questions.
Product Overview and Naming Confusion
Arsenic trichloride, also called arsenous trichloride or arsenic chloride, comes up in conversations about chemical synthesis and industrial research. You’ll see it referenced as AsCl3, and sometimes called butter of arsenic because of its oily, faintly yellow appearance. Its identity shifts depending on the context: a basic lab reagent, a precursor for pesticides, a chemical for etching glass. The multiple synonyms cause confusion, particularly for non-specialists faced with a list of chemical names that all nod toward the same corrosive, volatile liquid.
Physical and Chemical Character
A bottle of arsenic trichloride doesn’t look intimidating—just an oily, colorless to pale yellow liquid with a whiff of garlic or burning matches if you open it. That odor comes from its ability to hydrolyze in humid air, releasing both arsenic oxides and hydrogen chloride. Boiling starts around 130°C, and it weighs in at about 2.16 grams per cubic centimeter. The chemical doesn’t just burn; it reacts quickly with water or alcohol, producing heat and toxic fumes. Where some liquids evaporate quietly, arsenic trichloride fights back, eating through flesh and lung tissue with astonishing speed. This isn’t the sort of material a novice would try to handle at home.
Technical Specifications & Labeling: No Room for Error
Handling arsenic trichloride in any real-world facility means dealing with requirements that go beyond curiosity—strict labeling, secure packaging, no open handling allowed. Labels must warn about its acute toxicity, show proper hazard pictograms, and point to rigorous workplace exposure limits. Regions following GHS guidelines call out its corrosive and toxic nature, demanding workers suit up with gloves, goggles, and chemical aprons before they even approach the bottle. The European Union, the US EPA, and others classify arsenic trichloride as a substance requiring permits, inventory, and systems to prevent accidental release. An ounce of carelessness invites disaster.
Preparation: From Elementary Reactions to Laboratory Inspections
Making arsenic trichloride mostly involves reacting arsenic trioxide (As2O3) or pure arsenic metal with concentrated hydrochloric acid or chlorine gas. In small quantities, the process looks straightforward: add arsenic trioxide to hydrochloric acid under cooled, controlled conditions. Bubbling chlorine through molten arsenic trioxide also does the job, though it’s definitely not a kitchen-table project. As simple as the steps sound, the risks pile up fast, ranging from skin-burning splashes to clouds of poisonous vapor. Modern labs now focus on closed systems, exhausting vapors into scrubbers designed for absolute containment. Oversight from safety officers ensures protocols fit the danger.
Chemical Reactions & Modifications: Harnessing Volatility
The main draw of arsenic trichloride as a reagent is its strong affinity for forming bonds with other halides, metals, and even certain organic groups. It sits at the starting line for organoarsenic chemistry, helping create everything from pesticides to specialized semiconductors. Chemists use it to convert alcohols or amines into arsenic-containing analogs, many of which proved (sometimes tragically) useful in agriculture and medicine before their hazards became public knowledge. Even now, the ability to swap the chloride atoms with other functional groups opens up research possibilities, although restrictions limit the scope, given what science knows about the consequences.
Application Area: Progress and Pitfalls
Industrial history books say a lot about the role of arsenic trichloride in synthesizing organoarsenic compounds for herbicides and insecticides. I remember reading about its use in cotton defoliants or rat poison, back before stricter controls chased it out of broad commercial circulation. Some chemists keep exploiting its reactivity for etching glass and producing certain ceramics. Its importance in making semiconductors and specialized dyes continues in research labs, though concerns about waste disposal now overshadow these conversations. Each bottle in active use today signals a conflict between scientific innovation and regulatory oversight. The hazards outweigh most potential consumer applications, so use stays buried deep inside controlled environments.
Safety & Operational Standards: Keeping the Risks in Check
Experience working inside regulated chemical facilities taught me that, with arsenic compounds, routine becomes the enemy—complacency breeds danger. Tight controls around ventilation, sealed storage, and spill containment exist for a reason. Anyone interacting with arsenic trichloride has to understand what’s really at stake. The compound isn’t just corrosive; it causes multi-organ failure at low doses, whether through inhalation, skin absorption, or ingestion. Spill drills, fume hood use, and medical monitoring protocols shape every interaction. Waste management becomes a major challenge, given how quickly residues contaminate surfaces and drainage systems. Safety data sheets and refresher training stay mandatory, because a single shortcut could take out more than a career.
Research & Development: Progress with Caution
Laboratories chase new ways to harness arsenic trichloride’s reactivity, especially for synthesizing molecules not easily assembled from benign starting materials. Sometimes researchers try to tweak its chemistry, searching for less toxic intermediates or more selective reactions, all the while keeping regulatory compliance in sight. The focus in academic labs steers toward controlled micro-reactions and tight material tracking, so the shadow of past arsenic disasters still directs scientific priorities. Funding bodies demand risk assessments and disposal plans up front before funding any study that involves arsenic trichloride. While its central role in certain catalytic or etching reactions has not disappeared, demand for safer alternatives constantly drives innovation, though finding true replacements proves tough.
Toxicity Research: The Evidence is Overwhelming
Skepticism over chemical safety comes from decades wrapped in case reports, autopsies, and clinical data. Researchers found that arsenic trichloride can cause acute respiratory failure within hours of exposure, with chronic effects ranging from skin lesions and nerve damage to outright cancer. No known exposure route is safe, and no easy antidote exists. Long-term animal studies confirmed DNA damage and reproductive toxicity, which reinforced decisions to phase out many uses. Major public health agencies started recording and responding to these risks in earnest during the 20th century, tailoring policies to minimize occupational exposure. These days, any lab or factory choosing arsenic trichloride signs up for a burden of responsibility most industries would rather avoid.
Future Prospects: Hard Choices Ahead
New regulatory demands, consumer awareness, and stricter environmental controls all squeeze the options for arsenic trichloride’s future. The days of large-scale agricultural use now belong to history lessons. Chemical manufacturers now work on methods to reclaim or destroy arsenic waste before it leaves the plant. Some hope lingers for safer derivatives, although the periodic table’s chemistry puts limits on how gentle any arsenic process could become. Attention tilts toward digital record-keeping, real-time monitoring, and engineered containment that keeps harmful vapors behind layers of glass, steel, and specialized filters. As new professions grow up around sustainable chemistry and green engineering, the lesson arsenic trichloride offers is clear: the badge of progress carries a responsibility nobody can afford to slack on, especially when human lives and environmental health hang in the balance.
What is Arsenic Trichloride used for?
Real Uses in Chemistry and Industry
Arsenic trichloride shows up most often in laboratories and not so much in everyday life. Many chemists know the sharp, stinging odor when cracking open a bottle in a fume hood. Workers use it to make organoarsenic compounds, which end up in pesticides, pharmaceuticals, and even semiconductor manufacturing. It’s not a product people see on the shelves, but it quietly shapes modern industries behind the scenes.
Handling Risks Others Might Overlook
Most folks don’t think much about arsenic trichloride unless working in a lab or a plant. Those who do use it need to keep safety front and center. This stuff releases fumes that burn eyes, skin, and the lungs. Long-term exposure cranks up the risk for some forms of cancer, and spills don’t wash away like table salt. People who’ve worked with it often talk about the anxiety around possible leaks. Even with gloves, goggles, and that ugly chemical apron, there’s always a little worry that something could go wrong.
Driving Pharmaceutical Chemistry
The pharmaceutical world leans on arsenic trichloride to make compounds otherwise tough to prepare. My years moving between labs made it clear: sometimes the ugliest chemicals play a key role in cancer drugs and other treatments. Back in the early 2000s, discussions about making small quantities for research always included an extra layer of safety briefings. The benefit—sometimes a life-saving drug—came after a real risk assessment. Regulatory teams wanted heavy documentation before a single gram got shipped out.
Making Pesticides: A Long, Complicated Story
Long before most modern regulations, arsenic compounds powered farm chemicals across the globe. While some countries still use these formulations, the world moves steadily toward safer options. The push for change grew from new data showing persistent environmental and health risks. Living in a small rural town, I’ve seen worries pile up as testing revealed arsenic in local soil—a slow legacy of outdated products. Whole communities now deal with environmental cleanup, often paid for by taxpayers or through lawsuits.
Semiconductors and Tech Manufacturing
Arsenic trichloride pops up in tech manufacturing, sometimes to dope silicon with precise amounts of arsenic. The drive for faster and more efficient microchips depends on chemicals that push the limits. Working with folks in cleanrooms, I learned that companies spend huge sums to keep things safe—a big investment in local exhaust ventilation, spill control, and respiratory protection. Without strong government oversight, accidents could run up costs and cause real harm.
Solutions and a Safer Future
It’s time for tighter rules and better training. Government agencies need to push for alternatives, funding research into greener chemistry. Companies have to give workers solid information about the dangers, not just a pamphlet in a locker. I remember my own awkward questions on orientation day getting brushed aside—those conversations matter. Neighborhoods near chemical plants need regular updates and honest dialogue about what’s stored and why.
Science pushes forward, but vigilance keeps it from rolling over the people in the way. If we all insist on responsible handling and open discussion, arsenic trichloride’s legacy could end up as a tool for progress instead of one more industrial nightmare.
What are the safety precautions when handling Arsenic Trichloride?
Why Respect in Chemistry Matters
Anyone who’s spent time with hazardous substances knows that brushing off safety can turn a normal day into a trip to the hospital. Arsenic trichloride is no exception. The stuff deserves respect—pure and simple. It gives off fumes sharper than a punch in the nose, and it doesn’t play nicely with skin or lungs. Stuff like this isn’t some end-of-shelf cleaner; it’s a chemical that can do real, serious harm without much warning.
Keeping Exposure at Bay
I remember the first time I handled anything remotely as nasty; you learn real fast that breathing anything weird in a lab leaves your chest tight for days. Arsenic trichloride drifts as a gas almost at room temperature, and it reacts with water instantly, making hydrochloric and arsenous acids. That means even the humidity in the air is enough to stir up trouble.
Get yourself into a good, high-quality fume hood every single time. I’ve seen people trust in open windows or the idea that “just a minute” won’t matter, but it’s not worth the risk. Proper air control with a hood closed and functional saves more than your job—it can save your life.
Gloves don’t mean any standard rubber works. Nitrile and heavy-duty neoprene stand up better to the liquid, and even then, you want to change out gloves after every session and wash thoroughly. Goggles should wrap well at the sides. Forget about regular glasses; a splash could mean blindness or worse. Face shields offer an extra bit of comfort, especially when pouring or moving stocks.
Smart Storage and Clean-Up
Chemicals like this can break down plastic or glass, so check labels twice and stick with containers made of compatible materials. Label everything, and don’t trust it to memory. More than once, sloppy storage has led to panicked lockdowns and uncertain hospital stops. Fires and explosions happen when arsenic trichloride meets water or bases, so store away from sinks, humidifiers, or even cleaning stations with a mop bucket left nearby.
Spills bring out the worst. If this stuff hits a bench, a tiny amount of moisture can send up clouds. Sand or spill control agents work better than rags or paper towels, which just scatter droplets. After cleaning, treat all materials as toxic waste, and never toss them in a regular garbage bin. Waste that can leak or react in a dumpster has sent city workers into emergencies before.
Training and Emergency Fit
No amount of equipment matters if you don’t know what to do in a pinch. Training should go past the basics. Go through drills—real ones—not just day-one instruction. Make sure emergency showers work and eyewash stations aren’t blocked by cardboard boxes or old supplies. The buddy system, low-tech as it might sound, still saves lives. Keep a spill kit within easy reach, stocked and checked at least monthly.
The Facts Back It Up
The CDC and OSHA set limits for good reasons. Breathing in arsenic dust or fumes damages lungs, nerves, even kidneys. Arsenic exposure sits high on the list for cancer risks, according to the World Health Organization. Inhaling it, even in small doses over time, has led to fatal poisoning cases across industries—no scare tactics, just reality.
Culture Makes the Difference
At the end of the day, safety isn’t just about gear or warning labels. Labs where people speak up and double-check each other run fewer risks. The stubborn pride of skipping steps puts everyone at risk. Routine and a bit of humility work better than bravado every time.
Smart handling, respect, and a sense of responsibility go further than any instruction sheet. Arsenic trichloride holds no second chances.
What is the chemical formula and molecular weight of Arsenic Trichloride?
Chemistry at Work: The Formula of Arsenic Trichloride
Looking at a basic chemistry class, plenty of folks run into compounds with long names and even more puzzling formulas. Arsenic trichloride comes up on that list pretty quickly, especially if you’ve ever cracked open an old bottle or read through industrial safety guides. Its formula is AsCl3. At its core, the molecule sports one arsenic atom matched up with three chlorine atoms. That’s not just a jumble of letters to memorize – it’s a snapshot of how this compound fits together at the atomic level.
Getting the Numbers Right: Molecular Weight
Chemists and people who work with raw materials often need more than just the names and formulas. They want to know exactly how much a batch of something weighs, down to a tenth of a gram. Each molecule of arsenic trichloride weighs about 181.28 grams per mole. That number comes from adding up arsenic’s atomic weight, which is about 74.92, and three times chlorine’s atomic weight, clocking in at 35.45. Put those together, and each mole of AsCl3 tips the scale at just above 181 grams.
Why These Numbers Aren’t Just for Textbooks
Some might ask why the chemical formula and molecular weight matter if you’re not holed up in a lab. Here’s the truth: these facts shape actual work decisions. Factories using arsenic trichloride in the synthesis of organoarsenic compounds can’t fudge those numbers and still keep people safe. Accidents don’t care about intentions. Knowing the exact formula means you can guard against unexpected reactions, and pinning down the weight gives technicians the power to mix the right doses without guesswork.
My first chemistry job put these details into sharper focus. We prepped lab supplies using a checklist, double-checking compounds like arsenic trichloride because the risks weren’t just theoretical. Arsenic compounds can set off everything from chemical burns to toxic fumes. Dragging the wrong amount across the workbench might tip a small spill into a full-blown hazard. Every bit of data on AsCl3, from formula to molar mass, keeps that chain of mistakes from starting.
Risks and Responsibility
The dangers arsenic trichloride brings can’t be brushed aside. Its notorious toxicity has claimed more than a handful of headlines over the years, especially when spills or leaks hit local news. Each time people cut corners, someone else may end up breathing those chlorinated vapors. In many labs, the process grinds to a halt without double-checking the numbers and the paperwork before opening the valve.
Safety training, transparent labeling, and routine weighing keep mistakes at bay. There’s no room for carelessness – I’ve watched new hires freeze when the instructor explains that a splash from the wrong bottle means a ride to the hospital. Compounds like arsenic trichloride demand that same level of respect every time. Fact-based decisions – rooted in chemistry’s basics – protect both the work and the people in it.
Building Better Habits and Safer Spaces
Clear facts like “AsCl3 means arsenic plus three chlorines” and the number 181.28 underpin real-life lab safety and production efficiency. With dangerous chemicals, there’s no luxury of learning from hindsight. Relying on trusted scientific data and steady work habits doesn’t remove the risks, but it does keep them from turning into disasters. These aren’t just numbers for the sake of memorization; they help ensure you, your coworkers, and anyone downwind goes home safe each day.
How should Arsenic Trichloride be stored?
Why Handling This Chemical Isn’t Just for the Lab Pros
Anyone who’s ever stepped into a chemical store room knows how fast things can go wrong with the wrong setup. Arsenic trichloride stands out among the nastier compounds, mainly because it releases toxic fumes in humid environments and reacts so aggressively with water. Years ago, I watched an experienced chemist flinch as a single drop hit a damp glove, turning the air sharp and acrid. Every memory since then reminds me that the real risks lie in the details, not just the big rules posted on a door.
Straightforward Rules That Keep Hazards in Check
Storing arsenic trichloride safely starts with controlling moisture. This chemical breaks down in water, freeing up deadly hydrochloric acid and arsenic compounds. Dry, well-ventilated spaces make the difference between routine safety and dangerous accidents. You wouldn’t trust cardboard boxes or cabinets with leaky seals here. Choose airtight glass containers with tightly fitting Teflon-lined caps, because metals or loose plastic never hold up. I once saw an old rusty tin try to contain this chemical—within weeks, corrosion cut through, leaving a mess nobody wanted to clean.
Location Matters More Than Labels Suggest
I’ve known folks who figured a “hazardous” shelf would do, but not all flammable cabinets offer the right shields. Arsenic trichloride asks for a cool, dry, clearly labeled cabinet—preferably not stored near acids, water, or bases. Strong shelving and trays underneath prevent drips from spreading. A locked cabinet with secondary containment means less worry if that glass cracks by accident. Hazard symbols matter, but clear signage about toxic gases, asphyxiation, and corrosives stops mistakes before they start.
Don’t Rely on Just the Basics: Layers of Precaution
Chemical goggles, face shields, and thick gloves aren’t optional—ask anyone who’s gotten a whiff with just a dust mask on. Spill kits and emergency showers always need to be within arm’s reach. Proper ventilation never means using a domestic fan. Only professionally installed fume hoods vent away the fumes and lower the risk of inhalation. Emergency protocols should be clear so anyone, not just the most experienced tech, knows whom to call and what to do in case of exposure.
The Responsibility Goes Beyond the Workplace
Anyone handling arsenic trichloride holds a real trust: it’s not just about personal safety, but the well-being of everyone sharing the same roof. Regular inspections by trained eyes pick up cracks, leaks, and label fade before someone else pays the price. Regulatory agencies like OSHA and the EPA set standards for a reason, and records must stay updated—auditors don’t just look for paperwork, they check that proper respect is given to substances that can seriously injure or kill.
Pushing for Improvements in Everyday Practice
Safe storage isn’t a box-ticking chore. It takes teamwork and a willingness to question old habits. Mixing up training methods now and then wakes people up to risks they might miss out of routine. Asking for third-party safety audits brings fresh perspective. There’s no shame in admitting uncertainty—better to ask and store it right than regret a shortcut for the rest of your career. Real safety around arsenic trichloride starts with clear heads and the discipline to never assume yesterday’s safe setup will still be safe tomorrow.
What are the health hazards associated with Arsenic Trichloride exposure?
Everyday Hazards in Chemical Handling
Arsenic trichloride doesn’t get much press compared to asbestos or lead, but its risks are real. The minute this compound hits the air, it releases fumes that sting the throat and attack the eyes. Some of us have felt the way chlorine gas bites at the sinuses—imagine that with toxic arsenic mixed in. Chemists and factory workers in glass, electronics or pesticide plants bump up against this stuff in ways most of us never will.
Direct Exposure—What Actually Happens
A spill doesn’t just clear a lab. Liquified arsenic trichloride soaks in fast. Skin contact brings out blistering and redness on the hands and wrists before the clock hits five minutes. Inhalation goes deeper. Lungs burn, breathing feels tight, and within hours, headaches and dizziness settle in. This isn’t just a matter of comfort or discomfort—prolonged exposure means more arsenic in your organs, which can crank up the risk of liver and kidney dysfunction years down the road. High exposures sometimes show up as black “melanosis” spots on the skin, or white lines striating the fingernails. Medical researchers have linked workplace accidents with long-term toxin absorption, crawling into everyday life beyond the walls of the jobsite.
Swallowing, Even Small Amounts, Turns Dangerous
Ingesting arsenic trichloride—accidentally or otherwise—is another story altogether. Symptoms read like a worst-case list: vomiting, diarrhea, bursts of stomach pain followed by muscle cramps. These signals come loud and fast, sometimes within the hour. Without urgent treatment, arsenic circulates through the bloodstream, attacking nerves and shutting down basic organ function. Rates of neurological damage spike if help arrives late. In some regions still catching up to modern regulatory standards, contaminated water and improper chemical storage caused real deaths—especially in children.
Lessons from Lab Practice and Industry
Even with modern safety protocols, arsenic trichloride slips past basic gloves and masks. The vapors, heavier than air, can gather in low places where ventilation barely reaches. Anyone handling the material wears a full-face respirator, chemical splash goggles, and long-sleeved suits—not just as a rule, but because cutting corners leaves people sick. A friend working in an electronics plant mentioned how any spill immediately triggered alarms and stopped production for hours, all out of respect for what just a small amount could do.
Why Prevention and Education Matter
Basic facts shape better choices. The U.S. Occupational Safety and Health Administration (OSHA) caps workplace exposure at 0.01 mg/m3 over an eight-hour shift, knowing higher air concentrations have catastrophic effects. Hospitals keep chelation drugs like dimercaprol on hand, which can scrape some arsenic out of the blood if given early, but this approach only works after the fact. Shrinking risk depends on strong training, quick detection equipment, and solid reporting—all steps requiring buy-in from the ground floor up. In places without robust safety programs, community advocacy fills the gaps, raising awareness about what to look for and pressing companies to act fast after an incident.
Real Changes Start with Information
Decades back, stories from towns living downwind of chemical plants spurred action by workers and families. Easy-to-understand information, regular health checkups, and personal protective gear stack the odds in favor of safety. Shortcuts or unclear rules don’t just break regulations—they put lives on the line. Teach workers what arsenic trichloride does, outfit them right, double-check emergency plans, and communities see numbers trend positive. It comes down to digging past the formula on the label and pushing for fixes that outlast any one shift cycle.
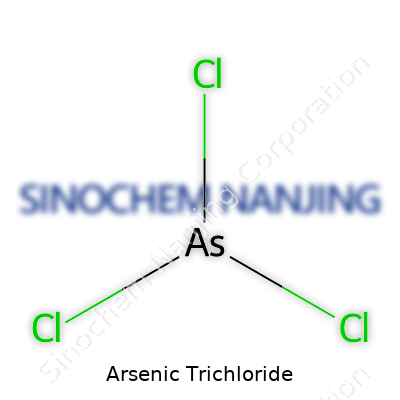
| Names | |
| Preferred IUPAC name | Trichloroarsane |
| Other names |
Arsenious chloride Arsenic(III) chloride Arsenic chloride Butter of arsenic |
| Pronunciation | /ˈɑː.sɪ.nɪk traɪˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 7784-34-1 |
| 3D model (JSmol) | `JSmol.loadInline('data:text/plain,C3\nAs 0.0000 0.0000 0.0000\nCl 1.9578 0.0000 0.0000\nCl -0.9789 1.6955 0.0000\nCl -0.9789 -1.6955 0.0000\n')` |
| Beilstein Reference | 358675 |
| ChEBI | CHEBI:30160 |
| ChEMBL | CHEMBL1230982 |
| ChemSpider | 23622 |
| DrugBank | DB11542 |
| ECHA InfoCard | 100.004.841 |
| EC Number | 231-858-9 |
| Gmelin Reference | 822 |
| KEGG | C01772 |
| MeSH | D001151 |
| PubChem CID | 24247 |
| RTECS number | CG0450000 |
| UNII | 6F5R77OQ39 |
| UN number | UN1558 |
| CompTox Dashboard (EPA) | DTXSID3020702 |
| Properties | |
| Chemical formula | AsCl3 |
| Molar mass | 181.28 g/mol |
| Appearance | Colorless, fuming liquid |
| Odor | Pungent |
| Density | 2.16 g/cm³ |
| Solubility in water | Decomposes |
| log P | 2.1 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 1.6 |
| Basicity (pKb) | 2.17 |
| Magnetic susceptibility (χ) | −53.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.545 |
| Viscosity | Liquid |
| Dipole moment | 1.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 328.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -305.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -581.2 kJ/mol |
| Pharmacology | |
| ATC code | V10BX01 |
| Hazards | |
| Main hazards | Highly toxic by inhalation, ingestion, or skin absorption; causes severe burns; emits highly irritating fumes; reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. H350: May cause cancer. |
| Precautionary statements | P260, P262, P264, P270, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P311, P312, P320, P330, P361, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-Acidos |
| Autoignition temperature | 200°C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LDLo oral human 93 mg/kg |
| LD50 (median dose) | LC50 inhalation rat 1,100 mg/m3 (1h) |
| NIOSH | T0155 |
| PEL (Permissible) | 0.01 ppm (as As) |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Arsenic trioxide Arsine Antimony trichloride Phosphorus trichloride |

