Antimony Trifluoride: From History to Future Prospects
The Early Story of Antimony Trifluoride
Antimony trifluoride didn’t just appear on the modern chemical stage. Centuries ago, chemists in Europe noticed its unique ability to react with other elements. In older texts, antimony itself sometimes got grouped with magical substances, but people soon discovered real, practical uses for its compounds. Two or three hundred years back, antimony salts began helping textile makers and ceramic workers get more consistent results. In this mix, antimony trifluoride stood out for its role in fluorination—before the modern era of finely controlled industrial chemistry made that routine. Back in the early 20th century, antimony trifluoride earned a reputation in the fledgling plastics and fluorochemical industries. Its long track record shows how older discoveries keep shaping modern science.
A Look at What Makes Antimony Trifluoride Stand Out
A lot of people only approach chemistry at the periodic table level, but understanding why antimony trifluoride matters means looking at the stuff up close. This substance forms as a fine white powder or sometimes as colorless crystals, faintly sweet-smelling and highly soluble in water. Its melting point, somewhere around 292°C, marks it as a stable solid by everyday standards, but it reacts strongly with moisture, producing corrosive hydrofluoric acid gas. Set its formula as SbF₃, and you see three fluorine atoms per antimony atom—a simple ratio that’s served millions of production tons over the decades. Its ability to exchange fluoride ions with organic or inorganic compounds places it at the center of several foundational reactions.
Inside the Lab: Technical Specs and Proper Labeling
Every serious chemist keeps one eye on technical standards. Antimony trifluoride typically reaches more than 99 percent purity for industrial-grade batches, with limits set for trace impurities like lead, arsenic, and sulfates. Container labels reflect these standards, stating content, lot numbers, and key hazards. Transporting SbF₃ calls for special containers because it corrodes most metals except lead and a few plastics. Lab workers recognize it from yellow hazard diamonds, wearing gloves and goggles every time they open a jar. Detailed specs matter less to the general public, but knowing this stuff can’t get shipped or handled free of oversight underscores how tightly modern society regulates its chemical backbone.
How It’s Made: Modern and Old-School Paths
Factories have settled on a few consistent ways to make antimony trifluoride. Most commonly, antimony trioxide or pure metallic antimony reacts with hydrofluoric acid gas. This pathway produces a wet paste, easily refined into the pure product by distillation or crystallization. In earlier days, chemists sometimes turned to dry distillation from certain minerals, but those routes gave wildly inconsistent yields. Using hydrofluoric acid started as a lab curiosity and grew into standard practice because it gave a cleaner, more predictable final product. Today, strict environmental controls surround this step, thanks to the toxicity and volatility of HF gas—historically, workers without proper safeguards paid a steep price.
Chemical Reactions: Key Transformations and Tweaks
Antimony trifluoride has a reputation as a reliable fluorinating agent. It swaps its fluoride ions into organic molecules, making it vital for producing specialty fluorocarbons. Chemists learned early that heating it with other halides or strong acids results in a frenzy of bonding changes, with SbF₃ often ending up as the source of highly reactive intermediates. The “Swarts reaction”—named after a 19th-century Belgian chemist—still gives industry a main route to fluorinated hydrocarbons. This one chemical has helped build the world’s stockpile of refrigerants, propellants, and even some pharmaceuticals. Plenty of research also looks into tweaking SbF₃ with modifiers or stabilizers to control its power, stretching its utility without causing runaway side reactions.
Synonyms & Product Names: What’s in a Name?
The world of chemicals overflows with nicknames and alternate labels—antimony trifluoride keeps company with “Swarts reagent,” “antimony(III) fluoride,” and sometimes simply shorthand “SbF3.” In industry catalogs, you’ll see all these. University researchers call it what’s familiar, but anyone buying or selling always checks for the right Chemical Abstracts Service number to track it across regions and uses. This nomenclature jungle can confuse newcomers, but the underlying chemistry remains as constant as ever.
Staying Safe: Handling Standards and Workplace Realities
Anyone working with antimony trifluoride needs nerves, reliable gloves, and well-ventilated labs. Exposure risks include skin burns and poisonous effects on organs—prolonged or careless handling brings on symptoms from irritation to systemic toxicity. National workplace standards require face shields, long sleeves, and emergency showers nearby. Workshops install exhaust hoods and monitor the air for HF vapor whenever SbF₃ processing goes on. Spills get contained with dry agents—never with water since dangerous hydrofluoric acid forms on contact. Proper training and strong personal discipline go a long way in keeping accidents rare. Environmental agencies also monitor disposal closely because antimony and fluorides both leave a long fingerprint in soil and water.
Where It Goes: Key Application Areas
SbF₃ has carved out a vital spot in several major industries. Fluorinated refrigerant production relies on it to build the backbone of modern air conditioning systems. Workers in the plastics sector use it to help create Teflon and other advanced polymers, shifting from the lab to the factory floor and back again. Ceramic and glass workers lean on antimony compounds for special colors and properties, and specialty metalwork sometimes borrows its reactive edge. Over the years, research triangles and industrial zones worldwide have called on SbF₃ to keep important processes flowing, making it a behind-the-scenes powerhouse in the built environment.
Inside the Lab: A Push for Research and New Breakthroughs
Antimony trifluoride research stretches far past its original applications. Universities and laboratories keep probing for new ways to push more fluorine into carbon frameworks, aiming for new drugs, ag chemicals, or advanced materials. Research teams have tested catalysts and additives that might lower the temperature or energy SbF₃ needs to work, chasing both cleaner chemistry and bigger yields. Some groups even look at recycling the spent reagent, closing the loop in high-value manufacturing. This slow, steady stream of research keeps pushing the old chemical into new realms, with each decade adding fresh chapters to an already colorful record.
The Hard Truths: Toxicity Research Stacks Up
Most old industrial chemicals come with a trail of cautionary tales, and antimony trifluoride is no exception. Inhalation, ingestion, or skin absorption can all bring risks—from acute burns right up to long-term systemic damage. Chronic exposure raises the odds for respiratory trouble, organ disruption, and in some cases, cancer. Public health agencies watch these numbers closely, especially in places with a long legacy of chemical waste sites. Modern research digs into the fine points of how SbF₃ and its contaminants move inside the body and the environment. Improved monitoring and better protective measures have lowered direct danger scores in the workplace, but medical studies keep reminding everyone that even trace exposure deserves respect.
Future Prospects: Where to Go Next
Looking ahead, SbF₃ faces a crossroads. Global industry still asks for reliable, affordable fluorinating power, especially as new refrigerants and specialty plastics phase out older, ozone-depleting options. At the same time, pressure mounts for safer, greener alternatives—scientists chase ways to build the same molecules without leaning on legacy toxins. Digital laboratories run simulations to map every bond, every reaction, in search of more sustainable paths. Meanwhile, stricter regulations march in, asking for zero-leak setups and full lifecycle reporting. The future may bring new fluorination agents, but there’s no quick route to replace a tool with so much history and entrenched knowledge. For now, SbF₃ stays on the frontline—with its long record and new environmental guidelines pulling it in new directions. The story of antimony trifluoride gives a snapshot of how science balances progress, safety, and adaptation, no matter how old the chemistry behind it.
What is Antimony Trifluoride used for?
What is Antimony Trifluoride?
Antimony trifluoride is a white, sometimes slightly grayish chemical powder with the formula SbF3. Most people never hear about it, but it plays a hidden role in shaping daily life. At its core, it comes from the element antimony pulled from ores, then processed with hydrofluoric acid to form this potent compound.
Creating Everyday Compounds
One of the biggest jobs for antimony trifluoride unfolds in the production of fluorinated organic compounds. Look at Teflon frying pans or the waterproof jackets in your closet – both rely heavily on fluorocarbons. Antimony trifluoride helps swap in fluorine atoms through a process called the Swarts reaction. Without this tool, we wouldn’t have non-stick cookware or all those handy stain-resistant fabrics. According to a study in the Journal of Fluorine Chemistry, industries manufacture several thousand tons of these chemicals annually, and nearly all depend on antimony trifluoride as the catalyst.
Boosting Everyday Materials
Manufacturers turn to antimony trifluoride for other industrial jobs, too. It plays a role in glassmaking, helping technicians polish glass to a clear, smooth shine. Some specialty glasses, especially those used in electronics or optics, gain extra durability and transparency with a little help from this substance. Even though most folks never see it on a label, it makes its mark on the final product.
Not All Shiny – Health and Environmental Risks
Everything comes with trade-offs. Antimony trifluoride’s sharp edge burns through more than just chemical bonds. It’s toxic if inhaled or swallowed, and contact with bare skin can cause nasty burns. Workers in chemical plants must take strong precautions, with gloves, goggles, and full respirators. Just a little dust in the air can cause coughing, shortness of breath, and long-term lung problems.
Waste control raises concern too. Improper handling or disposal sends this chemical into soil and water. Once in the environment, both antimony and fluoride ions can harm plant life, aquatic animals, and even people who depend on local water sources. The U.S. Environmental Protection Agency lists antimony compounds as a priority pollutant, tracking industrial releases and pushing for safer alternatives.
Searching for Better Paths Forward
Some companies look for substitutes, hoping to rely less on antimony. Safer fluorination agents and newer synthetic techniques inch forward in research labs. These alternatives lower the danger for workers and the planet but tend to cost more or require big changes to equipment. Until something better comes along, most manufacturers keep antimony trifluoride on the roster.
Another way to shrink risk: tougher rules for handling and disposal. Modern chemical plants use sealed equipment, scrubbers, and advanced filtration to keep fumes and waste out of the environment. Strong workplace training—coupled with regular monitoring—reduces exposure and keeps accidents rare. Public pressure and environmental watchdogs push companies to stay transparent about their chemical use and waste streams.
Looking Ahead
The story of antimony trifluoride doesn’t grab front pages, but it matters wherever chemistry opens new doors. It connects the high-tech world in our hands with the safety of the people and places behind the scenes. Knowing how these invisible chemicals shape our world—and what risks they bring—helps guide smarter choices for everyone.
Is Antimony Trifluoride hazardous or toxic?
Everyday Exposure, Long-term Risks
Antimony trifluoride lands on few people’s radar, but those who work with chemicals know its sharp sting. In the lab, this compound sits among the workhorses, turning up in fluorination reactions and helping produce other chemicals. Yet even some seasoned hands prefer to steer clear of it, and for good reason—its reputation carries plenty of weight.
Breathing in its dust or fumes delivers a harsh reminder of why safety goggles and gloves matter. Antimony trifluoride doesn’t settle quietly in the background; it irritates eyes, lungs, and skin within minutes. A few moments of contact might not sound dramatic at first, but years of exposure build up problems researchers and factory workers can’t ignore.
Science Behind the Concern
The toxic effects of antimony compounds get backed up by decades of studies. Occupational exposure has shown links to respiratory trouble, skin rashes, and even stomach pains and ulcers. Chronic exposure brings another layer of worry. Antimony compounds may upset the liver and heart. The International Agency for Research on Cancer (IARC) lists antimony trioxide, a close relative, as possibly carcinogenic—meaning scientists still debate antimony trifluoride’s cancer risk but don’t shrug it off.
This compound’s dust can irritate and damage lungs, especially without proper extraction systems. Antimony ions don’t abate quickly; traces stick around in soil and water for years after release. Ingesting even small amounts, whether in a lab accident or through contamination, leads to nausea, vomiting, or worse. It doesn’t belong anywhere near food or water.
Beyond the Lab: Environmental Worries
The problems extend past individual workers. Disposing of antimony trifluoride responsibly challenges plants across the world. Chemistry on paper and chemistry in the real world part ways when spills or leaks creep into local water supplies. Fish and aquatic life face toxicity at concentrations too small for the naked eye to notice. Seeing dead fish or signs of river pollution reminds us why chemical management matters.
In the wider ecosystem, persistent antimony can bioaccumulate. Small organisms take it in, then birds and animals farther up the food chain find themselves exposed. While research continues on the exact levels that harm wildlife, erring on the side of caution seems wise.
A Shared Responsibility
Chemistry keeps advancing, but the price of progress shouldn’t rest solely on workers or communities living near plants. Manufacturing practices should use engineering controls—think better ventilation and strict containment—not just basic gloves and goggles. Investing in automated systems reduces hands-on contact, trimming down the odds of mishaps.
Training staff to recognize early warning signs helps avoid health crises. Clear labeling and storage make all the difference, especially in busy warehouses where similar-looking drums can cause confusion. Environmental monitoring—regular soil and water testing—catches leaks before they spiral.
Lawmakers can press manufacturers for higher standards through regulation instead of relying on voluntary guidelines. The European Union keeps a close eye on antimony-based substances. Big countries now update safety data sheets and monitor manufacturing imports and exports, bringing global trade into smoother, safer territory.
Looking Ahead
Safer alternatives exist for some uses of antimony trifluoride, and more researchers have started to chase greener chemistry. That transition won’t happen overnight, but with growing awareness, better protection and transparent policies, the grip of these toxic byproducts on daily life can loosen. Getting there takes collaboration—between scientists, companies, communities, and regulators. Speaking from years of handling tricky materials, nothing beats a system where people prioritize safety as much as productivity.
What is the chemical formula of Antimony Trifluoride?
Chemistry’s Straightforward Truth: SbF3
Antimony trifluoride carries the formula SbF3. For anyone who spent real time in a chemistry lab, it stands out for more than just its letters and numbers. Three atoms of fluorine bond with one atom of antimony, and the result unlocks some distinctive reactions and gritty stories from the chemical world.
Behind the Letters: A Deep Dive into SbF3
Antimony, labeled Sb from its stibnite roots, gets married to fluorine in this compound. In my experience with synthesis and working alongside chemical engineers, SbF3 reminded me how some elements behave far differently together than in their solo states. Antimony by itself carries metalloid qualities and doesn’t set off alarm bells in daily conversation, yet combine it with fluorine—a highly reactive gas—and the compound becomes a powerful tool. This union isn’t just theoretical; it brings teeth to industrial uses, especially in fluorination.
Why SbF3 Makes a Difference in Industry
Antimony trifluoride shows up in fluorination, changing C-H bonds to C-F bonds in organic compounds. Chemists know fluorine bonds don’t break easily, so drugs and materials with fluorine in them tend to stick around under pressure. I’ve talked to pharmaceutical scientists who value SbF3 for these reasons: It changes the face of drug molecules and adds durability to materials, even working its way into the Teflon coatings you find on cookware.
There’s also a spot for SbF3 as a catalyst in certain reactions, especially for synthesizing some plastics. If you’ve handled manufacturing operations, there’s a good chance this chemical played a background role, keeping processes smooth and turning raw ingredients into everyday products.
Environmental and Safety Challenges
Using SbF3 always draws me back to safety protocols. It’s toxic, especially if inhaled or handled without the right gear. The risks aren’t just on paper—real accidents can happen. Take a story from an old industrial site I visited, where poor handling led to contaminated runoff. The result lingered in the soil for decades. Antimony compounds pose genuine threats to water and ecosystems, so getting rid of unused SbF3 calls for careful chemical waste practices.
Finding Smarter Ways Forward
Avoiding pollution takes effort. Some manufacturers invest in better containment and monitoring technology, both to protect their workers and to keep contaminants out of the community. I’ve seen improvements in waste neutralization—turning harmful chemicals into something less risky before disposal—and real dedication to training employees about what these compounds mean for personal and environmental safety.
On the research side, new catalysts and greener fluorination alternatives occasionally show up in industry journals. Yet SbF3 still earns its place on the shelf, especially where reliability matters. Scientists and engineers know the stakes, and they keep searching for sustainable replacements that won’t compromise on quality or safety. Until then, anyone who uses or manages SbF3 needs solid expertise, accountability, and a sharp eye for consequences.
How should Antimony Trifluoride be stored?
Respecting the Real Risks
Not every chemical draws attention like antimony trifluoride does in a lab or production space. Anyone who’s dealt with this compound can tell stories about the real dangers, both short-term and long-term. I remember my early days handling specialty chemicals: the safety briefings weren’t empty rituals—they kept bad days from turning into disasters. Antimony trifluoride, a white solid or sometimes a fuming powder, fits this bill. It reacts violently with water. Leave it around careless or lazy storage, and all it takes is a leaky pipe or a humid afternoon to trigger toxic fumes. You can’t afford shortcuts or improvisation.
Humidity and Moisture: The Hidden Enemy
Antimony trifluoride attracts water like a moth to a flame, and the results can send skilled professionals to the emergency room. Hydrolysis produces hydrogen fluoride gas (HF), which is highly corrosive and toxic. Even in trace amounts, HF attacks skin, bones, and even the calcium in your blood. Forgetting to seal a cap, missing a crack in a bottle—these mistakes invite accidents. From my own lab work, I always kept desiccators and airtight containers close. Silica gel or other drying agents work best as companions, especially in environments where you can taste moisture in the air.
Choosing the Right Container
Over the years, experience taught me to distrust old or recycled containers, no matter how much someone says they’re ‘just as good.’ Antimony trifluoride should only ever go in containers that resist corrosion. High-density polyethylene (HDPE) and certain types of glass offer the best chance at preventing leaks. Metal containers don’t mix well—they rust or break down, especially at connection points. You spot a crusty lid or a rusty ring, that’s a chemical incident waiting to happen. Check every container for tight seals, and never repurpose lids. Each time a new shipment arrives, I do my own inspection, since a rushed warehouse worker can overlook flaws.
Temperature Control Matters
Antimony trifluoride stays stable at room temperature, but storage rooms need real temperature consistency. Direct sunlight can heat up shelves and boost internal pressure in sealed bottles, possibly pushing cracks or seams to failure. Set your chemical stocks away from windows. If your area gets hot in the afternoon, invest in ventilation or climate control. Take it from someone who once spent an entire weekend cleaning up after a summer heatwave—temperature swings don’t just ruin product, they ruin safety plans.
Separation and Labeling Save Lives
Signals and labels should scream “hazard” before anyone even grabs the bottle. Every time I’ve seen a “clear” label get swapped out for a faintly printed one, the risk of mix-ups climbs. Use thick, clear labels detailing content, date received, and hazard symbols. Always separate antimony trifluoride from incompatible chemicals, especially water, acids, and bases. Organize shelves so this chemical stands apart from anything prone to leaking or spilling fluid. Small details—the shelf’s height, nearby drainpipes—shape the difference between safe storage and a dangerous oversight.
Emergency Planning Cannot Wait
No level of careful storage can replace readiness. Every facility storing antimony trifluoride should keep personal protective equipment nearby: gloves, goggles, face shields. Dedicated HF spill kits—calcium gluconate gel, neutralizing agents, absorbent pads—should sit close to storage areas, not three rooms away. I’ve run through drills where seconds mattered, fumbling through cluttered safety cabinets. With this chemical, you keep supplies visible and within reach.
Summary
Storing antimony trifluoride tests discipline. Ignore storage rules, trust outdated habits—you court real, often irreversible harm. Safe storage goes beyond paperwork. It lives in every small decision and the respect you give serious materials every single day.
What are the safety precautions when handling Antimony Trifluoride?
Understanding the Hazards
Antimony trifluoride can give seasoned chemists pause. Its reputation isn’t just academic: this compound stands out for its toxicity and its knack for reacting with moisture to form hydrofluoric acid. I remember the first safety briefing about this stuff and sensing the room go quiet—everyone knew this isn’t something to get careless around.
Personal Protective Equipment Really Matters
Face shields and gloves aren’t just for show. Getting antimony trifluoride on your skin can cause burns, and breathing in the dust or vapors can lead to nasty health effects—headaches, chest pain, worse if it turns into long-term exposure. Nitrile gloves hold up better than latex, and splash goggles keep your eyes clear. Lab coats buttoned to the collar and closed-toe shoes matter here. Sometimes it feels like overkill but I saw a tech once wash his hands too late after a splash; even minor contact leaves a lasting sting.
Ventilation and Containment Cut Down Risk
Every handler remembers the warning about fumes: “Never open the jar outside a hood.” Antimony trifluoride gives off fumes that can damage the lungs. Proper fume extraction draws out the invisible threat. A lab-grade chemical fume hood sets the gold standard. If the workspace doesn’t allow that, even using local exhaust fans or portable ventilation works better than nothing, but you shouldn’t rely on makeshift solutions for consistent safety.
Handling and Storage Demand Respect
Screw-capped bottles, properly labeled, tuck away in chemical cabinets rated for toxic materials. Dry storage is more than a guideline—moisture eats right through the compound and forms hydrofluoric acid gas. Desiccators with indicator gels help a lot. Even shelf placement matters; I always kept reactive materials like this on the lower shelf to stop a spill from spreading over a larger area. The simple act of keeping this stuff away from water and acids has probably saved more trouble than anything else.
Spill Response: Quick, Calm, and Informed
Nobody wants to test the emergency plan, but underestimating a spill causes more chaos. The key is knowing what acts quickly: evacuate anyone without protection, ventilate, then sweep up the dry powder—never add water. Specialized spill kits with calcium gluconate gel for hydrofluoric acid exposure belong in the same room. I once saw a poorly handled spill get into a mop bucket. That mistake added a layer of danger and complicated cleanup. Solid training and a calm head win the race.
Medical Awareness and Incident Prevention
Symptoms from exposure don’t always appear right away. Flu-like aches, breathing problems, and stomach upset point toward chemical contact. Fast reporting gets help in time, but the best scenario involves never reaching that point. It’s not paranoia to uphold checklists—routine reviews and refresher training drop accident rates sharply. The CDC tracks chemical incidents, and their reports show a strong link between regular drills and reduced injury.
Setting a Culture, Not Just Following Rules
Handling antimony trifluoride challenges anyone, seasoned or learning. Mistakes slip in if the group cuts corners. Sharing real stories and lessons, not just safety posters, turns rules into habits. A culture where people feel safe speaking up—“Hey, you missed your goggles”—grows trust and sharpens vigilance. Safety isn’t only about avoiding injuries; it shapes a workplace people trust.
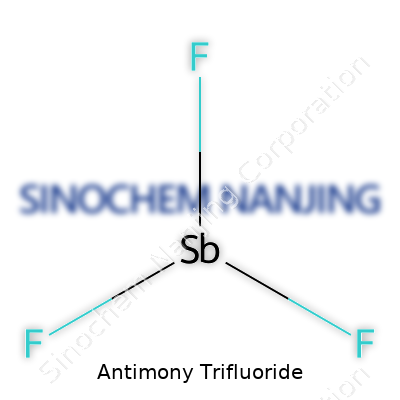
| Names | |
| Preferred IUPAC name | Trifluorostibane |
| Other names |
Antimony(III) fluoride Antimony fluoride Trifluoroantimony Antimonous fluoride |
| Pronunciation | /ænˈtɪməni traɪˈfluːəraɪd/ |
| Identifiers | |
| CAS Number | 7783-56-4 |
| Beilstein Reference | 3587156 |
| ChEBI | CHEBI:30427 |
| ChEMBL | CHEMBL1201586 |
| ChemSpider | 22422 |
| DrugBank | DB11103 |
| ECHA InfoCard | 100.028.395 |
| EC Number | 215-713-4 |
| Gmelin Reference | 60750 |
| KEGG | C06499 |
| MeSH | D000934 |
| PubChem CID | 24557 |
| RTECS number | BJ6300000 |
| UNII | QF6NX7U9P9 |
| UN number | UN2444 |
| CompTox Dashboard (EPA) | CompTox Dashboard (EPA) of product 'Antimony Trifluoride' is "DTXSID5044239 |
| Properties | |
| Chemical formula | SbF3 |
| Molar mass | 156.76 g/mol |
| Appearance | White to gray crystalline solid |
| Odor | Pungent |
| Density | 2.99 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.64 |
| Vapor pressure | 0.083 mmHg (25°C) |
| Acidity (pKa) | ~2.0 |
| Basicity (pKb) | 3.05 |
| Magnetic susceptibility (χ) | −62.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 8.2 mPa·s (at 15 °C) |
| Dipole moment | 3.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −1086 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1047.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB07 |
| Hazards | |
| Main hazards | Toxic by inhalation and ingestion; causes severe burns to skin, eyes, and mucous membranes; reacts violently with water and acids releasing toxic fumes. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | Precautionary statements of Antimony Trifluoride: "P261, P264, P270, P271, P272, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P307+P311, P308+P313, P321, P330, P362+P364, P405, P501 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 600 mg/kg |
| LD50 (median dose) | 132 mg/kg (rat, oral) |
| NIOSH | TF9010000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Antimony Trifluoride is "0.5 mg/m3 (as Sb) |
| REL (Recommended) | 2 mg/m3 |
| IDLH (Immediate danger) | 250 mg/m3 |
| Related compounds | |
| Related compounds |
Antimony trifluoromethanesulfonate Antimony pentafluoride Antimony(III) chloride Antimony trichloride Bismuth trifluoride Arsenic trifluoride |

