Antimony Trichloride: An Editorial Perspective
Understanding the Historical Journey
Antimony trichloride doesn’t land on most people’s radar, but this compound has a story that winds through chemistry’s timeline. Early alchemists in Europe fiddled with antimony salts, searching for the secrets of transmutation and remedies. Later, its chlorinated form popped up as glassmakers learned how it could clarify glass by removing greenish tints caused by iron. Over the centuries, these humble origins sparked industrial demand. Back in the day, analytical chemists reached for antimony trichloride to test for vitamin A, long before digital nutrient sensors existed. Its use in detection hints at its high reactivity, making it more than just a lab curiosity.
A Look at the Physical World
Antimony trichloride looks like a colorless, oily liquid or sometimes a soft, white solid, depending on how wet the air is. Its sharp, almost choking smell gives fair warning about its potency. It dissolves fast in solvents like chloroform and easily picks up water, reacting with it right out of the air. Start mixing it with water, and it smokes and fizzes as hydrochloric acid pops out. Folks handling this stuff in real lab settings learn respect fast—unlike many chemicals, it telegraphs risk in its appearance, smell, and aggressive nature.
Technical Specifications and Proper Labeling
Industries buying antimony trichloride usually want to know about its purity, water content, free acid levels, and sometimes trace metals. These aren’t trivial—small differences in purity shift reaction outcomes or the performance of finished materials. Proper labeling on chemical containers isn’t just red tape. It prevents confusion on the shop floor, keeps labs in compliance with safety laws, and lowers the risk of dangerous mix-ups. Folks in quality control remember how inadequate labels lead to costly mistakes, so the extra caution matters on every drum that rolls through the door.
Preparation Methods Borne of Hard-Won Experience
The standard preparation flows from reactions between high-purity antimony metal and chlorine gas or thorough mixing of antimony trioxide with concentrated hydrochloric acid. This sounds basic, but the devil’s in the details. Those who’ve run chemical reactors know it isn’t about pouring two things together and calling it a day. Extra attention to reaction temperatures, timing, and agitation shields against runaway reactions or nasty byproducts like antimony oxychloride. Production lines have to balance output with safety—older workers passing tips to new hands understand the value of precision, not just speed.
Reactive Personality in the Lab and Plant
The chemical temper of antimony trichloride comes through in its vigorous reactions. Douse it with water, and you get a haze of hydrochloric acid along with antimony oxychloride. Ship the liquid through pipes, and corrosion waits for anyone who skimps on maintenance. Its behavior as a Lewis acid lets it pull off tricks in organic synthesis, like adding chlorine to certain hydrocarbons. Chemists hunting new reactions take advantage of this adaptability. On the flip side, that flexibility calls for deep experience—misjudging conditions leads to byproducts or unexpected hazards. Any lab tech with an error story about spilling this stuff knows the sting of clean-up and review afterward.
Abyss of Names and Synonyms
Older books and chemical catalogs spill synonyms for antimony trichloride—antimonous chloride, butter of antimony, or even antimony(III) chloride. The phrase “butter of antimony” still gets a smirk from old-school glassmakers, a nod to the soft, spreadable texture the solid form takes. It’s worth sorting these labels out, since historic accident reports sometimes mention butter of antimony where modern documentation says antimony trichloride. Clarity on naming helps prevent confusion over storage or usage, especially where antique literature influences safety protocols.
Safety in the Real World
Safety protocols around antimony trichloride save skin, lungs, and ultimately lives. This compound soaks through gloves and stings the eyes and nose; there’s no room for casual attitudes. Respirators and chemical goggles aren’t extravagances—they’re a must. Spill response presents headaches; liquid leaks react with floor water, releasing hydrochloric acid fumes. Periodic safety drills and scenario planning decrease panic if something goes wrong; folks with long lab experience recall the days when these steps were skipped, and injuries followed. Lives taught by harsh lessons lobby for stricter handling standards and waste controls.
Industry and Research Applications
Antimony trichloride finds its way into a range of industries without much fanfare. Glassmakers and ceramics experts appreciate its clarifying properties, while organic chemists reach for it in halogenation reactions. Some folks I’ve known swear by it in early-stage chemical research—a reliable workhorse for measuring or catalyzing synthetic steps. As regulations tighten on fire retardants and specialty plastics, the compound’s reactivity keeps it in the mix for downstream applications. Within university research, innovation often means finding less hazardous substitutes, yet antimony trichloride’s accessibility and cost keep it relevant for experiments focused on chlorination.
Cutting-Edge Research and Ongoing Challenges
A steady stream of articles from chemical journals dives into new uses and safer handling frameworks. Some groups explore tweaking the molecule for more selective reactions, or swapping in greener solvents and less reactive partners to limit dangerous byproducts. Other researchers, motivated by environmental watchdogs, investigate recovery and recycling—turning spent trichloride streams into reusable resources. As governments grapple with rising toxicity thresholds and pollution controls, laboratory teams run pilot projects on closed-loop approaches. These efforts stem from both necessity and innovation, since waste costs and liability rank high on the ledger for any industry handling hazardous chemicals.
Toxicity Questions that Don’t Go Away
Plenty of toxicology studies warning about antimony trichloride’s risks line the walls of university libraries. Workers exposed to low levels track increased rates of lung and skin irritation, with chronic exposure linking to more severe symptoms. The compound’s ability to disrupt cellular processes makes regulators cautious. In the field, nobody shrugs off these warnings; personal experience dealing with chemical burns or persistent coughs stay with you. Advocacy groups press for transparent reporting and better worker health tracking, and many industry veterans side with them. No chemical, no matter how useful, deserves a blank check if it puts hands-on staff or the wider environment at risk.
Looking Ahead on Future Prospects
The conversation around antimony trichloride, and heavy metal reagents in general, sits at a crossroads. Advances in green chemistry bring hope for safer, less polluting substitutes, although adoption often stares down the reality of entrenched manufacturing lines and tight budgets. A continued push for more specific analytical tools spurs interest in tuning the compound’s reactivity to target only needed reactions, avoiding broad-spectrum harm. Government moves toward stricter disposal rules and emission caps challenge the status quo, pushing research toward more responsible use and clean alternatives. The prospects don’t hinge on abandoning tradition, but channeling experience—decades of hands-on labor, hard analysis, and collaboration between regulators, researchers, and industry workers. This patchwork legacy leaves space for optimism that safety, innovation, and accountability can reshape antimony trichloride’s next chapter.
What is Antimony Trichloride used for?
A Closer Look at Antimony Trichloride in Everyday Life
Antimony trichloride doesn't come up at most dinner tables, but it plays a quiet role in a bunch of products and processes. Most folks probably have never heard of it unless they’ve worked in chemistry, pharmaceuticals, or maybe in a fire lab. So why do labs and factories need this oddly named substance so much? It all comes down to what it can do to other materials — and the effects can be huge.
Fire Retardant Manufacturing
Factories making fire-resistant plastics and textiles often rely on antimony trichloride as a starting material for producing antimony oxides. These oxides end up blended into coatings and fibers, helping to make things less flammable. As a person who’s spent time around construction sites, I can tell you the fire rules get stricter every year. A treated plastic cable or curtain can slow down a fire, giving people precious extra moments to react. The facts are clear: fire-resistant materials in public buildings and transportation cut down injuries and deaths in emergencies. Antimony compounds still form a backbone in these critical safety upgrades, even as researchers keep looking for better alternatives.
The Chemistry Behind Testing and Analysis
Chemists leaned in on antimony trichloride long before modern machines did all the measuring. It has a knack for detecting vitamin A and similar compounds. If you remember old-school lab lessons, this chemical played a big role in colorimetric testing — basically, adding it to a sample, watching the solution change color, and learning a lot about what’s inside. Even now, some research labs choose antimony trichloride for these kinds of tests because of its reliability and the sharp clarity it brings during measurements.
Catalyst in Industrial Processes
Industrial chemists rely on antimony trichloride as a catalyst, especially in making plastics like polyethylene terephthalate. Most bottled water and soda containers are made from PET. The process isn’t as flashy as it sounds, but without the right catalysts, plastic manufacturing grinds to a halt or gets wildly expensive. My experience visiting a polymer plant made clear how production lines depend on every single step working precisely — antimony trichloride keeps things moving at scale.
The Glass and Dye Industry Connection
Glassmakers use it too. The chemical helps to remove coloring impurities, so clear glass stays clear. That’s why bottles, phone screens, and car windshields look the same all over the world — those industries count on chemicals like antimony trichloride to control the little details. Dye manufacturers use it as part of some specialty pigment processes. These pigments end up in inks, paints, and even cosmetics, so we all cross paths with its handiwork, even without realizing it.
Safety and Environmental Concerns
Handling antimony trichloride comes with risk. It reacts strongly with water and gives off heat and hazardous fumes. Workers need strong ventilation and training on handling dangerous chemicals. Regulators keep a close watch because run-off or spills can poison water sources, affecting both ecosystems and people. Documented cases show long-term exposure can cause health problems, so companies must invest in better systems or find safer alternatives. Upskilling workers, updating equipment, and enforcing proper disposal can ease the risk, but there’s still a long road for full transparency in how it’s used or discarded globally.
Changing the Way We Think About Hidden Chemicals
Many supply chains still depend on antimony trichloride, and it isn’t going away soon. Looking at its applications, some industries could shift to safer chemicals, but change moves slowly. People at home and work rarely see these hidden helpers, but knowing more about them can push demand for safer products. Laws, public awareness, and smarter design drive improvements, but those efforts need real investment from companies and governments. Chemical safety isn’t just for laboratory workers — it affects all of us, in ways we rarely see.
Is Antimony Trichloride hazardous to health?
The Real Risks of Antimony Trichloride
Dusty white powder, acrid fumes, and the kind of warning labels that make you pause—Antimony Trichloride doesn’t belong in a casual home lab. I’ve worked in environments where chemical safety isn’t just a footnote, it’s a daily check-off. In those settings, this stuff always calls for gloves, eye protection, good ventilation, respect.
Breathing in Antimony Trichloride vapors or letting it touch the skin isn’t just uncomfortable. Research from the European Chemicals Agency points out irritation starts quickly: burning in the nose, eyes that water and sting, coughing that stays with you. Skin contact brings on redness, blisters—the usual trouble with corrosive chemicals. During a busy day nearly a decade back, I watched a coworker pull off gloves too early after using this compound. Within minutes, his skin turned raw and angry-red. We kept an eye on him all shift. He ended up fine, but we all learned a lesson about shortcuts.
Chemical Exposure and Long-term Harm
Diving a little deeper, Antimony Trichloride doesn’t just sting in the short term. Studies published in journals like Regulatory Toxicology and Pharmacology point toward links with more serious problems. Long-term exposure—even at trace levels—may lead to lung damage, stomach pain, and lesions on the skin. The workers in old manufacturing plants, before strict regulations, sometimes showed signs of chronic bronchitis or persistent headaches. More care today means lower risk, but the record stands as a warning. The International Agency for Research on Cancer has also flagged antimony compounds for their possible cancer risks, though the data on trichloride is less clear than on other forms.
Some folks care most about environmental impact and here’s where the concern widens. Antimony compounds don’t break down easily in soil or water. That means spills or improper disposal stick around, drifting into food chains over time. Kids and pets playing around contaminated sites could be exposed more easily than adults, and that poses its own health questions.
Common Sense Goes a Long Way
Manufacturers and labs now have strict guidelines on storing and using Antimony Trichloride for a good reason. Eyes, skin, and lungs are too valuable to bet against proven hazards. Proper chemical fume hoods, disposable gloves, and chemical-resistant aprons cost a little extra, but they stop most accidents before they start. Safety training isn’t a joke—every minute spent learning those warnings could save a trip to the ER.
For folks outside science circles, the biggest advice is to avoid handling or storing this chemical. If an old container shows up during a garage or warehouse cleanout, treat it seriously. Hazardous waste disposal centers and local environmental agencies know exactly what to do, and they’d rather answer a phone call than see another poisoning statistic make the news.
Alternatives and Solutions
Some industries already moved away from using Antimony Trichloride, swapping in safer reagents. Green chemistry isn’t just a fancy buzzword—it’s how we cut down accidents and keep water supplies clean. Whenever a safer option exists, it draws fewer health complaints, less hazardous waste, and lighter safety overhead costs.
The moral feels pretty simple. Antimony Trichloride has uses in industry and laboratories, but the risks mean handling always deserves respect. For people working hands-on, up-to-date safety training and conscientious work habits form the best defense. For everyone else, steering clear keeps curiosity from turning into regret.
How should Antimony Trichloride be stored?
Why Care About Antimony Trichloride Storage?
I’ve seen enough cases of chemical mishaps to know that simple choices can stop a disaster before it happens. Antimony trichloride isn’t a household name. Even so, it finds its way into labs, metal processing, and the world of flame retardants. If left ignored on a shelf or tucked away in a careless spot, it’ll react with moisture in the air and give off hydrogen chloride gas—harsh on lungs, corrosive for metal. So the details of storage aren’t just a technical box-ticking exercise; this is about protecting workers, the public, and the environment.
The Real Risks: Corrosiveness and Fumes
Anyone who’s opened a bottle of this stuff will notice a sharp, chemical smell. Antimony trichloride goes after water vapor with real enthusiasm, churning out hydrogen chloride fumes. Give it a metal shelf or a damp storeroom, and it’ll chew right through either one. Exposure can sting the skin, eyes, and nose; worse accidents land folks in the emergency room with burns or breathing trouble. This isn’t just about following rules. Take these risks seriously and storage stops being an afterthought.
Solid Advice for Safe Storage
Start with a tightly sealed, chemical-resistant container. Glass stuck with a good PTFE-lined cap works far better than plastic, since stoppers and plastic often break down over time or can allow vapor to seep out. Keeping the lid screwed on tight isn’t nitpicking—every little bit of air sneaks in and can trigger a reaction.
Once contained, pick a spot away from any hint of water. Keep it off the floor and away from sinks, windows where rain might sneak in, or wherever a pipe could spring a leak. Even a humid corner spells trouble. One of the smarter moves is to use a dedicated dry box or store it in a cabinet fitted with desiccants—silica gel can pull out stray moisture, lessening the chance of an accidental reaction.
No one should store antimony trichloride near everyday acids, alkalis, or flammable stuff. If there’s a fire or spill, it can whip up a mess of unpredictable reactions. Mixing up chemicals in the same zone often leads to confusion if an emergency hits, making clean-ups riskier. Designate distinct spots for each high-hazard chemical. Label everything, and train every staff member so forgotten bottles don’t turn into a bigger risk later.
Ventilation counts. Every chemical store should move air fast enough to catch fumes before they build up. Simple exhaust fans can save lungs and prevent corrosive gases from lingering where people might breathe them in. I always encourage regular checks—filters clog, ducts rust, and bad systems can lull you into thinking you’re covered.
Keen Eyes and Solid Records Make a Difference
Smart storage comes down to clear protocols and a team that stays alert. Logging every use, checking labels for wear, and doing monthly visual inspections catch problems before they cost more than just time. Spills, leaky lids, or clouded glass all need a quick response to stop a problem from growing.
Protecting Real People, Not Just Checking Boxes
It’s easy to slip into the mindset where storage feels like nothing more than a compliance issue. In reality, keeping antimony trichloride safe is part of protecting every hand that might ever pick up the bottle. With well-thought-out steps and steady discipline, everyone can work or study with confidence, knowing the real risks have been addressed from the start.
What are the safety precautions when handling Antimony Trichloride?
Why Handling Antimony Trichloride Matters
Antimony trichloride isn’t your run-of-the-mill lab chemical. I’ve seen plenty of labs shy away from it after reading the hazard warnings—and with good reason. This colorless, fuming liquid gives off hydrochloric acid fumes and can eat through skin and metal. A small splash will burn or cause blisters, and breathing in its vapors will leave your throat raw. These hazards make safety gear and proper habits a must.
Personal Protection Isn't Optional
Before getting near the bottle, grab a real lab coat—not a thin painter’s smock. Choose gloves made of nitrile or neoprene; latex breaks down too fast. Wraparound goggles stop stinging fumes from irritating your eyes, and a proper face shield adds another layer against splashes. Shoes matter, too—a friend ignored this and learned the hard way that antimony trichloride eats right through socks.
Ventilate, Or You’ll Regret It
Antimony trichloride fumes get worse as the bottle sits open. Pour or mix it inside a fume hood, leaving the sash as low as possible. Cracking the lab window won't make a difference; only strong exhaust has a shot at keeping you from inhaling vapors. Workers in older labs often tape warning signs around fume hoods used for antimony trichloride, a habit worth keeping up since it sticks with people and prevents confusion.
Never Cut Corners on Storage
Leaving antimony trichloride out on a shelf turns a small job into an emergency. I always keep it in tightly sealed glass bottles, well away from water supplies. Moisture triggers a violent reaction, releasing hydrogen chloride gas. Once during a summer storm, I found drops landing on an open container—everybody evacuated faster than I’d ever seen over that invisible but deadly mix.
Simple But Firm Spill Rules
Sooner or later, a spill will happen. Neutralize right away using dry soda ash or lime; water only makes matters worse. Scoop the neutralized mess into a sealable bucket for hazardous waste pickup. Mop the area with more soda ash after. Every training I give, I stress this process—just grabbing a mop out of habit can send someone to the doctor.
Emergency Action: Know It Backwards And Forwards
First aid for antimony trichloride exposure is a race against the clock. Flush skin or eyes with running water for at least 15 minutes, even if it stings. If anyone breathes in the fumes, get them into fresh air. Every lab I’ve worked in keeps an emergency shower and an eyewash station close by. These have saved more than one careless student from permanent injury.
Training and Respect Go Hand-in-Hand
People handling antimony trichloride cannot wing it. Training comes first, then experience. Someone walking in without knowing spill response drills or where to find emergency wash stations is a danger to everyone. Secure written instructions and refreshers every few months. Even in busy labs, block out time for a quick rundown on dangerous chemicals—it keeps new workers safer and helps old hands avoid slipping up.
Solutions Beyond The Basics
Smart labs use electronic inventories to track who’s using hazardous chemicals and when. Label every container with clear hazard symbols and use QR codes for easy access to safety data sheets on mobile devices. Asking every person to sign off after training boosts accountability. These steps do more than tick boxes—they help create a mindset where everybody watches out for each other.
Having watched accidents happen despite good intentions, I won’t downplay the risks. Safety for antimony trichloride starts with respect, sharp habits, and knowing what you’re doing at every step. That’s a lesson you want to learn from a checklist—not from an Emergency Room trip.
What is the chemical formula and appearance of Antimony Trichloride?
What Makes Up Antimony Trichloride?
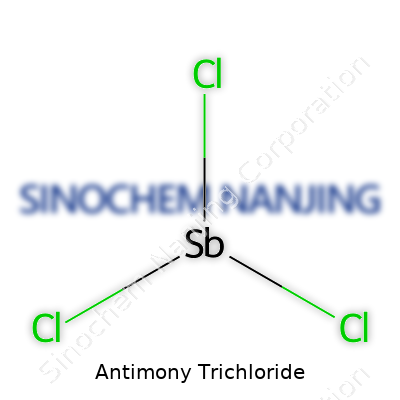
Antimony trichloride goes by the chemical formula SbCl3. This simple-sounding arrangement tells a bigger story. Antimony, a metal on the periodic table, bonds with three chlorine atoms. The arrangement isn’t just chemistry trivia—it shapes how people handle, use, and store the compound.
How Antimony Trichloride Looks in the Real World
Open a vial of antimony trichloride and you’ll notice a colorless, sometimes faintly yellow, crystalline solid. The crystals tend to look glassy, almost wax-like, and will melt in your hand if the room’s warm enough. SbCl3 melts at only 73°C (about 163°F). Pour it out and a fuming, corrosive liquid appears. Moisture hits and white fumes curl up—what you see is hydrochloric acid vapor and a sign to keep the lid tight and your hands dry.
Why Appearance and Formula Matter in Everyday Labs
In chemical labs, appearance and formula aren’t details to gloss over. The physical looks of SbCl3 give away clues about purity and freshness. Clear, colorless crystals shout “fresh.” A yellow tinge warns of decomposition or contamination. Experienced chemists check the color before using it because mistakes here mean messed-up experiments or even dangerous situations.
The chemical formula also shows just how reactive the material gets. SbCl3 breaks down with even a touch of water, releasing clouds of hydrochloric acid. A chemist learns to open bottles gently and under a fume hood. Forgetting these simple habits leads to ruined samples or, much worse, damaged lungs. I’ve seen more than one seasoned technician underestimate the fumes, only to dash out gasping, eyes running.
Recognizing Hazards Goes Beyond Science Textbooks
Handling antimony trichloride offers lessons you don’t find just reading research papers. The glassy appearance tricks people into treating it like salt or sugar, but its fierce reaction with water demands extra respect. Spills eat through floors, tables, and sometimes skin. The Occupational Safety and Health Administration (OSHA) tracks antimony compounds since chronic exposure damages lungs, skin, and even liver function. Anyone working closely with this chemical needs goggles, gloves, and a plan for accidents.
Paths Toward Safer Handling
Problems with accidents or spills tie back to two things: training and culture in the lab. Effective training doesn’t just come from laminated safety sheets—it builds from real stories, demonstrations, and, yes, sometimes learning from close calls. Setting up proper spill kits, using closed storage cabinets, and labeling bottles in bold, simple language stops most of the trouble before it starts. When supervisors treat safe habits as part of the routine, not an afterthought, the risks drop for everyone.
It isn’t only about following the rules. The look, smell, and behavior of antimony trichloride offer a daily reminder: chemistry’s basics save time, money, and health. The sooner every lab worker recognizes what’s in the bottle, down to the formula and the faint yellow shades, the sooner the workplace gets safer for everyone who walks through the door.
| Names | |
| Preferred IUPAC name | trichlorostibane |
| Other names |
Antimonous chloride Antimony(III) chloride Butter of antimony |
| Pronunciation | /ænˈtɪməni traɪˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 10025-91-9 |
| Beilstein Reference | 3588737 |
| ChEBI | CHEBI:30191 |
| ChEMBL | CHEMBL1201867 |
| ChemSpider | 20519 |
| DrugBank | DB11162 |
| ECHA InfoCard | 100.021.254 |
| EC Number | 231-868-0 |
| Gmelin Reference | 814 |
| KEGG | C18674 |
| MeSH | D000892 |
| PubChem CID | 24816 |
| RTECS number | BJ6475000 |
| UNII | V3D4300G1U |
| UN number | UN1733 |
| Properties | |
| Chemical formula | SbCl3 |
| Molar mass | 228.11 g/mol |
| Appearance | White crystalline solid |
| Odor | Pungent |
| Density | 3.14 g/cm³ |
| Solubility in water | Decomposes |
| log P | 0.92 |
| Vapor pressure | 0.0133 kPa (25°C) |
| Acidity (pKa) | -3 |
| Basicity (pKb) | -3.58 |
| Magnetic susceptibility (χ) | -63.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.740 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 364.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -401.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -504.90 kJ/mol |
| Pharmacology | |
| ATC code | V09CX03 |
| Hazards | |
| Main hazards | Corrosive, causes burns to skin and eyes, harmful if inhaled or swallowed, reacts violently with water, emits toxic fumes. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H314, H332 |
| Precautionary statements | P234, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P337+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Autoignition temperature | 223 °C (433 °F; 496 K) |
| Lethal dose or concentration | LD50 oral rat 1000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 3000 mg/kg |
| NIOSH | SN1225000 |
| PEL (Permissible) | PEL: 0.5 mg/m³ |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | 50 mg/m³ |
| Related compounds | |
| Related compounds |
Antimony pentachloride Bismuth trichloride Phosphorus trichloride Arsenic trichloride Antimony trifluoride |

