Looking Closely at Antimony Tribromide: A Chemical Under the Spotlight
Historical Development
Chemical history often reflects changing priorities and fresh perspectives. Antimony tribromide comes from a long tradition of chemistry adapting to new demands. Early chemists like Antoine Lavoisier helped shape modern methods through small advances—some by accident, some by intention. Antimony has roots in ancient medicine and metallurgy. Alchemists misunderstood its power for centuries. By the late 19th century, antimony compounds became vital in pigments, flame retardants, and laboratory experiments. Even in high school chemistry, demonstrations with antimony salts open eyes to how simple molecules spark big consequences. Today, the evolution of antimony tribromide points to the study of specialized chemicals whose use follows the beat of discovery and tough safety standards.
Product Overview
Antimony tribromide doesn’t stand out on a shelf, but its purpose stands solid in industrial and laboratory contexts. Often a white to pale yellow solid, this compound slips into processes that might not make headlines yet keep daily products safer and more reliable. Across plastics manufacturing, textile fireproofing, and analytical testing, this compound provides a way to control performance. Experience in applied chemistry shows the value of picking a chemical not for its looks or fame, but for its precise effect. Decisions about using antimony tribromide often come down to needing a tool that gets a specific job done—no more, no less.
Physical & Chemical Properties
In a laboratory setting, antimony tribromide usually appears as crystalline masses or powder, sensitive to moisture with the tendency to corrode surfaces over time. Handle it with dry gloves; exposure to water causes it to hydrolyze, producing hydrogen bromide fumes—definitely a wake-up call for anyone who’s ever underestimated the sting of a fume hood. Its melting point and solubility shift with atmospheric conditions, confirming that even a stable-seeming solid demands respect. Seeing it react with common solvents brings theory to life: the sharp smell, the haze, the unexpected warmth in your palm, all reinforce the importance of knowing your chemical inside out. The chemical reacts swiftly with bases, and its role as a Lewis acid often opens new pathways for researchers testing hypotheses about reactivity.
Technical Specifications & Labeling
Labels on chemical bottles tell a story any bench scientist learns to read the hard way. Purity grades set the tone—lower grades find homes in industrial-scale processes, higher grades in analytical labs. Reliable suppliers provide specifications covering trace water content, other halide presence, and measurement limits. Good labeling warns plainly about health risks—eye, skin, and lung irritation—marking antimony tribromide as something you take seriously in both handling and storage. Material that doesn’t meet expected benchmarks often signals a need for fresh training or a supplier review. Across my years in research settings, transparent labeling and trustworthy technical datasheets have often saved time and, sometimes, prevented close calls.
Preparation Method
Making antimony tribromide usually involves reacting antimony trioxide or metallic antimony with concentrated hydrobromic acid or elemental bromine. The procedure produces strong fumes, often requiring a good fume hood and careful temperature control. Watching team members mix the reactants, it’s easy to see why chemical manufacturing has drifted to expert hands; the release of hydrogen bromide can create bottlenecks if containment isn’t perfect. Reaction yields vary with reactant ratios and the rate of addition. Production speed and purity reach a balance set by both art and science—getting it right matters more than getting it fast. Operators who’ve spent years perfecting these steps always remind newer colleagues: respect the process, or pay for mistakes.
Chemical Reactions & Modifications
Chemical flexibility sits at the heart of how antimony tribromide finds its way into different products. The compound enters substitution and addition reactions, forming complexes with organics or other halides. In the lab, its reactivity with amines and esters creates intermediates for plastics, dyes, and even pharmaceuticals. If put under heat or in the presence of water, it decomposes—releasing not just bromine but also toxic antimony trioxide. This behavior prompts steady improvements. Sometimes researchers tweak reaction conditions, trying to minimize byproducts, improve yields, or cut down hazardous offgassing. Living through the grind of multi-step syntheses, chemists pick up on which small modifications—in temperature, pressure, or stirring—make a big difference to the final result.
Synonyms & Product Names
In chemical catalogs, antimony tribromide goes by various names—often as stibium bromide, or simply SbBr3. Sometimes it appears in product lists as antimonic bromide or tribromostibane. Awareness of synonyms keeps projects running smoothly. Missing a name or misreading a reference can derail an experiment. Over the years, both in academic and industrial labs, I’ve watched project leads spend whole afternoons tracing synonyms through scientific papers—painstaking but essential work. Journal articles, import documents, and regulatory filings often juggle multiple names and not all of them cross over neatly between languages and regions. Recognizing alternate names helps teams avoid confusion when ordering or reporting on results.
Safety & Operational Standards
Antimony tribromide presents real risks if handled without care. Strict ventilation rules apply — fume hoods, sealed containers, and full PPE remain the baseline. Even one lapse, like a dropped glove or missed respirator, can end with someone in the medical unit. Breathing in fumes or letting powder touch your skin brings immediate stinging and lasting irritation. Long-term, exposure to antimony compounds connects to chronic respiratory issues and potential toxicity. Past incidents across factories and labs show how easily a routine procedure slips into an emergency if shortcuts creep in. Training, regular drills, labeled storage, and a no-exceptions approach all play their parts. Through years in shared workspaces, the best safety culture always comes from hands-on mentoring — teaching, not just telling, sets the bar higher.
Application Area
Stepping outside the lab, you’d find antimony tribromide influencing modern life far more than most realize. Plastics and textiles gain crucial flame-retardant features thanks to its chemistry. The danger of fast-spreading fires in homes or public spaces changes when treated materials use antimony-based additives. Analytical chemists employ SbBr3 to test for chloride ions, and its role in producing other antimony compounds extends into batteries, semiconductors, and specialty glass. Each application brings its own demands around purity, stability, and safe use. Years spent helping clients troubleshoot manufacturing hiccups have shown me that even single-digit variations in component performance—driven by small impurities or uncalibrated dosing—can send final products off-spec. Quality control, attention to process detail, and regular review make the difference.
Research & Development
Antimony tribromide remains a topic of close R&D focus. New rules around flame retardancy push companies to seek alternatives or modify existing compounds for better environmental impact. Researchers probe for ways to limit toxic byproducts while keeping fire protection strong. Real movement happens in collaborations between industry and academic labs, where teams test greener halides or tweak synthesis routes for less hazardous emissions. Sometimes the biggest breakthroughs arrive from the edges—graduate students or upstart companies taking risks traditional players shy away from. My time reviewing grant proposals shows that incremental gains matter—lowering byproducts, boosting yields, or shrinking energy use all add up to meaningful improvements over time, even if progress crawls by line by line.
Toxicity Research
Antimony in general, and antimony tribromide in particular, sparks ongoing health research. Animal studies point to lung and skin problems; human data ties antimony exposure to respiratory and cardiovascular effects after chronic contact. Safety standards from agencies like OSHA and the European Chemicals Agency set limits—thresholds, not guarantees. Ongoing toxicity research means recommendations sometimes shift as fresh evidence surfaces. Medical staff at chemical plants and universities monitor team health, reviewing new studies to update workplace standards. My own colleagues often voice frustrations at regulatory lag—where clear health concerns move faster than rule changes. Still, the trend bends toward more caution, tighter exposure limits, and deeper investment in worker safety programs.
Future Prospects
Antimony tribromide’s road ahead depends on how the world weighs industrial need against emerging health and environmental priorities. Regulatory squeeze will push firms to develop safer handling, substitute chemicals, or better recycling processes. As new materials technology grows, demand could shift—safety in electronics and construction sometimes runs up against toxicity concerns. Green chemistry holds out hope—researchers send out trial balloons with organics, silicates, or other alternatives, gauging which might eventually unseat antimony compounds in applications like fire retardants. Collaboration across disciplines and industries proves critical to speeding this transition. Based on past industry cycles, companies and regulators rarely move in straight lines; progress often looks like two steps forward, a stumble back, and then a leap ahead, driven by public expectation, scientific progress, and market forces pressing for real change.
What is Antimony Tribromide used for?
What It Does in Industry
Factories like antimony tribromide for its fire safety perks. Toss this compound into plastics or textiles, and it helps slow down flames. Years back, I started a job where the building codes kept changing to demand better flame-resistance, especially after a string of warehouse fires. Looking over the materials list, antimony tribromide kept popping up, especially in electronics and tough synthetic fabrics.
That reliance on this chemical has kept damage from electrical fires short in a lot of old apartment blocks. Add just a pinch to a plastic casing or the insulation around wiring, the stuff suddenly resists burning. A few grams can make wiring less likely to melt or burst into flame, which matters if kids end up poking behind the sofa, as they do.
Antimony Tribromide in Labs and Chemical Processes
Lab techs keep it on the shelf for research and specialty products. Mix it with other chemicals, and it works as a catalyst or makes other specialty flame retardants. Older books on glassmaking talk about antimony compounds for certain optical glasses—those ones that need to balance durability and clarity for special lenses.
Risks and Health Warnings
Here’s the other angle: this stuff isn’t gentle on people or the planet. Wind can pick up fine dust from factories, and fumes sometimes slip past older ventilation. If someone inhales particles or handles it without gloves, rashes or lung irritation start cropping up fast. Toxicology reports warn that ingestion or chronic exposure may mess with the liver, skin, or respiratory system.
That concern goes double for workers. Laws about handling antimony-based compounds get tougher every year for a reason. Europe and the US both flag it for safety. In factories where air filtration works poorly, I’ve seen companies scramble after a surprise inspection, swapping out older flame-retardant mixtures for safer blends as soon as they can manage the cost.
Why Keep Using It?
A lot of manufacturers still weigh cost, availability, and legal requirements. Cheaper flame retardants do show up sometimes, but they don’t always work as well, or companies worry about other toxic byproducts. Aluminum trihydrate and magnesium hydroxide get some use, but for some products, especially small electronics, switching gets expensive.
Safer Alternatives and Industry Pressure
Industry watchdogs push for plant upgrades, better personal protection, and, where it’s affordable, total phase-out. More and more companies take the extra step to invest in research for safer flame-retardant options. Proper handling—full gloves, ventilation, closed mixing stations—slows down health complaints. Responsible firms list every toxic input, run exhaust scrubbers, and plan regular medical checks for staff.
Beyond workspaces, governments force household goods makers to limit how much antimony ends up in things like children’s pajamas. Public pressure makes a difference. Stories about contamination spark faster change than company press releases. From my experience, real reform takes a mix of regulation, transparency, and plain old public awareness.
Looking Ahead
Demand for fire safety isn’t going anywhere. At the same time, communities want fewer chemicals in the products around their families and the folks who make them. Chasing better solutions—whether with new chemistry or tighter rules—doesn’t just help workers. It builds trust and keeps problems from showing up down the line, which is exactly where everyone wants to be.
What is the chemical formula and structure of Antimony Tribromide?
Recognizing the Formula
Antimony tribromide shows up in the world of chemistry with the formula SbBr3. This compound brings together one antimony (Sb) atom and three bromine (Br) atoms. For people handling chemicals or working on inorganic compounds, this formula flags a compound with practical uses and unique traits.
Structure That Shapes Its Behavior
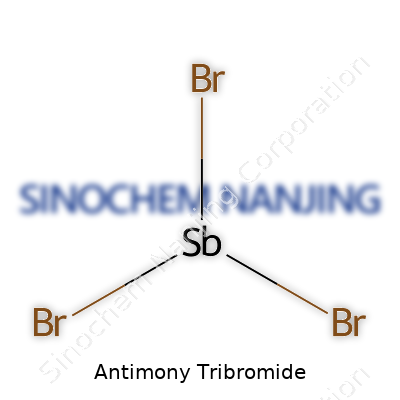
SbBr3 takes on a pyramidal shape. Chemistry classes may sketch this up as a central antimony atom surrounded by three bromine atoms, leaving one spot for a lone pair of electrons. That lone pair keeps the structure unsymmetrical, which affects how SbBr3 reacts with other substances. Bond lengths between antimony and bromine measure around 2.5 Ångströms, a detail highlighted in spectroscopic and crystallographic studies.
This arrangement gives the compound a reputation for volatility and makes it quite reactive with water. Dip a sample in water, and it hydrolyzes, giving off a thick white cloud of antimony trioxide (Sb2O3) and hydrogen bromide (HBr), both of which demand safety protocols. It’s a classic illustration of how a structure’s shape can decide both application and handling in the real world.
Why Identifying SbBr3 Matters in the Lab and Industry
I’ve seen chemists pull SbBr3 out of their chemical cabinets to prepare flame retardants, or to work in dye manufacturing and analytical chemistry. Any mistake in identifying the compound or misunderstanding its behavior invites hazardous outcomes. For instance, its toxic nature calls for ventilation, gloves, and goggles, not just in the research lab but in any facility where larger quantities get handled.
Mislabeled or contaminated antimony tribromide has huge potential for harm, especially since hydrogen bromide gas irritates the lungs and eyes. Regulatory bodies like OSHA keep tight tabs on how facilities store and handle SbBr3. In one of my old chemistry classes, a student mix-up triggered a minor evacuation—clear proof that every gram deserves respect.
Supporting Safe Practice with Science
Scientists often turn to X-ray crystallography and gas-phase electron diffraction to confirm the structure of SbBr3. Results match theory: the lone pair on antimony pushes bromine atoms aside, leaving a three-legged pyramid, not a flat triangle.
It’s tempting for newcomers to see chemistry as a game of memorization, but recognizing the molecular geometry directly links to safe lab habits. Miss that lone electron pair, and you miss the reason antimony tribromide reacts with moisture more aggressively than, say, sodium chloride.
Addressing the Risks with Knowledge
Better labeling, ongoing training, and clear safety data sheets help keep incidents with SbBr3 in check. Employers need to keep these materials updated and push for regular drills. Laboratories must check for leaks and manage storage to keep reagents upright and sealed. Online forums and professional organizations exchange stories about near misses, building a culture of safety that goes beyond formal text.
Understanding antimony tribromide boils down to combining textbook knowledge with hands-on respect. Recognizing SbBr3 isn’t just an exercise for exams—it determines the outcome of reactions and the health of everyone nearby.
Is Antimony Tribromide hazardous or toxic?
Understanding the Risks of Antimony Tribromide
People rarely hear about antimony tribromide unless they work in chemical processing, flame retardant manufacturing, or some niche corner of scientific research. Still, this compound raises important health and safety questions.
In my work with chemistry educators and labs, antimony compounds usually attracted a certain kind of respect, partly from stories passed around class, but mostly because the safety data sheets brought out the red flags. Antimony tribromide is no different. Anyone who’s spent some time reading up on chemical hazards learns pretty fast that skin, eyes, and breathing suffer most from sloppy handling or accident.
Direct contact quickly irritates skin and eyes. Workers exposed in poorly ventilated spaces—through powder spills, leaky storage, or heating—risk breathing in fumes that cause coughing, shortness of breath, and chest discomfort. The poison control centers back up those reports with case files. Ingesting antimony compounds can cause vomiting and stomach cramps, and overexposure may even damage the liver, kidneys, or heart with repeated incidents. These aren’t theoretical risks; they are real for anyone handling or disposing of the compound without care.
Then there’s the long game. Long-term exposure to dust or fumes may lead to chronic breathing issues. The International Agency for Research on Cancer lists certain antimony compounds as possibly carcinogenic. While evidence specifically linking antimony tribromide to cancer in humans remains limited, the overall risk affects industries that use and dispose of antimony substances.
One situation that always stuck with me came from a recycling plant. Improperly discarded industrial containers released residue. Workers cleaned it up without protective gear, thinking the risk minor. The headaches, throat irritation, and rash that followed turned heads quicker than any pamphlet. An on-site safety audit found that people lacked information, not just equipment.
Tackling the Hazards
Companies and researchers won’t walk away from antimony tribromide anytime soon, so tackling the hazards calls for real controls. Ventilation isn’t just about open windows. Proper extraction and filtered hoods cut down on airborne exposure. The best labs lock up antimony compounds, check containers for leaks, and train every new worker about spills and first aid. Even at small scales, goggles, gloves, and aprons should be standard.
Monitoring helps too. Breakrooms aren’t lab spaces. Tracking who comes in contact with the stuff can stop chronic exposure before it starts. In my view, real-time air quality monitors don’t replace smart habits, but they backstop unexpected events. Some companies run regular medical checks for staff, watching for early signs of toxicity. Policy works only if the everyday worker trusts the system and knows how to spot trouble.
Rules matter, but personal vigilance saves lives. Anyone who spends time in a space where chemicals gather learns respect quickly. Reporting spills immediately, never improvising protection, and recognizing symptoms early make a difference. The wider world needs to understand these chemicals don’t just live in textbooks—they’re in workplaces across the globe. Making that connection can push decision-makers to invest in safer substitutes or push for stricter regulation on disposal and recycling.
Building Safer Environments Together
Any industry that keeps using antimony tribromide has a responsibility. Protecting people from its risks isn’t about ticking boxes or waiting for an incident. With transparency, frequent training, and a willingness to change old habits, everyone can build a healthier workspace. Respecting science, sharing stories, and demanding accountability keep these hazards from leading to long-term harm. Every lesson learned adds up to safer days for those of us who handle not just this compound, but every dangerous material in the toolkit.
How should Antimony Tribromide be stored and handled?
Why Antimony Tribromide Gets Special Attention
Plenty of chemicals demand respect, and antimony tribromide ranks high on the list. This stuff comes with real risks for your health and for the environment. Inhaling it or getting it on your skin can kick off serious problems—imagine shortness of breath, chest pain, or even burns. It’s not just people who face trouble either. Leaks or spills have the power to wreak havoc on water and soil if left unchecked.
Getting Storage Right
Anyone storing antimony tribromide cannot treat it like common cleaning chemicals or solvents. Dryness matters. Keep the containers in a cool, well-ventilated room, nowhere near sources of moisture since antimony tribromide reacts fiercely with water. This reaction spits out hydrogen bromide, which stings the eyes and lungs. A tight seal on containers stops leaks and keeps out air and water. Glass, polyethylene, and polypropylene containers stand up well against this compound.
Temperature swings spell trouble. A steady, moderate temperature helps prevent pressure buildup, which can turn a simple bottle into an accident waiting to happen. Sunlight can also speed up reactions, so stash the containers away from direct rays. Use clear labels with hazard warnings. People in the area shouldn’t have any doubt about what’s in the drum or bottle.
Handling: Suit Up and Slow Down
Anyone who’s worked in a lab knows shortcuts lead to accidents. No bare skin gets near antimony tribromide—lab coats, gloves (nitrile or neoprene work best), and chemical goggles offer good coverage. A face shield steps things up when pouring or transferring the material. Going without proper gear often results in lasting effects, and recovery’s rarely quick.
Spilling this stuff means cleaning up with care, not with paper towels or rags. Specialized absorbents help bind the chemical. Get rid of any cleanup material through hazardous waste channels—never down a drain or into a landfill. I’ve seen the mess that comes from cutting corners, and long after the bottles get put away, contaminated surfaces stay risky.
Sensible Precautions Keep Everyone Safe
Before working with antimony tribromide, go over safety data sheets. Training and drills help workers stay calm if the worst happens. Easy access to eyewash stations and emergency showers can make all the difference. In my own experience, those minutes after an exposure sort out who ends up with minor irritation or something much worse.
Fire safety takes on new meaning with antimony tribromide. Water will make things worse, so keep dry powders handy for small fires. Ventilation should draw fumes away from workspaces, not circulate them around the building. No one appreciates a pounding headache or burning lungs after a misplaced splash or puff of vapor.
Responsible Choices Beyond the Workplace
Effective rules and proper facilities matter, but personal responsibility carries a heavy weight. What goes down the drain or ends up in regular trash dumps doesn’t just disappear; it circles back. Mistakes with antimony tribromide leave scars on people and places. Investing in training, gear, and time pays off long term by keeping everyone out of the emergency room and the headlines.
Nothing beats care and respect for the task at hand. Whether it’s a drum in an industrial warehouse or a vial in a research lab, each container deserves undivided attention. Mistakes can last lifetimes, but steady habits and practical setups write a better story—for people and for the planet.
What are the common applications of Antimony Tribromide in industry?
Fire Retardancy: Protecting Materials from Flames
Factories dealing with plastics or textiles know the hazard that fire brings. Antimony tribromide acts as a flame retardant in many consumer and industrial products. During my visit to a mid-sized plastics manufacturer, I saw firsthand how safety officers treated raw polymer granules with fire-retardant additives. Antimony tribromide, thanks to its bromine content, breaks up the combustion reaction at the molecular level, which cuts down how quickly flames spread. A report from the American Chemistry Council confirms this, pointing out that antimony-based compounds slow ignition and reduce smoke in furniture foam or building panels. Workers benefit, too—houses, cars, and public places become a little safer because the materials inside fight fire instead of fueling it.
Enhancing Optical Devices and Glass
Many folks overlook what goes into high-quality glass—yet the glass business relies heavily on chemical additives for great results. Antimony tribromide gives specialty glass its crystal-clear shine and improved resistance to clouding. Fiber optic cables, for instance, need perfect clarity to transmit data at high speeds. My time in a research lab opened my eyes to how the right additive transforms ordinary sand and limestone into glass that powers tech. In the world’s top electronics, fiber optics enable internet speeds that drive YouTube, Zoom, and cloud storage. Even basic architectural glass in skyscrapers sometimes receives treatment for extra durability.
Catalysis in Dye and Chemical Synthesis
The color in your favorite t-shirt or the finish on printed packaging starts in a chemistry lab. Specialty dyes and pigments require precision, and companies often reach for antimony tribromide as a catalyst. Its presence helps chemical reactions flow to completion, bringing vibrant, stable colors faster and cheaper than untreated processes. Data from industry sources like the Chemical Book suggest companies prefer antimony compounds for their ability to streamline synthesis, reduce waste, and support consistent batches.
Role in Analytical Laboratories
Testing precious metals, arsenic, and a host of other elements in environmental and quality-control labs calls for accuracy. During early days at my university chemistry department, reagent prep involved many odd-sounding compounds, including antimony tribromide. Analytical chemists still pick it for specific tests, such as the Gutzeit method for detecting arsenic. Lab staff value how it brings out sharp, reliable color changes so results aren’t left open to guesswork.
Risks and Responsible Use
Despite these advantages, conversations about antimony compounds never ignore health and environmental risks. The U.S. Environmental Protection Agency lists antimony bromides among substances that may harm aquatic life or pose chronic dangers to people exposed daily. Plating shops and recycling centers must handle all brominated flame retardants with strict safety plans. Cleaner production lines, better worker training, and transparent labeling for end consumers keep antimony applications on the responsible side.
Charting a Safer Industrial Future
Industry experts aim to develop new, less hazardous flame retardants and glass additives but still rely on antimony tribromide for now. Research points to greener formulas, including phosphorus-based chemistry and advances in nanotechnology, that serve the same purpose with less long-term risk. From a worker’s viewpoint, updated protocols, smart ventilation, and strict waste management bring chemical processes into the 21st century. Safer cities, homes, and jobs come from better science and honest workplace practices.
| Names | |
| Preferred IUPAC name | Tribromostibane |
| Other names |
Antimonous tribromide Antimony(III) bromide Tribromantimony Antimony bromide |
| Pronunciation | /ænˈtɪməˌni traɪˈbroʊmaɪd/ |
| Identifiers | |
| CAS Number | 7789-61-9 |
| Beilstein Reference | 3589869 |
| ChEBI | CHEBI:30443 |
| ChEMBL | CHEMBL1201576 |
| ChemSpider | 21171060 |
| DrugBank | DB13804 |
| ECHA InfoCard | ECHA InfoCard: 100.028.261 |
| EC Number | 215-729-3 |
| Gmelin Reference | 82020 |
| KEGG | C14457 |
| MeSH | D001166 |
| PubChem CID | 24403 |
| RTECS number | BR1225000 |
| UNII | SY7Q9UCG4D |
| UN number | UN1436 |
| Properties | |
| Chemical formula | SbBr3 |
| Molar mass | 445.37 g/mol |
| Appearance | White to gray granules or crystalline solid |
| Odor | Odorless |
| Density | 4.15 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.78 |
| Vapor pressure | 0.4 mmHg (150°C) |
| Acidity (pKa) | -2.1 |
| Basicity (pKb) | 4.46 |
| Magnetic susceptibility (χ) | -78.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 2.253 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | SbBr3: 342.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -88.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -392.9 kJ/mol |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | P201, P261, P264, P270, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-0-NA |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1000 mg/kg |
| NIOSH | SA1960000 |
| PEL (Permissible) | PEL: 0.5 mg/m3 |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Antimony trichloride Antimony trifluoride Antimony triiodide Bismuth tribromide Arsenic tribromide |

