Antimony Potassium Tartrate: A Chemical With a Long Shadow
Looking Back: Historical Development
Antimony potassium tartrate shows up in the footnotes of pharmaceutical history, and every time it does, it carries lessons about the double-edged nature of technological progress. In the 17th and 18th centuries, doctors searched for ways to treat diseases that we now manage with antibiotics or vaccines. Antimony potassium tartrate, known then as “tartar emetic,” offered a kind of hope—though often a dangerous one—in treating fevers and certain parasitic diseases. In places from colonial Europe to parts of Asia, this compound built a reputation for both healing and harm. It served as one of the mainstream approaches for schistosomiasis into the mid-1900s because few alternatives worked better. As with many chemicals, success demanded a careful eye on dosing and observation, because the line between therapy and poison in this field is razor thin. Even after safer drugs arrived, the story of antimony potassium tartrate forms part of the backbone in the evolution of medicines—like a time capsule, it shows scientists’ readiness to push boundaries for a solution, even at high risk.
Product Overview and Identity
Known by quite a few synonyms—tartar emetic, emetic tartar, antimonyl potassium tartrate—this compound’s range of names shows how chemistry gets tangled with language and tradition. Sometimes, communities or labs referred to it simply as “antimony tartar.” What matters for science is not only what you call it, but how you use it, and what knowledge guides that use. Anyone who’s cracked open an old chemistry textbook or read about 19th-century medicine will spot antimony potassium tartrate as a recurring compound, popping up in early pharmaceutical manufacturing and in some analytical lab procedures. As people refined production process, they landed on more reliable ways to obtain it in crystalline or powder form, adjusting to scientific advancements as well as stricter rules for chemical handling. Old and new, the names stick around—proof of the compound’s lingering influence.
Physical & Chemical Properties
Antimony potassium tartrate doesn't look threatening—a white, crystalline substance, it blends into the background of a lab shelf. Looks deceive, especially here. With a molecular structure that binds antimony to tartrate ions, this chemical exhibits good solubility in water and high reactivity in acidic environments. Anyone who has worked in a chemistry lab will remember the way humidity or heat can affect substance handling, and potassium antimony tartrate is no exception. It melts at a moderate temperature and dissolves relatively easily, making it suitable for applications that require a consistent solution. The substance’s density and water solubility mean that proper storage plays a bigger role than many would guess, especially where careless handling leads to contamination.
Technical Specifications & Labeling
Care with chemicals starts the moment you read a label. Antimony potassium tartrate typically comes with thorough technical documentation, spelling out details such as molecular weight, empirical formula, level of purity, and special storage recommendations. These details help prevent mistakes in dosing or handling. Regulatory standards demand clear hazard pictograms, storage instructions, and warnings, mainly due to known toxicological risks. My own experience with laboratory chemicals taught me never to trust a faded or incomplete label, especially when the risks include acute toxicity or environmental contamination. In teaching or industrial settings, strict labeling anchors safety, helping both experienced and new users avoid costly errors.
Preparation Method
Preparation of antimony potassium tartrate requires more than routine mixing—this isn’t a compound for amateur chemistry. Manufacturers start with antimony trioxide and react it with potassium hydrogen tartrate in hot water, a process that relies on full control over temperature and purity of reactants. The solution crystallizes after cooling; the crystalline solid is then isolated, washed, and dried. Variations in the method reflect changes in available starting materials, scale of production, and prevailing safety standards, but the core principle—strict control to prevent toxic dust or volatilized antimony—remains constant for anyone serious about safety.
Chemical Reactions & Modifications
As a chemical reagent, antimony potassium tartrate finds use in redox reactions and as a catalyst in some organic syntheses. Its ability to act as an oxidizing or reducing agent, depending on conditions, makes it valuable in qualitative analysis, especially in the detection of certain ions. Modifications involving substitution in the tartrate moiety or further coordination with organic ligands have been explored to improve its chemical stability or reduce its toxicity. Researchers sometimes tweak formulations in search of safer alternatives, often driven by tightening regulations and a better understanding of chronic toxicity.
Synonyms & Product Names
Names like “emetic tartar” or “antimonyl potassium tartrate trihydrate” still turn up in medical history papers and laboratory stock lists. Pharmaceutical catalogues in the early 20th century listed it as “tartar emetic USP” or similar, and regulatory guidelines today use its systematic nomenclature—potassium antimony(III) tartrate—to reduce ambiguity. These multiple names reflect regional, disciplinary, and historical differences, which can create confusion unless proper context is given. Clarity matters; I’ve seen researchers lose time chasing synonyms through old papers, a reminder that language in science needs precision for safety and reliable results.
Safety & Operational Standards
No amount of transparency in documentation can make handling antimony potassium tartrate risk-free. Acute exposure risks include severe gastrointestinal distress, cardiac complications, and organ toxicity. Regulators have set workplace exposure limits, but the safe route involves personal protective equipment, solid training, and strict adherence to storage protocols. Laboratories use fume hoods, eye protection, and gloves to prevent inhalation and skin contact. Disposal rules follow hazardous waste guidelines because antimony compounds can pose environmental dangers, persisting in soil and water. I’ve seen too many near-misses with poorly stored chemicals; regular audits and clear procedures are what keep accidents from turning into tragedies. Safety culture depends on leadership setting an example at every level.
Application Area
Antimony potassium tartrate once had a larger presence in the pharmaceutical world. Its most infamous use: an antiprotozoal agent, mainly for treating schistosomiasis and leishmaniasis, diseases that threatened communities with few medical resources. Hospitals and clinics phased out its use as safer drugs took over, but the compound’s story did not stop there. Chemists and biologists kept it around as a reagent in labs, often for analytical determinations, including investigations in materials science or the quality testing of other chemicals. Its catalytic and analytical applications extend beyond medicine—for example, in textile processing and as a mordant in dyeing. My encounters with this chemical came mostly in teaching labs, where instructors showed how easily its properties rendered it both useful and hazardous, blending the line between learning and practicing real-world risk management.
Research & Development
Academic and industrial researchers continue to re-examine compounds like antimony potassium tartrate, especially in the context of green chemistry and toxicology. Ongoing work looks at developing derivatives with lower toxicity or replacing this compound in cases where better alternatives exist. Periodic reviews in research literature highlight the balance between preserving valuable chemical properties and achieving modern safety standards. There’s a push in analytical chemistry to adopt less hazardous reagents without losing accuracy or efficiency. The struggle to modernize these long-established practices touches on broader debates in science policy—when should history give way to innovation, and how to achieve that without erasing what scientists have learned from mistakes and successes alike.
Toxicity Research
The toxicity of antimony compounds, including potassium tartrate, draws increasing attention from toxicologists, who point to both acute poisoning and the risks of chronic exposure. That reality shaped medical emergency protocols; in the past, overexposure led to cases of vomiting, cardiac arrhythmias, and even death. Modern research now explores how such compounds affect the environment, entering water systems or accumulating in soils, with evidence showing slow but troubling patterns of bioaccumulation. Regulatory agencies use animal studies as well as epidemiological data to develop better occupational standards. For those of us who have seen the effects of poor chemical management, these guidelines represent real progress but also a stern reminder: some tools carry legacies far beyond their initial use.
Future Prospects
Few expect antimony potassium tartrate to return to the center of pharmaceutical practice, but its legacy continues, especially in academic research and regulatory policy. Emerging green chemistry movements see compounds like this as models for what to avoid or reformulate, pushing for reagents with lower environmental and human health risks. Research into antimony alternatives and safe disposal methods gains steam, paralleled by tighter workplace safety laws worldwide. In my view, progress will come from combining historical knowledge with new technology, investing in education about chemical dangers, and keeping alive the memory of mistakes made along the way. The compound’s story isn’t just a chemical equation or an entry in a reference manual—it's a reminder that every tool can turn into a hazard if care slips for even a moment.
What are the common uses of Antimony Potassium Tartrate?
Old Chemistry, Ongoing Roles
Once you’ve set foot in a chemistry lab, the sharp scent and deep color of antimony potassium tartrate—often called tartar emetic—stays in memory. This compound played a big part in clinical and industrial history. It’s no longer common in medicine, but the lessons from its uses matter, and concerns over safety still linger.
Lab Testing and Research
Antimony potassium tartrate takes the spotlight during chemical experiments in university labs. For anyone who’s worked on analytical chemistry projects, it shows up as a reagent, especially in redox titrations. It helps scientists determine small amounts of certain ions in water or soil—a role that keeps showing up in environmental science. Its predictability and sharp color changes give measurable results. Anyone curious about water quality or soil contamination will run into this compound in a report or a sample bottle.
Veterinary Use and the Changing Landscape
Older textbooks talk about tartar emetic as a treatment for parasitic infections in animals. Livestock, especially in places with limited options, used to get doses to fight off worms and other parasites. Reports from the early twentieth century describe these methods as standard. Over time, safer and more effective drugs replaced it, but some small veterinary clinics in developing regions still rely on these old-school compounds in tough situations.
Role in the Textile and Leather World
Factories that process animal hides or color textiles lean on antimony potassium tartrate for its mordant properties. Anyone who’s visited a tannery or textile plant might remember the pink and purple tones left behind on batches of cloth and leather. Here, tartar emetic helps dyes stick and last through years of use and washing. Artisans and manufacturers, especially outside of high-tech centers, keep older recipes with this compound because they know how it reacts, and they trust those results.
Emetic History and Modern Medicine
Years ago, people at hospitals or clinics reached for tartar emetic to cause vomiting in poison emergencies or to treat certain kinds of schistosomiasis. Patients who tried these treatments probably remember powerful nausea and side effects. These days, the risks—from severe vomiting to potential toxicity—drove doctors toward safer options. But it helps to remember how desperate situations shaped medicine, and how antimony potassium tartrate often stood at the edge of last resort in older hospitals.
Hazards and the Push For Safer Alternatives
People who’ve handled antimony potassium tartrate, myself included, respect its dangers. Mishandling can mean tremors, organ damage, or worse. The World Health Organization lists compounds like these among hazardous materials needing strict control. Research by groups like NIOSH shows how these risks shaped handling protocols for both workers and the environment. Industries transitioning to less toxic materials show what’s possible once people factor in long-term safety for both humans and the earth.
Seeking Solutions
Switching over to safer alternatives changes labs, farms, and factories. While some researchers and craftspeople rely on old habits, training on updated protocols and sharing safer substitutes make progress possible. Open conversations—without hiding legacy uses—let a community weigh risks and pick smarter options. People in industry or health who push for these changes show leadership by caring for the next generation instead of just repeating the status quo.
What are the potential health hazards of handling Antimony Potassium Tartrate?
A Closer Look at Risks in Daily Handling
Antimony potassium tartrate has found a place in labs and industry workspaces, yet its dangers often hide in plain sight. Anyone who has worked with chemical reagents knows how easy it is to underestimate risks during routine handling. Over years spent in labs, I’ve seen more people worried about acids or solvents than about this white, crystalline powder. Yet, mishandling even small amounts can cause real harm, especially if habits slip into carelessness.
Routes of Exposure and What Happens
Skin isn’t a reliable barrier against this compound. Dust can sit on gloves or cuffs, only to be carried off onto the skin or even food. I’ve watched a colleague develop skin rashes from not changing gloves after a spill they brushed aside as harmless. Skin absorption or frequent contact sets up irritation and even systemic poisoning, depending on how often and how much someone gets on themselves. Eyes burn fiercely if exposed, and you don’t forget the redness or pain afterwards.
Breathing in dust kicks off headaches, coughing, and shortness of breath. Chronic inhalation becomes a bigger concern. Some old reports and hospital archives document persistent lung effects after regular exposure. On the rare occasion someone swallows a small amount—from an unwashed hand or a contaminated bottle—the results are much nastier. Abdominal pain, persistent vomiting, diarrhea, then dehydration. Severe cases run the risk of collapsing the circulatory system. Doctors look for irregular heartbeats and kidney trouble soon after poisoning. This isn’t a minor irritant; even acute exposure triggers noticeable symptoms.
Long-Term Health Dangers
Long-term work around antimony compounds comes with its own reputation. Industrial hygiene texts trace links between repeated exposure and problems with the liver and kidneys. Some studies worry about the heart too; those exposed at work over months show changes in electrocardiograms. The International Agency for Research on Cancer classifies antimony trioxide as possibly carcinogenic, reflecting biological uncertainty about the long-term risks. Potassium tartrate combines both the antimony and the chemical’s ability to dissolve readily in water, so its body absorption matters even more when people fail to take precautions.
Toward Safer Handling: Recommendations That Work
Safety won’t come from rules alone—it takes personal vigilance. Using gloves and lab coats matters, but regular hand washing, especially before meals, kept many of us out of the medical office. Clean-up habits become a big deal: use a HEPA vacuum for powder spills, never your bare hand or an old brush. Fume hoods should always stay on during use, not just during weigh-ins, because airborne particles linger longer than most think. Splash goggles, not just safety glasses, keep the eyes irritation-free.
Training and reminders help bridge knowledge gaps. In shared labs, reminders stapled near weighing stations boost compliance. Easy access to Material Safety Data Sheets makes a difference—people actually use them if copies aren’t locked in drawers. At the organizational level, substituting antimony compounds with safer alternatives whenever possible cuts risks off at their source. I’ve seen some chemistry groups shift toward less hazardous reagents without sacrificing results.
Respect for the Material and for Colleagues
Taking antimony potassium tartrate seriously speaks to the wider health culture in any workplace. Treating every chemical with respect protects not just the user, but everyone else who walks through the door. I trust experience—and the lessons of past medical cases—more than luck. Rapid emergency response can make a big difference too, because early symptoms escalate quickly. Encourage quick action, and you keep small mistakes from turning into tragedies.
How should Antimony Potassium Tartrate be stored safely?
Understanding the Risks
Antimony potassium tartrate doesn’t draw much attention in the news, but it deserves a close look, especially in labs and storage rooms. Handling chemicals often gets brushed aside as routine — I have seen this too often, and it brings trouble more times than not. This compound shows up in lab reagents and old-school medical settings, but its toxicity presents a real hazard: every spill and careless mistake creates the real chance for harm. Swallowing or breathing in dust or vapors can lead to nausea, cardiac issues, and in some cases even death. There’s no reason to court these risks, when simple steps can make all the difference.
Practical Steps for Storing the Compound
I always kept Antimony potassium tartrate in a locked chemical storage cabinet, spaced well away from acids, combustible materials, and anything that could spark a reaction. It takes more than just tossing the jar on a shelf — a solid glass container with a tight-fitting lid stops moisture and keeps the air free from dust that can trigger toxic exposure. I learned early on to label containers as clearly as possible, using both the full chemical name and prominent hazard warnings, since too many labels read like code to tired eyes at the end of a shift. There’s an old joke about ‘mystery bottles’ forgotten in the corner, but no one ever laughs when someone accidentally uses or moves one of these without knowing what’s inside.
Control of Temperature and Humidity
Most chemicals, Antimony potassium tartrate included, keep best at room temperature with as little humidity as possible. Damp air causes clumping, releases more dust, and speeds up the breakdown of chemicals — this is where headaches begin for both safety and the quality of the results in the lab. I always tell new lab techs to avoid storing chemicals near sinks, windows, or any area prone to leaks and high moisture. The few extra steps it takes to stash hazardous chemicals in a dry, stable spot can prevent poisonous fumes and exposure later.
Personal Protection: The Human Factor
Proper handling goes hand in hand with proper storage. Gloves, goggles, and lab coats are standard, but they need to be right by the storage site to keep anyone handling the compound from skipping protection. I made it a point to run quick spot checks for damaged gloves and worn-out safety glasses — many people cut corners when no one is watching, but a few minutes here beats a lifetime dealing with chemical burns or chronic health problems. Beyond the gear, having a written policy on spill response with easy access to spill kits and dedicated disposal containers keeps emergencies small rather than disastrous. Training can’t be a one-off thing, either. Regular reminders and drills build habits that stick, even under stress.
The Importance of Policies and Oversight
Labs and schools must review storage practices as part of routine safety audits. Some of the worst situations I’ve seen started with good intentions but fell apart thanks to forgetfulness and a sense of routine. Checklists, labeling rules, and strict sign-out procedures create a paper trail that helps spot mistakes before they cost someone their health. The reality is, a chemical like Antimony potassium tartrate will never be completely safe, but doing the basics right — every time — keeps the workplace far safer. Government guidelines, safety data sheets, and institutional memory (from those who have learned the hard way) all support safer storage habits, which pay off every day the compound stays where it belongs, sealed and controlled, well out of reach of trouble.
What is the chemical formula and structure of Antimony Potassium Tartrate?
The Facts on Antimony Potassium Tartrate
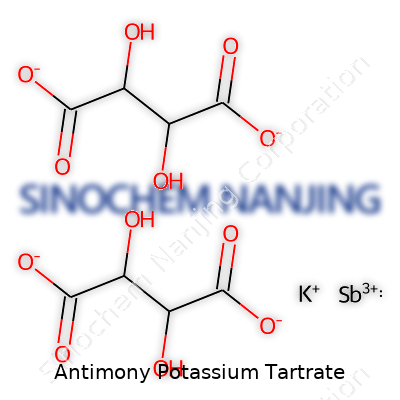
Some chemicals carry strange names, and antimony potassium tartrate stands out for both its mouthful of a title and its history in science and medicine. Chemists often call it “tartar emetic”. Its formula—K2Sb2(C4H2O6)2·3H2O—reflects just how complex things get even with elements that feel basic in high school chemistry. Once you piece the letters and numbers together, you spot potassium (K), antimony (Sb), and tartrate, which grows out of tartaric acid. Water rounds out the structure as a hydrate, giving extra stability and making the crystals look clear and shiny.
Putting the Molecule Together
The science behind this compound isn’t just good for trivia night—it stands as a glimpse into just how creative chemistry gets. Two antimony atoms snuggle up with two tartrate groups, all balanced by two potassium ions. Each tartrate group anchors the metallic antimony, keeping it locked in a specific shape. Throw in water molecules, and you have a structure that packs stability and helps the powder dissolve easily in liquids. I remember using a model kit for similar compounds in chemistry labs. Getting the bonds right made everyone’s head spin—organic parts bend and loop, metals sit locked in position. Tartaric acid forms the backbone, with loads of carbon and oxygen forming rings. Antimony, as a metal, brings density, while potassium’s main job is to keep the whole structure electrically balanced.
Historical Impacts and Modern Concerns
I read stories of old-time medicine where antimony potassium tartrate entered as a common remedy despite its risks. Used in the fight against parasites like schistosomiasis, it once saved lives. People didn’t have gentler drugs, so doctors gambled, trading a dangerous substance for a shot at survival. That risk shows just how much chemistry pushed medicine forward, for better or worse. We now know this compound can wreck the heart, liver, and kidneys if misused. Today’s doctors left it behind for safer options, and in the lab, handling requires real care—nobody wants to mess with antimony without gloves and a hood. After handling, thorough hand washing and proper chemical waste disposal come second nature in a good lab.
Why Chemical Knowledge Still Matters
Curiosity about molecular structure isn’t only for students. Knowing chemical formulas and shapes lets people working in labs, hospitals, and even factories keep risks in check and understand just what they’re dealing with. Accidental exposure has consequences; health and safety training drills the importance of knowing your compounds. If a spill happens, you don’t want someone guessing how antimony and tartrates might react. Environmental risk looms for improper disposal, as antimony leaches into water and soil.
Possibilities for Improvement
More transparency in chemical manufacturing and better labeling make a world of difference. My time in research taught me that even basic education about chemical structures can prevent accidents and environmental harm. Digital databases, QR codes on bottles, and interactive molecular models could help workers learn on the fly. Regulators, like the EPA and OSHA, offer guidance, but real change starts where people use chemicals every day. Better substitution for hazardous compounds and clear communication channel the promise of chemistry without repeating mistakes from the past.
What precautions should be taken when disposing of Antimony Potassium Tartrate?
The Dangers Lurking in a Laboratory Staple
Antimony potassium tartrate doesn’t usually get headlines, but anyone who has worked in a chemistry classroom or a factory knows that even the most ordinary compounds can hide serious risks. I learned this lesson the tough way in my early days working as a lab assistant. One oversight, such as ignoring the small print on a chemical’s data sheet, can end up causing more than a mess—it can put people and the environment in danger. Antimony compounds, in particular, pose both acute and long-term health threats. Inhaling or ingesting dust particles can cause headaches, vomiting, and even organ failure over extended exposure. Mishandled disposal often causes toxic buildups in water and soil, threatening wildlife and downstream communities.
Why Dumping Doesn't Cut It
Some folks downplay the hassle, thinking the occasional small spill or improper rinse won’t matter much. That’s far from the truth. Antimony in waterways can disrupt aquatic life and enter the food chain, eventually ending up on dinner plates. My friend who works at a waste treatment facility once described discovering unmarked barrels that set off alarms—not just with detectors, but also with the local health department. Nobody wanted that call at 2 AM, especially once the legal department stepped in. Stories like this underline the point: quick fixes on disposal often come back to haunt.
Smart Steps for Disposing of Antimony Potassium Tartrate
Safe disposal starts with small habits—label everything, never mix unknown chemicals, and keep updated records. In practice, that means securing waste in dedicated, corrosion-resistant containers. A sturdy polyethylene drum with a screw-top works better than the battered paint can I once saw used as a “temporary” fix. Always store waste containers in marked areas, away from ordinary trash, and never near drains. Antimony shouldn’t wind up in municipal landfills or run down the sink. Instead, collection by a licensed hazardous waste hauler must be the default. It’s tempting to cut corners, especially if budgets run tight or management prioritizes speed over safety. That shortcut always carries bigger consequences later on.
Personal protective equipment—gloves, goggles, fitted masks—should never get skipped. Even after waste gets transferred out of your hands, everyone in the chain relies on your diligence. Make sure every container sent out has a clear label. In labs and factories, regular training sessions help staff understand why these routines matter. I once saw how a team’s yearly safety refresher cut down on incidents and raised everyone’s awareness about even the smallest risks.
The Call for Better Systems
Accidents and pollution often trace back to poor planning or lack of resources. Governments could step in with grants or incentives to help smaller outfits access proper disposal services. Regular audits and strict penalties for illegal dumping remain important, but so does educating newcomers and experienced staff alike. Clear signage, better waste logs, and partnerships with credible disposal contractors turn what feels like extra work into everyday good practice.
The goal isn’t just following rules. Protecting colleagues, the public, and the environment should become a point of pride. As someone who learned through trial and error, small steps—attention to containers, reluctance to improvise, solid training—really do pay off. Especially with chemicals like antimony, the best habit is treating every disposal like it could matter years from now, because sometimes, it does.
| Names | |
| Preferred IUPAC name | Dipotassium 2,3-dihydroxybutanedioate antimonate(III) |
| Other names |
Tartar emetic Potassium antimonyl tartrate Emetic tartar Antimony potassium tartrate trihydrate Antimonyl potassium tartrate |
| Pronunciation | /ænˈtɪm.ə.ni pəˈteɪ.si.əm tɑːˈtreɪt/ |
| Identifiers | |
| CAS Number | 28300-74-5 |
| Beilstein Reference | 4096806 |
| ChEBI | CHEBI:61330 |
| ChEMBL | CHEMBL1356 |
| ChemSpider | 18560 |
| DrugBank | DB14107 |
| ECHA InfoCard | 100.028.230 |
| EC Number | 231-555-9 |
| Gmelin Reference | Gm.2.205 |
| KEGG | D03212 |
| MeSH | D001166 |
| PubChem CID | 24856 |
| RTECS number | WT2625000 |
| UNII | M4I0D6VV5M |
| UN number | UN2694 |
| Properties | |
| Chemical formula | K2Sb2(C4H2O6)2·3H2O |
| Molar mass | 667.87 g/mol |
| Appearance | Colorless or white crystalline powder |
| Odor | Odorless |
| Density | 2.607 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.15 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.68 |
| Basicity (pKb) | 6.0 |
| Magnetic susceptibility (χ) | -35.5e-6 cm³/mol |
| Refractive index (nD) | 1.593 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 510.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2077.1 kJ/mol |
| Pharmacology | |
| ATC code | S01XA02 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes damage to organs. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P330, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 3-2-2-NA |
| Autoignition temperature | > 400 °C (752 °F; 673 K) |
| Lethal dose or concentration | LD50 oral rat 115 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 100 mg/kg |
| NIOSH | SN16500 |
| PEL (Permissible) | PEL: 0.5 mg/m³ |
| REL (Recommended) | 0.7 mg/kg |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Potassium Tartrate Sodium Tartrate Tartaric Acid Antimony Trichloride Antimony Triacetate |

