Antimony Lactate: Yesterday’s Curiosity, Today’s Chemical Workhorse
The Journey of Antimony Lactate from Alchemy to Modern Chemistry
Antimony compounds have never quite had the limelight like gold or copper, yet the story of antimony lactate runs deep through the veins of both historical industry and modern scientific exploration. Early European alchemists toyed with antimony’s glassy ores centuries ago, always just one clever experiment away from stumbling on compounds more useful than the mysterious black sulphides they coveted. As the nineteenth century dawned, developments in organic chemistry set the stage for the union of antimony with lactic acid, giving birth to antimony lactate—a salt neither as flashy as antimony trichloride nor as notorious as antimony potassium tartrate, but practical for coats, catalysts, and beyond. From German dyeworks to textile mills in Britain, those first batches of antimony lactate quickly found their roles in modifying fibers and processing finishes. The chemical quietly supported industry and medicine while sidestepping the bombast of more famous reagents. In many ways, this history paved the way for the careful approach chemists and safety officers take with antimony salts today.
Product Overview: What Sets Antimony Lactate Apart
Formed when antimony oxide takes a chemical stroll through lactic acid, antimony lactate emerges as a solution or crystalline powder, depending on how it is prepared. Unlike some antimony compounds that come with a severe hazard tag, antimony lactate offers a milder approach thanks to the buffering action of lactic acid. Its physical appearance—often a pale, cloudy liquid or white powder—masks the complexity of reactions going on at a molecular level. Instead of releasing aggressive fumes like antimony trichloride or causing violent outbursts like metallic antimony in contact with halogens, antimony lactate behaves in a controlled manner, dissolving smoothly in water or alcohol and bringing manageable reactivity to the table.
Getting to Know Its Properties and Character
Antimony lactate carries both the amphoteric nature of antimony and the mild acidity from lactic acid. This duality lets it take part in a variety of reactions without veering into unpredictable territory. It typically appears as a viscous liquid or crystalline powder, depending on humidity and preparation conditions. Its solubility in water stands out, making it much easier to introduce into chemical reactions or industrial processes compared to some salts that cling to beakers or settle out at the bottom of mixing tanks. The pH often skews mildly acidic, but not enough to pose a risk to common labware or skin on brief contact. One can smell a faint tang, more metallic than sour, enough to hint at its metallic roots without overwhelming the senses. High-purity forms tend to have a relatively narrow melting point and a consistency that supports accurate dosing for research purposes. All in all, what you get is a chemical that plays nicely during storage and transport, standing ready for action without unnecessary risks—or unnecessary surprises.
Technical and Labeling Standards: What the Details Reveal
Product specifications for antimony lactate tend to focus on antimony content, percentage purity, and water content. Regulatory agencies expect clear declarations of these elements on labels, both for the sake of workers’ safety and for downstream analyzers tracking heavy metal residues. The CAS number connects handlers firmly to the chemical’s unique identity, helping avoid confusion with lookalike salts or related antimonates. In the EU, the REACH framework asks for hazard statements concerning chronic toxicity, and classification aligns with global harmonized systems for chemical handling. Labels give users a quick but critical picture: how much antimony sits in the mixture, what potential hazards come with repeated contact, and what safety gear—gloves, goggles, masks—should guard workers’ health. Full compliance with GHS rules is not just red tape; those hazard icons on a drum signal to the next shift what lies inside and what preparation the material demands. Regular audits by chemical inspectors can turn up surprises in labeling compliance, so most large players make sure to stick rigidly to international standards rather than risk shipment delays or fines.
The Pathway to Synthesis: Putting Knowledge into Practice
Making antimony lactate involves a few deliberate choices, none of which need to reinvent the wheel. The method that stands up to repeat testing starts with antimony trioxide, which gets suspended in water to form a slurry. By slowly introducing lactic acid under gentle heat, one nudges antimony into a soluble salt state. Control of pH, temperature, and mixing speed separates an operator’s product from a bad batch full of unreacted lumps. Industrial reactors scale this up, using jacketed vessels or continuous flow setups that keep temperatures and concentrations steady over hours of production. Purification, most often by filtration and evaporation, clears out excess lactic acid and side products. The result leaves a relatively clean antimony lactate mixture, ready for bottle or barrel.
Chemical Reactions and Spinoff Compounds
Within a laboratory, antimony lactate transitions easily into new forms. Reacting with strong oxidizers can yield pentavalent antimony compounds, each with their own industrial quirks. Gentle heating can strip away the organic partner, turning antimony lactate back into oxides or leaving a residue of soft, white antimonic oxide. A base like sodium hydroxide nudges the salt into precipitation mode, pushing antimony into less soluble hydroxide forms. Lactic acid itself secures a stable grip on antimony, which prevents wild swings in reactivity. This chemical’s balance—neither too aggressive nor too passive—has made it a cornerstone intermediate for synthesizing other organoantimony salts, dyes, and specialty materials.
Names in the Field: Synonyms and Aliases
Chemistry’s tradition of inventing several names for the same molecule gets no break here. On supply lists, antimony lactate sometimes appears as antimony(III) lactate, lactic acid antimony salt, or simply antimonious lactate. In patent databases, the phrase antimony(III) 2-hydroxypropanoate turns up when international applicants try to cut through ambiguity. There’s a risk with all these synonyms—students and fresh researchers can lose track of which name links back to which test result. Solid communication from supplier to scientist offers a crucial path through the jargon. The key is focus: don’t get caught up in labels, keep your eye on chemical behavior.
Staying Safe: What Daily Handling Teaches Us
Anyone who spends time with antimony compounds develops a deep respect for both their utility and their health risks. Antimony lactate bottoms out with a lower toxicity than many of its cousins, but repeated or careless exposure escalates risks of skin irritation, respiratory distress, and chronic effects documented in heavy industry. Safety goggles and nitrile gloves mark the baseline, along with careful fume handling, especially if heating or mixing with acids. Waste must go to controlled facilities, not down the drain—environmental persistence looms large in regions with strict water quality goals. Safety data sheets sit open on benches and get signed off before every new process. Frequent monitoring for airborne dust and dissolved antimony cements a culture of safety that leaves no room for shortcuts.
Where and Why It Gets Used
Antimony lactate’s cycle of usefulness turns up most in flame retardant manufacture, polyester resin production, and specialized catalysts for textile finishing. Some dye processes depend on just this salt to lock pigments in synthetic fibers, supporting a global fabric market that prioritizes durability over disposability. Plastics processors add antimony lactate to improve flame resistance, keeping in line with consumer safety codes from Tokyo to Toronto. Medical researchers have also eyed antimony lactate as a vehicle for controlled delivery of antimony ions, but the trend has shifted away from heavy metal treatments in human health. In the classroom, advanced chemistry courses might introduce the salt as a gateway to organometallics, giving students a window into both lab safety and industrial insight in a single lesson. Sustainability-conscious designers keep an eye on how antimony slips into recycled plastics, and regulators periodically scrutinize its end-of-life environmental fate, inviting a back-and-forth between innovation and environmental caution.
Upstream in the Lab: Research and What’s on the Horizon
Wider chemical research teams look beyond the established uses of antimony lactate, puzzling over its coordination chemistry and its role in advanced catalysis. At conferences, poster sessions on green chemistry sometimes feature this salt as a springboard for new water-based syntheses, minimizing noxious byproducts. Instrumental methods keep growing more refined, offering sharper quantitative analysis of trace antimony in complex mixtures, tighter than what could be tracked a decade ago. Researchers in polymer chemistry envision antimony lactate as a tunable node, customizing both polymer structure and properties without breaking the bank. As new products and composites move toward biocompatibility and lower toxicity, the interplay between traditional antimony chemistry and green design philosophy keeps the research world guessing.
Toxicity: A Balanced Perspective from Field and Lab
No amount of industrial optimism erases the reality: antimony lactate belongs in the family of heavy metal compounds that demand long-term scrutiny. Epidemiological studies trace long-term exposure in factory workers to respiratory issues, gastrointestinal complaints, and subtle changes in liver function. In aquatic environments, antimony ions can linger and build up in water-dwelling life, underscoring the need for tight emission controls. Short-term toxicity lands well below that of more notorious antimony salts but doesn’t warrant complacency. Scientists probe for chronic thresholds that regulators and companies can use to retool exposure standards, and public health professionals push for transparency about any new industrial process that could add to cumulative environmental burden.
Looking Ahead: Antimony Lactate’s Place in Modern Industry
Industrial cycles never stop evolving, and the future for antimony lactate depends on a mix of scientific insight, regulatory push, and sustainable design priorities. The growing demand for flame-retardant fibers, recyclable plastics, and specialty catalytic materials all suggest that antimony lactate keeps its foothold in major supply chains worldwide. Resource constraints, shifting consumer demand, and environmental activism all put a premium on research for safer substitutes and closed-loop recycling approaches. Scientists keep searching for more benign, organic catalytic systems, yet nothing offers the same mix of cost, reactivity, and reliability in certain commercial settings as antimony lactate. Investment in greener manufacturing, improved worker safety training, and comprehensive toxicity studies all deserve priority, both for the health of today’s workers and the ecosystems downstream of tomorrow’s products.
What is Antimony Lactate used for?
What Draws Industry to Antimony Lactate?
Every time a manufacturer adds color to plastic, improves flame resistance, or even looks for a chemical catalyst to speed up certain reactions, someone in that chain probably thinks of antimony lactate. This chemical doesn’t land in headlines or consumer conversations, but its reach stretches across everyday products. I’ve watched labs tweak with dozens of compounds trying to enhance plastic safety, and antimony compounds never leave the shortlist.
Fire Safety That Makes a Mark
Fire retardants often sound like a dull subject until you see how plastics without them react in a burn test. Antimony lactate gets called up in plastic industries—especially where polyesters or PVC show up—because it works with halogens to slow flames. Take electrical installations, server rooms, or even the cladding material in public buildings—fire codes put the squeeze on what can be used, and antimony lactate helps manufacturers hit those marks. It’s not the sole fire shield, but it makes a difference in slowing the spread, buying those crucial seconds for people to react in case something goes wrong.
How Color Comes to Life in Plastics
Making colored plastics might sound like it’s all about dyes, but there’s more chemistry under the hood. In my experience working alongside techs creating batch after batch of colored polyesters, antimony lactate showed up as a trusted catalyst. Without it, the reaction called polymerization drags out, wasting time and energy. A small tweak with this chemical, and suddenly, production moves faster and more predictably. Plus, formulas are stable and the color holds up under sunlight or during recycling efforts.
The Textile Angle
If you’ve pulled on athletic wear or even sifted through office carpet tiles, polyester has been part of your day. Polyester producers lean heavily on antimony lactate during spinning processes. This compound delivers fibers that can resist heat and take up dyes evenly—a must for both quality and long-lasting color. On factory tours, I’ve noticed engineers reaching for this additive almost by instinct: it’s become second nature and the industry trusts its results.
Tackling Antimony Lactate’s Safety Concerns
Scrutiny tends to zoom in on any chemical used in bulk, and antimony-laced compounds attract that microscope. Official reports (like those out of the European Chemicals Agency) trace how much ends up in the environment or consumer products. Some studies show risks if workers inhale dust or if residues build up, so safety matters at every step. Responsible handling and updated filtration systems in factories go a long way. This creates an urge for more research into both human health and different disposal techniques—real issues that industry groups and regulators should prioritize.
Room for Smarter Chemistry
Alternatives do exist, but antimony lactate sticks around because it delivers reliable performance at a price other methods struggle to match. That said, the right move includes more investment in sustainable options. Bio-based polymers or non-toxic flame retardants hold promise, but scaling them up demands more R&D budgets. By learning from past over-reliance on certain additives, professionals can drive change that matches safety, environmental, and commercial needs. Keeping updated data, honest risk assessments, and stronger safety gear will protect both the people working hands-on and folks using finished products.
References:- European Chemicals Agency – Antimony compounds assessment
- International Chemical Safety Cards – Antimony Lactate
- Polyester production literature, American Chemical Society
What is the chemical formula of Antimony Lactate?
Digging Into Antimony Lactate
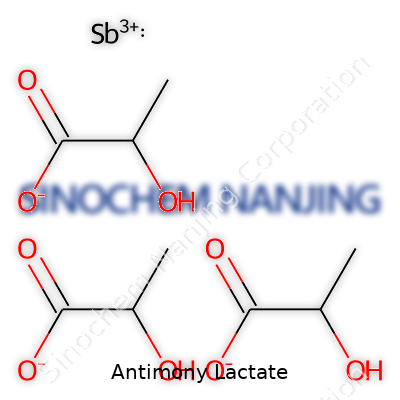
Science on your shelf often looks more complicated than it really is. Antimony lactate falls squarely into this camp. Its chemical formula, C6H9O6Sb, represents a bond between antimony—a soft, silvery metal found mostly in stibnite ore—and the byproduct of sour milk: lactic acid. Some industries may glaze over the details, but getting this formula right sets the stage for accuracy in so many areas, from manufacturing to research.
Why C6H9O6Sb Stands Out
Not all compounds behave the same way. Antimony, sitting just below arsenic on the periodic table, quickly gets mixed up in conversations around safety or toxicity. When merged with lactic acid, its properties shift and take on a different form than raw antimony or separate lactic acid would offer. Fitness powder labels, for example, tend to gloss over chemical distinctions, but miss even a single element, and you might end up with an entirely different substance—one with risks or lost benefits. According to OSHA guidelines, antimony compounds can present health hazards, so clarity about what exactly sits in a container helps keep everybody safe and well-informed.
The Real Implications for Industry and Health
Many modern plastics use antimony compounds—often antimony trioxide. But switch the ligand, or change just a slice of the molecule, and you influence everything from heat stability to toxicity profile. Antimony lactate finds its calling mostly in textile and polymer manufacturing, often acting as a catalyst. For those of us with hands in paints or chemical engineering, knowing exactly what’s in the mixture is non-negotiable—mistakes can cost money, health, or even lives. I learned early in my chemistry days that shorthand never stands in for accuracy. My old professor would dock a point for every stray letter in a chemical equation. It wasn’t about nitpicking; it was about training us for safety and sound results down the line.
Room for Solutions
Better labeling and education start with chemists and manufacturers. A well-printed chemical label does more than tick a regulatory box. It warns, instructs, and empowers folks to use, store, or dispose of a substance responsibly. For small labs, open access to verified chemical information can keep standards just as tight as the big names. Digital databases like PubChem or ChemSpider list compound details, including C6H9O6Sb for antimony lactate, so folks can double-check specifications before use.
Industry and research both move quickly, but setting strong habits around these foundations is long overdue. No one enjoys side-stepping standards, and users have a right to clear, precise information—especially when working with compounds that affect health or stability in their final products. Next time you pick up a container marked antimony lactate, don’t just squint at the name. Track the formula. Cross-check the health advice. Detail-driven practices stick, whether mixing up a batch in the lab or scaling up on the plant floor. In science, as in life, a single detail can be the difference between safety and risk, success and mishap.
Is Antimony Lactate hazardous or toxic?
What Is Antimony Lactate?
Antimony lactate shows up in the chemical industry as a salt containing antimony and lactic acid. Factories put it to work mostly as a catalyst for making plastics like PET. If you’ve used a clear plastic water bottle or handled synthetic fabric, chances are you’ve had a brush with the results of antimony compounds in action.
Understanding Its Hazards
People get nervous about antimony compounds, and for good reason. Antimony itself isn’t safe in large doses. In its pure form or in many salts, it brings risk. Breathing dust or fumes tied to antimony compounds can trigger lung issues, skin irritation or, over time, something far worse, such as damage to the liver or heart. Now, does that translate one-to-one with antimony lactate? Not quite. The lactate salt dissolves well in water, so it can move from containers to the environment easier compared to some other forms.
A deep dive into the data reveals that antimony compounds, including lactates, get classified as potential carcinogens. Chronic exposure means trouble. Workers who handle them—machinists, maintenance techs, or anyone in the plastic supply chain—end up with the most risk. Breathing in even low amounts, day-in-day-out, has been tied with lung problems, stomach pain, and even changes to blood chemistry.
Environmental Risks
Antimony compounds don’t stay put. When they wash off into waterways, fish and plants soak them up. The U.S. Environmental Protection Agency sets strict limits for antimony in drinking water for this reason. The science points towards bioaccumulation and risk to aquatic life. That includes ants, fish, and everything that relies on clean streams. It’s not just a worksite problem; heavy use at factories can mean low levels spread to nearby places, inching their way into the food chain and the soil.
What About Safe Handling?
Experience in chemical plants taught me one thing: you can’t slack on safety, not even for a day. Antimony lactate wants to stick to gloves or float through the air if spilled. Respirators, ventilation, and strong protective routines make all the difference. I’ve seen what happens when shortcuts get taken—itchy eyes, burns, coughing fits. Supervisors worth their salt invest in gear and enforce clean-up drills. Proper labeling, sealed storage, and routine waste checks set apart the safe shops from the sloppy ones.
Antimony compounds demand respect because even tiny amounts can cause harm long after you think they’re gone. Everyone—operators, environmental teams, and even folks outside the fence—benefits from careful handling and informed training.
Safer Alternatives and Solutions
A big trend now: companies looking for antimony-free ways to make plastic. Some replace it with titanium-based catalysts. These options need testing and buy-in, so progress moves slow. But reports show that strong interest from both regulatory agencies and consumers keeps pushing change.
Clear reporting and regular medical checks go a long way, too. Anyone working with antimony compounds deserves yearly health monitoring and honest conversations about the risk and steps taken to keep them safe. Factories who join industry safety groups or stay current on the latest science adapt faster and protect more people.
I’ve found that nothing beats a culture of transparency and education. The risks around antimony lactate can’t be ignored, but with the right steps, most problems aren’t inevitable. Cleaner replacements, good training, and environmental controls all add up.
How should Antimony Lactate be stored?
Understanding the Risks
Antimony lactate shows up in laboratories and certain manufacturing spaces because it acts as a catalyst and stabilizer. I’ve seen folks treat it with less respect than it deserves, probably because it doesn’t come with the kind of warnings plastered across something like sulfuric acid. Still, this stuff brings real risks, both to your health and to your workplace safety. If you’ve ever dealt with the headache that follows a chemical leak, you already know the value of storing chemicals right from the start.
Storage: Keeping People and Product Safe
Leave antimony lactate in an open container, and you’re risking contamination or accidental contact. Most suppliers deliver it in tightly sealed drums or plastic jugs, and that’s no accident. Even a trace of moisture sneaking in can change the chemical’s properties, making it less reliable for any process that counts on consistency. A friend working in a textile lab once found out the hard way that a batch left open ended up costing thousands in wasted fabric and delays.
Antimony compounds do not tolerate high humidity or abrupt temperature changes. Keep the area cool and dry. I’ve lost count of how many labs use storage rooms with basic air conditioning to handle seasonal shifts. Even minor shifts cause degradation. Those molecules aren’t forgiving — stability drops fast.
The Human Factor: Health and Compliance
Let’s talk health. Antimony compounds, including the lactate, irritate skin and eyes. Inhaling dust or vapor leaves your lungs worse off. Without gloves, eye protection, and a decent lab coat, it’s easy to slip up. I remember seeing a technician end up with a nasty rash just from handling bottles that had spilled traces on the outside. Rules about personal protective equipment exist for a reason, not just to tick boxes.
Spills on the lab bench or floor make a mess, but they also mean wasted money and hazardous cleanup. I’ve seen facilities fined for failing inspections, especially around hazardous chemical storage. Regulators pay close attention to labels, secure lids, and proper segregations by class of chemical. In some places, antimony lactate sits in the “hazardous but not acutely toxic” category, which is only safe if handled correctly.
How a Good System Prevents Problems
Label every container clearly. Even if you’re the only one using it, a detailed label avoids mistakes if someone else steps in. I’ve been in more than one work room where a missing label led to hours wasted tracking down the mystery substance.
Keep containers off the ground, away from sunlight, and in dedicated cabinets. Chemical safety cabinets cost more, but they earn their keep the first time a container leaks or breaks. Shelves with secure lips or bins help keep things upright, and nobody wants to mop up a pool of antimony lactate.
Planning for the Unexpected
Every storage area should hold spill cleanup supplies. At a minimum, absorbent pads and neutralizing agents belong close at hand. Make sure a detailed safety data sheet hangs nearby or sits in an easy-to-find binder. Training everyone who works near these materials saves trouble — I’ve run drills where the most junior staff handled an emergency faster than their managers, simply because they knew what to do.
Antimony lactate may not grab headlines, but its potential hazards demand respect. Smart storage means fewer accidents, less waste, and better compliance. No shortcut matches the peace of mind that comes from putting the basics in place.
What are the safe handling procedures for Antimony Lactate?
Looking at the Risks
Antimony lactate isn’t something most people keep in their cabinet. It’s a chemical used in specialty industries, like producing flame retardants or certain plastics. Stories of mishaps and exposure don’t make big headlines, but those working around it know you can't let your guard down. My years in industrial settings hammered in the lesson: taking shortcuts with chemicals catches up to you. Even small missteps create lifelong problems, especially with substances that fly under the radar.
Personal Protective Gear: The First Line of Defense
Gloves and goggles should never gather dust. Simple latex or nitrile gloves usually block skin contact well, since antimony lactate can irritate or burn. Splashes toward the eyes create a different danger, and safety goggles keep accidents from turning into permanent injuries. I’ve watched people skip face shields to “get it done fast”—regret always followed. Investing a few minutes in protective gear never once felt wasted to me.
Clean Work Spaces Save Lives
Spills happen more often than manufacturers admit. Concrete flooring absorbs chemicals, and improper ventilation allows fumes to linger. Good ventilation systems make a shop bearable and prevent airborne exposures. Vent hoods and open-door policies—literally letting air move through—make a real difference. Regular cleaning routines also prevent buildup and cross-contamination between chemical batches. From what I’ve seen, those who keep things tidy have fewer illnesses and fewer “close calls.”
Proper Storage Matters More Than Labels
Labels matter, but so does storing containers somewhere dry and cool. I once ran into an issue at a small factory where heat caused leaky drums—the damage cost more than the year’s supply of antimony lactate. Leak-proof, sealed containers set on non-porous shelves beat cardboard boxes on concrete every time. Segregation from food, acids, and oxidizers cuts risks nearly in half. Even the smallest leaks require action right away; one mop-up with proper absorbent materials will prevent a month of headaches down the line.
Training Isn’t an Afterthought
No one learns safe chemical handling from a pamphlet. In the real world, hands-on training, frequent safety meetings, and updated protocols built real workplace safety. People remember drills and checklists. From the stories I hear, even the most experienced workers stay out of trouble by reviewing emergencies regularly—no one feels above a refresher course, and walking through the “what-ifs” keeps everyone on the same page. Practical, hands-on rehearsals stick with people longer than reading a document on a screen.
Waste Disposal: A Crucial Step
Throwing antimony lactate down the sink will cost anyone their business, not just their health. Safe disposal means sealed, clearly marked containers and pickup by licensed professionals. I’ve witnessed fines and environmental damage from neglecting this step, so following EPA and local rules avoids a world of legal trouble. Recordkeeping also protects workers and companies years down the line. Tracking every ounce disposed builds transparency, which becomes handy during audits or after unexpected incidents.
Final Thoughts
Antimony lactate presents real risks, but a culture built on respect, practical knowledge, and real safety habits protects everyone involved. Experience shows that fixing problems early, using proper gear, and keeping spaces clean benefit both workers and the business bottom line. Taking extra steps saves lives and keeps the environment cleaner for everyone.
| Names | |
| Preferred IUPAC name | Antimony(3+) tris(2-hydroxypropanoate) |
| Other names |
Antimony(III) lactate Lactic acid antimony(III) salt Antimony trilactate |
| Pronunciation | /ˈæn.tɪˌmoʊ.ni ˈlæk.teɪt/ |
| Identifiers | |
| CAS Number | 18621-17-5 |
| Beilstein Reference | 1322109 |
| ChEBI | CHEBI:91233 |
| ChEMBL | CHEMBL1201580 |
| ChemSpider | 120910 |
| DrugBank | DB13520 |
| ECHA InfoCard | ECHA InfoCard: 03-2119956954-33-0000 |
| EC Number | 247-384-1 |
| Gmelin Reference | 9302 |
| KEGG | C18636 |
| MeSH | D017700 |
| PubChem CID | 166830 |
| RTECS number | AH8400000 |
| UNII | YUH8C7FD6O |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C6H9O6Sb |
| Molar mass | 410.90 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.85 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.49 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.5 |
| Magnetic susceptibility (χ) | -78.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.56 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | Std molar entropy (S⦵298) of Antimony Lactate: 445.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | no data |
| Pharmacology | |
| ATC code | V03AB05 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin and eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P501 |
| Lethal dose or concentration | LD50 oral rat 990 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1600 mg/kg |
| NIOSH | SN45200 |
| PEL (Permissible) | 0.5 mg/m3 |
| REL (Recommended) | 0.5-3.0% |
| Related compounds | |
| Related compounds |
Antimony(III) oxide Antimony(III) acetate Antimony potassium tartrate |

