Antimony Diisopropyldithiophosphate: Past, Present, and Beyond
Origins and the Long Road to Modern Antiwear Additives
Industrial chemistry has seen eras defined by quiet revolutions. Among them, the development of metal dithiophosphate complexes drove huge progress in lubrication over the twentieth century. In the early decades, engineers faced early wear and corrosive breakdown. They needed new additives to protect machinery, especially in automobiles and heavy industry. Researchers began tuning organophosphate ligands onto different metals, discovering that antimony compounds could stop metal-on-metal contact and cut wear dramatically. Out of this creativity grew Antimony Diisopropyldithiophosphate, a specialty chemical with a mouthful of a name but a simple reputation in part shops and lubricant supply rooms: it keeps things running longer.
What Sets This Additive Apart?
The chemistry behind Antimony Diisopropyldithiophosphate isn't talked about outside research labs, though it’s straightforward in form. This molecule carries both antimony, a metallic element with a storied industrial pedigree, and organophosphate groups branched with isopropyls. This balance lets it dissolve well in oil, acting as both a wear-fighter and a secondary antioxidant. Its chemical neighbors in the same category often partner zinc, but antimony brings a different suite of effects, with lower reactivity compared to traditional zinc additives. Viscous, amber to brown, with a faint musty odor matching many metal dithiophosphates, this liquid resists hydrolysis better than early variants, a much-appreciated trait if you’ve ever dealt with oil breakdown.
How Manufacturers Get from Raw Elements to Finished Antiwear Agent
The process churns through antimony trioxide, transforming it slowly in reactors with diisopropyl phosphorodithioic acid under heat and the presence of solvents. Mixing is tricky. Too much heat and the additive turns sludgy; not enough and it barely reacts. Operators watch pH and measure sulfur, tuning each batch in real time—a messy art more than an exact science. Some labs purify these mixtures, chasing out metallic grit and excess sulfur, leaving behind a clear, potent concentrate destined for blending plants. Each batch marches out under restrictive environmental and workplace standards; spills pose cleanup challenges, and waste streams get tested before disposal.
What Happens Inside the Engine: Chemistry in Action
Pour this additive into motor oil, and the story changes fast. High temperatures and loads in engines trigger reactions between iron on engine parts and the additive. A protective layer forms, almost glassy—just a few molecules thick—stopping direct contact. This film also slows the march of oxidation, meaning oil lasts longer and keeps working beyond its typical life. In extreme conditions, such as racing or diesel trucks under punishing loads, this difference shapes whether equipment hits service intervals or faces sudden failure. To put it plainly, the chemistry turns disaster into routine maintenance, and that reliability is hard to overstate.
The Safety Puzzle and Ongoing Challenges
Anyone handling antimony compounds knows the stakes. Inhalation, accidental contact, and environmental spills run real risks. Chronic exposure to antimony has drawn scrutiny for respiratory and cardiac impacts. Regulatory groups warn about aquatic toxicity—freshwater algae and fish can't handle even small doses dumped down the drain. Workers keep to protective suits and ventilation, labs audit air and effluent, and producers look for ways to keep antimony out of rivers and soil. Many countries tightly limit how much can appear in finished lubricants and mandate child-resistant packaging. These steps haven't erased accidents, but they keep routine jobs much safer than in the past.
Where It’s Used, and Why It Stays Relevant
Antimony Diisopropyldithiophosphate found a niche in crankcase and hydraulic fluids where traditional zinc additives face regulatory limits or performance drawbacks. Some modern engines, with their aftertreatment and strict emission requirements, need additives that don't poison catalytic converters or particulate filters. Here, antimony’s performance edge still stands out, especially for long-drain intervals in mining, marine engines, and off-highway vehicles. Gear oils, industrial greases, metal forming fluids—all see use cases where additive performance makes or breaks million-dollar machinery over the course of months. For maintenance managers, choosing additives with proven track records often means fewer headaches and surprises, translating to predictable budgets and safer work environments.
Synonyms, Labels, and Avoiding Confusion
Walk through a warehouse or scan a shipment manifest and this compound shows up under several related names: Antimony O,O'-diisopropyldithiophosphate, sometimes just “Sb-DTP,” or as an “antimony dialkyldithiophosphate” in supplier catalogs. The key shared feature—antimony linked through sulfur to dialkyl phosphates—remains the fingerprint no matter the alias. Some marketers emphasize antimony in the product name, others spotlight the alkyl groups to target specific customers. This patchwork of labels has led to mix-ups, especially among buyers dealing with new suppliers. Accurate hazard labeling and technical sheets cut these problems, though confusion pops up in new markets where regulatory names or local translation miss the point.
Research and What Science Tells Us So Far
Most work on this additive now revolves around two questions: can it perform better, and can it pose less environmental risk? Scientific reviews published in the past few years show steady progress using advanced surface analysis to watch the additive’s protection films develop, vanish, and reform cycle after cycle. Laboratory friction testing lets chemists tweak formulas by using different alkyl substituents or by blending antimony with molybdenum, boron, or lower-toxicity metals. Environmental toxicology teams follow the fate of antimony—how it persists in soil or water, how fast it degrades, and which forms turn hazardous over time. The trade-off sits between performance and safety, a tightrope act made tougher by tightening global standards. Still, the quest for a perfect balance keeps teams in business and research labs busy.
Toxicity: Weighing the Evidence and Seeking Better Options
Regulators take a hard stance on antimony exposure, with limits in workplace air and product content inching downward as new evidence emerges. Large-scale studies of workers exposed to antimony over decades suggest increased lung and heart problems, though causal links get tangled with co-exposures. Wildlife studies show effects at low concentrations in aquatic systems, raising concerns over long-term accumulation. Some forms of antimony can transform in the environment, moving from relatively stable to more mobile and potentially harmful ones as they encounter sunlight or microbes. Scrubbing these risks means not just technical fixes inside the factory gate—better personal protection, sealed transfer lines, and more advanced effluent treatment—but also research into substitutes and ways to recover or recycle antimony from waste.
Where the Industry Goes from Here
I’ve seen plenty of chatter about moving away from metals in lubrication, especially as electric engines and sustainability targets rewrite the rules. Still, field experience and lab results both say the world isn’t done with antimony-based additives just yet. For critical applications where reliability outweighs all else, and where regulatory hurdles remain manageable, Antimony Diisopropyldithiophosphate stands its ground. Yet the next decade may look different. Companies are exploring phosphorus-free antiwear systems, advanced organic friction modifiers, and ways to keep metals out of waste streams. Next-generation products, built off years of hard lessons and tough restrictions, look set to reshape not just performance but the entire supply chain. Anyone invested in the machinery that powers industries will want to keep a close watch on these shifts, balancing what works today against what’s coming tomorrow.
What is Antimony Diisopropyldithiophosphate used for?
The Role in Everyday Machinery
Anyone with a foot in the machinery world will bump into the name Antimony Diisopropyldithiophosphate at some point. This chemical acts like a helper in keeping engines from grinding themselves to dust. Primarily, it goes into lubricants as an anti-wear and extreme pressure additive. If you own a car, ride a motorbike, or handle heavy equipment, chances are, you have benefited from this additive without knowing it.
How It Protects Metal
Friction wears parts down fast. Additives based on antimony create a protective film right on the metal’s surface, which keeps gears and pistons running smoother for longer. Machines working under high stress, like construction vehicles or factory presses, rely on this little bit of chemistry to tackle high heat and crushing forces.
People sometimes ask why not just use zinc-based additives, which are cheaper and common. Zinc dialkyldithiophosphate dominated the field for decades. The reason antimony versions step in is because they can do the job where zinc faces limits, such as when regulations cap the amount of phosphorus or sulfur in the oil, both of which zinc compounds deliver. Antimony steps up to the plate and balances out the performance without busting regulatory limits.
Staying Safe and Green
I keep an eye on how chemicals move through the environment. Antimony compounds, in general, have raised some eyebrows over toxicity concerns. Long-term exposure, especially in high concentrations, can be a risk. The trick for manufacturers and users is to use these additives responsibly—take care with disposal, keep up with the latest research, and make sure to meet workplace safety measures.
Looking for more environmentally friendly options runs parallel with using what works right now. Researchers press on with new additives that try to outdo antimony in performance and environmental footprint. Phosphorus-free and sulfur-free variants keep popping up, powered by a mix of necessity and innovation. While some big names in industrial oils and lubricants chase greener alternatives, antimony-based solutions stay popular because, for tough jobs, they still check off all the right boxes.
The Economics Behind Usage
Choices in industrial chemistry don’t boil down to science alone. Decisions in factories and garages often revolve around cost, dependability, and availability. For a mid-sized machine shop or an industrial engine manufacturer, shifting away from proven additives takes considerable investment. Some industries, such as mining or railroad maintenance, stick with antimony blends because they know how the machinery reacts to them through years of trial, error, and broken parts.
It always comes back to this: reliable machinery saves money and keeps businesses running. Whether hauling lumber, laying pavement, or digging minerals, downtime eats into profits faster than most realize. Using a tough, reliable chemical additive, even a mouthful like antimony diisopropyldithiophosphate, buys time, stretches service intervals, and can be the difference between a summer of steady work and weeks of expensive repairs.
Checks, Balances, and the Path Forward
Strong safety protocols, smart recycling, and constant scrutiny from health authorities keep this chemical in check. Demand for better alternatives stays high, and everyone with a stake in the industry watches shifts in regulations and research results closely. For now, though, antimony diisopropyldithiophosphate plays a central role where heavy-duty performance matters most.
What are the safety precautions when handling Antimony Diisopropyldithiophosphate?
Why Safety Doesn’t Take a Back Seat
Handling chemicals like antimony diisopropyldithiophosphate calls for respect and experience. This compound finds use in specialized lubricants and industrial additives. Over the years, I’ve seen plenty of situations where ignoring basic precautions creates bigger problems with chemicals like this. Skin, eyes, and lungs can all end up at risk if safety slips up. Accidents might not just cause discomfort—they can bring long hospital visits, or even worse.
The Risks—And How to Dodge Them
This chemical can irritate the skin and eyes on contact. Breathing in its vapors or dust irritates your throat, nose, and lungs. Repeated exposure over time, even in small amounts, may trigger more serious health issues, including neurological effects. People who work in close proximity to it get exposed to fumes during mixing, pouring, or other transfers, so extra caution comes in handy.
Suit Up: Personal Protective Equipment Really Matters
Goggles offer solid protection for your eyes—that’s a non-negotiable. Splashing once is all it takes to do damage. Nitrile or neoprene gloves work better than bare hands and stand up to both the chemical and the solvents you might pair it with. A chemical-resistant apron blocks drips and splashes from tagging clothes and skin. Closed shoes with covers round out the bare minimum.
If you’re working with large batches, or anywhere airflow turns stale, a respirator with organic vapor cartridges often makes sense. Even in smaller volumes, the smell and fumes can feel tough. Don’t skip this protection because the immediate risk doesn’t always show up right away. Once, a teammate thought the low odor meant low risk, but ended up with headaches for the rest of the day.
Good Habits Make a Difference
Always handle this stuff in a space with solid ventilation. An open window likely won’t cut it. A chemical fume hood or, at least, an exhaust fan helps move fumes away from faces and lungs. Don’t eat, drink, or touch your face in the area you’re working. Little habits like washing hands before leaving the lab or workshop set the foundation for long-term health. I’ve found that building these routines early on pays off far more than strong warnings or lectures.
Storage and Spills—Handle with Thought
Store antimony diisopropyldithiophosphate in tightly sealed containers in cool, dry areas. Keep it far from direct sunlight, strong acids, or oxidizers. Label the containers clearly and never reuse them for anything else. Training for emergency spills should go further than a quick video or a posted chart. Have spill kits nearby with absorbent pads and neutralizers that actually suit these types of compounds. Absorb the spill, contain the waste, and avoid tracking residue beyond the workspace.
The Human Factor
Sometimes it’s easy to think gloves or a label will do most of the work. Yet, the key to real safety sits in constant awareness. Chemical safety training helps, but only if people pay attention. Checks before and after each use, having fresh gear, and never cutting corners build trust—not just in safety systems, but in each other. Respecting the hidden risks leads to a safer workplace and healthier workers in the long run. Antimony diisopropyldithiophosphate can stay useful without causing harm, as long as people bring care and common sense into each step.
What is the chemical formula of Antimony Diisopropyldithiophosphate?
Understanding the Chemical Formula
Antimony diisopropyldithiophosphate carries the chemical formula Sb[S2P(OC3H7)2]3. This compound brings together the element antimony and organic phosphorus groups known as diisopropyldithiophosphate ligands. Each ligand contains two isopropyl branches, which stem from the backbone of dithiophosphoric acid and pair with sulfur atoms. What you get is a molecule where one antimony atom coordinates with three diisopropyldithiophosphate ligands, tying together organic and inorganic worlds.
Why the Formula Matters for Industry and Research
Few chemicals spark as much curiosity among lubrication experts as antimony diisopropyldithiophosphate. Consider how it improves the anti-wear properties of oils and greases in heavy-duty machinery. I’ve seen seasoned engineers swear by additives built around this compound, especially in applications where high pressure and temperature threaten to break down lesser oil blends. Antimony in this formula brings unique friction-reducing and oxidation-resisting capabilities, helping machines run longer with less downtime. The ligands contribute their own strengths, forming a protective layer on metal surfaces that stops wear before it starts.
Digging through tribology journals, you’ll find that compounds like this one are regularly tested alongside more well-known zinc dialkyldithiophosphate (ZDDP) additives. The science points out that antimony complexes, particularly when bonded to diisopropyldithiophosphate, give comparable or better results in terms of anti-wear and extreme pressure performance. This directly connects to better operational safety in gearboxes and other critical equipment. High-wear environments call for innovation, and chemistry often leads the charge.
What Sets This Compound Apart
This isn’t just a story about a chemical formula. Each atom in Sb[S2P(OC3H7)2]3 plays a unique role. The antimony center deals with load-bearing. Its heavy atomic mass supports the creation of a solid lubricating film. The diisopropyl groups and sulfur atoms ramp up the chemical’s ability to connect with metal and deliver a dual layer of protection. Hands-on lab results show that this type of compound reduces the metal-to-metal contact that wears down engine and hydraulic components. There’s a reason industries like mining and marine engineering keep a close eye on developments in anti-wear additive science.
Progress and Challenges
Of course, every promising solution faces scrutiny. Environmental researchers have raised questions about the long-term effects of compounds containing antimony and organophosphate groups. Health and environmental risks push scientists and regulation bodies to quantify exposure and set safe use standards. I’ve watched teams balance the needs of machinery—where maintenance costs and downtime can spiral out of control—against the need to keep workplaces and our water supplies safe from contamination. Progress here depends on transparency and rigorous testing, not short-term fixes.
Paths Toward Responsible Use
Sustainability isn’t a side issue any more. Laboratories continue to investigate new additives based on the antimony diisopropyldithiophosphate structure, but with tweaks aimed at lower toxicity and easier biodegradation. In the meantime, engineers implement closed-loop systems and spend more resources on safe disposal and recycling of used lubricants. By keeping close tabs on research and openly sharing safety data, companies and regulators can work together to refine both products and policies. Smart steps now mean durable protection for machines—and for the environments they operate in.
How should Antimony Diisopropyldithiophosphate be stored?
Real Safety for a Real Hazard
Antimony Diisopropyldithiophosphate sounds like a mouthful, and it definitely packs the punch of a challenging industrial chemical. Folks working with this stuff know it brings hazards most people can’t picture unless they’ve seen the label: toxic, flammable, possibly corrosive. I’ve been around enough niche chemicals to learn that ignoring good storage practice never ends well — you don’t forget the day you see a minor spill turn into an emergency.
Keeping antimony compounds in order starts with solid respect for their risks. This chemical doesn’t belong in a cluttered storeroom with no climate control. Every seasoned lab tech knows to read the manufacturer’s safety data sheet before even signing for a delivery. Antimony Diisopropyldithiophosphate usually demands a cool, dry, well-ventilated space. Direct sunlight or heat sources tempt fate, so find a storage spot out of their path.
Why You Can't Cut Corners on Containers
Plenty of problems come from careless handling of original packaging. I’ve seen glass and high-quality plastic containers survive years because they never got swapped out for something less sturdy. The lid must screw tight, with no cracks or warps that let moisture creep in, because this chemical can break down or release fumes if water finds a way inside. Store the container upright and avoid stacking heavy items on top, which keeps the seal healthy.
Some labs use extra containment, like sealed tubs or bins lined with absorbent pads, in case of leaks. This step might sound like extra work, but a cleanup job from a missed drip takes twice as long. Taking time now saves big headaches later, especially when working in shared facilities.
Location Makes All the Difference
I’ve watched enough chemical audits to spot preventable mistakes. Antimony Diisopropyldithiophosphate plays poorly with strong acids, oxidizers, or moisture-rich substances. It pays off to sort your shelves so these chemicals never sit side-by-side. Separate corrosive storage cabinets from organic compound shelves. If you store large quantities, adding a spill containment tray beneath the containers works as extra insurance.
In my experience, real risk crops up during moves or renovations, when labels fall off or incompatible chemicals accidentally get combined. Inspect labels regularly, rewrite them if they fade or peel, and tape on a legible warning with the hazard class. If possible, digitize your inventory so you don’t lose track of containers stashed behind newer deliveries.
Why Training Can’t Be Optional
No storage setup replaces hands-on training. Even the best ventilation won’t matter much if someone opens a bottle in a cramped, steamy closet. Newcomers benefit most from seeing how a respected technician organizes each shelf. Chemical know-how carries through generations by storytelling, not just paperwork. If you catch a mistake early, use it as a teaching moment — share examples of near-misses so the lesson sinks in.
Emergency plans deserve attention too. Don’t wait for an accident before figuring out where your eye wash and spill kit actually live. This isn’t about paranoia; it’s a matter of having everyone on the same page. Good habits protect careers and keep those lab doors open year after year.
What are the physical and chemical properties of Antimony Diisopropyldithiophosphate?
A Closer Look at the Substance
Antimony Diisopropyldithiophosphate brings together antimony, phosphorus, sulfur, and organic groups in a single molecule. People usually spot this compound as a yellow-brown oily liquid, sometimes with a faint sulfurous smell. It doesn’t dissolve in water. Instead, it mixes well with lubricating oils and other non-polar solvents. Some labs report the density around 1.1 to 1.2 g/cm³, depending on the exact formulation. Compared with many other metal-organic chemicals, it tends to show robust thermal stability, often handling temperatures up to 180°C without breaking down.
Getting to the Chemistry
This compound has a phosphorodithioate backbone, and antimony attaches by forming bonds with sulfur atoms. The diisopropyl groups give extra bulk, offering resistance against unwanted reactions with oxygen or moisture. Instead of staying inert, this structure interacts with metals in useful ways. The sulfur atoms carry a lot of importance—they cling to metal surfaces and sometimes react to form protective layers. Experiences in the lab show that these layers can reduce wear and tear on machine parts.
Antimony Diisopropyldithiophosphate doesn’t explode or catch fire easily, though it can break down and give off unpleasant fumes if someone heats it above its safe range. That’s a crucial point for anyone handling it in workshops or industrial plants. Even old safety data sheets remind workers to avoid breathing the fumes and suggest using gloves and eye protection. This compound doesn’t splash around on a workbench—it sticks to oily fingers and equipment, so good cleanup practices make a real difference.
Using the Properties in Real Life
Over the years, companies have leaned on this compound for its anti-wear and antioxidant abilities. The main claim to fame comes from use as an oil additive, where it lengthens the lifespan of gears and engines. The story goes beyond simple lubrication. In auto repair shops and big manufacturing plants, breakdowns due to metal wear can grind everything to a halt. By putting this chemical in the mix, mechanics and technicians see smoother operation and less frequent servicing.
There’s always a trade-off, though. Antimony is a heavy metal; some authorities worry about its long-term impact on the environment. Waste oils and residues pose a disposal challenge, so strict handling and waste management plans matter. European regulators and some U.S. states keep an eye on antimony compounds, calling for proper containment and review. Engineers and environmental specialists often work together, searching for ways to capture or recycle oily residues. Some labs research biodegradable alternatives, but few match the raw durability this molecule offers.
Finding a Middle Ground
Researchers and industry pros try to balance performance and environmental care. While the demand for reliable anti-wear additives isn’t going anywhere soon, pressure builds to limit heavy metal use. In response, some companies experiment with lower concentrations of antimony-based additives or explore hybrid packages with zinc or molybdenum. Others invest in better oil filtration and recovery. Frontline workers, from mechanics to plant engineers, contribute plenty of ideas for practical day-to-day improvements.
The conversation about Antimony Diisopropyldithiophosphate shows a bigger truth: every chemistry decision shapes more than machinery. It affects workers, communities, and the health of natural systems. Moving forward, building safer, cleaner, and cost-effective technologies stands as both challenge and opportunity.
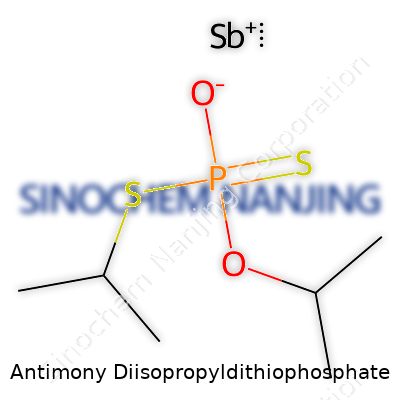

| Names | |
| Preferred IUPAC name | Diisopropyldithiophosphatoantimony |
| Other names |
Phosphorodithioic acid, O,O-diisopropyl ester, antimony(3+) salt Antimony O,O-diisopropyl phosphorodithioate Antimony diisopropyl dithiophosphate Diisopropyldithiophosphoric acid antimony(III) salt |
| Pronunciation | /ˌæn.tɪˌmoʊni daɪ.aɪˌsɒ.prəˌpiːl daɪˌθaɪ.oʊˈfɒs.feɪt/ |
| Identifiers | |
| CAS Number | 27249-40-7 |
| Beilstein Reference | 6568773 |
| ChEBI | CHEBI:38875 |
| ChEMBL | CHEMBL3334645 |
| ChemSpider | 22459878 |
| DrugBank | DB11437 |
| ECHA InfoCard | 20-211-742-219 |
| EC Number | 238-854-8 |
| Gmelin Reference | 70816 |
| KEGG | C18909 |
| MeSH | D010583 |
| PubChem CID | 87649 |
| RTECS number | WT3150000 |
| UNII | WB9VV3WT17 |
| UN number | UN3334 |
| CompTox Dashboard (EPA) | DJ2FD465P0 string |
| Properties | |
| Chemical formula | C6H15O2PS2Sb |
| Molar mass | 399.43 g/mol |
| Appearance | Yellow liquid |
| Odor | Slight mercaptan |
| Density | 1.14 g/cm³ |
| Solubility in water | Insoluble in water |
| log P | 1.47 |
| Vapor pressure | 0.23 hPa (20 °C) |
| Acidity (pKa) | 2.2 |
| Basicity (pKb) | 6.03 |
| Magnetic susceptibility (χ) | -4.31E-6 cm³/mol |
| Refractive index (nD) | 1.573 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 603.6 J/mol·K |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H332, H413 |
| Precautionary statements | P210, P260, P273, P301+P310, P302+P352, P305+P351+P338, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0, Special: -- |
| Flash point | 89°C (192°F) |
| Lethal dose or concentration | LD50 (oral, rat): 8200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: > 5,000 mg/kg |
| NIOSH | S8007 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Antimony tris(dipropyldithiophosphate) Zinc diisopropyldithiophosphate Antimony triphenyldithiophosphate Antimony tris(O-alkyldithiophosphate) Bismuth diisopropyldithiophosphate |

