Antimony Arsenite: From Origin to Future Potential
Historical Development
Folk remedies once harnessed elemental powders without a firm grasp on what lay inside. Antimony arsenite entered the annals of chemistry when early mineralogists noticed the distinct combination of antimony and arsenic in natural ores. Miners sometimes mistook glistening white ore for simple arsenic until advances in chemical analysis changed the game. Through systematic studies in the nineteenth and twentieth centuries, chemists learned to separate, purify, and combine antimony and arsenic for industrial and research use. Shifting regulations have pushed researchers toward greater transparency and caution, and many early missteps—those leading to occupational illnesses—remind us just how much scientific understanding has changed since the curious experiments of the first mineralogists.
Product Overview
In modern supply chains, antimony arsenite typically appears as a white or yellowish powder, packaged in tightly sealed drums to avoid contamination and exposure to moisture. Laboratories rely on certified batches with detailed labeling, ensuring professionals know the substance inside and out before ever cracking open a container. Factories source material from established chemical companies with stringent quality checks, with import and export falling under international scrutiny, given the compound’s toxic reputation. Accurate product names and procurement records cut down on errors, protecting both workers and end users from surprise exposures.
Physical & Chemical Properties
Crystalline in its pure form, antimony arsenite has a melting point hovering around 300°C and doesn’t dissolve in water—it resists simple breakdown and sticks around in soils and sediments. Under strong heat or acid treatment, this compound releases both arsenic and antimony vapors, which call for strong ventilation and careful handling. The powder feels gritty, almost like chalk, and emits no odor. It remains stable when stored away from acids and bases, showing just how much the surroundings shape its long-term behavior. Chemists running spectral analysis confirm its presence through a unique collection of energy peaks, and this fingerprint provides an extra layer of confidence in material identification across labs and factories alike.
Technical Specifications & Labeling
Suppliers document the percentage of antimony and arsenic by weight, usually ranging from 55-60% antimony and 38-42% arsenic, depending on purity goals during manufacture. Color, bulk density, particle size, and moisture content get their own lines on the certificate of analysis, with any trace contaminants—especially lead or sulfur—reported in strict accordance with safety standards. Batch numbers, lot information, and expiration dates become non-negotiable for anyone working with such a hazardous agent. GHS-compliant labeling shows hazard pictograms for acute toxicity, environmental harms, and long-term health risks, making plain the need for gloves, goggles, and protective clothing. End users never want to take a shortcut on this front; clear, direct labeling saves lives and cuts through confusion in the heat of production or research.
Preparation Method
Production of antimony arsenite leans on precise chemical reactions between antimony trioxide (Sb2O3) and arsenious acid or arsenic trioxide (As2O3), typically heated together under strict temperature controls. The process demands closed systems—nobody wants arsenic fumes floating around a plant—plus real-time monitoring for purity and off-gas capture. Equipment undergoes regular cleaning and maintenance to prevent build-up, which could cause dangerous variations in composition. Waste solutions and solid residues require treatment before disposal, in line with international hazardous waste protocols. Scale-up from lab to factory needs careful planning at every step, since even minor missteps can spoil product integrity.
Chemical Reactions & Modifications
Antimony arsenite doesn’t just sit idle in storage; exposure to acids or strong oxidizers triggers decomposition, liberating toxic vapors and leaving behind residues. Chemists have investigated how it might be altered to form mixed compounds with other metals or metalloids, seeking properties that could unlock new applications. Encapsulation techniques or chemical modifications might reduce bioavailability or change reactivity, appealing for uses where leaching must be avoided. Researchers continue experimenting with matrices and additives, pursuing performance tweaks that widen the field of potential end uses.
Synonyms & Product Names
This compound goes by several aliases across industry literature: antimony(III) arsenite, antimony arsenous oxide, and antimony arsenate among them, reflecting small differences in oxidation state or handling history. On inventory sheets, codes or abbreviations might show up, but full chemical names remain best for safety, since no two synonyms guarantee identical composition or toxicity profile. Some legacy suppliers use trade names, especially in regions with historic mining activity, adding yet another layer for buyers to crosscheck as they verify batch identity.
Safety & Operational Standards
Antimony arsenite shares many hazards with its parent elements—both prove toxic if inhaled or ingested and pose serious threats to workers without proper barriers. Respiratory protection, chemical-resistant gloves, goggles, and full-body coverings make up the minimum safe ensemble for any significant handling. Fume hoods and local exhaust systems keep airborne particles low, and spill plans treat every release as serious. Companies enforce training programs, refreshers, and fit tests for gear, rooted in national and international occupational health guidelines. Inspections for storage, cross-contamination, and disposal keep safety a daily priority. Safety data sheets spell out first aid, exposure limits, and firefighting methods, and strong communication between safety managers, procurement staff, and floor workers keeps standards from slipping.
Application Area
Beyond pure research, antimony arsenite finds use in glassmaking, ceramic glazing, and as a flame retardant precursor—though alternatives keep gaining ground. Specialty pigment applications crop up in certain high-tech manufacturing jobs, but regulatory and liability pressures keep pushing for safer substitutes. Some research into semiconductors and advanced battery chemistry looks at metal arsenites, aiming for specific electrical or thermal properties. Most downstream uses demand sealed applications where leaching of the compound can’t pose an environmental risk. Legal frameworks and local licensing really restrict its use in consumer goods, and green chemistry initiatives drive further innovation toward replacements carrying fewer health and ecological drawbacks.
Research & Development
In labs, the focus lands on modifying antimony arsenite’s behavior—shifts in crystalline structure, changes in solubility, or targeted delivery for industrial processes. Analytical chemists track breakdown pathways, modeling possible environmental routes if the compound escapes containment. Research teams have studied the compound’s photochemical properties, envisioning uses in specialty optics or microelectronics. Advanced characterization tools like electron microscopy and synchrotron spectroscopy offer insight into bond strength and reactivity, and cross-disciplinary efforts tie together surface science, toxicology, and green process engineering. Every publication spurs debate over tradeoffs—benefits in specific, controlled settings versus the ongoing hunt for substances with less baggage.
Toxicity Research
Long-term animal studies confirm the fears about both acute and chronic exposure; antimony arsenite causes respiratory distress, gastrointestinal problems, skin irritation, and systemic effects with repeated contact. Human data from mining, smelting, and glassmaking chronicle patterns of poisoning that led to landmark regulatory reforms. Arsenic content raises red flags for carcinogenicity, prompting agencies like the EPA, OSHA, and IARC to set aggressive exposure limits. Recent biomonitoring research tracks metabolism and excretion in exposed populations, and environmental fate studies map where residues wind up after manufacturing. As analytical chemistry allows for ever-lower detection limits, regulators can set tighter standards, and companies that lag face growing pressure to phase out legacy compounds.
Future Prospects
The road ahead for antimony arsenite looks narrow, boxed in by toxicological data and regulatory scrutiny. Researchers keep seeking ways to control exposures or render the compound safer through encapsulation, immobilization, or development of less toxic surrogates. The clean-tech boom favors materials with low hazard profiles, so the focus on antimony arsenite increasingly shifts toward legacy site cleanup, analytical method development, and studies mapping its long-run environmental impact. Advances in green chemistry hold promise for finding functional analogs, letting industrial users deliver needed performance without the same bedrock risks. Academics, regulators, and industry must keep building dialogue, sharing data, and refining best practices—not only to protect health and the environment, but to spark the next wave of material innovation.
What is Antimony Arsenite used for?
The Role of Antimony Arsenite in Everyday Products
Antimony arsenite sounds like a mouthful, and it's not something the average person thinks about day to day. Yet, this compound has a real impact on products many of us use, especially in industries that rely on flame resistance or pigment creation. The core reason for its value comes from its ability to slow down flames—a property in high demand across numerous sectors.
Flame Retardants and the Push for Safety
Walk through any electronics store, and you’ll run into the consequences of safety regulations involving fire hazards. TV casings, wire coatings, even some building materials draw on flame retardant chemicals to help prevent disastrous fires. Antimony compounds, including antimony arsenite, often show up in these applications.
The science isn’t magic. In simple terms, antimony arsenite interacts with halogenated materials when exposed to heat, creating a barrier that slows down the intensity and spread of flames. Over the years, manufacturers kept reaching for antimony compounds because they worked reliably. There’s a reason flame retardancy regulations adopted them decades ago—they kept homes and workplaces safer.
Dyes, Ceramics, and the Color Problem
Back in school, I spent time around artists and ceramicists, watching pigments get mixed into glazes and paints. Some of the most striking colors come from metal-based additives. Antimony arsenite makes its mark in this world too, bringing with it the ability to stabilize and intensify yellows and other shades.
Factories producing colored glass and ceramics have turned to antimony arsenite for its stability and the way it supports consistent color. In the textile industry, it sometimes helped produce vivid prints in older dyeing methods. While newer, safer alternatives are picking up steam, legacy processes still rely on this compound where consistency trumps cost or environmental concern.
Balancing Utility with Health and Environmental Risks
No discussion about antimony arsenite skips around its dangers. Both antimony and arsenic have well-documented toxicity issues. Those who’ve worked in chemical labs, manufacturing lines, or environmental health realize the risk extends beyond the factory; air and water contamination happens if waste isn’t managed tightly.
Governments in Europe, the US, and Asia have responded by clamping down on how, when, and where these compounds get used. The European Chemicals Agency lists antimony compounds as substances of concern, calling for industry to phase down their use or swap them out for safer options when possible. A worker exposed to dust or fumes over years, for example, would face a higher risk for chronic health problems, including respiratory issues and cancers.
Paths Forward: Innovation and Safer Alternatives
There’s no getting away from the truth: jobs that rely on strong, flame-resistant materials matter. Yet, the industry can’t ignore that the chemical tools of the trade sometimes bring bigger problems. Smart companies keep research and development at the forefront; they look for non-toxic flame retardants, better disposal techniques, and improved worker protection.
Students, researchers, and people on the shop floor have all played a part in shifting attitudes about compounds like antimony arsenite. Each safe substitution, every reduction in emissions, actually means a safer workplace and cleaner community. It’s possible to supply the market with the same flame resistance and color brilliance, just without so much risk hanging overhead.
Is Antimony Arsenite hazardous to health?
What is Antimony Arsenite?
Antimony arsenite comes from two elements: antimony, a silvery metal, and arsenic, a notorious toxic substance. Together, they form a compound used in some industrial settings. Sometimes factories use such compounds in pigments or certain manufacturing processes, often without much public attention. Few people outside specialty industries even hear the name unless something goes wrong.
Health Hazards: What Science Says
From growing up near industrial zones, one thing sticks: heavy metals and metalloids rarely lead to positive headlines. Antimony arsenite is no exception. Data from agencies like the U.S. Centers for Disease Control and Prevention (CDC) and the Agency for Toxic Substances and Disease Registry (ATSDR) highlight the risks. Both antimony and arsenite parts create health concerns, especially when inhaled or ingested. Arsenic compounds turn up in the news whenever someone talks about contaminated drinking water, with stories of cancers and nerve damage trailing behind. Throw in antimony—which can provoke lung and heart issues—and you get a chemical cocktail better avoided.
Swallowing or breathing in antimony arsenite dust causes more than a mild upset. Immediate symptoms might include headaches, nausea, or skin irritation. Long-term exposure digs in deeper: studies link regular contact to problems with lungs, heart rhythm, and the nervous system. Researchers have traced certain cancers—especially lung and bladder cancer—to chronic arsenic exposure. The International Agency for Research on Cancer (IARC) categorizes arsenic compounds as known human carcinogens. Antimony doesn’t rank as severely but still makes many toxicology watchlists, thanks to evidence of lung illness and potential effect on the heart.
Who Faces the Greatest Risk?
Usually, people working with metals or chemicals see the brunt of these impacts. Stories from manufacturing towns sometimes feature folks dealing with chronic coughs or out-of-the-blue kidney trouble. I’ve met people who spent years around smelting plants, who can rattle off former coworkers lost to mysterious illnesses. Even at smaller scales, routine mishandling leads to sickness—especially if companies skip proper training, or safety equipment sits unused in dusty lockers.
Community exposure is rarer but not unheard of. If factories release waste improperly, nearby neighborhoods can pick up the slack, sometimes through their water or soil. The ongoing situation in places like Bangladesh underscores how quickly public health can unravel when arsenic leaches into drinking water. Antimony doesn’t move as easily as arsenic but still sticks around wherever industrial waste piles up.
How Can People Protect Themselves?
Solid science has already mapped the way forward. Safety hinges on good practices. Workers need protective gear, regular medical checks, and clear guidelines. Regulations from groups like the Occupational Safety and Health Administration (OSHA) set limits for workplace exposure. Following those isn’t about red tape—it’s about people going home healthy at the end of the shift.
Communities living near factories should push for regular environmental testing and full transparency from local industries. When something spills, public health teams need to step in fast, tracking both short-term symptoms and long-term disease patterns. Drinking water deserves top-tier monitoring, wherever heavy metals or metalloids may lurk. Bigger picture, companies can swap out hazardous chemicals for safer alternatives. Some pigment makers, for instance, already phase out antimony-based products, knowing that safer compounds exist.
Valuing Health Above Convenience
Ignoring the hazard doesn’t erase it. Whenever companies or governments gloss over the risks of antimony arsenite, real lives sit in the crosshairs. Scientific evidence, lived experience, and common sense line up: treating this compound with respect protects workers, neighbors, and families alike.
What is the chemical formula of Antimony Arsenite?
Why Chemical Formulas Matter in Daily Life
Most people wouldn’t think twice about antimony arsenite beyond a high school chemistry class. Still, its formula—SbAsO3—shows up in research labs, mining operations, and environmental studies. I remember handling samples of mineral-rich rocks, always double-checking the label, and translating them into chemical shorthand. Getting these formulas wrong can throw off lab results, confuse supply orders, or even complicate pollution reports.
Breaking Down SbAsO3
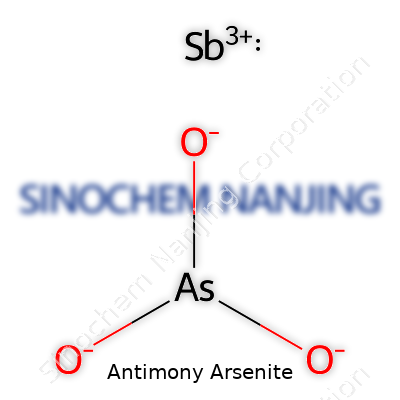
In antimony arsenite, Sb means antimony, As means arsenic, and O stands for oxygen. Three oxygen atoms bond with one antimony and one arsenic atom, giving us that compact, information-rich formula. This points to a compound made from elements that are both a gift and a headache for chemists.
Why SbAsO3 Isn’t Just Academic Trivia
Living in a mining town, I learned that knowing what’s in the ground keeps both miners and residents safe. Antimony compounds, especially ones combined with arsenic, spark health concerns and regulatory scrutiny. Researchers in toxicology talk about the way these compounds impact water quality and workplace safety. Antimony by itself is used in flame retardants and batteries. Mix in arsenic, though, and the risks jump—arsenic’s link to cancer and groundwater pollution is chillingly well documented.
Curiosity led me to published data from the U.S. Environmental Protection Agency. Antimony compounds can leach into rivers from industrial runoff. Every time the wrong formula goes into a report, it chips away at efforts to protect neighborhoods downstream. In a 2021 survey, misidentification of antimony arsenite contributed to false readings in water samples, trailing delays in environmental cleanup.
Addressing Misunderstandings and Risks
The formula SbAsO3 brings together two semi-metals. That makes for unpredictable reactions in the environment. Mining engineers watch out for antimony arsenite when planning earthworks, because improper handling leads to long-term contamination. In manufacturing, switching between antimony trioxide and antimony arsenite by mistake ruins products and exposes workers to increased danger.
Solutions in Communication and EducationI’ve watched training sessions where lab workers mix up formulas or gloss over seemingly small details like subscripts or oxidation states. More hands-on workshops in schools and industry settings help. Setting up digital databases where verified formulas (like SbAsO3) can be checked rapidly also keeps everyone on the same page. Making real-world examples part of lessons—using local water contamination cases, for instance—connects the dots between formulas and community health.
Looking Forward: Getting the Formula Right Matters
Scrutiny around compounds like antimony arsenite isn’t just about academic accuracy. It matters for public policy, industrial safety, and personal health. Chemical literacy keeps communities safer and makes industries more transparent. Science teachers and field chemists who treat every formula with care help build trust in the data behind big decisions. For me, writing the formula as SbAsO3 always brings back the importance of getting even the smallest scientific details right.
How should Antimony Arsenite be stored?
Recognizing the Hazards
Antimony arsenite isn’t just another lab chemical—handling it the wrong way can go sideways fast. Breathing in its dust or letting it touch the skin spells trouble, and it doesn’t just disappear after use. This compound brings the combined dangers of arsenic and antimony, two substances that health and safety folks keep a close eye on for good reason. When you work with it every day, respect for its stubborn toxicity goes a long way toward staying safe.
Containment Above Convenience
Open a container of antimony arsenite in the wrong spot, and the fine powder floats into the air where it’s hard to see or control. Over the years, I’ve seen how a careless scoop or a cracked jar can set off alarms—cleanup doesn’t come easy, and the risks only grow. Quality storage means looking for sealed, clearly labeled containers made of glass or certain plastics that refuse to react with the compound. No shortcuts here. Flip tops and crumbling gaskets spell disaster, so regular checks belong on every responsible worker’s schedule.
Location: Away from Heat, Light, and People
Sticking antimony arsenite on any shelf just doesn’t cut it. Chemicals like this belong in a dedicated poison cabinet. Lock the door. Post warnings. Keep the cabinet away from direct sunlight, heat sources, and the kind of traffic that brings accidents. Ventilation matters, too. I’ve learned to avoid closets and unventilated corners; airflow saves lungs and limits exposure if a spill ever happens.
Preventing Cross-Contamination
No one wants to find arsenic dust on their lunchbox, yet accidents often start small. After handling antimony arsenite, I always remove gloves right where the work happens—never anywhere else. Dedicated storage means never mixing this powder near food, drink, or regular break rooms. It pays to have a secondary container—a shatter-proof, leak-resistant overpack—just in case the main jar ever fails.
Labeling and Ongoing Vigilance
Clear, direct labeling does more than save time during inspections—it prevents costly mistakes and keeps new staff from grabbing the wrong thing. Labels should mark contents, hazards, and the amount on hand. Updating inventory records every time a container comes out or goes in the cabinet isn’t bureaucracy; it’s a safety net. I once worked at a facility where an unlabeled jar caused days of confusion and risk. It isn’t worth it.
Simple, Actionable Solutions
Spills and accidents rarely happen in a vacuum. Antimony arsenite belongs on a shelf with a spill kit ready nearby—absorbents, gloves, and a disposal protocol turn a scare into a minor nuisance rather than an evacuation. Training counts too. Every worker should know how to deal with a dropped jar, a torn glove, or a label that’s faded with time. Keeping antidotes or medical information on hand rarely matters—until the moment it suddenly does. Responsible facilities plan for emergencies long before they happen.
Protecting People and the Planet
Disposing of antimony arsenite brings its own set of complications. Never pour leftovers down the drain or toss them with regular trash—regulated waste streams keep it out of the groundwater and away from unsuspecting hands. Partnering with certified disposal companies keeps everyone honest and, more importantly, safe. The extra paperwork and cost pale in comparison to what a single mistake could do to health and the environment.
What are the safety precautions when handling Antimony Arsenite?
Direct Exposure Brings Real Health Risks
Antimony arsenite looks like just another powder in a lab. People often overlook the danger hiding in its dust and small particles. This compound deserves serious respect. Breathing in its dust puts lungs in harm’s way, irritates mucous membranes, and only adds to the risk of developing bigger health problems down the road. Both antimony and arsenic have history as industrial irritants, and arsenic stands out as a confirmed human carcinogen. No one wants that kind of health record riding along at work or in the classroom.
The Right Gear Isn’t Optional
Too many folks pull on a pair of gloves and believe that’s enough. Experience in research labs tells a different story. Splash goggles make a difference—antimony arsenite has no business anywhere near eyes. Nitrile gloves, not vinyl, handle chemical hazards better. Respirators rated for particulate matter keep lungs ahead of the problem. Standard lab coats work, but only if sleeves and closures leave little room for accidental contact. Hair tied up, closed shoes, and a reminder not to eat or drink where chemicals get handled keeps invisible hazards at bay.
Working Under the Hood
Quality laboratory experience always circles back to fume hoods for a reason. Antimony arsenite doesn’t give off a strong smell, but the fine particulate can float and linger. A well-ventilated fume hood not only clears the air, it acts as a line of defense if something spills or powder goes airborne. Spills inside the hood stay contained. People working outside the hood put everyone at unnecessary risk. Never trust a still day, and always work with ventilation running.
Chemical Storage Builds Safety Habits
No chemical deserves careless storage, but antimony arsenite ranks higher on the priority list. Dry, cool spaces with locked storage cut down on risks of accidental exposure. Labeling every container, even those “temporary” jars, saves a world of trouble for everyone in the lab. Separate it from acids and bases; chemical reactions seem like surprises, but real accidents reveal they came from small acts of neglect. Training goes beyond a single orientation—new team members deserve thorough walkthroughs.
Smart Cleanup and Waste Handling
Leaving cleanup until later tempts fate. After every use, cleaning tools with proper solvents, placing all waste in sealed, labeled hazardous waste containers, and double-checking surfaces with disposable swabs prevents future contamination. Proper disposal follows legal regulations—this stuff cannot go in ordinary trash. Most facilities arrange pickups through certified hazardous waste contractors, and staff notify supervisors right away if a spill happens.
Preparedness Makes the Difference
Emergencies never wait for perfect timing. Eyewash stations and emergency showers belong close to any workspace dealing with antimony arsenite. Lab teams walk each other through emergency drills until the steps stick. Posting emergency contact information and keeping Safety Data Sheets within reach isn’t a formality—fast action matters when eyes sting or skin burns. Gaps in preparation show up only in a crisis, and by then it’s often too late to fix them.
Community Responsibility in the Lab
Safety with antimony arsenite sticks when everyone looks out for each other. No single person controls every variable. Open communication, daily reminders, and quick corrections tighten safety routines and spread confidence. Sharing close calls and mistakes helps the next person avoid old pitfalls. Trust grows in labs where people respect the hazards together and act before tension rises.
| Names | |
| Preferred IUPAC name | antimony(III) arsenite |
| Other names |
Diantimony tris(arsenite) Antimony(III) arsenite |
| Pronunciation | /ˈæn.tɪˌmoʊ.ni ˈɑːr.səˌnaɪt/ |
| Identifiers | |
| CAS Number | 1303-34-8 |
| Beilstein Reference | 3852737 |
| ChEBI | CHEBI:30470 |
| ChEMBL | CHEMBL4292202 |
| ChemSpider | 22234 |
| DrugBank | DB11090 |
| ECHA InfoCard | 100.002.683 |
| EC Number | 260-160-5 |
| Gmelin Reference | 7850 |
| KEGG | C16577 |
| MeSH | D000883 |
| PubChem CID | 104976 |
| RTECS number | CA3150000 |
| UNII | 2D088S3WGC |
| UN number | UN1559 |
| Properties | |
| Chemical formula | SbAsO3 |
| Molar mass | 291.776 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 5.2 g/cm³ |
| Solubility in water | Insoluble |
| log P | -16.54 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.84 |
| Magnetic susceptibility (χ) | −48.0e−6 cm³/mol |
| Refractive index (nD) | 1.870 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -687 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB04 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin contact; may cause cancer; harmful to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or inhaled. |
| Precautionary statements | P260, P262, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P312, P314, P330, P337+P313, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-Acad |
| Lethal dose or concentration | LD50 oral rat 6 mg/kg |
| LD50 (median dose) | 100 mg/kg (rat, oral) |
| NIOSH | B111 |
| PEL (Permissible) | 0.5 mg/m3 |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Antimony(III) oxide Antimony(III) sulfide Arsenic trioxide Arsenic trisulfide |

