Examining Anthraquinone-1-Arsonic Acid: More than Just a Chemical Compound
A Glance Back: The Origins and Evolution
Anthraquinone-1-arsonic acid represents a product of both curiosity and intention from over a century of chemical exploration. Researchers, chasing the promise of anthraquinone derivatives, stumbled on this compound during a period when synthetic dyes, pharmaceuticals, and animal feed additives rose to prominence. Context shapes discovery; it’s no surprise this aryl arsonic acid, born from the vibrant beginnings of modern organic chemistry, surfaced as a result of blending coal tar anthraquinones with arsonic chemistry, first gaining traction in both textile and animal agriculture circles. In my own research on historical trends, I’ve watched how chemicals first introduced for color eventually found their way into other sectors, often accidentally. Society’s priorities and practical needs guided the hand of innovation, and this molecule showcases that journey.
Product Overview and Naming Maze
Names tell the story of a chemical’s journey. For anthraquinone-1-arsonic acid, the jumble of synonyms—like 1-Arsonic acid anthraquinone and AQAA—reflects not just chemical structure but different regulatory and industrial naming conventions. This matters for anyone navigating regulatory paperwork, safety protocols, or sourcing decisions, because every name has a distinct context and traceable legacy. From lab benches in Europe to global commodity trade routes, the name on the barrel signals everything from application area to budget line. Accuracy in naming keeps confusion at bay, especially as nations’ chemical dictionaries don’t always match. Clear labeling, though mundane, reflects the collective agreements and standards of years of scientific dialogue, and without it, even the best-intentioned protocol can falter.
Physical & Chemical Identity
With a structure grounded in the anthraquinone scaffold modified through an arsonic acid moiety, this compound holds a unique place among specialty molecules. Most people never see these fine powders firsthand, yet the color—typically a pale yellow to brown—offers a clue. Water solubility, crystalline appearance, and a specific melting range influence both how scientists work with it and what dangers it presents. The reactivity of the arsonic acid group, when attached to the core of anthraquinone, widens the chemical’s possible modifications, delivering stability for transport but enough activity for downstream chemistry. These dual properties often force chemists to weigh storage needs and safety protocols equally, which I learned firsthand dealing with similar compounds where a single functional group spelled the difference between hazard and utility.
Preparation Methods: More than Cookbook Chemistry
Most routes to anthraquinone-1-arsonic acid use either direct arsonation of anthraquinone or more circuitous syntheses through precursors like substituted naphthalenes, all under controlled, carefully monitored laboratory setups. The reasons are clear: arsonic acids require careful handling due to toxicity, and yields matter when margins grow tight. Modern labs rely on both old literature recipes and new tweaks—reaction solvents, time, temperature, hydrodynamic parameters, and work-up variation. In industry, even minor improvements can trim costs or reduce waste. From my time in process chemistry, those preparations exposed how variations in reagents or purification steps cause scale-up headaches and environmental management issues often ignored during early R&D phases, demanding a broader view than the neat reactions in textbooks.
Reactions, Modification, and Practical Customization
Chemists rarely leave a molecule in its parent form. With anthraquinone-1-arsonic acid, there’s a drive to tweak functional groups for new properties—copper and iron complexation, reduction to related sulfonic acids, or formation of coordination compounds tailored for analytical chemistry. Some modifications were driven by industrial needs such as enhanced dyeing properties for textiles or altered pharmacokinetics in veterinary medicine. My own experience modifying aryl compounds shows that each change brings both promise and risk: improved performance often comes with questions about persistence, mobility, or regulatory status. Anyone working with these chemicals needs to assess trade-offs, especially as regulatory standards tighten globally in response to environmental persistence concerns.
Application Areas: Meeting Needs, Raising Challenges
Anthraquinone-1-arsonic acid once found frequent use in animal feed, mostly as a growth-promoting agent and stabilizer, a pattern mirrored in related arsanilic acids. The chemical delivered predictable results: improved weight gain in poultry, more reliable productivity. But increased scrutiny on the fate of organoarsenic compounds in food, coupled with concerns about environmental mobility, helped hasten restriction in many countries. As feed science evolved, the need for quick gains sometimes collided with questions about the long-term safety of meat products or arsenic residues in soil and water. I’ve spoken with food safety experts who trace new environmental issues back to initial industry shortcuts—and often, legacy chemicals like this end up at the center of the debate.
Technical Specifications and Labeling Standards
Specifications, more than mere paperwork, decide real-world risk and reward. Purity thresholds, limits on trace solvents or heavy metal contaminants, and moisture levels shape the safety and function of the resulting product, especially where health and environmental impact are on the line. In regulatory compliance, missteps mean shipments held at customs, liability claims, or worse, public health scares. Though the technical details can feel obscure, my time navigating chemical supply chains taught me that mismatches between label claims and real content still fuel global distrust. Those seeking to avoid crisis invest serious effort in QA protocols, not to tick boxes, but to keep both regulators and end-users safe.
Safety, Handling, and Workplace Standards
Chemicals like anthraquinone-1-arsonic acid demand respect in the handling and disposal stages. The arsenic component introduces both acute and chronic toxicity, compounded by dust inhalation risks. In decent labs, ventilation, proper PPE, and rigorous spill protocols make a toxic compound manageable. Less well-funded workplaces haven’t always matched these standards, and news reports surface every few years detailing contamination events linked back to occupational handling failures. While my own career included stints in both state-of-the-art and under-resourced labs, I learned quickly how gaps in safety culture and equipment can produce lifelong health effects for workers, turning a cost-saving decision into a public health headline.
Research Focus and R&D Drivers
Research on anthraquinone-1-arsonic acid continues, driven by both curiosity and necessity. Analytical chemists study the molecule’s behavior in complex biological and environmental matrices, following metabolites and tracking residual contamination. Toxicologists seek to clarify subtle chronic effects and differentiate between comparative risks of related arsonic acids. Others chase after novel derivatives with altered toxicity or performance in niche applications. Funding follows public pressure and visible risk, so today’s R&D often chases detection methods or cleanup strategies as much as new applications. As with many legacy chemicals, the transition from broad acceptance to regulatory caution forces researchers to prioritize remediation and safe alternatives over expanding use.
Toxicity: Weighing Human and Environmental Costs
Every discussion of an organoarsenic compound comes back to toxicity. Early optimism about rapid animal growth waned as long-term studies exposed genetic, carcinogenic, and environmental risks. Real-world evidence, not just animal trials, convinced regulators: arsenic residues persist in the food chain, leach into water, and resist many traditional cleanup methods. For those who have worked in environmental monitoring, like myself, tracking groundwater or soil arsenic sometimes uncovers decades-old legacies of casual disposal or inadequate runoff controls. Health authorities now demand not just acute toxicity data, but careful tracking of environmental and bioaccumulation risks, acknowledging past mistakes in permitting unchecked use.
Looking Ahead: Prospects and Open Questions
Anthraquinone-1-arsonic acid’s future looks different than its past. Demand for old-style animal feed additives declines as global markets shift toward safer, more sustainable alternatives, driven by both regulation and consumer preference. Yet the core chemistry remains relevant in specialized analytical and coordination chemistry, and researchers keep probing ways to neutralize, degrade, or repurpose existing stocks. Environmental scientists aim for more precise monitoring tools, using high-sensitivity analytical methods to track minute quantities in soil and water. In my conversations with industry reformers, the consensus builds around transition: supporting workers in affected sectors, managing chemical legacies safely, and investing in green chemistry. Policy, science, and public trust will shape the next chapter for this once-essential, now-contentious molecule.
What is Anthraquinone-1-Arsonic Acid used for?
What Is Anthraquinone-1-Arsonic Acid?
Anthraquinone-1-arsonic acid doesn’t grab headlines like some synthetic chemicals, yet it finds its way into many agricultural settings. Most people don’t come across this compound at the grocery store or pharmacy, but its impact quietly shapes parts of the food chain—especially in animal agriculture.
Role in Animal Farming
Farmers looking to boost feed efficiency in poultry have reached for anthraquinone-1-arsonic acid as a feed additive over the years. The idea behind such additives is simple: help birds gain weight faster, ward off digestive problems, and increase output. It delivers faster growth so commercial producers can move flocks through the system at a quicker pace. This might sound helpful from a business side, but it comes with a list of side effects people worry about—including residues in meat, the potential exposure of farmworkers to hazardous substances, and the challenge of keeping environmental runoff in check.
Health and Environmental Risks
I grew up in a rural part of the Midwest surrounded by fields and livestock operations, and the local community didn’t often talk about feed ingredients in detail. Yet, stories circulate—farmers sharing news about new regulations or odd results in lab reports. The issue with Anthraquinone-1-arsonic acid isn't just about animal growth, but about what sticks around after its use.
Arsenic-based compounds like this one grab the attention of scientists and public health advocates for good reason. If residues linger in chicken meat or eggs, families eating these products could face health problems after years of exposure. Runoff from farms has also led to concerns about drinking water quality, since arsenic can settle in soil and find its way into nearby rivers and groundwater. Research from groups like the Environmental Working Group points out that even low levels of arsenic exposure over time increase the risk of certain cancers and skin issues.
Regulation and Consumer Pressure
Regulators haven’t ignored these risks. In recent years, more countries have moved to restrict or phase out the use of arsenic-based feed additives. The U.S. Food and Drug Administration, for instance, began tightening rules on similar compounds after tests showed residue in finished chicken products. Consumers are more tuned in now, too, demanding cleaner labels and asking questions about where their food comes from. This momentum pushes producers to look for safer, cost-effective ways to keep their birds healthy—without leaning on questionable chemicals.
Moving Toward Safer Solutions
Farmers often need reliable tools to protect animal welfare and bump up yields, but it’s hard to ignore medical studies connecting long-term arsenic exposure and human health. Alternatives exist, and more producers rely on improved nutrition, probiotics, and targeted vaccines. These methods might take more time or investment up front, though they give peace of mind to families and help keep public trust in the food system.
My own experience with farming families tells me that most want to do right by their land, livestock, and buyers—even if regulatory pressure plays a role in their choices. Ultimately, keeping up with research, sharing knowledge, and giving a voice to consumers, farmers, and scientists alike paves the way toward better outcomes—on dinner plates and in communities across the country.
Is Anthraquinone-1-Arsonic Acid hazardous or toxic?
The Substance in Question
Anthraquinone-1-arsonic acid might sound unfamiliar, but chemicals like this have been part of agriculture for decades. Historically, this compound popped up in feed additives for livestock, pushed by a goal to help animals grow faster. Some markets also saw it included in dyes or specialty industrial processes, harnessed for its chemistry rather than its character. The moment arsenic sneaks into a molecule, science needs to take a closer look.
Looking at Safety Concerns
Personal experiences with feed formulations have taught many in agriculture to read ingredient labels with skepticism, especially when any form of arsenic shows up. Arsenic, whether organic or inorganic, never gets a free pass. Decades ago, nobody batted an eyelash at organic arsenicals like this one. It sounded less scary than metallic arsenic or infamous poisons. Over time, studies showed that the "organic" label doesn’t guarantee safety—and the tide began to turn.
The issue rests on metabolic breakdown. Animals don’t keep anthraquinone-1-arsonic acid in its original state. Once inside a chicken or pig, certain forms of organic arsenic can convert into inorganic forms. Inorganic arsenic is toxic to humans, linked to skin lesions, cardiovascular disease, or cancer, just to name a few health risks. People worry about residue: When the feed passes through the animal, does dangerous arsenic end up in meat or waste, and ultimately the environment?
Regulatory Actions and Studies
Regulators in the US, Canada, and the EU have paid attention. The FDA, for instance, withdrew approval for products containing roxarsone, a similar organic arsenic compound, over these toxicity concerns. In the EU, anthraquinone-1-arsonic acid isn’t seen as a safe feed additive for animals, let alone humans. Research followed the regulatory action, with studies showing measurable levels of inorganic arsenic in both treated animals and runoff from farms where waste gets used as fertilizer.
These studies changed how veterinarians and farmers handle animal feed. The evidence points back to one lesson: animal feed has to go through more scrutiny, especially if it contains legacy chemicals like arsenic-based compounds. There’s plenty of proof that most consumers want their food supply to be free from even the hint of contamination.
Risks Beyond the Farm
Working in agricultural settings, I learned that contamination doesn’t stop at the farm. Waste with arsenic compounds works its way into water systems, soil, and eventually food plants. It’s easy to see why environmentalists keep raising red flags. Kids who drink well water from areas where this kind of waste was spread decades ago still face chronic arsenic exposure risks. No one can joke about cancer clusters.
Solutions and Safer Pathways
There are alternatives on the table. Growth promotion in livestock no longer leans on chemical crutches as often. Improvements in nutrition, better farm hygiene, and vaccination programs lower disease levels and boost productivity without chemical shortcuts. Feed manufacturers now opt for mineral mixes with trace elements carefully monitored—iron, zinc, selenium—minus the dangers. It’s a win when both livestock and people benefit.
Ongoing vigilance matters. Each new study into the safety of animal feed ingredients should get a fair hearing. Scientists, regulators, and producers do best when they keep a skeptical eye and ask if anything in the food chain could harm people down the road. The days of assuming “organic” arsenicals are safe because they once got regulatory approval belong in the past.
What are the storage conditions for Anthraquinone-1-Arsonic Acid?
Getting Straight to the Point about Chemical Storage
Working with chemicals, things can turn risky quickly if storage goes sideways. Anthraquinone-1-Arsonic Acid, used in research labs and sometimes in specialty manufacturing, asks for special respect. If someone treats it like a regular powder, trouble finds them. I’ve spent time in labs and seen how sloppy habits lead to costly messes or, worse, safety scares. Proper storage isn’t overkill—it keeps folks healthy and stops expensive materials from going bad.
What’s Special about This Compound
Anthraquinone-1-Arsonic Acid brings both organic and arsenic chemistry together. The arsenic part should put everyone on alert. Dust or spills from this stuff can be toxic, and long-term exposure stacks up problems for both people and the environment. Reports on chemical compatibility highlight its sensitivity to moisture and need for controlled storage. Ignoring these facts costs more than money—it risks lives and wrecks reputations.
Everyday Best Practices from the Lab Floor
Locking up Anthraquinone-1-Arsonic Acid in a dry, cool spot with a tight-sealing container keeps it stable. Humidity brings caking or chemical changes that ruin batches. Heat speeds up decomposition, shifting the chemistry and sometimes releasing toxic fumes. Refrigeration usually isn’t right—unless a supplier’s very clear about it—because condensation can slip in each time someone opens a container. A shelf in a chemical cabinet away from sunlight, moisture, and sources of heat—simple, but it works.
Labels matter just as much as the right shelf. I’ve run across old jars with faded writing, and no one could say what waited inside. Label everything, list purchase dates, and note hazards. Updating an inventory spreadsheet helps, but only if someone checks it and tosses outdated or questionable stock. The EPA and OSHA both give clear guidance: don’t wait until regulators ask questions—keep chemicals tracked from day one.
Personal Safety and Environmental Responsibility
No one gains by skipping gloves or eye protection. Trained hands and the right safety gear cut the risk of exposure. Dust from this acid is no joke—skin, eyes, and lungs all need protection. If a spill happens, grabbing the right cleanup kit and knowing the emergency protocol makes the difference between a five-minute hiccup and an all-day hazard mitigation event. Waste goes into sealed, labeled hazardous containers, not down the drain or in the regular trash. Ignoring disposal rules invites heavy fines and community backlash.
Making Storage Work for Everyone
The temptation to put rare chemicals at the back of a forgotten shelf runs strong in busy labs. But discipline pays off. Regular checks for leaks, lid tightness, and expiration dates catch trouble early. Team training sessions—every year or when turnover happens—help everyone remember the right way to store and handle these materials. Investing in purpose-built chemical cabinets with ventilation brings a one-time cost but protects everyone in the long run.
Practical Steps Forward
Treating Anthraquinone-1-Arsonic Acid with the respect it deserves keeps chemistry safe and effective. The next time someone buys or opens a new bottle, double-checking the storage area and updating labels only takes a minute and saves a world of hurt. This isn’t just about compliance—it’s about protecting the people who make research and industry run.
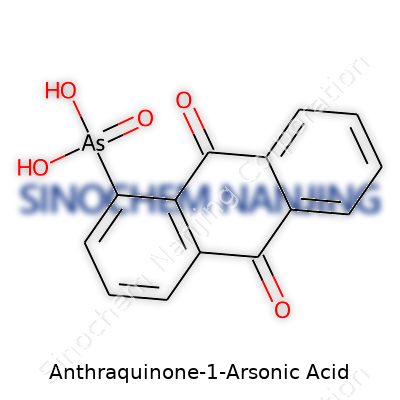
What is the chemical structure of Anthraquinone-1-Arsonic Acid?
A Simple Breakdown of a Complex Molecule
Anthraquinone-1-arsonic acid often gets attention from chemists for its unique combination of aromatic rings and elements not found in everyday molecules. If you picture a chemical structure as a city map, this compound stacks its “buildings” in a way that speaks to creativity and a little bit of risk. It starts with the anthraquinone backbone, which looks like three benzene rings fused together in a row. That’s a tried-and-true setup for dyes, pigments, and other colorants. The name gives away an important detail: this molecule wears an arsonic acid group at position 1. In chemical terms, that means an –AsO(OH)2 group is attached to the first position on one end of the three-ringed core.
The Atoms and Bonds
Zooming in, you meet carbon, hydrogen, and oxygen from the anthraquinone part, and then arsenic, more oxygen, and a bit of hydrogen from the arsonic acid group. The anthraquinone segment offers up a flat, stable wallpaper that helps the molecule slide into biological spaces or materials. Its two ketone groups double up the oxygen count, reinforcing the molecule’s toughness. Throw in the arsonic acid appendage, and suddenly, you’ve got an acid that behaves differently from common ones like sulfuric or phosphoric.
Why This Structure Matters
Throughout chemistry, anthraquinone-based molecules have left a mark. Natural anthraquinones show up in plants and mold, coloring everything from rhubarb to lichens. They’re not just pretty though: those fused rings can produce reactive oxygen species, kickstart electron transfers, and even act as scaffolds for cancer drugs. Slapping an arsonic acid group onto this skeleton changes its game. Arsenic groups bring toxicity and medicinal power. For years, arsonic acid derivatives popped up in animal feed additives and old-school anti-infective drugs. Anthraquinone-1-arsonic acid walks a narrow path, mixing pharmacology with risk.
The Risks and Lessons from Experience
Handling arsenic compounds seems easy in textbooks but, in any real lab, you learn to treat them with deep respect. Even a slight dusting in the air calls for fume hoods and gloves. I once saw a colleague develop a rash after a quick spill, even though the exposure barely lasted a minute. Long-term, chronic exposure links directly to cancer and other serious health issues. Looking at anthraquinone-1-arsonic acid, I remember hearing stories about how these arsenic-laced feed additives ended up in groundwater, putting nearby communities at risk.
Responsible Use—Drawing the Line Between Benefit and Harm
Chemists play a larger role than just building new compounds. Their knowledge of structure translates directly into public safety. Anthraquinone-1-arsonic acid might have helped boost growth in animals but left a chemical legacy in farm soil and waterways. The answer isn’t to ban all innovation, but to reduce the environmental and health costs wherever possible. Researchers now look for alternative growth promoters and strict disposal protocols. Transparent labeling, responsible sourcing, and ongoing monitoring make a difference.
Keeping Pace with Discovery
Wider awareness of a molecule’s structure helps society weigh both its promise and its pitfalls. Reading a skeletal formula isn’t just for chemists. When people realize what an arsenic group means, they’re better prepared to ask questions about food safety, soil chemistry, and water quality. With science moving quickly, the story behind anthraquinone-1-arsonic acid reminds us that every atom in a structure matters, shaping the world in ways we might not notice at first.
How should Anthraquinone-1-Arsonic Acid be handled safely?
What’s At Stake With This Chemical?
Anthraquinone-1-arsonic acid shows up across certain research and manufacturing spaces. It doesn’t belong anywhere near folks who respect clean air, safe drinking water, or basic skin health without solid safety habits. Breathing in particles or fumes, getting this stuff on the skin, or handling it with bare hands leads to irritation, sometimes toxicity, and a handful of risks that stretch far outside personal discomfort. Real exposure brings on headaches, rashes, and in tough cases, organ stress. When arsonic acid compounds slip out of controlled zones, even the local environment catches a hit. Arsenic-based chemicals stick around, don’t break down easily, and raise trouble for aquatic life and anyone downstream.
Everyday Handling: Gloves, Goggles, and Respect
Men and women working in labs or around powdered chemicals often hear reminders about safety gear. For anthraquinone-1-arsonic acid, there’s no room for forgetfulness. Nitrile gloves stop the acid from slipping through to the skin. Safety goggles stop splashes and dust from heading for the eyes. Even in well-ventilated spots, a fitted mask blocks inhalation of any fine dust. It’s tempting to skip this when busy or running behind, but that shortcut just puts everyone at risk. I'm always reminded of one hot summer on a research project—one slip led to an hours-long cleanup and a near-disaster nobody wanted to repeat.
Making Clean-Up Part of the Routine
Spills of dangerous powders or solutions create more problems than most people expect. Water, soap, and plenty of patience help, but only if the right supplies are within arm’s reach. For anthraquinone-1-arsonic acid, dry spills need careful collection with damp towels or specialized vacuums so dust does not become airborne. Disposal means sealed containers—nothing flushed or tossed in the trash. At a facility I visited, each lab station kept its own spill kit. No waiting, no searching, just straight to work cleaning up the moment something hit the counter. That small investment made a mess stay small and kept everyone calm. It’s never about overkill—it’s just respect for risk.
Safe Storage: Keeping Risk Where It Belongs
Anthraquinone-1-arsonic acid isn’t the kind of item you store next to coffee or shared office supplies. Dry, labeled containers sit behind locked doors, away from damp, heat, or sunlight. Clear, honest labeling fills shelves across the safest labs. Forgetting a single name or skipping a warning gets someone hurt. I spent afternoons double-checking shelves, never minding the repetition—if it takes another minute to stay safe, that’s one minute well spent.
Training: Sharing Wisdom Beats Injury
Veterans in any field know: knowledge keeps you upright and free from emergency showers or hospital trips. Training means more than signing a paper or watching a faded video. Walking through procedures, showing where spill kits hide, and practicing glove removal without smearing chemicals feels tedious. That hands-on time sticks. It fills in the gaps textbooks leave open. One-on-one training, honest discussions about mistakes, and open eyes toward new information make safety second nature.
Potential Improvements
Not every workspace starts with best practices. Some companies still overlook updated material safety data sheets or ignore local rules about chemical disposal. Sharing info, setting up regular audits, and using real-life stories to show what goes wrong helps shift habits. In my work, a quick refresher every few months proves more useful than once-a-year box-checking. Encouraging staff to point out safety gaps—without blame—spreads watchfulness and trust.
Protecting People Means Protecting the Mission
No chemistry breakthrough, manufacturing run, or data collection justifies risking a team’s well-being. That’s true for anthraquinone-1-arsonic acid and every other potent compound lining the back shelves of research and industry. Care creates more than a record of safety. It lets everyone head to their next challenge—healthy, whole, and even a bit wiser.
| Names | |
| Preferred IUPAC name | 1-arsonylanthracene-9,10-dione |
| Pronunciation | /ˌænθrəkwɪˈnoʊn wʌn ɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 492-19-1 |
| Beilstein Reference | 2858730 |
| ChEBI | CHEBI:85685 |
| ChEMBL | CHEMBL141117 |
| ChemSpider | 13963820 |
| DrugBank | DB12803 |
| ECHA InfoCard | 100.004.697 |
| EC Number | 207-796-9 |
| Gmelin Reference | Gmelin Reference: 167979 |
| KEGG | C18697 |
| MeSH | D000882 |
| PubChem CID | 69250 |
| RTECS number | AY9625000 |
| UNII | F6KB92T9P7 |
| UN number | UN3438 |
| Properties | |
| Chemical formula | C14H9AsO5 |
| Molar mass | 367.14 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.84 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -0.14 |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | 2.66 |
| Magnetic susceptibility (χ) | -85.0×10⁻⁶ cm³/mol |
| Dipole moment | 5.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 228 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1010.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2538 kJ/mol |
| Pharmacology | |
| ATC code | A16AX04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P314, P362+P364, P391, P501 |
| Lethal dose or concentration | LD50 oral rat 46 mg/kg |
| LD50 (median dose) | LD50: 2000 mg/kg (rat, oral) |
| NIOSH | BZ8750000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m3 |

