Aniline Phthalate: A Commentary on Its Past, Present, and Where It Could Go
Historical Development
People started combining chemistry’s building blocks in all sorts of ways as far back as the nineteenth century, racing to invent dyes, medicines, and plastics. Aniline phthalate falls into this story because it comes from two cornerstone chemicals: aniline, which once colored the world with the first synthetic dyes, and phthalic acid or anhydride, a base for paints and plastics. Back in the early days of organic chemistry, benches buzzed with experiments on what would happen if you put these two together. Out of that curiosity came aniline phthalate, a compound that carved out a spot in R&D and manufacturing, without ever grabbing the same headlines as its parent chemicals. It has quietly supported progress, riding on the back of fundamental chemistry that changes how people live and work.
Product Overview
Not a household name, aniline phthalate tends to keep a low profile, making its way into specialty chemical blends, research labs, and places where modification of molecular structure can bring benefits to coatings or materials. Sitting at the intersection of aromatic amines and aromatic acids, this compound represents how simple molecular tweaks often open doors for material scientists and production chemists. Most people don’t talk about it at dinner tables, but its presence shows up in technical spaces looking to harness its unique chemical behavior.
Physical & Chemical Properties
This compound doesn’t give much away with its looks. Usually you’ll find it as a solid, sometimes a powder or crystalline chunk, and it doesn’t have a flashy color. The melting point registers high compared to many organic intermediates, pointing to a strong molecular structure held together by aromatic rings. Put it near a flame or solvent, and it reacts much like you’d expect from phthalates and anilines: resistant to water, eager to dissolve in organics, and sensitive to strong acids or bases. The slight odor—a sharp, chemical tang—reminds you that aniline derivatives never stray far from their roots.
Technical Specifications & Labeling
Anyone handling chemicals day to day knows that specifications make all the difference between lab success and trouble. Aniline phthalate doesn’t throw curveballs if sourced from reputable suppliers. Most suppliers stake their name on purity above 98 percent, free of heavy metals and low on water content. On a warehouse shelf, expect to see labeling that highlights hazards related to aniline’s known toxicity, so nobody risks breathing in dust or letting it touch bare skin. Even with routine use, controlling exposure marks a line between a well-run workspace and an accident waiting to happen.
Preparation Method
Chemistry textbooks draw a straight line from starting compounds to aniline phthalate through condensation reactions—heat up aniline and phthalic anhydride under controlled conditions, often with a catalyst or solvent, and the product comes in solid form. This method has roots going back decades, and while manufacturers have optimized it for reliability, it still needs tight control over temperature, reaction time, and cleaning steps. Small lapses leave impurities or lower yields, and nobody wants to waste expensive precursors. Most labs treating this reaction see first-hand how even a few degrees too hot or too cool change results, proving that art and science share the work.
Chemical Reactions & Modifications
Once on hand, aniline phthalate acts as a handy building block. Its reactive centers open doors for further changes—nitration, halogenation, sulfonation, or coupling for dyes and intermediates. Industrial chemists treat it as a starting position, not a finish line. The aromatic ring stands up well to mild reactions but goes through transformations with harsher conditions, letting researchers swap groups or tack on side chains for entirely new molecules. This kind of flexibility draws out the creative side of organic synthesis, giving room for those who like to tweak and experiment.
Synonyms & Product Names
In the world of chemicals, nothing collects more names than an intermediate that fits in multiple processes. Aniline phthalate also appears as N-phenylphthalamic acid or phthalamic acid anilide in older literature. Sometimes, catalogues list it under trade identifiers based on company codes, but chemists keep to the IUPAC or logical names to make sure everyone’s speaking the same language. Tracking down research articles or patents sometimes means searching across these different terms, an extra chore for anyone hunting for references.
Safety & Operational Standards
Even in labs that’ve seen it all, aniline phthalate demands respect. Its connections to aniline bring real concerns about inhalation and skin contact. Decades of safety studies on aromatic amines show what happens with careless exposure: headaches, methemoglobinemia, and in some cases more severe effects. Regulations often require gloves, fume hoods, and disposal protocols to keep risks contained. Facilities storing any significant amounts keep training updated and audit procedures, because a single missed step can cause lasting harm. Best practices here don’t just check regulatory boxes—they keep people healthy.
Application Area
Industry and academia alike use aniline phthalate for research, especially where new polymers or active materials are in the works. Dye chemists might use it for exploring color fastness or molecular binding. In certain coatings or additives, it has a way of changing final textures or improved solubility. Analytical chemistry has put it to use as a reagent for detecting minor ions, where its specific reactions with certain metals or other compounds help develop rapid diagnostic tests. Anyone who’s spent time optimizing a process will tell you: sometimes a little-known intermediate becomes a secret weapon for new products or smarter production.
Research & Development
Across academic publications and industrial research, aniline phthalate gets attention for its versatility. Polymer chemists push its limits, checking how it influences chain structures or affects thermal stability. Environmental researchers dig into how it breaks down or transforms in wastewater, trying to make sure processes stay green and safe. Universities sometimes explore new derivatives as potential pharmaceuticals or catalysts, each experiment spinning out more knowledge or a wild card result. Even with all these efforts, the chemical still holds some mystery, waiting for a curious mind to stumble on a fresh angle.
Toxicity Research
Aniline holding onto its reputation as a health hazard means any related compound deserves a close look. Researchers keep coming back to chronic exposure studies, acute toxicity reports, and environmental impact statements. Data from animal models highlights risks of organ toxicity if poorly handled, driving home that just because a chemical proves useful, it shouldn’t become casual. Policy makers and industrial safety engineers work together to set threshold exposure limits and improve monitoring, using past evidence to inform present precautions. Focusing on education at every level, from new students to experienced operators, shows that knowledge makes the difference between routine use and regret.
Future Prospects
Looking ahead, aniline phthalate stands as a representative of compounds caught between opportunity and caution. New material science keeps opening doors for molecules like this, especially as industries chase smarter, lighter, or more durable materials. Green chemistry efforts might one day bring improved synthesis or safer versions, reducing reliance on hazardous parents like aniline. Research circles expect tweaks in the molecular structures could produce brighter pigments, stronger coatings, or innovative medical devices. Real breakthroughs depend on people willing to dig deep, test boundaries, and weigh risks. Whether or not it ever becomes the centerpiece of manufacturing, it will keep working quietly in labs and pilot plants, shaping progress one reaction at a time.
What is Aniline Phthalate used for?
Understanding Aniline Phthalate
Aniline phthalate rarely gets the attention of the bigger chemicals that shape our daily products, but in certain circles, it has made a real mark. This compound comes about when aniline meets phthalic acid, and anyone who’s spent time in a chemistry lab has probably seen it referenced in older textbooks. In my college days, we actually used it in organic chemistry, not because it was flashy, but because it helped us with a basic yet critical test: the detection of hydroquinone.
The Spot Test—A Classroom Staple
For many years, aniline phthalate served as a reagent in chemical spot tests. Specifically, labs used it to pick out tiny bits of hydroquinone, which pops up in everything from photography chemicals to hair dyes. The test works thanks to a simple color reaction: touch a paper strip with some hydroquinone, add aniline phthalate, and a distinctive blue or green color appears. For students and scientists alike, this quick test gave a clear ‘yes’ or ‘no’ about the presence of this compound, and no fancy instruments were needed.
Why is this so important? In fields like environmental science and forensic chemistry, answers need to be fast. Plus, in places where machines like gas chromatographs aren’t available, a strip test can mean the difference between finding a contaminant or missing it. Spot tests with aniline phthalate, while a bit old-school now, opened doors before technology caught up.
Industry and Research Use
Aniline phthalate isn’t just a relic of textbook experiments. In chemical research, it acts as a building block for other reactions. Its ease of preparation and predictable behavior give scientists a way to study how certain aromatic compounds interact and react. Sometimes researchers dive into similar compounds just to see how the chemistry shifts—a method that leads to new discoveries, not only in dyes and pigments but also in developing better analytical reagents.
Health and Environmental Concerns
With anything that sounds like it belongs in an old lab drawer, questions about safety pop up. Aniline, from which this compound starts, comes with real health concerns. Extended contact or careless handling can lead to skin and respiratory problems. Phthalates, on the other hand, tug at a much bigger debate. Many phthalate chemicals have been linked to human health risks and environmental harm, especially after they enter waterways. Studies show that some phthalates disrupt hormone functions in both animals and humans. Dropping aniline phthalate into the water table could create trouble in the long run.
I always tell young lab workers, “Don’t treat any chemical as harmless,” no matter how familiar it feels. Better training and tighter rules around disposal have trimmed some of these risks, but small labs and developing regions often struggle to keep up with best practices.
Finding Safer and Smarter Ways Forward
Today, scientists push to cut down on using risky substances without sacrificing accuracy or access to results. Alternatives to aniline phthalate spot tests keep rolling out. Now, portable spectrometers and colorimetric kits offer safer chemistry, using less hazardous chemicals and clearer disposal guidelines. These newer tools still cost more and aren’t always easy to find, but they offer hope for future generations of scientists and students.
Until technology closes the gap the rest of the way, clear labeling, staff education, and investment in better protective gear can shrink the practical hazards where older chemistry remains in use. Those simple steps help make sure knowledge—and safety—gets passed along with the experiments themselves.
What are the safety precautions when handling Aniline Phthalate?
Understanding the Hazards
Aniline phthalate doesn’t belong anywhere near your skin or lungs. This chemical, found in some dyes and plastics, brings danger with its ease of absorption and toxic fumes. Many lab workers, including myself, remember the choking scent and the unmistakable headaches that follow even brief exposure. The risks go much deeper: both aniline and phthalates are linked to blood disorders, nervous system trouble, and in some studies, cancer. Handling this stuff with care isn’t extra credit—it’s required for protecting your health.
Preparation Starts Before You Open the Bottle
No one should walk blindly into a lab and handle aniline phthalate for the first time. Training matters here, not only for obeying the rules, but also for keeping yourself and everyone nearby safe. Before I ever worked with it, I learned what to do if it splashed on my clothes, or how to recognize vapor leaks. Safety Data Sheets (SDS) need to be close at hand, reviewed, and actually read—not skipped over. Know the symptoms of exposure: blue lips, confusion, and rapid heartbeat call for an immediate response, not blind guessing.
It’s All About the Right Gear
Standard cotton lab coats don’t cut it. I usually grab a chemical-resistant apron, proper nitrile gloves, and full goggles. Aniline phthalate eats through thinner gloves quickly, so double gloving works best for me, especially when moving bigger quantities. A fume hood is not optional; its fans pull dangerous vapors away before they get a chance to enter your lungs. Even with decent ventilation, I always test for leaks and check the airflow before starting any transfer.
Spills Happen—Plan for Them
I once watched a colleague knock over a bottle, and I’m glad the spill kit sat right on the counter instead of locked away somewhere. Proper cleanup gear includes absorbent pads, neutralizing agents, and sealed waste bags. Don’t pick up glass shards by hand, and never pour what’s left into the sink. Clean and decontaminate surfaces, and leave the contaminated gloves and clothing for proper disposal, not for reuse. Emergencies aren’t the time to improvise—knowing the location of eyewash stations and showers saves precious seconds.
Storing and Labeling Like a Pro
Storage matters just as much as day-to-day handling. Keep aniline phthalate in tightly sealed containers, away from acids, oxidizers, and direct sunlight. I found it saves time and headaches to double-check container labels, using dates and hazard warnings that everyone can read at a glance. Old or unclear labels only invite mistakes. Only keep the smallest amounts needed for a project on hand; the rest belongs in a secure, monitored chemical cabinet.
What Can We Do To Make It Safer?
Safer substitutions are gaining ground, as some labs switch away from aniline-based chemicals altogether. Automation helps too: remote-controlled transfer systems lower direct contact risk. Teamwork plays a key role—look out for newer colleagues, check each other's gear, and speak up if something looks off. Periodic training sessions and open conversations prevent complacency, which remains the real enemy in the lab. Safety culture isn’t a checklist—it’s a habit built over time, supported by everyone from new hires to senior researchers.
What is the chemical formula of Aniline Phthalate?
The Chemical Composition
Aniline phthalate stands out with the chemical formula C6H5NH3+·C8H5O4-. This combination pairs the aromatic amine aniline and phthalic acid to form a salt. In my time handling chemistry at the bench, writing this formula wasn’t just about memorizing; it’s about recognizing the way chemicals interact to create new materials we rely on every day.
Connecting Chemistry to Daily Life
You encounter aniline phthalate most where color meets fabric. Textile labs and classrooms favor it for identifying and isolating aniline in solutions. Its utility comes through in spot tests because of its unique crystalline form, which is easier to see and collect than aniline oil. Working in a teaching lab, I saw how students better grasped acid-base reactions when they watched colorless aniline convert to solid, distinguishable crystals. It’s proof that chemistry can move from invisible to unmistakable with just the right reagent.
Why Exact Formula Matters
Getting a formula right isn’t only for exam points. If you mislabel, you risk introducing errors or hazards in the lab. Aniline by itself, C6H5NH2, poses toxicity risks and appears as a colorless to slightly brown oil. Phthalic acid, with a formula C8H6O4, showcases its own functional possibilities. Joining the two turns something potentially hazardous into something easier to handle and identify. Knowing the formula lets professionals order, store, and dispose of the salt with clear safety steps, minimizing the risk of exposure.
Supporting Science with Reliable Sources
Textbooks backed up by peer-reviewed research firmly list the formula as C6H5NH3+·C8H5O4-. Chemists at the bench and in industry take this information seriously, using standards like Merck Index or CRC Handbook of Chemistry and Physics. The value here isn’t in one book, but in a track record of accuracy built over decades.
Thinking About Safety and Sustainability
Every handling of aniline or its derivatives comes with a heavy lesson in chemical safety. I remember reading stories from old dye factories, where health took a back seat to productivity. Today, material safety data sheets and workplace regulations spotlight safe storage and waste management. In labs where aniline phthalate gets used, staff train on gloves, goggles, and spill kits as much as they do on test tubes and pipettes. Safer methods and alternative dyes, based on greener chemistry, are cropping up to cut down on workplace hazards while maintaining the precision aniline phthalate offers in tests.
Looking Forward
As technology keeps changing, chemistry doesn’t wait around. Researchers search for less toxic options for both educational use and industrial application. The goal isn’t just better performance, but health and sustainability for the next generation of scientists. Aniline phthalate’s formula will likely stick around textbooks, but future labs may lean toward compounds with fewer risks.
How should Aniline Phthalate be stored?
Why Proper Storage Matters
Anyone who has spent time in a lab understands that some chemicals come with risks many outside the room barely notice. Aniline phthalate fits this description. I remember my first interaction with it during college—my gloves got slightly tacky, and a faint chemical smell lingered on my hands even after washing up. Those little reminders go beyond comfort; exposure carries real health risks. Safety steps with this chemical aren’t just about ticking boxes. They protect skin, lungs, and years of your life.
Get the Details Right in Storage
Open containers or poorly closed caps set everyone up for trouble. Aniline phthalate does not fancy air or excess moisture. Keep it in a tightly sealed container, preferably glass or something certified chemical-resistant. Avoid using metal cans, since these can corrode or even react over time. Clear, permanent labeling is more than a courtesy for night-shift techs—it helps stop mix-ups that lead to exposure or accidents, especially under stress.
I’ve found that shelves should not sit out in the open. Use a cool, dry, and well-ventilated spot, far from sunlight and direct heat sources. Humid spaces or steamy storage rooms become a risk, as the chemical’s properties can shift with excess damp or warmth. A locked cabinet rated for hazardous chemicals solves more problems than it costs to install. Storing large amounts right next to regular solvents or organic peroxides does not end well. That’s more than just a textbook warning: cross-contamination or incompatible pairings have set off fires before.
Watch for Health Risks and Legal Rules
Breathing in aniline phthalate over months or years—no matter how much you think you’re used to it—takes a toll on blood, nerves, and health. Skin contact can cause irritation, and even a small accidental spill cannot go ignored. NIOSH and OSHA set clear handling and exposure guidelines. Facilities with a chemical hygiene plan protect everyone, not just the folks listed as “chemical handlers.”
Waste disposal can’t fall into the “just pour it down the drain” habit. Work with certified hazardous waste collection. Most local regulations require a log of usage and disposal, which can seem tedious but prevents fines and surprises. It’s worth having a spill kit nearby—one stocked with absorbent pads, gloves, and safety goggles. Training everyone who may step near the storage area takes an afternoon but saves endless headaches in the long run.
Building Good Habits, Not Just Following Rules
It’s easy to stick a container in a box marked “hazardous” and move on. What protects people isn’t the label or the log sheet—it’s the repeated habit of checking seals, separating storage, reading up on regulations, and keeping a spill kit handy. Labs and stockrooms where accidents rise share one trait: over-confidence and corner-cutting. Those I’ve seen thrive encourage quick reporting, use clear process guides, and invest in proper storage setups.
Everyone benefits from a little humility and the discipline to check, not guess. Severe chemical incidents rarely start with a clear warning. They happen after complacency sets in. That old lesson from the lab, the one that made my gloves tacky and set the rules for handling, turns out to be true in every workplace: People matter more than convenience, and safety is a daily habit, not a chore.
Is Aniline Phthalate hazardous to health?
Understanding What We’re Dealing With
Aniline phthalate is a chemical compound used in laboratories and, on rare occasions, in industrial settings. People might run into it during academic research or chemical testing. Not many outside of chemistry circles would recognize it, but the conversation around chemical safety doesn’t hinge on familiarity alone. Health matters touch everyone, regardless of background.
The Hazards: What Science Shows
Getting near aniline phthalate means you’re dealing with the byproducts of both aniline and phthalic acid. Aniline by itself has shown dangers when inhaled, swallowed, or absorbed by skin. Doctors and toxicologists have documented cases of methemoglobinemia, a condition where blood can’t carry oxygen efficiently, after significant aniline exposure. People describe dizziness, headache, and sometimes much worse outcomes in intense cases.
Phthalates draw attention for a different set of reasons. This group includes chemicals used to soften plastics and has raised red flags worldwide. Some studies connect long-term phthalate exposure to hormone disruption and developmental effects. Though not all phthalates spell trouble at the same level, it’s impossible to ignore these reports.
So, handling a compound formed from both aniline and phthalic acid, you notice a pattern: higher potential for harmful effects, especially if safety standards drop or workers cut corners. Having spent time in research labs, I’ve seen how lax attitudes around even low-use chemicals, like aniline phthalate, spark health issues sometimes dismissed as “just a headache.” Over time, repeated contact can add up. The more you know, the less you want to take chances.
Risk Doesn’t Mean Panic
No one benefits from panic, but dismissing chemical risk also gets people hurt. Regulatory bodies—including OSHA and the European Chemicals Agency—push for strict exposure limits with chemicals bearing aniline’s name. Aniline phthalate hasn’t been studied as extensively as its building blocks, but workplaces using it often apply equivalent safety measures: gloves, eye protection, plenty of ventilation. Spills or inhalation can still happen if people let their guard down. Stories of workers taken to the hospital for cyanosis due to aniline-related compounds exist for a reason.
I’ve learned that risk awareness means treating every unknown like a real threat—at least until solid research says otherwise. Most health professionals support the idea: better safe than sorry, especially with symptoms that show up in vague ways, such as fatigue or nausea that can easily be dismissed. Employees want to work in environments where their long-term well-being matters. If you work with aniline phthalate, you should expect reporting systems and regular health checks.
Moving Toward Practical Safety
Some people argue the main problem rests with misuse, not the chemical itself. That only gets half the story right. Training plays a lead role, but oversight and updated research matter just as much. Wherever aniline phthalate shows up, employers ought to provide material safety data sheets, give detailed instructions for handling, and ensure everyone on the floor knows what symptoms to watch for. Institutions can support research into safer alternatives and greener chemistry techniques that replace questionable ingredients with something less risky.
It’s not about painting every chemical as a villain. It’s about keeping people safe, following facts, and refusing to sweep health complaints under the rug. The track record of related chemicals urges respect and vigilance. The right to a safe workspace never takes a back seat, no matter how niche the compound.
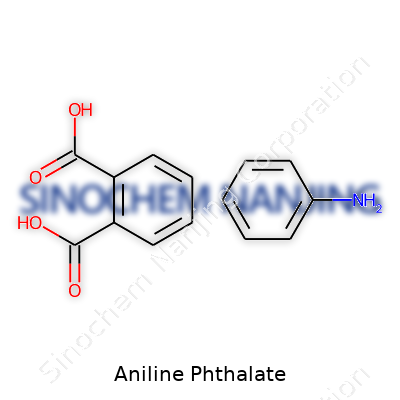
| Names | |
| Preferred IUPAC name | N-phenylaniline-1,2-dicarboxylate |
| Other names |
Anilinium phthalate |
| Pronunciation | /ˈæn.ɪ.liːn ˈθæl.eɪt/ |
| Identifiers | |
| CAS Number | 101-16-6 |
| Beilstein Reference | 82278 |
| ChEBI | CHEBI:134766 |
| ChEMBL | CHEMBL1234947 |
| ChemSpider | 27497 |
| DrugBank | DB14299 |
| ECHA InfoCard | 100.017.840 |
| EC Number | 202-425-9 |
| Gmelin Reference | Gmelin 8359 |
| KEGG | C07284 |
| MeSH | D000886 |
| PubChem CID | 8434 |
| RTECS number | BZ9625000 |
| UNII | 1A3KJL5Z3F |
| UN number | UN1547 |
| CompTox Dashboard (EPA) | DTXSID8020633 |
| Properties | |
| Chemical formula | C8H9NO2C8H4O4 |
| Molar mass | 227.24 g/mol |
| Appearance | Light yellow crystalline solid |
| Odor | Amine-like |
| Density | 1.23 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.96 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.96 |
| Basicity (pKb) | 3.98 |
| Magnetic susceptibility (χ) | -64.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.586 |
| Viscosity | 1.438 cP (20°C) |
| Dipole moment | 3.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.5 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -448.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3227 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V04CH05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes skin and eye irritation; may cause respiratory irritation; possible carcinogen. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P302+P352, P305+P351+P338, P311, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 149°C |
| Autoignition temperature | 650°C |
| Lethal dose or concentration | LDLo (oral, human): 285 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 650 mg/kg |
| NIOSH | UR6350000 |
| PEL (Permissible) | 2 ppm |
| REL (Recommended) | REL (Recommended): 0.5 mg/m³ |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Aniline Phthalic acid Phthalic anhydride Acetanilide Phenylhydrazine |

