Aniline Nitrate: A Closer Look at History, Chemistry, Safety, and the Road Ahead
Historical Development
People have mixed chemicals for centuries, sometimes out of curiosity, sometimes with clear intent. In the 19th century, as the chemical industry moved out from backroom curiosity into industrial maturity, compounds like aniline nitrate emerged as byproducts of efforts to derive more from coal tar and other organic sources. Aniline nitrate gained attention in a period when the lines between dyes, medicines, and explosives blurred. For decades, researchers tinkered around with aniline and nitric acid, reporting explosive results, both literal and in the sense of rapidly expanding knowledge. By World War periods, chemical engineers pressured teams to find alternatives and improvements for propellants, munitions, and industrial formulations, so substances like aniline nitrate found a niche, even as safety concerns made broad industrial adoption a challenge. Still, the versatility of the aniline core ensured its chemistry popped up in patents from wartime innovation through the boom of synthetic dyes and pharmaceuticals.
Product Overview
Aniline nitrate blends one part aromatic amine (aniline) with a potent nitrate component. Chemically, that means carrying characteristics of both world's: the reactivity and aromaticity of aniline, and the energetic, oxidizing push from nitrate. This kind of dual nature creates the risk and opportunity people see in such chemicals. In practice, labs and companies have sold aniline nitrate in the form of colorless crystals or occasionally as mixed solutions, mostly when small complex reactions require a strong nitrating—and sometimes reducing—agent. Today, its direct commercial use runs fairly narrow, but academic and specialty research keeps the substance in the catalog for those who know what they're handling. Beyond chemical circles, the general public rarely hears the name, so it flies under the radar despite its interesting properties.
Physical & Chemical Properties
Aniline nitrate presents as transparent to pale crystalline material, not too hygroscopic but certainly with an edge toward sensitivity—especially heat and shock. Heat gets the material jumpy quickly, decomposing, possibly violently, below what many might assume is a safe temperature. Water solubility registers as moderate, and like other nitrates, mixing with organic material deserves care. From a chemical angle, it keeps the lone pair on nitrogen from aniline, yet acts as a salt pairing with oxidizing nitrate, leading to instabilities. The smell of aniline comes through, a telltale sweet but stubbornly chemical odor, and like many aromatic amines, raises alerts for toxicity and environmental hazard. These physical and chemical realities force rigid lab controls for anyone working with the stuff.
Technical Specifications & Labeling
Producers who sell aniline nitrate typically offer crystalline product rated for research and analysis, packaged securely to avoid accidental shock and moisture. Containers run from small amber glass bottles to sealed plastic-lined drums, with prominent hazard pictograms mandated by international GHS standards. Manufacturers' documentation includes purity grading, moisture levels, and batch traceability—a must for anything classed as hazardous, let alone with explosive potential. Labeling spells out acute toxicity, oxidizer class, and emergency measures, sticking closely to REACH, OSHA, and comparable regional chemical safety frameworks. Occasionally, documentation shows approved use cases, defaulting to research and not-for-human-consumption disclaimers.
Preparation Method
Techs synthesize aniline nitrate by reacting carefully measured aniline with concentrated nitric acid under cooled conditions. Keeping the temperature in check takes skill, as overheating sets off dangerous side reactions. The chemist runs the nitration in an inert or water-ice bath, neutralizes afterward with a base if required, isolates the product by slow evaporation or gentle vacuum filtration, then rinses and dries under tightly controlled lab conditions. Early researchers learned hard lessons with improper ratios or poor mixing, so today’s published methods stress accurate dosing and patient cooling. One misstep, and yields can drop—or worse, the flask can run away toward hazardous decomposition.
Chemical Reactions & Modifications
With its energetic nitrate group linked to an aromatic amine, aniline nitrate plays a reactive role in more advanced synthesis. Chemically, it can act as a donor or acceptor, participate in redox cycles, and even generate further aromatic substitution under controlled catalysis. For example, researchers have explored its use in nitrogen-based bond-forming, though always with solid risk mitigation. In the explosives field, aniline nitrate’s instability sees it mixed into composite materials rather than used alone. Some experiments feed it as a stepping-stone to more elaborate pharmaceutical intermediates or modeling compounds that stand in for environmental hazard studies. Each modification aims to harness the unique blend of aniline and nitrate, while working around the limitations of volatility and toxicity.
Synonyms & Product Names
Aniline nitrate shows up in journals and catalogs under a handful of names, sometimes confusing generations of chemists. Some call it "phenylammonium nitrate," others "aminobenzene nitrate," and more rarely, "benzenamine nitrate." The chemical structure can also appear as C6H5NH2·HNO3 or C6H7N3O3. Trade catalogs prefer straightforward “aniline nitrate” for simplicity, but documentation stresses the connection to both nitro and amine groups, with hazard registers sometimes calling it out under both headings. Each name underlines its dual chemical lineage and attempts to flag its risk as well as potential utility.
Safety & Operational Standards
Aniline nitrate deserves respect. Small missteps spell big trouble, so anyone handling it must invest in proper PPE: gloves, splash-proof goggles, resistive lab coats, and always, a controlled ventilation setup to keep vapors in check. Storage demands cool, dry, and shock-free settings—labs close to the source should never stockpile. If production or disposal hits scale, regional and global rules intervene: transport classifies as hazardous, often forbidden by standard postal or commercial air. Emergency response plans form a non-negotiable element of any standard operating protocol, as spill or fire scenarios risk rapid escalation. In-house training, regular reviews, and tight access controls represent the baseline for anyone bringing aniline nitrate into the workspace.
Application Area
Despite its notoriety, aniline nitrate’s day-to-day industrial use has contracted over time. Specialty labs may request it to replicate old dye chemistry, or as a chemical intermediate in synthesizing complex organic molecules. Some military and pyrotechnic testing still examines its properties in composite energetic materials, though more stable alternatives steal the spotlight today. Environmental chemists and toxicity researchers use it as a reference compound, modeling degradation pathways or simulating nitrate-amino mixing in polluted systems. Beyond these, applications trend narrow because the blend of hazard and modest economic benefit tends not to pay off for broad industry.
Research & Development
Academic chemists continue to probe the limits of aniline nitrate in reaction design—especially where a dual-function nitrate and amine starter could enable shorter, greener synthesis routes for complex aromatic molecules. Some effort focuses on designer energetics, tweaking decomposition rates for controlled initiation or tuning release profiles in specialty propellants. Toxicologists and environmental scientists also use aniline nitrate as a benchmark in screening for nitrated aromatic contaminants. Each new paper nudges the boundaries of knowledge, but most researchers recognize the safety ceiling, sometimes choosing to develop theoretical models rather than large-scale hands-on experimentation.
Toxicity Research
Toxicity stands as an unavoidable reality for aniline nitrate. The parent aniline already clears warning levels for both acute and chronic effects on blood, liver, and nervous system. The nitrate part carries its own oxidative stress load, potentially complicating symptoms for both accidental exposure and planned toxicology trials. Lab animals experience methemoglobinemia and respiratory distress at moderate doses, underlining strict regulatory controls even for potential environmental release. Water treatment professionals also worry about downstream products; both aniline and nitrates have persistence and reactivity in aquatic systems. Regulators, as a result, place severe limits on occupational and environmental exposure, pushing handlers to keep quantity low and protective processes rigorous.
Future Prospects
With risk awareness rising and green chemistry maturing, the long-term clock may be running out for wide use of aniline nitrate outside closely monitored labs. Though the compound remains a fascinating case for reaction chemistry and a necessary part of some forensic or historical testing, safer alternatives with similar synthetic value keep arriving. Some universities keep a sample on hand for specialists exploring old reaction pathways or training hazmat teams, but industry continues a march away from high-toxicity/high-volatility chemicals like this one. The future likely involves more modeling, simulation, and substitution, leveraging legacy knowledge while pushing for cleaner, lower-risk chemical solutions. Progress in analytical chemistry, computational modeling, and process innovation keeps the compounds' legacy alive while limiting its front-line role to a shrinking pool of experts.
What is Aniline Nitrate used for?
The Chemical’s Many Faces
From early days in the chemical storage business, it’s easy to see how certain compounds carry more baggage than others. Aniline nitrate rarely comes up at backyard barbecues, but it has a reputation among folks in manufacturing and research circles. As a product of aniline and nitric acid, this compound looks like just another chemical on a ledger. In reality, it supports everything from specialized dyes to the tough world of explosives.
Behind the Scenes in Industry
Most people never see aniline nitrate on a store shelf or in a delivery truck. That’s because it does its best work behind the scenes. Chemical labs and certain industries draw on its properties to move research forward or build new materials. Its most prominent use lands squarely in explosive manufacturing, giving a punch to demolition charges and mining ops that demand reliable blast power. Safety protocols get strict around it, not only because of its volatility but also its potential health risks. Awareness isn’t optional—it’s the thin line between a successful operation and disaster.
The Science Connection
University years exposed me to labs that counted on chemical reactions with tight margins for error. Where others saw only risk, knowledgeable folks saw tools—if handled well. In research, aniline nitrate occasionally finds a role in making specialty chemicals. Sometimes, you’ll catch it in niche synthesis work. Outside of textbooks, its imprint shows up in applications like dye manufacturing, especially for tough-to-make shades and specialty inks. Scattering its use across smaller batches lets companies avoid the headaches that come from larger-scale risks.
Facts and Cautions
No point pretending the risks are minor. The National Institute for Occupational Safety and Health (NIOSH) labels aniline nitrate as potentially hazardous, linking it to health issues from skin contact, inhalation, or accidental ingestion. Symptoms may include headaches, dizziness, and even more severe outcomes after major exposures. Over the years, regulations forced tighter controls on its shipping and storage for good reason. A friend’s family ran a transport business that once handled shipments of chemicals like this. Even the steeliest truckers respected the strict separation of reactive substances. Spill drills got rehearsed more than once.
Problems and Possible Steps Forward
One stubborn problem with aniline nitrate remains waste disposal and environmental impact. Dumping waste or spilled material results in soil and water contamination, raising environmental and regulatory headaches. Fines stack up; local wildlife pays a bigger price. Factories and storage facilities can move forward by investing in training for handling hazardous materials and by improving fire suppression systems. Updating containment protocols and running regular safety audits catches problems before they spiral.
Room for Improvement
Chemical producers and users could benefit from more transparent reporting and communication with local communities. People living near large plants rarely get a say, yet they have the most at stake if something goes wrong. Raising awareness, not just in boardrooms but out in neighborhoods, makes long-term sense for both safety and trust. Looking back, clear language and honest communication formed the backbone of any solid safety culture. The bottom line: aniline nitrate keeps proving useful, but only if respect for its hazards and impact stays front and center.
Is Aniline Nitrate hazardous or toxic?
What Is Aniline Nitrate?
Aniline nitrate belongs to a group of chemicals that often see use in the lab, especially in industries dealing with dyes and explosives. This compound fuses two distinct substances: aniline, which comes from petroleum and plays a role in making dyes, and nitrate, which reacts strongly with many materials. That blend makes aniline nitrate a hot topic for safety concerns.
Hazardous Properties
One major issue with aniline nitrate comes from the fact that it contains both a fuel and an oxidizer. People have used nitrates in explosives for over a century, and aniline by itself has shown dangerous and toxic qualities. Mix the two, and you have a substance that doesn’t just irritate skin or eyes but poses real risk in the air, especially if it’s heated or if dust gets loose.
Chemical incidents in history have shown the importance of proper handling. Explosions caused by mishandling nitrate compounds have taken lives, destroyed properties, and left long-term environmental scars. Even smaller incidents—such as spills—put workers and first responders at risk. Aniline’s toxic nature adds another layer here. If inhaled or absorbed, it can cause serious symptoms: headaches, blue skin (from lack of oxygen in the blood), confusion, or even unconsciousness. This isn’t fearmongering; these are established outcomes from real life.
Environmental and Health Risks
No one wants to end up exposed to a compound that interferes with how blood carries oxygen. I’ve seen factory workers complain of dizziness and unexplained weakness after aniline exposure. There’s evidence that long-term exposure can hurt the liver and kidneys, too.
Beyond individual risk, there’s a community angle. Leaking aniline nitrate into the soil messes with local ecosystems. Fish die. Water sources that serve whole towns get contaminated. Emergency response teams have protocols for good reason: prevention saves lives and reduces harm long before cleanup becomes a nightmare.
Regulation and Safe Practice
Countries like the United States have legal limits on how much aniline workers can touch during an eight-hour shift. OSHA sets these limits for a reason, not just for the sake of red tape. In my visits to chemical plants, I’ve seen strict safety rules in place. Protective gear, eye washes, spill containment kits—these aren’t there for show. They help prevent emergency room visits.
Some companies invest in training, run drills, and make sure everyone recognizes the smell and signs of exposure. That works in their favor: trained workers respond quickly, report dangers, and avoid shortcuts that lead to disasters. The real danger comes when shortcuts are taken or safety gear gets left in the locker.
What Makes a Difference
Calling aniline nitrate a routine industrial chemical ignores what happens if something goes wrong. Having clear labeling, proper ventilation, and strict accountability in place makes a world of difference. Technology helps—real-time air monitors, digital tracking of storage amounts—but lasting safety comes down to everyday habits.
Communities need reporting systems for leaks and incidents, and companies need real consequences for safety violations. That keeps everyone alert. Those living near storage facilities should know who to call and what action to take in case of a spill. Education doesn’t just belong in the lab—it belongs in living rooms if hazardous chemicals are stored nearby.
How should Aniline Nitrate be stored safely?
Treating Aniline Nitrate With Respect
Aniline nitrate carries a reputation for volatility. Anyone who's handled chemicals in a lab or warehouse knows certain risks are worth close attention, and this compound sits near the top of the list. Over the years, I've seen shortcuts lead straight to disaster, often because someone thought a simple label was "enough" warning. Storage mistakes turn dangerous fast with this material, and stories from plant managers or emergency responders back that up. Few consider the small stuff, but it's the day-to-day diligence that makes the difference.
Recognizing the Hazards
Exposure brings real harm—skin burns, toxic vapors, and the threat of explosion in poor conditions. Its dual nature, sharing traits with both oxidizers and toxins, pushes people to treat it with double caution. The U.S. Occupational Safety and Health Administration calls for strict protocols; smart companies put them at the front of every storage plan. Statistics from hazardous materials incidents remind us how easily bad luck strikes once basic steps get skipped.
Storage Strategy That Works
The right container stands up to chemicals, seals tightly, and resists heat and shock. Stainless steel holds up well, and I've seen heavy-duty polyethylene work for some handlers, provided the drums show zero cracks. Keeping containers full, not partially empty, limits airspace, reducing the explosive risk. Serious operators tag every vessel with a hazard symbol; after walking through more than one storeroom where this step fell through the cracks, I've learned labels save lives as much as physical barriers do.
Controlling the Environment
Temperature matters more than some think. Rooms never climb above room temperature. Good ventilation keeps vapors from pooling, which prevents fires and protects anyone working nearby. I recommend separate, dedicated storage away from other chemicals, since nitrogen compounds and incompatible acids spell trouble when mixed. Fireproofing around the area keeps accidents from jumping to the rest of a facility. Locking the entrance and restricting access only to those trained for hazardous materials forms the last line of defense.
Regular Checks and Emergency Planning
The unsung hero in chemical storage comes down to routine inspection. Scheduled walkthroughs spot corrosion or leaks before they get out of hand. Spills or vapor leaks require a rapid, mapped-out response; every worker ought to know the real steps, not just some training-room theory. Keeping neutralizing agents and protective gear close by speaks for itself after you've seen a spill or unexpected reaction. The smartest companies run practice drills and keep emergency phone numbers posted in plain sight.
Investing in Knowledge and Equipment
Savings from bargain containers or skimping on training vanish with one slip-up. I can point to countless stories where small investments in air-quality monitors or new tubs paid for themselves the moment a fault appeared. Involving experts from chemical safety boards or consulting firms brings an extra set of experienced eyes to any storage setup. Safety data sheets shouldn't gather dust—a printed and digital copy belongs in every workspace where aniline nitrate plays a role.
Staying Ahead of the Curve
Regulations shift as new incidents teach fresh lessons. Keeping up-to-date on required signage, local fire codes, and reporting rules doesn't just prevent fines; it saves lives. Networking with others in the field and sharing best practices spreads hard-won wisdom, which has a way of turning up just when you need it. Chemical storage isn't glamorous, but the discipline pays off every single day nothing goes wrong. That’s real peace of mind you can’t put a price on.
What are the handling precautions for Aniline Nitrate?
Understanding the Risks
Aniline nitrate does not mess around. As someone who’s spent plenty of hours in chemistry labs—always double-checking a label—I know the first lesson is respect. This compound brings a strong mix of toxicity and reactivity. Skin contact stings, and even brief exposure to the dust or fumes lingers on your mind—and your lungs. If you're not careful, it burns before you see the flames.
Why Personal Protection Comes First
Think of gloves, goggles, and a thick lab coat as your first line of defense. Nitrile or neoprene gloves work better than others because aniline slips through latex like water through a sieve. Fume hoods do not just keep that signature bitter smell out of the room; they shield your lungs from more than a nasty surprise. Lab culture sometimes breeds shortcuts, but with aniline nitrate, one time without gear can haunt you with chemical burns or respiratory pain.
Storage That Makes Sense
Dry, cool storage keeps aniline nitrate stable. Folks sometimes underestimate how quickly this compound heats up or reacts with the wrong container. Avoid metal shelves—corrosion is real with even a tiny bit of moisture around. Stick to tight-sealing glass or thick polyethylene, with dedicated cabinets for oxidizers and away from anything flammable. A small spill on a shelf sparks more than cleanup trouble; it invites fire.
Ventilation Matters More Than You Think
Every lab tech has a ventilation story. Aniline nitrate gives off nitrous fumes—unseen, but dangerous. Good airflow saves lab mates from headaches or worse. I’ve seen the difference between cramped rooms with stagnant air and updated setups with real fume hoods: people feel safer, accidents happen less often, and the workplace hums with less of that sickly-sweet edge in the air.
Spill Plans: React, Don’t Freeze
Preparedness counts for more than bravado. Quick access to spill kits means the risk shrinks fast. Neutralizing agents like soda ash tame small messes, but you always want to have enough absorbent on hand, and a route to safety. After a close call in a crowded stockroom, I stopped trusting intuition alone. A posted spill plan and regular drills mean people act with certainty, not panic.
Disposal: No Room for Guesswork
Aniline nitrate doesn’t belong down the drain or in the trash. Local hazardous waste services know how to handle this stuff, and skipping that step leads to more trouble than you want. I’ve seen shops fined and shut down for improper waste disposal—don’t let a shortcut undo months of safe work.
Training Keeps Mistakes Rare
People look out for each other when everyone knows the risks. New lab workers shadow experienced hands and absorb these habits early. Regular training on chemical hazards builds confidence, not just caution. Opening the door to questions—about safety sheets or emergency stops—can keep a bad day from turning into an emergency.
Final Thoughts: Safety Culture Overrules Speed
Rushing with aniline nitrate never pays off. Like most chemical hazards, a “just this once” moment can leave lifelong consequences. Safe work slows things down, but in labs where everyone speaks up and follows the rules, accidents stay rare. That’s experience worth sharing.
What is the chemical formula and CAS number of Aniline Nitrate?
Basic Profile: Chemical Formula and Identification
Aniline nitrate carries a chemical formula of C6H5NH2·HNO3, which essentially means one molecule of aniline and one molecule of nitric acid combine to form a salt. Its CAS number is 3545-69-9. This registration ensures scientists, medical workers, and suppliers speak a common language when referencing this substance, avoiding confusion or dangerous mix-ups.
The Real Picture: Why Knowing These Details Matters
In the chemical industry and across research labs, details like formulas and CAS numbers do more than fill out paperwork. Picture a busy lab bench with bottles carrying dozens of labels—one slip, and a small typo can send safety measures off the rails. Using the right formula and ID number makes sure that everyone, from sourcing to handling and storage, stays on the same page.
My own years in academic research taught me the value of such identifiers. Graduate students and experienced chemists alike double-check CAS numbers before initiating a new experiment or ordering raw materials. The stakes are high; an error could mean a failed synthesis, or worse, a hazardous reaction. It might sound dry, but these details keep people safe and experiments reproducible.
Practical Uses and Hazards
Aniline nitrate has some niche uses—mainly in making explosives and certain dyes—but it doesn't get top billing in most chemical catalogs or textbooks. This has a real-world impact: rarer substances often fly under regulatory radars, or folks overlook proper training and storage. The chemical has a track record of instability, especially when it’s damp or stored too long. Its breakdown can produce toxic products, so quick reactions and responsible disposal matter.
The potential for accidents brings up solid concerns. Regulations exist, but not every facility has strong oversight. Production in low-resource settings or unregulated sectors ramps up the risk of storage mishaps. There are documentaries and case studies where old barrels—never meant to last years—have led to fires, contamination, or even deaths. It's not overblown; an ounce of prevention really is worth more than chasing a spill or fire after the fact.
Building Safer Practices and Clearer Information
The strongest foundation starts with good information. Reliable databases like PubChem and the National Institutes of Health’s resources remain the go-to for up-to-date chemical data, safety sheets, and emergency protocols. Peer-to-peer learning, such as experienced chemists walking new staff through storage procedures, beats online modules any day.
Outside of lab or factory floors, regulatory bodies should enforce inventory tracking for less common chemicals. Even small-batch users can keep better logs—simple checklists on a clipboard, physical or digital, show a lot about what’s in stock and who touched it last.
Better transparency, more robust labeling, and regular checks can head off most emergencies. Companies and universities benefit from involving workers in safety updates, not just sending emails. If every chemical gets the respect—and the clear labeling—it deserves, many disasters become stories never told.
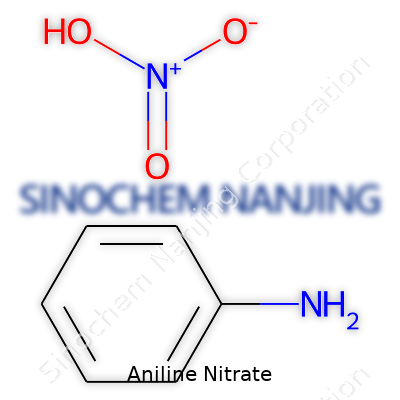
| Names | |
| Preferred IUPAC name | Phenylaminium nitrate |
| Other names |
Aniline nitrate Aniline, nitrate Benzenamine, nitrate |
| Pronunciation | /ˈæn.ɪ.liːn ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 86-88-4 |
| Beilstein Reference | 608942 |
| ChEBI | CHEBI:82230 |
| ChEMBL | CHEMBL3650545 |
| ChemSpider | 80146 |
| DrugBank | DB11472 |
| ECHA InfoCard | 100.013.883 |
| EC Number | 209-317-3 |
| Gmelin Reference | 83628 |
| KEGG | C19315 |
| MeSH | D000875 |
| PubChem CID | 86465 |
| RTECS number | BA9625000 |
| UNII | 0114533H7Z |
| UN number | UN1547 |
| Properties | |
| Chemical formula | C6H5NH2·HNO3 |
| Molar mass | 124.11 g/mol |
| Appearance | White crystalline solid |
| Odor | ammonia-like |
| Density | 1.44 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.20 |
| Vapor pressure | 0.04 mmHg (at 20 °C) |
| Acidity (pKa) | pKa = 4.6 |
| Basicity (pKb) | 9.4 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.552 |
| Viscosity | 2.2 cP (at 25 °C) |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 283.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -287.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1106 kJ/mol |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | May explode by shock, friction, fire or other sources of ignition. Toxic if swallowed, inhaled or absorbed through skin. Causes burns. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS02, GHS06, GHS03 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H411: Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P220, P221, P264, P270, P273, P280, P301+P310, P305+P351+P338, P306+P360, P308+P311, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2 3 1 OX |
| Autoignition temperature | 260 °C (500 °F) |
| Explosive limits | Lower: 1.4%, Upper: 12.6% |
| Lethal dose or concentration | LD50 oral rat 940 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 950 mg/kg |
| NIOSH | LT5425000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Aniline Aniline hydrochloride Nitroaniline Nitrobenzene Phenylhydrazine Aniline sulfate |

