Aniline Acetate: A Grounded Commentary on Its Past, Present, and Prospects
Historical Development
Chemicals often tell stories about human curiosity. Aniline acetate’s trail started in the years when organic chemistry cracked open the secrets behind color and synthetic materials. By the late 19th century, after the isolation of aniline and the birth of organics as a field, acetate derivatives started drawing attention. Scientists realized that combining acetic acid with aniline, a substance sourced from coal tar and plants like indigo, allowed for new classes of indicators and reagents. Over decades, as chemistry advanced from smoky labs to precision-driven research, aniline acetate showed up in classrooms and industrial processes, playing a steady—if understated—role in analytical work, especially in detecting wood lignin and aldehydes. These roots remind us that even compounds that aren’t blockbusters contributed to the proof-and-error cycles that built today’s chemical toolbox.
Product Overview
Aniline acetate isn’t a high-profile chemical in most labs. Most folks run into it as a specialized reagent. It comes as a white or pale yellow solid, well suited to bench work. The compound dissolves in polar solvents like water and alcohol, so it mixes into solutions quickly. Given its stability under ambient conditions, it stores without fuss if kept dry and cool. Industries see its value mostly as a diagnostic tool—particularly when working with plant material, where it helps flag the presence of certain aldehydes or lignin thanks to a color reaction.
Physical & Chemical Properties
Solid at room temperature, aniline acetate melts at modest heat, a little below the boiling point of water. Like its parent compounds, it floats a faintly sweet, slightly musty odor. Its molecular weight, by my calculations, lands just above 180 g/mol. In water, it proves reasonably soluble—a must for anyone creating test solutions. Chemistry teachers like that it gives a fast color readout in reactions, turning papers pink to show certain lignins or aldehydes exist in a test sample. That simplicity and visual response put it ahead of some more complicated reagents.
Technical Specifications & Labeling
Bottles of aniline acetate need clear, robust labeling. The trade expects purity not less than 97%, and solutions, often made at 0.1-1% concentration, land squarely in technical labs. Labels carry the UN number, batch code, synthesis date, and warnings about toxicity and flammability. I won’t forget handling a faded bottle at an old college; clear information helps avoid slip-ups in multitasking labs.
Preparation Method
Chemists prepare aniline acetate by letting aniline react with acetic acid or its derivatives under moderate conditions. Often, labs add glacial acetic acid to a cooled solution of aniline, mix well, and let crystalline aniline acetate form upon standing or gentle cooling. Filtration and drying finish the process. The whole setup reflects typical acid-base salt formation—basic aniline accepts a proton from acidic acetic acid—yielding a product with little fuss, though solid technique still matters to avoid waste.
Chemical Reactions & Modifications
Aniline acetate enters reactions as both a source of aniline and as a mild acid. It supports diazotization work, a classic process for creating azo dyes or indicators. The compound also finds use where chemists want to protect or modify the aniline group gently. It reacts predictably, breaking down to its components under alkaline or strong heating conditions. Researchers keep things simple with this chemical—there’s little risk of runaway reactions or tricky side products.
Synonyms & Product Names
Across catalogs and journals, aniline acetate picks up a few aliases—aniline ethanoate, acetic acid anilinium salt. In older textbooks from the early 1900s, it sometimes went by “aniline acetix” or “anilinacetat.” Those digging through patents or scientific articles spot these alternative labels, which helps avoid confusion, especially with other aniline derivatives that might look or sound close but work quite differently.
Safety & Operational Standards
Everyone working with aniline acetate faces potential hazards. From firsthand experience, gloves and goggles aren’t negotiable. Aniline, being aromatic, can pass through skin, affecting the blood’s ability to carry oxygen. Acetate salts tend to be less dramatic, but risk increases with concentration. Avoid inhaling dust and spills, work in ventilated spaces, and know where the eyewash station sits. MSDS sheets demand respect for a reason. Lab managers check storage often—dry, dark cabinets far from acids or bases stop accidents before they start.
Application Area
The major arena for aniline acetate is detection: spot tests for wood fibers, especially verifying lignin presence in paper pulp, and checking aldehydes in organic samples. While a customer may find fancier, more modern analytical tools, small outfits still rely on this salt. Historians dug up old records showing its use in identifying plant fiber in archaeological finds; conservators in museums also use it for rapid field diagnostics. Paper mills lean on color reactions to tweak bleaching or pulping processes—old school but cost-effective. In some teaching labs, students still learn to observe sheet color changes to infer chemical composition, gaining hands-on respect for practical chemistry.
Research & Development
Academic groups keep finding novel applications for aniline acetate. Analytical chemistry hasn’t replaced rapid visual indicators, especially in cash-strapped locations or in the field. Researchers combine it with smartphone apps that do automatic color detection, turning a hundred-year-old test into a tool for the digital age. Investigations also look at using modified acetate salts for selective recognition—think microfluidic chips or sensors for environmental aldehydes. The baseline chemistry remains the same, but it gets reimagined wherever agility and speed outweigh the complete analytical readout from big machines.
Toxicity Research
Aniline itself triggers ongoing discussion. It can cause methemoglobinemia when mishandled—oxygen can’t travel in the blood as it should. Animal studies confirm that even at low exposures, risk lurks if sick days stretch or safety corners cut. Strong oversight reduced accidents in most commercial labs, but students or small-scale operations still sometimes skip precautions. Testing continues to pinpoint safe handling and smarter antidotes, especially in developing countries where industrial controls sometimes lag. The acetate form, while somewhat milder, calls for the same respect—errors are rare in well-run labs, but stories from colleagues highlight that every mishap carries lessons forward.
Future Prospects
Looking ahead, aniline acetate may not take the spotlight in new industries, but it won’t vanish soon. As labs push into greener, less toxic chemistry, new salts or safer detection methods may eventually sideline it. For now, especially in education, quick analysis, and in heritage science, its simplicity supports learning and low-cost operations. Synthetic chemists and analysts still hunt for non-toxic alternatives, an effort that — like the history of aniline acetate itself — shows how progress depends on a grounded grasp of the chemical’s role, its risk, and the demands of real work at the bench.
What is aniline acetate used for?
Understanding Aniline Acetate’s Purpose
Aniline acetate rarely pops up in everyday conversations, but it holds a quiet importance in chemical labs and manufacturing floors. Chemistry students run into this compound during classic paper chromatography tests, often using it as a reagent to check for specific substances. In the real world, aniline acetate works best for detecting certain sugars—pentoses especially—because it reacts and gives a bright color that tells scientists what’s present. I remember the first time I watched that pink hue appear on a test strip in a college course. It made the abstract world of chemical structures feel tangible and direct.
Behind the Lab Bench—Why Chemists Choose Aniline Acetate
The test for pentoses, known as the Bial’s test, takes a little vial, a bit of the right chemistry, and suddenly a clear answer reveals itself. That reaction only happens because aniline acetate has the right structure and behavior to spot pentoses out of a mix of other sugars. Lab techs run dozens of these assays every day in clinical labs, plant science research, and even forensic analysis. Consistent results matter, especially when health decisions or crop yields depend on catching the right molecule.
Stepping Outside the Laboratory
Aniline acetate serves more than academic curiosity. Chemical manufacturers use it to produce dyes and pigments for everything from textiles to plastics. These industries lean on precise color reactions, and few things beat a well-tried solution to tweak fading, fastness, or shade fidelity. Textile workers sometimes handle finished fabric that owes its color to those chemical steps, although they rarely see the ingredients firsthand. Back in college, I worked a summer job packaging textiles. I didn’t realize then how precise chemistry guided every batch's color. Learning about these processes later put that hands-on work in a new light.
Questions Around Safety and Handling
While aniline acetate can make tests easy, safety prompts concerns for both workers and the environment. Aniline by itself has a tough reputation—it’s toxic, can cause methemoglobinemia in high doses, and requires protective gear in handling. Downstream effects matter. Water treatment plants need to know about chemicals like aniline and its compounds in industrial run-off, since these materials don’t always break down easily in nature. Regulatory bodies like the EPA set strict thresholds, pushing companies to capture or neutralize waste before discharge. Employees in chemical plants keep gloves, eye protection, and fume hoods close by. Every safety briefing drills home the message of caution and respect around such chemicals.
What Could Move the Industry Forward?
Many labs and factories look for substitutes or updated safety measures, but aniline acetate’s reliability makes it hard to swap out. Tweaking ingredient lists or introducing automation, though, could lower risks. Academic researchers sometimes try “green chemistry” angles—using alternative reagents or recycling more. The push for safer processes blends practical chemistry with public health goals, showing that old-school chemistry doesn't need to mean old-school hazards. I’ve talked with scientists passionate about not just solving technical problems, but about shaping jobs and workplaces that respect both the workers and the wider community.
From school science kits to industry floors, aniline acetate knots together tradition, usefulness, and a call for responsibility. Scientists stay curious, patients deserve safe answers, and workers want to go home healthy. With attention and a little innovation, chemical staples like aniline acetate can keep their purpose—while losing some of their sting.
How should aniline acetate be stored?
Recognizing the Real Hazards
Aniline acetate feels like just another clear liquid in a lab bottle, but I learned early that ignoring its downsides can set up trouble. The chemical world isn’t forgiving. Spills, contamination, or fumes circulating in a shared workspace often catch folks off-guard. People tend to think it’s the big, dramatic accidents causing trouble, but slow leaks, improper labeling, or forgetting to check expiration dates ramp up the risks far more often.
Choosing the Right Spot
Cool, dry corners far away from sunlight fit best for aniline acetate. Heat and humidity do more than ruin the compound—they accelerate its breakdown and create by-products nobody wants to breathe or touch. My old lab partner used to stash things by the window to keep them handy, but we ended up replacing stacks of ruined samples and venting out an entire classroom more than once. Temperature swings and sunlight don’t just threaten purity; they can drive up vapor pressure and leak flammable fumes.
Protection from Contamination
Sometimes, little bits of moisture can sneak into poorly sealed bottles. Moisture builds up, turning a safe chemical into an unwanted experiment with acidity or degradation. Glass works better than plastic for aniline acetate—plastic containers sometimes react or, worse, deform if the compound sits long enough. I always reach for well-capped, amber glass bottles since they cut down on light exposure and slow down chemical breakdown. Tight stoppers keep excess air and water out, which reduces the chance for odd reactions. If you ever spot cloudiness or a color shift in storage, don’t risk it—replace the sample and clean that area thoroughly.
Clear Labeling Never Fails
Lab mishaps usually start with something as simple as a missing or faded label. A sharpie and acid-resistant tape don’t cost much, and they’re worth their weight in headaches saved. Keep the chemical name, date of receipt, and concentration marked right on the container. Out-of-date chemicals produce unpredictable results—nobody wants contaminated data or strange reactions. I run an informal “shelf check” once a month, crossing off anything that doesn’t look or smell right, and replacing labels when the writing fades.
Ventilation and Spillage Preparedness
Fume hoods do more than just suck away annoying smells. Any vapor from aniline acetate becomes a problem for eyes and lungs, especially in small or busy spaces. Even if direct toxicity seems low on paper, long-term exposure wrecks concentration and health. Spill kits should sit close by, stocked with absorbent pads and gloves. Nobody ever expects spills—the right supplies catch the problem before it spreads. A thick lab apron never goes unused in my workspace. Gloves and goggles aren’t just for “big reactions” either—I’ve seen glass bottles slip and break during simple shelf checks.
Training Each New Pair of Hands
Relying on memory or habit works until it doesn’t. Each year brings a new set of students or techs, and every group needs a fresh rundown of safe storage routines. Walkthroughs and practical demos help more than printed posters. I bring out old containers, let the group sniff out the bad samples, and show where the spill kit hides. Sharing real-life mistakes, including my own, sparks better questions and improves safety much faster than any poster or manual. Real stories stick with people long after the lecture ends.
What are the safety precautions for handling aniline acetate?
What Aniline Acetate Means for Everyday Work
Anyone who spends time in a chemistry or research lab has come across substances that need respect. Aniline acetate is no exception. With its toxic fumes and risks for skin and respiratory exposure, safety isn't just a formality—it's the difference between a normal day and a trip to the emergency room. Some years back, I handled chemicals daily in an academic setting, and the lessons learned stick with me. You have to put people first. No shortcut is worth an injury. Protecting yourself isn’t just about following a bunch of rules—it's about guarding your health and your team.
PPE: The Absolute Base Layer
Aniline acetate can irritate the skin and eyes, and inhaling its vapors leads to far bigger issues than a cough. Goggle up every time you handle it. The sting in your eyes isn’t something you want to gamble with, so forget about regular glasses—they don’t seal. Gloves aren’t optional. Nitrile does the trick far better than latex, and disposable gloves mean you can toss them if there’s a spill. A decent lab coat works as a basic shield, and don’t forget the closed-toe shoes—just one drip, and you’ll understand why sandals have no place in a chemistry lab.
Why Good Ventilation Matters
No matter how careful you are, fumes can sneak up on you. A couple minutes working with aniline acetate in an open room, and the air feels different—your nose knows before your brain does. Always work in a fume hood. I once ignored this advice for a quick task, and a headache followed for hours. A ventilated workspace cuts down that risk, and if a spill happens, you’re better off having those fumes whisked away instead of hanging around you and everyone else.
Storage and the Unseen Dangers
A lot of risk comes after the experiment ends. Leaving aniline acetate in open containers exposes you to contamination and leaks. Store it in a well-labeled, tightly sealed bottle, and tuck it away from heat or sunlight. I learned the hard way that storing chemicals too close to incompatible substances can mean extra trouble—fires, toxic gases, and ruined research. It pays to brush up on chemical compatibility charts even if you think you remember. Fact: improper storage stands high on the list of lab accidents reported worldwide, so don’t add your story to that statistic.
Spills and Disposal
Spills do happen. You need to know what to do before they happen. My routine includes a ready-to-go spill kit that has absorbent materials, gloves, and a container for quick clean up. Small spills get cleaned immediately, but bigger messes need a step back to make sure everyone knows what’s happened. Disposal relies on following local hazardous waste policies. Pouring leftover aniline acetate down the drain risks environmental damage and legal fines. It takes extra effort to contact certified waste handlers or the campus EH&S office, but it’s non-negotiable if you care about community safety.
Training and Routine Checks
Every year, thousands of lab injuries spring from simple mistakes or gaps in training. No matter your experience, refreshers and walk-throughs catch little things like leaky bottles or worn gloves. Sometimes, the safest labs are those where people check in with each other, talk through their process, and aren’t afraid to call out a problem before it happens. I’ve seen near-misses avoided just because someone took a minute to ask a question.
Safe handling of aniline acetate isn’t rocket science. It’s habits, vigilance, and respect for yourself and your coworkers. Every person deserves to go home healthy at the end of their shift.
What is the chemical formula of aniline acetate?
So, What’s Aniline Acetate?
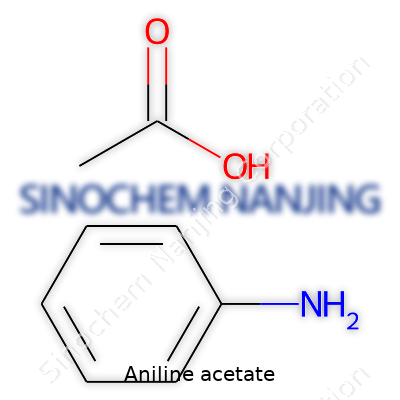
Aniline acetate is the salt formed when aniline reacts with acetic acid. The chemical formula tells you exactly how those atoms come together: C6H5NH3+ CH3COO-. Written more simply, you’ll see it as C8H11NO2. That formula crops up in chemistry labs and textbooks, helping chemists track down what’s in the bottle.
Why It Matters
Having the exact formula isn’t just something that matters to folks doing chemistry homework. In an industry lab, mistakes with a chemical like aniline acetate can mean ruined products or worse. Aniline itself gets used to make dyes, medicines, and rubber additives. If you’re making something where precision counts, like pharmaceuticals or certain pigments, you can’t afford to guess on chemical identity. People have learned that the hard way—labs that mix up compounds run into serious safety issues and regulatory headaches. The formula is a promise: C8H11NO2 means you know what you’re working with.
What’s at Stake with Chemical Accuracy
Every chemist runs into the risk of using the wrong substance because a bottle is mislabeled or misunderstood. The world saw what happened with the thalidomide disaster in the late 1950s and early 1960s—small changes in chemical structure have big impacts. Though that wasn't about aniline acetate specifically, the lesson sticks with anyone who’s spent time in a lab. Precision in naming and formula keeps products safe for consumers, and safeguards workers in the lab.
How Chemists Use This Formula
In teaching, professors lean on correct formulas to show students how chemical reactions produce predictable results. With aniline acetate, you’ve got a classic example of acid-base chemistry. Mix aniline (C6H5NH2) with acetic acid (CH3COOH), and you watch the proton from the acid hop over to aniline, creating a salt. That gives you the formula for aniline acetate.
In research, chemists check these formulas before mixing chemicals or scaling up reactions. One overlooked detail can derail a whole line of work, and in my own experience, nothing stops a project dead like realizing halfway you grabbed the wrong starting material. Clear formulas, backed by reputable databases and standard safety practices, cut down on mistakes.
Solutions to Common Issues
A lot of trouble in labs traces back to poor labeling or confusion over chemical names. Strong communication helps. Include formulas right on chemical labels, double-check bottles before use, and keep databases up to date. In some labs, color-coded systems or digital tracking help chemists match the right substance to the right procedure. On the regulatory side, mandatory safety data sheets need to provide these formulas clearly and in multiple places.
In university labs, training new chemists to look for full formulas (and not just the name) builds good habits early. Mistakes still happen—in my time, I’ve seen everything from spilled acids to entire research batches go down the drain because of a swapped reagent. Each time, it circles back to one core message: Exact formulas keep science honest and safe.
Facts for Reference
Aniline acetate lives at the border between basic chemistry and real-world application. It acts as a reagent to detect certain sugars in paper chromatography. The well-known chemical publications detail its melting point of about 178°C and its solubility in water and ethanol. Knowing the formula, C8H11NO2, offers a quick checkpoint for anyone involved in handling or research.
Is aniline acetate toxic or hazardous to health?
What We Know About Aniline Acetate
Anyone with some lab experience has probably come across aniline acetate, especially in color reaction tests for chemistry work. This compound, formed from aniline and acetic acid, appears in many scientific protocols. The name might sound ordinary, but a quick scan of its chemical structure hints that it carries the track record of both its ingredients. Many folks working in teaching labs or industrial settings want to know how risky aniline acetate really is.
Digging Into the Hazards
Aniline alone drew plenty of regulatory scrutiny over the years. Several studies have shown aniline can cause methemoglobinemia—a condition where blood loses some of its ability to carry oxygen—after skin contact or inhalation. We’re not talking about rare, worst-case events. People exposed to aniline-laden air reported headaches, dizziness, palpitations, and more. Chronic exposure even raised flags for cancer risk. Add acetic acid into the mix and the result, aniline acetate, keeps some of these warning signs. Most hazard summaries describe aniline acetate as poisonous to humans if swallowed or if it comes in contact with skin. Symptoms tend to mirror those of aniline: blue-tinged lips, fatigue, even shortness of breath. Animal studies reinforce these concerns, as high doses cause nervous system problems in test rats and rabbits. It doesn’t take much—reports show health effects at levels barely above routine lab exposures.
Risk in Real-World Use
I’ve worked with university students who use aniline acetate in spot tests for paper chromatography or to help identify sugars. They often ask if gloves and goggles are enough. Gloves do help, but an accidental splash is more common than most students expect. Spilled material on a wrist or forearm can cause tingling and numbness in minutes. Even a whiff of dust rising when weighing the solid can trigger coughing and a sore throat. Some folks shrug off small splashes or minor spills, but this attitude leads to trouble. I remember a technician who ignored a drop on the back of her hand—four hours later, she was in the campus clinic with a severe headache and nausea. Years of anecdotal stories line up with the statements in regulatory guides: don’t underestimate the risks.
Protecting Against Exposure
The best protection still remains old-fashioned attention to technique. Using chemical splash goggles, nitrile gloves, and working under a fume hood guard against splashes, vapors, and fine dust. Skin exposure deserves quick action: rinse the spot right away with plenty of water. Never eat or drink in the work area. I always remind students and coworkers to take Material Safety Data Sheets seriously, even if the chemical shows up in basic lab kits. Keeping containers tightly sealed and cleaning up spills immediately also pays off.
What Else Matters?
Environmental impact often falls by the wayside in the rush to finish experiments, but aniline acetate doesn’t just vanish. Spills on a bench top find their way into drains. Waste programs in research labs keep this chemical out of municipal water—but smaller facilities aren’t always as careful. Regulatory agencies classify aniline compounds as hazardous waste because they threaten fish and other aquatic life. If you’re using this compound, proper disposal matters as much as keeping your hands clean.
Staying Safe and Smart
No chemical in a bottle deserves blind trust. Label warnings, data sheets, and workplace rules exist because folks learned these lessons the hard way. Safer alternatives exist for some tests, but where aniline acetate is necessary, knowledge and habits make the biggest difference. Respect the risks, stay alert, and this compound stays useful without causing harm.
| Names | |
| Preferred IUPAC name | N-phenylethanamide |
| Other names |
Acetic acid anilide Phenyl acetate Anilinium acetate Acetanilide N-Phenylacetamide |
| Pronunciation | /ˈæn.ɪ.liːn ˈæs.ɪ.teɪt/ |
| Identifiers | |
| CAS Number | [541-58-2] |
| Beilstein Reference | 603088 |
| ChEBI | CHEBI:64052 |
| ChEMBL | CHEMBL2106236 |
| ChemSpider | 10477 |
| DrugBank | DB14087 |
| ECHA InfoCard | 100.013.800 |
| EC Number | 292-548-5 |
| Gmelin Reference | 136839 |
| KEGG | C01880 |
| MeSH | D000883 |
| PubChem CID | 8093 |
| RTECS number | AJ3675000 |
| UNII | 0E3E6G1S5B |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID2020783 |
| Properties | |
| Chemical formula | C8H9NO2 |
| Molar mass | 135.17 g/mol |
| Appearance | White crystalline powder |
| Odor | Faint, slightly aromatic |
| Density | 1.22 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.90 |
| Vapor pressure | 0.00016 mmHg (25°C) |
| Acidity (pKa) | 4.6 |
| Basicity (pKb) | 9.4 |
| Magnetic susceptibility (χ) | -61 x 10^-6 cm³/mol |
| Refractive index (nD) | 1.572 |
| Viscosity | 1.02 mPa·s (25 °C) |
| Dipole moment | 2.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 256.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -210.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3226.3 kJ/mol |
| Pharmacology | |
| ATC code | V04CP01 |
| Hazards | |
| Main hazards | Harmful if swallowed, inhaled or absorbed through skin; causes skin, eye and respiratory irritation; may cause methemoglobinemia and affect blood. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H319: Causes serious eye irritation. H351: Suspected of causing cancer. |
| Precautionary statements | P261, P280, P301+P310, P305+P351+P338, P405, P501 |
| Flash point | 70°C |
| Autoignition temperature | 730°F (387°C) |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1625 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1620 mg/kg |
| NIOSH | BX8225000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Aniline Acetic acid Acetanilide Phenyl acetate N-Phenylacetamide |

