Ammonium Periodate: Looking Beyond the Chemistry
Historical Development
Ammonium periodate rarely takes center stage, overshadowed by more common oxidizers like potassium permanganate or sodium chlorate. Old laboratory notes trace its earliest use to the late 1800s, a time of relentless curiosity. Chemists in Europe experimented with the chemical as they tried to uncover unknown stories in the world of periodates. These early discoveries emerged from an era when every new compound brought both excitement and risk. Back then, nobody babysat researchers with modern safety rules or equipment. Flasks boiled over, and folks found out the hard way what an energetic oxidizer could do. Today, its story connects to generations of exploration and careful study that helps keep labs safer than they were in those rough-and-tumble days.
Product Overview
Unlike common household chemicals, ammonium periodate holds its place on the shelf thanks to its unique reactivity. Often sold as a crystalline solid, it calls for respect from anyone handling it. Aside from a handful of niche suppliers, few people outside the chemical sciences have even seen this material. It’s not sitting around in drugstores or grocery aisles. While its peers head straight into fertilizers or swimming pools, ammonium periodate gets tucked away for analytical chemistry, research, or specialized synthesis where its oxidizing strength actually matters.
Physical & Chemical Properties
Handle this salt without care, and you’ll risk more than just ruined glassware. Ammonium periodate usually appears as a white-to-pale yellow crystalline powder. It packs a punch as one of the most potent oxidizing agents in its class. Water dissolves it, but it doesn’t just stir up and sit quietly—watch for fizz and color change if you start mixing it with the wrong things. Heat or friction? You’re courting disaster. Like many periodates, it’s sensitive. Standing around too long in sunlight or warmth won’t do it any favors. These crystals can break down, sometimes violently, splitting apart into iodine and other byproducts. From a chemical standpoint, the compound seems mild, but in the real world, one slip can escalate.
Technical Specifications & Labeling
In my years digging through old storerooms and picking up obscure bottles, I’ve seen less-than-perfect labeling more than once. Industry aims for tight specs: high purity, exacting particle size, and proper moisture content. Nobody wants impurities messing with research results or causing surprise reactions. Reliable suppliers label with UN numbers, hazard pictograms, and detailed instructions—those warnings aren’t just for show. The oxidizing capability must be clear. Researchers expect strict documentation on every bottle, from lot number to storage advice, especially given its volatility.
Preparation Method
Iodine remains at the center of ammonium periodate’s creation. Chemists typically produce it by carefully reacting solutions of sodium periodate with ammonium salts, sometimes adjusting conditions to control the final product’s purity and yield. Temperature, order of addition, and pH: all play a role. Sloppy workup leads to products prone to decomposition or those packed with contaminants. Using cheap starting materials, shortcutting washing steps, or skipping purification usually backfires. If you’re after a reliable oxidizing agent, you learn that every step—from mixing to drying—carries weight.
Chemical Reactions & Modifications
The bond between its ammonia group and periodate anion gives it some attitude. Add it to organics and certain metals, and watch aggressive oxidation follow. Any researcher running a synthesis that needs heavy-duty oxygen transfer knows its importance. Ammonium periodate can split certain bonds that less powerful oxidizers leave untouched. Modifications—like swapping in different cations—shift its solubility or reactivity, tweaking how it plays out in the flask. I’ve seen colleagues try to temper its force by diluting or complexing it, but those tweaks never fully take away its readiness to react. Each reaction tells you something about its limits and how you might harness or avoid its explosiveness.
Synonyms & Product Names
One thing that’s always a hassle: the endless names for a chemical. Ammonium periodate shows up in old catalogs as ammonium metaperiodate, periodic acid ammonium salt, or even obscure trade names when companies try to brand it. Such aliases trip up even the best-trained chemists. Mistaken identity doesn’t just derail a synthesis—it can lead to safety lapses if proper handling info slips by. Standardized nomenclature in chemical databases cuts some of the confusion, but there’s no substitute for double-checking the label anytime you pull a bottle from the shelf.
Safety & Operational Standards
Every new lab tech asks the same question: How dangerous is this stuff? The answer depends on respect and routine, learned from real-world mishaps. I’ve seen gloves pitted after a spill and glassware shatter from a mix-up in reactions. Anyone opening a bottle needs to review safety sheets and suit up with gloves, goggles, and lab coats. Any experienced chemist won’t keep ammonium periodate near flammable solvents or open flames. Storage means cool, dry, and locked down. Waste disposal is another sore point. Down the sink isn’t an option; hazardous waste teams haul it away under strict protocols. Companies and researchers lean on international guidelines like those from OSHA, IATA, and the EU CLP to keep workspaces and transport as safe as possible.
Application Area
The compound finds fans among scientific researchers. Analytical chemists put it to work as an oxidizer in specific reactions, especially if gentler agents can’t finish the job. Chemical synthesis sometimes needs brute force to break bonds or nudge a sluggish transformation. I rarely see ammonium periodate outside specialized research, and scaling up for industrial purposes feels fraught with risk and expensive workarounds. Individual curiosity about niche oxidizers keeps this reagent in circulation, even as safer options crowd the bench. Its use in the field outside labs is slim—no huge manufacturing processes depend on it.
Research & Development
People working in R&D juggle the need for powerful chemicals like ammonium periodate against health, safety, and environmental concerns. In my own experience, most teams want to squeeze as much performance from a reagent before retiring it for something less hazardous. Researchers explore derivatives, stability hacks, and new reaction mechanisms to make operations smoother and safer. There’s a hunger to find greener chemistry solutions. Some folks experiment with encapsulation or immobilization to prevent runaway reactions, with mixed results. Publications drive attention to newly discovered uses or prep methods, but real innovation looks for substitutions that won’t devastate lab safety records.
Toxicity Research
Toxicology around potent oxidizers always draws attention, and ammonium periodate fits that bill. Toxic effects stem from direct contact—risking severe burns—or from inhaling dust. Environmental exposure spells trouble, since released periodates interact strongly with organic material and aquatic systems. Animal studies and in vitro work have shown harmful impacts at relatively low doses. I’ve seen enough cautionary tales among toxicologists to approach this chemical with a full measure of respect. Some researchers, especially those working on new reaction pathways, push for more thorough long-term studies, especially on chronic exposure and breakdown products. Until better data appears, folks tend to err on the side of over-protection instead of rolling the dice.
Future Prospects
No one expects ammonium periodate to explode into mainstream use, but it won’t disappear either. As research shifts toward sustainable and safer reagents, chemicals like this one have their future shaped by regulation and collective caution. At universities and in industry, the push is on for alternatives that do the heavy lifting without bringing such serious hazards. Some advances in chemical engineering may eventually carve out new roles for it, particularly if scientists manage to harness its oxidizing power in controlled environments or find unexpected applications in high-tech synthesis. Until then, ammonium periodate’s contributions to chemistry come with a constant hum of vigilance, a reminder that real progress always weighs benefit against risk.
What is Ammonium Periodate used for?
A Closer Look at Ammonium Periodate
Ammonium periodate doesn’t draw much attention outside research labs, but it packs a punch in chemistry circles. It’s mostly a white or off-white powder, pretty stable when dry, but can be reactive under the right conditions. The unique thing is its high oxidizing power. This alone makes it stand out as a tool rather than something seen in mass markets.
Why Chemists Care About Oxidizing Power
High school chemistry labs rarely touch ammonium periodate. That’s not a coincidence. The compound contains periodate ions, which can break down molecules quickly. Chemists use it to strip electrons from other substances. In real terms, that means they use ammonium periodate to rip apart bonds inside organic molecules.
Think about synthesizing new medicines. Some steps need to trim away pieces of bigger molecules, making them simpler or shaping them for the next phase. That’s when something like ammonium periodate steps in. It’s strong enough to tackle jobs that weaker oxidizers can’t. Pharmaceutical researchers lean on this when a tricky change in molecular structure could unlock a new treatment or a cleaner way to make a known compound.
It’s Not for the Home Lab
Handling ammonium periodate comes with risks. It can spark reactions if things get even a bit out of control. That’s the reason for heavy restrictions outside labs and production plants. The explosive potential, especially mixed with combustible stuff, keeps it off retail shelves. I’ve heard of seasoned chemists flinching a bit when handling it for the first time, double-checking their workspace and equipment more than usual.
Uses Besides Synthesis
Ammonium periodate isn’t just for making new molecules. In analytical chemistry, it helps break down complicated samples. Imagine a lab wanting to detect tiny amounts of metals or organic contaminants. By using ammonium periodate to tear apart the sample, tests run faster since you’re left with just the bare bones of what you want to measure.
Another area that sometimes pops up is teaching about redox (reduction-oxidation) reactions. While teaching, professors choose dramatic reactions that grab attention. Ammonium periodate delivers there too, turning colorless mixtures vivid or generating gases. It’s hands-on proof that big changes can come from what looks like dry powder at the bottom of a vial.
Environmental Impact and Safety
Nothing this reactive can be considered without thought for the waste it creates. Periodate ions won’t always break down in just water and sunlight, so labs handle disposal with care. There’s a push in research toward finding “greener” alternatives. Some chemists are exploring catalysts or enzymes that work just as well, but so far, ammonium periodate has its place in certain must-do experiments and manufacturing steps.
What’s Next?
People will probably keep looking for safer and more sustainable options. Until then, ammonium periodate stays on the shelf for those tough reactions where few other chemicals can compete. It’s never about using the most powerful tool just because it exists—good chemists think twice, weigh the risks, and respect the energy locked inside each spoonful of this compound.
Is Ammonium Periodate hazardous or toxic?
Understanding Ammonium Periodate: More Than Just a Chemical Name
Ammonium periodate doesn’t often pop up in everyday conversation, but in lab settings, it tells a different story. This compound holds a reputation for being tricky to handle, and not because of its name. Chemists recognize it as an oxidizer with extra punch. The compound’s main job usually involves acting as a source of iodine in certain syntheses, sometimes in analytical work or as a reagent in organic labs. It gets its power from the periodate part of its structure, lending it both instability and reactivity.
Real Hazards on the Bench
On paper, ammonium periodate looks like just another reagent. In practice, its ability to release oxygen and iodine under the right conditions deserves respect. The compound decomposes easily, which means it can break down violently when disturbed, heated, or brought into contact with organic materials. Stories from experienced chemists often mention surprise flashes, sometimes smoke, and cleanup headaches. In my own lab time, the protocol came with bold, underlined warnings—never mix ammonium periodate with combustibles or reducing agents. You don’t want to discover by accident that it sets off a reaction big enough to damage glassware or worse.
Inhalation is another concern. Dust or fumes from this compound can irritate the nose and throat; at higher doses or over time, that irritation steps up to more serious health worries for the lungs. The eyes and skin can also react, sometimes with lasting stinging and rashes. Some researchers recall stinging sensations that lingered long after lab coats got turned in for the day.
Facts Behind the Risk
Research and safety data sheets agree: ammonium periodate isn’t for casual handling. The National Institute for Occupational Safety and Health (NIOSH) points out how oxidizers like this amplify fire risks, even with substances that usually burn slowly or not at all. Even common dust or cotton wadding can turn flammable when this salt lands on it. Calls to poison control centers after unintentionally breathing in or touching similar oxidizers happen every year. Most incidents result from storage mistakes or a rushed cleanup job.
Chronic exposure data on ammonium periodate stays limited, but the potential for tissue damage and respiratory distress shows up in case reports and animal studies. It fits the broader category of iodine-based oxidizers, many of which leave lasting impacts if protocols slip.
Building Safer Labs and Workplaces
Mistakes with ammonium periodate often come down to missing awareness or skipped steps. The solution is built around education and a culture that values labeling, ventilation, and proper protective equipment: goggles, gloves, solid ventilation, and training that sticks. Storage in a cool, dry, separated spot makes sense—not just because the law demands it, but because workers go home safer. Keeping incompatible chemicals well apart, keeping the handling bench tidy, and never working alone with unstable substances are practical steps.
Water-based cleanup and emergency kits belong near any bench using ammonium periodate. Quick action can make the difference between a minor story and a hospital visit. Sharing stories inside lab teams about close-calls and lessons learned makes the warnings real rather than abstract.
Smart Questions for Everyday Use
Anyone working with unfamiliar chemicals should ask supervisors for a walk-through and push for updated safety protocols as new information comes in. It’s not just about preventing a single explosion or a rash; it’s about making every workday safer and sending everyone home at the end of each shift.
How should Ammonium Periodate be stored?
Treating a Sensitive Chemical with Respect
Ammonium periodate doesn’t catch much spotlight, but for anyone who handles oxidizers in a lab or industrial setting, this compound leaves little room for mistakes. I remember the first time I saw this white, seemingly harmless powder. A seasoned chemist simply said, “Keep it dry, keep it cool, and don’t mess around.” That advice stuck with me, and it’s grounded in the very real risks involved.
Dryness Is Safety
Humidity and ammonium periodate make a bad combination. Exposing it to even a bit of moisture nudges it closer to breaking down or—even worse—causing a fire. This is not a compound to leave in a glass jar with a snap lid on a shelf. Use glass or sturdy plastic containers with tight, chemical-resistant seals. Double-bagging inside a desiccator, away from any wet reagents or leaky pipes, sets a strong first line of defense against unwanted reactions.
Temperature—Keep It Low, Steady, and Out of Sunlight
Heat speeds up chemical reactions. With ammonium periodate, a rise in temperature can set off decomposition. For folks used to tossing chemicals onto the top shelf, this routine spells trouble. Aim for a storage space below room temperature—usually in the 2 to 8 °C range. That’s the domain of temperature-monitored refrigerators used only for chemicals, never for food or drinks. Sunlight offers no help here, as UV light also chips away at stability. A dark, clearly labeled cabinet does the job.
No Crowding with Incompatibles
It’s tempting to stack bottles and jars together, but oxidizers demand respect. Keep ammonium periodate far away from anything flammable, organic solvents, acids, or reducing agents. Cluttered chemical shelves hide dangers. I’ve seen someone absent-mindedly store nitric acid and a combustible solvent side by side—not a relaxing coffee break follows that discovery. Separate oxidizers in their own, isolated section with clear warning signs. If organization gets tight, most labs benefit from a quick stock inventory to clear out forgotten or expired materials.
Label Everything and Train People Well
In the rush of busy work, labels sometimes get overlooked or faded. There’s no room for mystery or assumption when storing chemicals like ammonium periodate. Clear, permanent labels listing name, hazard warnings, and date of receipt help prevent mix-ups. I like to add “Extreme oxidizer—keep dry and cool” on each container. Training matters even more. Everyone handling or storing the compound must understand the real-world consequences of simple mistakes, not just theoretical risks.
Emergency Plans Matter
Accidents don’t give warnings. Ammonium periodate’s hazards call for ready-to-go emergency procedures. Fire extinguishers for chemical fires, spill kits for oxidizers, and easy access to Material Safety Data Sheets turn confusion into action. I keep an extra copy of emergency contacts right on the cabinet door. Routine drills and honest reviews after near-misses catch gaps before catastrophe hits.
Support from Honest Information
Trustworthy resources save lives. The National Institutes of Health, OSHA, and chemical suppliers regularly update storage recommendations based on new incidents, research, and reported near-misses. I check their guidance every year before reviewing storage protocols. Relying on solid, up-to-date information forms the backbone of any responsible chemical management system.
What is the chemical formula of Ammonium Periodate?
The Formula and Meaning Behind Ammonium Periodate
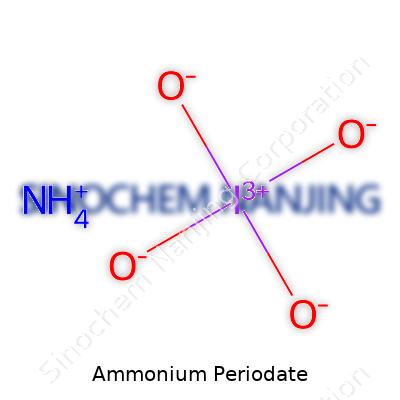
Chemistry likes a bit of puzzle-solving, and ammonium periodate scratches that itch. Its chemical formula, NH4IO4, combines ammonium and periodate ions. On paper, it looks simple: the ammonium ion (NH4+) and the periodate ion (IO4-). The formula pulls together something with historical curiosity, hidden power, and a touch of unpredictability.
Chemical Context: Why Bother With Ammonium Periodate?
Ammonium periodate does not often make headlines in mainstream science news. You won’t find it under the sink or on the supermarket shelves. Labs use it mainly for its oxidizing abilities. The periodate ion loves to grab electrons, which means it can break down or transform other chemicals. In organic chemistry, that has real weight: it can split up molecules in exact ways, especially when it comes to cleaving bonds in sugars, alcohols, or certain organic compounds.
In the hands of a skilled chemist, ammonium periodate helps strip away layers of complex molecules. Researchers have reported how periodates help open up chains of molecules, making it easier to analyze their fragments.
Practical Importance and Safety Reality
Safety counts every time even a spoonful of a strong oxidizer like ammonium periodate comes out. It doesn’t demand respect through its appearance—usually a white solid or powder—but it doesn’t forgive mistakes, either. In the early days of my chemistry training, I watched a demonstration where periodate salts rapidly discolored and broke down a piece of organic matter within seconds. That teaches a lesson more than a textbook ever will.
Reactivity means potential danger. Oxidizers can cause fires or explosions if they come in contact with organic materials or reducing agents. In my own experience, a poorly cleaned beaker, still carrying a bit of residue, can turn into a small hazard when mixed with a bit of periodate. This underlines the need for clean glassware and careful handling anytime ammonium periodate hits the benchtop.
The Environmental and Ethical Side
Many periodate compounds don’t linger in stories about chemical spills or pollution, but that doesn’t mean they don’t pose risks. Heavy metals in their elemental forms can affect groundwater. Even with a chemical that doesn’t leave piles of visible waste, disposal matters. Disposal in the drain without neutralization is out of the question—proper lab protocols and local regulations must guide every step. Strict adherence to safety and environmental guidelines ensures these reactive materials don’t escape into the wider world unnoticed.
Sparking Curiosity: Possibilities and Responsibility
Chemicals like ammonium periodate remind us how even the most niche compounds can have wide-ranging effects. Its formula looks dry on a page, but in a beaker, under the right conditions, it teaches lessons about chemistry’s power. This connection between formula and function sits at the heart of what makes scientific discovery thrilling—and, just as often, humbling. Proper training and respect for these substances open doors to new research without compromising safety.
Final Thoughts
Ammonium periodate illustrates how chemical formulas tie into real challenges and possibilities, inside and outside the lab. Knowledge paired with respect for the risks means safer, more meaningful science, whether you’re breaking bonds or just reading up on what makes these compounds tick.
What safety precautions are needed when handling Ammonium Periodate?
Understanding What You’re Working With
Ammonium periodate is not something you find in an average supply closet. You're dealing with an energetic oxidizer that brings a real risk of fire and explosion if handled carelessly. I learned pretty early in my chemistry days that handling strong oxidizers is nothing like messing with table salt. They have a mind of their own, especially if their environment turns unpredictable. In chemistry, facts have a way of making themselves known—sometimes quickly, sometimes catastrophically.
Personal Protective Equipment Is Step One
It’s easy to say “wear protection,” but it really saves skin and eyesight. Goggles? Absolutely. Not safety glasses—full splash proof goggles. Your eyes can’t grow back. Go for a lab coat made from flame-resistant material, not just cotton. Nitrile gloves work, but double them up, because break-throughs happen. Cover any exposed skin and tie back loose hair. One moment of distraction, and you could end up with a chemical burn or worse.
Keep Water Away—It Changes Everything
Water is not harmless around ammonium periodate. Even a little moisture can set off decomposition, which releases oxygen and heat—possible fire right there. I made a big mistake once by wiping a benchtop with a damp cloth after handling periodates. Nothing happened, but I only realized later how lucky I was. Clean and dry hands, dry surfaces, and a dry atmosphere cut down the risk. Store the powder in tightly sealed containers with desiccants. Forget about the general chemical cabinet. Dedicated dry storage only.
Mixing—Not a DIY Adventure
Mixing ammonium periodate with other chemicals—especially organics, acids, or reducing agents—just creates a recipe for disaster. The compound reacts violently, and sometimes unpredictably, with fuels or common lab solvents. You’ll see warnings about chlorinated solvents; those aren’t just written for liability. Static, friction, or even a spontaneous shock could trigger an explosion if it’s contaminated. Anytime I see someone grinding powders together, it gives me chills—friction and oxidizers don’t mix. Use a plastic spatula, not a metal one, and never grind or triturate this stuff.
Ventilation and Location Matter
Normal lab air systems aren’t enough here. The workspace needs exhaustive airflow, direct to the outside, way above head. Set up a fume hood with a flow monitor. The fumes and dust are hazardous if inhaled. Never lean over the hood to get a “better look.” Inhalation can cause a whole cascade of respiratory trouble. Store only small quantities where you work, and keep enough space between containers to prevent a chain reaction if one goes.
Disposal Isn’t as Simple as the Drain
Disposing of ammonium periodate or anything contaminated with it takes special procedures. Treat the waste as both a strong oxidizer and a possible explosive. Label everything so that nobody else cleans up messes blindly. In the labs where I worked, nobody ever dumped leftovers down the sink. We worked with hazardous waste professionals, following all legal requirements. Neutralizing periodates before disposal—using proven protocols—gave everyone peace of mind.
Preparation Beats Regret
Handling a compound like ammonium periodate rewards careful preparation. From wearing the right gear to labeling your workspace, every step makes a big difference. Rushing through routine or “knowing better” leads to accidents every year across labs around the world. Stick to what the safety data sheet says, respect the compound, stay focused, and don’t cut corners. Your health and your team’s safety depend on it.
| Names | |
| Preferred IUPAC name | Ammonium periodate |
| Other names |
Ammonium metaperiodate Ammonium salt of periodic acid Ammonium periodate(VII) |
| Pronunciation | /əˈmoʊniəm pɪˈrɪ.əˌdeɪt/ |
| Identifiers | |
| CAS Number | 7790-28-5 |
| Beilstein Reference | 3581806 |
| ChEBI | CHEBI:37104 |
| ChEMBL | CHEMBL1201584 |
| ChemSpider | 21568245 |
| DrugBank | DB14130 |
| ECHA InfoCard | 100.029.969 |
| EC Number | 236-647-2 |
| Gmelin Reference | 8498 |
| KEGG | C18716 |
| MeSH | D000678 |
| PubChem CID | 25056 |
| RTECS number | SD7520000 |
| UNII | 3A0MI3756F |
| UN number | UN1479 |
| Properties | |
| Chemical formula | NH4IO4 |
| Molar mass | 284.94 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 3.382 g/cm³ |
| Solubility in water | Insoluble |
| log P | -4.89 |
| Acidity (pKa) | 10.2 |
| Basicity (pKb) | 11.87 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.821 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -298 kJ/mol |
| Pharmacology | |
| ATC code | V03AB47 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes skin and eye irritation |
| GHS labelling | GHS02, GHS05, GHS07, GHS08, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | P221, P220, P273, P280, P305+P351+P338, P337+P313, P370+P378 |
| NFPA 704 (fire diamond) | 2 0 3 OX |
| NIOSH | SU2545000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of Ammonium Periodate: "0.7 mg/m3 |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Sodium periodate Potassium periodate Periodic acid |

