Ammonium Perchlorate: Science, Safety, and the Story Behind the Chemistry
How Ammonium Perchlorate Shaped Technology and Industry
Ammonium perchlorate has played a larger role in the modern world than most people realize. Its journey began in the early twentieth century in research labs hungry for new oxidizers. During the rocket boom of the Cold War, it came to define solid rocket propulsion. Suddenly, the future relied on this white, crystalline powder. Think about the Apollo missions and how every stage demanded materials that could deliver power, stability, and predictability. Ammonium perchlorate met those demands. Engineers wanted a strong oxidizer that wouldn’t break the bank and wouldn’t fall apart under harsh conditions. Its ability to store oxygen in a compact form made it a staple for both military and civilian applications, powering everything from missile systems to the fireworks that fill the summer sky. As the Space Race roared ahead, the people in labs and plants producing this chemical shared a sense of urgency matched only by those who would one day ride the rockets their work enabled. Today, ammonium perchlorate stands as both a legacy and a challenge—one grounded in engineering, health, and environmental stewardship.
Inside the Chemistry—What Really Makes It Work
If you look at ammonium perchlorate’s structure, with its blend of ammonium and perchlorate ions, what jumps out is how easily it sheds oxygen. That pattern translates directly into its explosive potential and its usefulness for propulsion. On a shelf, it appears as a colorless or white powder—unassuming until you understand its reactive personality. Its solubility in water and reactivity with organic compounds turn it into both a building block and a hazard. Heat it up and a violent reaction fills the air with gasses—ammonia, water vapor, hydrogen chloride, and oxygen—powering rockets or shaking the ground in controlled demolitions. These reactions keep researchers concerned, because even a slight deviation in handling or storage creates the risk for accidental detonation. The chemical world calls it NH4ClO4, but plant workers and researchers pay more attention to its behavior than its label. The need for strict environmental controls around ammonium perchlorate has only grown since stories emerged of groundwater contamination near old production sites. This chemical brings both great promise and real risk.
Getting It From Raw Materials to High-Tech Uses
The route to ammonium perchlorate starts with reacting ammonia with perchloric acid, a process that sounds simple until you factor in the need for precise conditions and rigorous controls on purity. Equipment must resist corrosion, and every batch must meet demanding quality thresholds, because impurities can compromise everything from stability to performance. The end product goes through filtering, washing, and drying, often followed by controlled grinding to reach the particle size called for by different rocket or pyrotechnic applications. Those steps carry hazards at every stage—spontaneous ignition, exposure to toxic fumes, runoff of hazardous waste—and require skilled workers and robust safety protocols. Everyone on the floor understands that the rules exist for a reason. The drive to improve production methods also continues to shape academic research. Scientists explore greener synthesis routes and better waste treatment options, mindful of both history and the need for public trust.
Names and Labels: More Than Just Jargon
People in the field know ammonium perchlorate by several names. Whether it’s called AP, ammonium chlorate(VII), or given a chemical shorthand like NH4ClO4, the substance stays the same, but context matters. Military suppliers, environmental regulators, and fire safety officers each speak about it in their own dialects, but everyone understands what’s at stake—safe containment, strict quantity limits, mandatory labeling, and documentation every step of the way. Labeling requirements in countries like the United States and throughout Europe have become stricter over the years. Workers need to know not just what they’re dealing with, but how to detect mishandling or degradation before things go wrong. Cases of ammonium perchlorate incidents have forced changes to these protocols, prompting the industry to reexamine every assumption.
Application in Life and Technology
Looking beyond the lab, ammonium perchlorate powers much more than rockets. Firefighters rely on it in certain emergency signaling devices. Law enforcement and the military need it for controlled demolition and specialty munitions. Pyrotechnicians turn to its predictable burn for dazzling firework displays. Each of these sectors faces questions about lifecycle management: What happens to leftover chemicals? How do you mitigate long-term environmental harm? These questions carry real urgency for workers and communities close to manufacturing and storage sites. Environmental activists and public health watchdogs demand transparency and accountability. Meanwhile, aerospace engineers continue to demand even higher performance, stricter tolerances, and deeper environmental analysis. Every successful launch owes something to the behind-the-scenes work ensuring ammonium perchlorate does the job it’s meant to do.
Research, Modification, and the Push for a Safer Future
Today, ammonium perchlorate sits at the center of intense research. Universities and national labs pursue ways to improve the chemical’s burn characteristics, storage safety, and environmental impact. Some groups experiment with surface coatings or blending with additives to control the rate of reaction and reduce toxic byproducts. Others look at recycling spent material or designing new ways to neutralize perchlorates in waste streams. The toxicity of perchlorate salts—especially their interference with human thyroid function—has made scientists and regulators cautious. Streams and aquifers contaminated with perchlorate raise alarms and push the industry to crack down on accidental releases. The push for alternative green propellants grows year by year, fueled by both regulation and advances in materials science. Ammonium perchlorate’s future now depends as much on environmental innovation as on performance in final products.
Ammonium Perchlorate’s Lasting Impact and Challenges
Wherever ammonium perchlorate goes, debate follows. The advances it powered for global space programs, national security, and public celebrations aren’t in doubt. At the same time, critics highlight the environmental and health trade-offs, especially in groundwater safety around manufacturing hubs. Solutions come from science and regular pressure—from the inside and the outside—to improve production, monitoring, disposal, and cleanup. Transitioning away from ammonium perchlorate is not simple; no direct substitute matches all its properties yet, and scientists caution against replacing one problem with another. Industry and lawmakers keep looking to close the gap with innovation, stricter regulation, and better public oversight. Any real progress demands more cooperation, more transparency, and a willingness to invest in safer, cleaner technology for both people and planet.
What is ammonium perchlorate used for?
Fueling Rockets and Fireworks
Growing up, I often watched rockets take off on TV, cutting through the sky with raw power. Years later, I realized what actually fuels that blast: ammonium perchlorate. This chemical packs a punch, providing the crucial oxidizer in solid rocket propellant, both for NASA’s solid boosters and for missiles. Most commercial and government launches stick with solid propellants for reliability, and the main oxidizer in these products is ammonium perchlorate. It reacts fast with fuel, creating the gases and energy needed to push heavy loads beyond our atmosphere. Military missiles rely on it, too; for them, time and reliability are a matter of life and death.
Outside space and defense, ammonium perchlorate also brings excitement to the Fourth of July. Firework manufacturers turn to it for the same reason: a powerful burn that makes colors pop high overhead. Any time you catch that thunderclap and flash during a celebration, chances are you’re witnessing the chemistry of this salt in action.
The Darker Side: Health and Environmental Risks
I live near a former military site where old rocket fuel got buried decades ago. Local concern spiked when perchlorate contamination appeared in the groundwater. Even small amounts mess with the thyroid gland because perchlorate blocks iodine’s role in hormone production. That’s not something to take lightly, especially for kids or pregnant women. Towns like ours have faced uneasy choices: pay millions for filtration, or risk long-term problems for future generations.
Cleanup isn’t simple. Ammonium perchlorate dissolves in water and moves quickly, making it hard to trap once it’s out. California, Texas, and some parts of the Midwest have dealt with headaches from leaking ammunition plants and fireworks factories that left their mark decades ago. Health experts recommend strict monitoring of drinking water around known sites. The EPA and state regulators face a tough job—sometimes stuck between industry pressure and public health.
Industry Reliance and the Demand for Safer Practices
Even with the risks, space missions and military defense show few signs of shifting away from ammonium perchlorate anytime soon. I talked to a few chemists in the aerospace sector; replacing it isn’t just about swapping out a chemical. Big rockets need stability and lots of power on demand. So far, no one’s found an alternative that handles stress, vibrations, and high heat so well. Research in green propellants, like hydroxylammonium nitrate or sodium nitrate, keeps moving, but nothing has become mainstream for major launches.
Some companies invest in better handling and disposal. They run closed-loop systems to capture runoff and keep contamination off the books. Enforcement and routine safety checks play a huge role. Small steps also matter, like improved storage or replacing outdated fireworks with greener tech for shows.
Balancing Progress and Responsibility
Ammonium perchlorate stands as a reminder that what fuels progress also carries baggage. There’s no denying its importance to rockets, fireworks, and national security. Ignoring water safety and long-term health brings too many risks. Communities near manufacturers or old disposal sites—often folks who’ve lived there for generations—deserve transparency and honest assessment of the threat. More research into alternatives, plus real investment in cleanup, gives everyone a shot at enjoying the spectacle without looking over their shoulder. Progress doesn’t always come easy, but standing still helps no one.
Is ammonium perchlorate dangerous or explosive?
Real Dangers Hiding in Plain Sight
Ammonium perchlorate turns up in headlines from time to time—usually after something goes wrong at a chemical plant or fireworks factory. Growing up near a fireworks manufacturer, caution around chemicals like these became second nature in my community. People who use ammonium perchlorate rarely take its risks lightly. The compound isn’t some everyday powder sitting quietly on a shelf; it powers rockets, missiles, and some industrial applications. All that punch comes at a cost.
What Makes This Stuff Tick?
The reputation for volatility comes from its chemistry. Ammonium perchlorate breaks down when heated, releasing oxygen fast. That trait propels rockets into space, but it also means a mishap or accidental mixture with fuel and heat could set off a rapid—and dangerous—reaction. Unlike gasoline or propane, ammonium perchlorate doesn’t just burn, it acts as an oxidizer, intensifying any fire or explosion it joins.
Accidents Don’t Happen in a Vacuum
Stories from the 1988 Henderson, Nevada plant disaster don’t fade easily. More than 3,000 tons of ammonium perchlorate erupted in an explosion heard for miles. The blast flattened buildings and left a visible mark on safety regulations. Investigators later pointed toward lapses in storage practices as a core reason the event turned so catastrophic. Even decades later, regulators and fire departments use this tragedy as a teaching moment.
Material data from the National Fire Protection Association and the U.S. Chemical Safety Board support those lived experiences. Shortcuts or ignorance about basic precautions risk lives, property, and the environment. Runoff from spills can also contaminate water supplies with perchlorate ions, putting health at risk.
Balancing Use With Responsibility
Despite risks, rocket launches and defense systems depend on ammonium perchlorate. Stopping use altogether isn’t going to happen. Human spaceflight, satellite launches, and even some weather rockets all put trust in its power. Instead, safety comes from discipline and clear ground rules. Trained professionals handle and store the chemical in dry, cool, and well-ventilated facilities, keeping it far from sparks, fuels, or anything that might trigger a reaction.
Transparency helps too. Factories and warehouses serving communities should open up about what they store and train emergency crews effectively. Communication and accountability keep neighbors informed and first responders prepared.
Steps Toward Safer Handling
Stricter regulations make a difference. The Federal Emergency Management Agency lists ammonium perchlorate as a hazardous material, which triggers tighter guidelines for manufacturing, transport, and disposal. Plant operators now run regular safety drills and maintain automatic fire suppression systems. In the past, corners sometimes got cut. Today, the stakes and standards both run higher.
On the research side, scientists keep searching for alternate propellants with fewer dangers to health and the environment. Some new rocket ventures lean on liquid fuels or hybrid designs. Still, nothing in wide use matches ammonium perchlorate’s unique blend of power and reliability in solid rocket motors. Until something better comes along, stewardship—not fear—guides safer applications.
The Bottom Line
Ammonium perchlorate brings speed, energy, and risk in equal measure. Past disasters and daily safety routines both show the need for respect and constant vigilance. Anyone living near an industrial site or working with bonded chemicals knows the uneasy truth: accidents often wait for a single oversight. Serious training and responsible oversight remain the strongest safeguards against explosive consequences.
How should ammonium perchlorate be stored safely?
Getting Real About a Powerful Chemical
Ammonium perchlorate shows up in rocket fuel, fireworks, and airbag systems. Safe storage isn’t just a checklist item for compliance or insurance; it’s personal. One mistake with this material, and consequences don’t just end with property damage—they reach lives, families, and entire communities.
Simple Steps, Serious Stakes
From working near industrial sites, I’ve seen what going through the motions looks like. Pallets stacked too close. Bags stored under leaking rooftops. Companies get comfortable, and then everyone pays for that comfort when a spark turns into a blast. In my experience, clear rules only stop disaster if people follow them with their eyes open.
What Matters Most
For ammonium perchlorate, dry conditions make a huge difference. So does temperature. Storing this chemical in a warehouse that sees wild swings from freezing to blazing amplifies risks. Humidity can cause the material to clump or react. Dirt, oil, and even stray bits of metal set the stage for something bigger than a scare. Think about a storage area that stays below 30°C, away from sunlight and naked bulbs. Fire-resistant walls and non-sparking floors cut down the odds, too.
Companies sometimes focus so much on controlling temperature and humidity, they forget about air flow. A stuffy room filled with ammonium perchlorate builds up fumes and dust. I once oversaw a site where we installed ceiling vents and mechanical fans so air stayed fresh, with sensors that flagged poor air before it built up into a bigger problem.
Getting serious about safety means thinking about who gets near the storage area. Trained staff understand what protective gear they need. They know not to bring matches, phones, or metal tools inside. I’ve worked on teams that ran drills repeatedly, making sure everyone had emergency numbers memorized and exits clear of clutter.
Labeling and Segregation
Every drum, carton, or container should shout what’s inside. Legible labels and safety sheets at the door save time during emergencies. Nearby chemicals matter, too. Ammonium perchlorate shouldn’t share a room with fuels, acids, or combustibles. Back in my early days, someone stored cleaning supplies in the same area. We fixed that overnight, splitting incompatible materials after learning the hard way in a close call.
Monitoring and Inspection
It’s tempting to set up shelves, sign off on a storage procedure, and move on. But chemicals shift over time. Regular inspections flag leaks, corrosion, or caked powder that signal trouble. In places I’ve worked, we used digital logs for weekly walkthroughs. Even small changes—powder around the lid, a strange smell, cracks in packaging—meant time to act.
Building a Culture of Safety
Government rules spell out minimum expectations. Good companies go beyond that. Earning trust among staff and neighbors means open records, visible training programs, and a willingness to invest in improvements. Storage practices should evolve after every incident, even those you hear about from other cities or countries.
Safe ammonium perchlorate storage is never finished. Every step, from dry floors to inspected exits, carries real weight. It’s about respect for the danger and the people whose lives hang in the balance.
What is the chemical formula of ammonium perchlorate?
Understanding Ammonium Perchlorate’s Formula
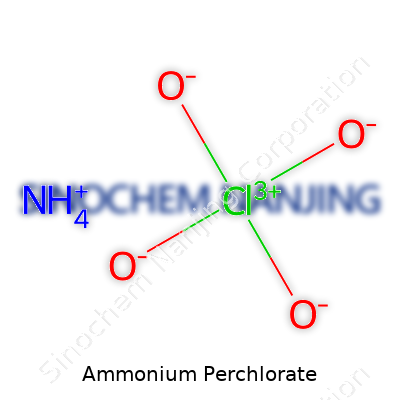
Ammonium perchlorate lands in the spotlight with the chemical formula NH4ClO4. It forms from ammonium ions and perchlorate ions bonded together. That simple combination leads to powerful outcomes. You get a white, crystalline solid that dissolves well in water and acts as a strong oxidizer.
I ran into ammonium perchlorate for the first time during a summer internship at a small aerospace lab. Watching engineers weigh out the powder in a cleanroom drove home just how potent this material can be. A crew of experts—chemists, physicists, and trained techs—wore lab coats and gloves like it was second nature. Even a pinch means serious business.
Why the Formula Matters Beyond the Lab
NH4ClO4 never stays a niche formula for long. Solid rocket boosters, airbag inflation systems, and pyrotechnics wouldn’t deliver the same performance without it. With solid fuels, a balanced reaction between ammonium (NH4+) and perchlorate (ClO4-) turbocharges thrust. The shuttle launches most folks watched as kids ran on this kind of chemistry.
Numbers from NASA point out that ammonium perchlorate made up around 70% of the propellant mix inside the boosters of the legendary Space Shuttle. It doesn’t just help get rockets off the ground. Rescue flares in fishing boats, mine rescue kits, and smaller fireworks shows also rely on controlled energy releases that trace back to this formula.
Handling Risks: Health and Environment
Every expert who works with NH4ClO4 keeps a close eye on safety. Exposure brings risk. The perchlorate ion, when released into drinking water, can disrupt thyroid function, especially in kids and pregnant women. Once, a cleanup specialist told me about groundwater screening near manufacturing plants. High levels meant round-the-clock testing and new water lines for nearby families.
Reports from the U.S. Environmental Protection Agency note that some communities deal with lingering contamination after decades of industrial use. In places like Southern California, tap water testing and updated water treatment systems became part of daily life to avoid health issues. This isn’t just a lab hazard—it’s a community problem, too.
Finding Better Solutions
Some rocket and defense companies started phasing out NH4ClO4 in favor of more environmentally-aware alternatives. Researchers have experimented with energetically similar compounds that break down into safer byproducts. But matching performance from ammonium perchlorate isn’t simple—the formula’s power is hard to beat, both technically and economically.
One promising idea comes from advanced filtration using ion exchange resins and bioremediation. I’ve spoken with municipal engineers working on “green” cleanups. They combine nutrients and bacteria to help natural systems break perchlorates down underground. It takes patience, equipment, and long-term investment, but it shows that chemical formulas can be tackled with creativity—not just brute force chemistry.
Moving Forward with Science and Care
NH4ClO4 brings a mix of promise and responsibility. Science gives us powerful tools—but it also asks tough questions about health, safety, and sustainability. Every time the simple formula of ammonium perchlorate appears on a chalkboard, it represents not just fuel for rockets, but a challenge to balance innovation with care for people and places.
Is ammonium perchlorate regulated or restricted for purchase?
What’s the Big Deal with Ammonium Perchlorate?
Ammonium perchlorate finds its way into some of the world’s most powerful fireworks and rocket motors. The energetic qualities come from a simple chemical reaction — combine it just right and you get a force that can lift satellites or paint the sky in July. This same property also turns it into a sought-after ingredient for those wanting to do harm. A blast fueled by this compound doesn’t rely on fancy engineering; just access and know-how. That raises eyebrows in places where safety and security matter.
Rules and Red Tape: A Real Part of the Story
Walk into a big box store, and you won’t spot ammonium perchlorate on any shelf. The reason ties right into federal oversight. In the United States, the Department of Homeland Security classifies it among chemicals of interest. The reason isn’t hard to guess: manufacture and storage can support serious misuse. DEA and ATF regulations put up more barriers for anyone claiming a legitimate need. This is the stuff governments don’t leave unchecked. Record-keeping requirements and site inspections become part of the price for access. Small-scale users—think amateur rocketeers—often face background checks, licenses, and lengthy waiting periods.
The European Union keeps a similar grip on ammonium perchlorate. Public sales vanished after incidents linked to explosives. Now, only businesses with clear justification can legally acquire it. In India and China, sales go through government agencies and require paperwork from end-users. That’s a lot of hoops to jump, whether you’re a researcher or supplier.
Why All the Hassle? Lessons from the Field
I remember speaking with a pyrotechnics specialist after a local display. He pointed out how regulations shaped their work. Extra cost and time went into tracking each chemical shipment, with audits at random intervals. Fail to meet storage standards and the operation shuts down. Small mistakes carry heavy consequences. For enthusiasts, these rules can feel unfair. From the outside, it’s easy to wonder if all the caution pays off. Then stories arise—like thefts from industrial warehouses or makeshift labs found in quiet suburbs—that drive home why safeguards matter.
It’s also about protecting water and soil. Improper disposal turns perchlorate into a widespread contaminant. That’s not just a theoretical risk. The EPA flagged elevated perchlorate levels in some water supplies, citing run-off from defense sites and abandoned manufacturing plants. This affects thyroid function, with long-term health implications for communities that depend on well water. Managing this risk starts long before the substance reaches a buyer.
Room for Improvement in Oversight
Industry veterans often talk about striking the right balance. Amateurs and educational groups easily get caught up in red tape, discouraging positive discovery and innovation. At the same time, loopholes in online sales or lax enforcement in some regions let bad actors slip through. Better tracking from supplier to end-user could help, as could community education around safe handling and legal consequences. Some countries pioneered electronic registries, tying individual purchases to real identities. These measures didn’t stop legitimate research; they shrank the grey market.
What lands in the headlines tends to focus on the spectacular: fireworks mishaps or terror threats. Yet, for most of us, the story is quieter but no less important—about control, accountability, and health. In the end, ammonium perchlorate’s regulation serves not just to keep explosives out of the wrong hands, but to keep drinking water safe and towns free from contamination. The process isn’t perfect, but loosening the reins in the name of convenience risks a lot more than personal freedom.

| Names | |
| Preferred IUPAC name | azanium perchlorate |
| Other names |
Perchloric acid, ammonium salt AP Ammonium perchlorate(VII) Perchloric acid ammonium salt Nitric acid, ammonium salt (incorrect) Ammonium salt of perchloric acid |
| Pronunciation | /əˌmoʊniəm pərˈklɔːrieɪt/ |
| Identifiers | |
| CAS Number | 7790-98-9 |
| Beilstein Reference | 146081 |
| ChEBI | CHEBI:63038 |
| ChEMBL | CHEMBL1085827 |
| ChemSpider | 22215 |
| DrugBank | DB11110 |
| ECHA InfoCard | 100.028.763 |
| EC Number | 231-512-4 |
| Gmelin Reference | 8788 |
| KEGG | C16538 |
| MeSH | D000650 |
| PubChem CID | 24541 |
| RTECS number | SC7520000 |
| UNII | E2FMI96556 |
| UN number | 1481 |
| Properties | |
| Chemical formula | NH4ClO4 |
| Molar mass | 117.49 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.95 g/cm³ |
| Solubility in water | 24.5 g/100 mL (25 °C) |
| log P | -4.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -10 (for HClO₄, conjugate acid) |
| Basicity (pKb) | 14.62 |
| Magnetic susceptibility (χ) | -57.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 223.5 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -295.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -295.72 kcal/mol |
| Pharmacology | |
| ATC code | V04CX |
| Hazards | |
| Main hazards | Oxidizer, may explode if heated, fire and explosion hazard, harmful if swallowed, causes irritation to skin, eyes, and respiratory tract |
| GHS labelling | GHS02, GHS03, GHS07, GHS09 |
| Pictograms | GHS01, GHS03, GHS07 |
| Signal word | Danger |
| Hazard statements | H272: May intensify fire; oxidizer. H302: Harmful if swallowed. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-4-OX |
| Autoignition temperature | > 240 °C (464 °F) |
| Lethal dose or concentration | LD50 oral rat 362 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1,950 mg/kg |
| NIOSH | SN3075000 |
| PEL (Permissible) | 0.7 mg/m³ |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium chlorate Potassium perchlorate Sodium perchlorate Perchloric acid |

