Ammonium Nitrite: An In-Depth Commentary
Historical Development
People have looked at ammonium nitrite with a mix of curiosity and caution since the late 19th century. Chemists experimented with this salt long before its quirks were nailed down, making it one of those compounds that popped up in labs mostly by accident. Some early records came from attempts to understand nitrogen cycling in nature, and by the time scientists realized its instability, ammonium nitrite had already become part of the basic chemical toolkit. It has served as both a textbook example of an unstable nitrogen compound and a hard lesson for chemistry classrooms about safe laboratory practices.
Product Overview
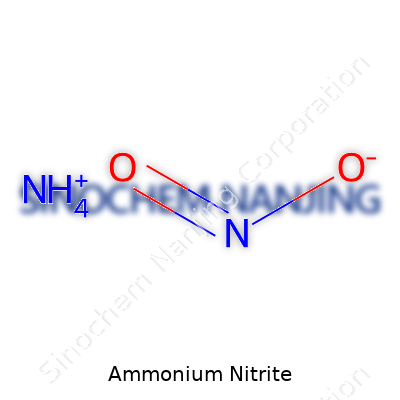
Ammonium nitrite carries the formula NH4NO2, which describes a white or pale-yellow crystalline powder that starts breaking down at the hint of heat or time. Its solution is colorless but not harmless. The dual presence of ammonium and nitrite ions gives it a foot in two worlds: it reflects both the ability to donate and accept nitrogen, something chemists appreciate for laboratory syntheses. Few folks outside chemical or analytical labs keep it around, and for good reason—it’s not a compound to store and forget.
Physical & Chemical Properties
This compound weighs in at 64.06 g/mol and dissolves easily in water, making solution preparation simple, but its stability falls apart beyond 70°C. Moist conditions only make things worse. The powder releases a mild ammonia odor, a hint it isn’t just sitting quietly on the shelf. If you’ve handled it, you know not to shake or heat it unnecessarily, since ammonium nitrite breaks down to nitrogen, water, and a fierce risk of explosion. Chemically, it’s both an oxidizer and a reducing agent, and that makes for some interesting reactions under the right triggers.
Technical Specifications & Labeling
Regulations around ammonium nitrite run strict. Shipping labels warn about its oxidative capacity and its instability. Storage calls for cool spaces out of sunlight and sealed containers, with plenty of organizational accountability. The material safety data sheet highlights its reactivity and toxic potential, especially under accident conditions. Laboratories tracking ammonium nitrite mark containers with clear hazard symbols and track every gram. In practice, if a business works with this compound, it puts serious protocols in place—any shortcut raises the risk of harmful incidents.
Preparation Method
Most labs prepare ammonium nitrite by neutralizing nitrous acid (itself unstable) with ammonium hydroxide under cold temperatures. Some routes use double decomposition from sodium nitrite and ammonium chloride, but all reliable methods steer clear of heat and proceed at temperatures not much above freezing. Years back, I watched a chemist handle the mixing and cooling process with a caution you rarely see elsewhere. It underscored the need to respect both starting materials and final product: a lapse in attention can lead to violent decomposition, which most lab veterans have heard stories about, if not witnessed firsthand.
Chemical Reactions & Modifications
Put ammonium nitrite in contact with acids and you get nitrous oxide, which acts as both an anesthetic and a greenhouse gas. Mild warming prompts the substance to break down into nitrogen and water, a reaction that seems simple but has caught chemists off guard plenty of times. Its reactivity lies at the heart of both its usefulness and its danger: while ammonium nitrite can provide a ready source of nitrogen, it also means that under uncontrolled conditions, disasters follow rapidly. Modifications aren’t common; the compound’s instability leaves little wiggle room for tweaking its structure without losing control.
Synonyms & Product Names
Chemists sometimes call it nitrous acid, ammonium salt, or use its systematic name: azanium nitrite. Industrial suppliers label it as ammonium nitrite, NH4NO2, and some regulation manuals refer to it under its UN identification number for hazardous materials. This variety in names doesn’t change realities in handling—the risks remain, regardless of label.
Safety & Operational Standards
Handling ammonium nitrite means adopting habits you don’t soon forget. Trained personnel require goggles, gloves, lab coats, and access to well-ventilated hoods. All work happens behind shields, with spill kits and first aid materials ready. Accidental exposure calls for immediate cleanup using excess water to dilute residual material. Years ago, a well-tuned emergency protocol saved a colleague after a glass bottle cracked and dropped ammonium nitrite onto the lab bench. Responders knew exactly how to act, and those routines made all the difference. Any business or school lab using this chemical follows strict operating procedures: frequent audits, logs for every transaction, and never storing excess amounts. Fire departments inspect facilities that list ammonium nitrite as present for good reason.
Application Area
Most people never need ammonium nitrite. Analytical chemists might use it for short-lived reagents in the field. In some niche scenarios, it helps generate nitrous oxide for specific industrial reactions or in certain pharmaceutical syntheses. Beyond that, its dangers overshadow convenience, so it’s rare to see it stocked in bulk. Modern technology leans toward safer, longer-lived alternatives whenever possible. The few who do use it develop serious expertise, and they teach new users both the utility and the risk through hands-on supervision, not just reading safety manuals.
Research & Development
The research spotlight rarely falls on ammonium nitrite these days, but a few teams look for safer preparation routes and decomposition inhibitors. Some have worked to better understand its role as an intermediate in nitrogen cycling, especially in environmental chemistry. I’ve read studies tracking nitrite’s transformation in natural systems, where unexpected traces of ammonium nitrite help piece together how water and soil manage nitrogen loads. Experiments often involve just micrograms, with plenty of fail-safes watching every step. There’s ongoing curiosity about how this chemical can model unstable intermediates elsewhere, but most ambitious projects redirect their focus to safer ground.
Toxicity Research
Toxicology data paint a clear picture: ammonium nitrite is dangerous both as a source of ammonia (which irritates mucous membranes, skin, and lungs) and as a nitrite (which interferes with hemoglobin’s oxygen-carrying ability in the blood). Acute exposure leads to symptoms like headaches, dizziness, low blood pressure, and, if untreated, the dreaded blue skin—methemoglobinemia—in severe cases. Long-term research notes no safe threshold for routine handling outside of controlled laboratory environments. In my experience, colleagues who take its toxicity lightly rarely repeat the mistake. Scientists studying nitrites in food warn about accidental conversion, especially since ingesting nitrites links to methemoglobinemia in infants and potential cancer risks in adults.
Future Prospects
Ammonium nitrite will probably remain a teaching tool in chemistry and a minor laboratory curiosity everywhere else. Researchers will likely keep looking for stable alternatives for analytical uses or for safe, in-situ production in closed-loop systems. Environmental research may turn up new roles for this compound as the technology to monitor tiny concentrations improves. Whatever small window for expanded application exists will demand not just better chemical understanding but also robust risk management, since its dangers refuse to fade with time. Enthusiasts and professionals will always value safety as the core lesson drawn from ammonium nitrite’s history and properties; any new uses will have to show they’re not trading convenience for serious risk.
What is ammonium nitrite used for?
What Makes Ammonium Nitrite Stand Out
Most people won’t hear about ammonium nitrite in daily life. This chemical doesn’t sit on store shelves or pop up in chat with your neighbors. Still, folks working in labs pay attention to it. Ammonium nitrite, made by mixing ammonia and nitrous acid, has a practical side, but it also demands some respect because it can break down in an unsafe way if it’s not handled carefully.
Where It Gets Used
Chemists use ammonium nitrite mainly for its role in making other chemicals. If you walked into a university research lab or some manufacturing operations, you’d see it as a short-lived substance. It’s key for creating nitrogen gas on demand right inside sealed containers. Nitrogen gas gets used in experiments where oxygen can mess things up, so creating a safe, oxygen-free environment matters. In my own experience, I saw a chemistry professor mix up a solution with this compound to quickly force out all the oxygen without buying and storing heavy gas cylinders.
Another use pops up during synthesis of azo dyes and other specialty chemicals. The chemical helps out when scientists want to build more complex molecules or create ingredients for certain industrial products. Still, its touchy nature keeps it from going mainstream; companies often substitute with more stable materials whenever possible.
Safety and Real-World Concerns
Anyone who’s mixed up ammonium nitrite knows it doesn’t like to stay put. The stuff breaks apart, sometimes with enough energy to cause real trouble. This isn’t just a “handle with care” substance. If it dries out, there’s a risk it could explode. Most labs keep only tiny amounts on hand, making it right before use. Good ventilation and strict temperature control make a huge difference. In one workplace story, a small spill sent everyone out of the lab—nobody hurt, but the scare was real. That’s why making ammonium nitrite on-site, in diluted form, keeps people safer.
Safety guidelines come from painful past lessons. I remember a story passed down from a retired technician about a near miss caused by storing the crystals too long. After that, the rules tightened. The National Institute for Occupational Safety and Health (NIOSH) keeps ammonium nitrite on its list of chemicals with strict handling advice. Even with close attention, spills and breakdown events can happen, so the best defense stays rooted in preparation and training.
Looking to the Future
Factories and researchers keep searching for safer ways to get the same job done. New chemical processes sometimes edge out ammonium nitrite for routines where the risk outweighs the reward. Safer substitutes for some nitrite functions, like organic nitrites or other nitrogen sources, grow more common each year. There’s a trade-off—some alternatives cost more, or don’t deliver the same quick reaction.
In fields that can’t switch away easily, more investment goes into worker training, updated storage systems, and stricter rules. I’ve sat through more safety refreshers on this compound than I can count, and every year the checklists grow longer. Awareness, backed up with regular inspections, helps push down the risk as much as science and common sense allow.
Why This Matters
Chemicals like ammonium nitrite remind us that progress in science carries real-world responsibility. Generating nitrogen gas in a pinch holds value, but the demands of safety, clear reporting, and respect for history shape how folks move forward. If you’re working with materials that need ammonium nitrite, stay sharp, talk to the veterans, and follow good habits—mistakes tend to punish carelessness the hardest. For most folks, awareness and seasoned respect for the chemistry pays off in the long run.
Is ammonium nitrite stable and safe to handle?
Understanding What’s On the Workbench
Ammonium nitrite might sound like another bit of harmless chemistry kit, but experience in both school labs and industrial sites tells a different story. This chemical, made from ammonia and nitrous acid, often shows up in textbooks next to warning labels for good reasons. In small academic demos, teachers rightly raise their voices about safety, and professionals in chemical plants don’t take it lightly either.
Why Ammonium Nitrite Stirs Worry
This compound isn’t happy hanging around on a shelf. It breaks down easily, literally decomposing even at room temperature. That’s not just a chemistry oddity: this decomposition can give off nitrogen oxides and heat. Some of these gases can choke a lab space and harm folks inside, while the heat can lead to fire—or worse if enough material is packed in one place.
Years working with regulated substances have drilled in a simple point: if a material releases toxic gases or triggers a violent reaction with just a gentle nudge, it deserves respect and clear-eyed risk assessment. Reports from the mid-20th century mention explosions caused by tiny sparks, friction, or just exposure to sunlight. Even in small amounts, ammonium nitrite leaves no room for sloppy handling.
Safety Steps—No Room for Shortcuts
Curiosity and scientific progress might put people close to ammonium nitrite, but practical safety comes first. Lab veterans store even tiny amounts in cool, dark conditions, away from anything organic or flammable. No mixing with acids, bases, or combustibles gets allowed. Even glassware or tools used with ammonium nitrite need careful cleaning. Loose procedures result in damaged equipment, at best—or serious injuries at worst.
Government guidelines line up with this caution. Industrial facilities rarely keep more than what’s needed for immediate use, and strict rules govern shipping and abuse. Personal experience working with energetic compounds teaches that emergency plans must stay up to date and that protective gear—face shields, gloves, ventilation—is no joke.
Experience Over Hearsay
Stories circulate in the safety community: someone skipped a step, tried storing a jar for longer than needed, or didn’t notice unexpected heat during a demonstration. The result? Near-misses or full-blown accidents that become local legends. It’s never some fluke—missing a warning sign nearly always traces to ignoring best practices or underestimating risk.
Paths Forward for Safer Chemistry
Higher education, research labs, and industry put strong safeguards in place. Automation takes human error out where possible. Smaller-scale substitutes get used instead of ammonium nitrite for teaching basic concepts. Documentation and training take up a chunk of the work week, not because bureaucracy insists, but because past disasters left scars and lessons.
Those who work hands-on with these substances tend to welcome scrutiny and updated protocols. Bringing down the number of incidents comes from a willingness to adapt as new research and real-world case studies shed light on weak spots.
Looking Out for Each Other
Colleagues watch each other’s backs. After seeing a fume hood filter clog or a glove develop a pinhole, nobody shrugs off even small safety slips. This attitude isn’t negativity—it’s smart stewardship of tools and lives. The best chemists and technicians keep risk front of mind every time ammonium nitrite goes on the task list, proving safe work is possible, but never a guarantee.
How should ammonium nitrite be stored?
Understanding Ammonium Nitrite's Reputation
Ammonium nitrite doesn’t get much attention in everyday conversations, but anyone working near chemicals or in a lab environment hears about it plenty. I still remember my college chemistry professor telling stories about its unpredictable nature. He hammered home safety for good reason—it’s famously unstable and unforgiving. Ammonium nitrite breaks down at ordinary temperatures, and its decomposition releases nitrogen gases and water. Left unchecked, this decomposition can intensify quickly, causing pressure build-up or even explosions.
Keeping ammonium nitrite around means understanding real risks, not just ticking boxes on a safety list. It can be tempting to assume that a cool, dry cabinet solves everything, yet this mindset overlooks the bigger picture: temperature, contamination, and fire risk never really go away.
Risks Demanding Respect
History teaches hard lessons. In more than one lab around the world, improper storage triggered events nobody wants to relive. Once, while working at a research facility, one ill-sealed jar nearly made someone lose their hand when moisture crept in. Even a small amount of humidity acts like a time bomb because water can accelerate decomposition. Without proper containment and handling, ammonium nitrite goes from stable to hazardous without warning.
Static discharge is another hazard that rarely gets the attention it deserves. Loose packaging or high friction from moving containers can spark a reaction. Even light jostling in the wrong humid environment starts a dangerous chain reaction. The fact ammonium nitrite isn’t sold freely in consumer markets speaks volumes: experts realize the stakes.
Practical Storage Solutions
Sound storage habits save lives. From experience and industry guidelines, a few rules stand out. Always store ammonium nitrite in tightly sealed, chemical-resistant containers. Polyethylene or glass offers solid protection. Metal containers invite corrosion, which only pushes the risk higher.
A dry, cool, well-ventilated storage space is non-negotiable. Aim for locations far from sources of heat or open flames. Flammable substances and acids cannot share shelf space with ammonium nitrite—this isn’t a situation for shortcuts. Even the presence of dust, sawdust, or oil can act as a catalyst. In the labs where I've worked, everyone double-checks labels and isolation before signing off at the end of a shift.
Firefighting measures should be nearby and tailored for chemical fires, not just the standard water extinguishers found in most offices. Water breaks down ammonium nitrite further, producing additional risk if used incorrectly.
Only Trust Trained Hands
Training and written protocols matter. I’ve watched new hires try to rush, only to realize that rushing near ammonium nitrite leads to regret. Only qualified staff should handle and transport it. Emergency procedures need regular review, as new people join and departments change.
Several regulatory agencies, including OSHA and the CDC, publish guidance about storing and handling reactive substances. Basing procedures on science and hard evidence—plus reviewing those after every safety drill—keeps everyone ahead of trouble. I still keep a mental checklist from my early years on the job, and most seasoned chemists can recite theirs by heart.
An awareness of the dangers and proven commitment to safe storage keeps laboratories and workers out of the headlines. Respect for ammonium nitrite’s strength isn’t just a technical point—it’s the difference between a routine day and one nobody forgets for all the wrong reasons.
What are the hazards associated with ammonium nitrite?
Easy to Overlook, Hard to Ignore
Ammonium nitrite sits on a long list of chemicals people use in labs and industries. At first glance, it seems like just another white powder with a fancy name. Underneath that label, though, hides a track record of danger, especially if it slips out of careful hands. The stuff packs a punch because it contains both a fuel (ammonium) and an oxidizer (nitrite) in a single, unstable package.
Explosion Risk Isn’t Theory—It’s Real
It’s tempting to gloss over warnings, especially after years of safe handling, but stories from experienced chemists reveal why small mistakes attract big consequences here. Ammonium nitrite breaks down quickly, sometimes unpredictably. This breakdown releases nitrogen gas and water, and under the right conditions throws off enough energy to blow a hole in your expectations—or the floor beneath your feet. Dentists don’t pop this stuff on shelves, and schools don’t touch it for good reason.
Looking at past incidents, explosion risks jump out as the main headache for those working with the material. When the temperature rises or contamination sneaks in (a drop of oil, a sprinkle of dust), ammonium nitrite can decompose explosively. Back in 2001, a research facility faced major damage during routine disposal. Emergency crews found out the hard way that “controlled” doesn’t always guarantee “safe.”
Toxic Fumes Make a Bad Day Worse
Blasts grab headlines, but there’s an invisible threat too: toxic fumes. When ammonium nitrite decomposes, it can release nitrogen oxides. Breathing these gases leaves lungs raw and can send workers to the hospital. Cases in industrial environments, often during spill cleanups or accidental heating, remind us the fumes trail behind even after the bang.
Water and Heat—Triggers Hiding in Plain Sight
Ammonium nitrite doesn’t survive long in water, but that’s not always a good thing. Mixing the material with water in the wrong container or at the wrong temperature triggers those nasty gases—nobody wants to huff that by mistake. Laboratory reports stress that storage matters. Cool, dry, and airtight is the only way to dodge trouble. A forgotten bottle on a warm shelf can turn into a ticking problem.
Keeping Safe Means Taking Responsibility
When I think back to the safety training days, ammonium nitrite always drew extra caution. Not just for its ability to react. The main thing is its unpredictability. PPE—personal protective equipment—needs more than lip service; gloves, goggles, solid ventilation. Sharps warnings, fire extinguishers nearby, and spill kits prepped for both solids and gases keep troubles contained as much as possible. Every person on team shifts from “just another day at work” to “vigilance mode” once ammonium nitrite hits the bench.
Practical Steps Lower the Risk
Solutions exist: limit the amount stored, use fresh batches instead of old stock, and keep meticulous logs to avoid surprises. Double-check before mixing with anything, even simple solvents. Label containers with both chemical names and clear hazard warnings, not just codes. Some shops now use safer alternatives for common tasks, trading convenience for well-earned peace of mind.
Everyone benefits from an honest approach to risk. More transparency in reporting accidents, sharing data between labs, and teaching new workers the ugly stories along with the textbook facts could prevent the next big mishap. Ammonium nitrite reminds us that safety shouldn’t live in the back room—it belongs on the frontline, backed by both personal experience and scientific evidence.
Can ammonium nitrite be shipped internationally?
Looking at the Realities and Risks
Ammonium nitrite may not be the most talked-about chemical, but folks in science or industry know its name well. People hear about ammonium nitrates and associate them with fertilizers and, sadly, explosives. Ammonium nitrite fits into a similar story — it comes from mixing ammonium and nitrite ions, and it’s far more unstable than its cousin, the nitrate salt.
Ask anyone who’s worked in a chemical stockroom about dumping ammonium nitrite in a box and mailing it across borders — you’ll likely get a sharp look and maybe a story about bad accidents chewing up budgets and even injuring people. Some years back, I saw lab shipments rejected simply because of the faintest suspicion that a box might contain this compound. Even in steady environments at room temperature, ammonium nitrite can decompose, leaving a mix of nitrogen gas and water vapor. Extra heat or a rough journey ramps the risk even higher.
Laws and Tight Regulations: Not Just Red Tape
Shipping regulations for dangerous goods aren’t cooked up in a vacuum. The international rulebook — known as the International Air Transport Association (IATA) Dangerous Goods Regulations and the United Nations’ “Orange Book” — straight up list ammonium nitrite as forbidden for air cargo and ships. The Department of Transportation in the US and the ADR in Europe take a hard line, too. The phrase “not permitted for transport” pops up in category after category. If an exporter wants to move a tiny vial across a border, they face a regulatory brick wall.
For years, experts have linked similar chemicals to industrial explosions. Awareness soared after the Beirut port disaster in 2020, although ammonium nitrate took the blame there, not the nitrite. Still, public fear and government caution grew. Every chemical incident pushes tighter laws. Even universities and licensed facilities must handle ammonium nitrite in chilled, dilute solutions — usually made right as it’s needed and destroyed after use, all because of its raw instability.
Why the Attention Matters
Public safety isn't just a buzzword — it’s actually the bottom line when regulators clamp down. If you think laws are just overkill, remember the Oklahoma City bombing and how much tighter restrictions became after that tragedy. The memory of shipping disasters like the Texas City explosion still lingers with regulators. One small slip or careless packaging job can mean disaster.
For supply chains, researchers, and anyone hoping to study or use ammonium nitrite, the clampdown presents real frustration. I’ve seen brilliant projects held back because a grant team couldn’t source fresh material in time, or because import paperwork turned into a months-long slog. Still, every frustrated phone call or rejected shipment speaks to the larger cost — a single accident could wipe out more than a year’s research.
What Would Help
Creative workarounds have emerged. Most chemists now prepare ammonium nitrite on-site, mixing dilute solutions from available precursors. Some countries encourage labs to use substitutes where possible. If you can build collaboration across borders, it makes sense to set up experiments locally rather than try to import the chemical. Trends in science-smart logistics could someday allow safer, smarter, temperature-controlled “micro-batches”—assuming laws can keep up.
If you’re reading this as someone caught between a supplier, a customs agent, and a deadline, don’t just assume you can ship ammonium nitrite internationally. The rules are built from real-world tragedies and serious science. And for now, the only secure shipment most can count on stays close to home, under strict supervision, and with careful records at every step.
| Names | |
| Preferred IUPAC name | Ammonium nitrite |
| Other names |
Nitrous acid ammonium salt Ammonium dinitrate |
| Pronunciation | /əˈmoʊniəm ˈnaɪtraɪt/ |
| Identifiers | |
| CAS Number | 13446-48-5 |
| Beilstein Reference | 0118731 |
| ChEBI | CHEBI:62993 |
| ChEMBL | CHEMBL1287332 |
| ChemSpider | 13685645 |
| DrugBank | DB14162 |
| ECHA InfoCard | ECHA InfoCard: 03-2119981065-39-0000 |
| EC Number | 229-276-8 |
| Gmelin Reference | 67621 |
| KEGG | C14110 |
| MeSH | D000648 |
| PubChem CID | 24547 |
| RTECS number | BR7410000 |
| UNII | 7QF2A4300L |
| UN number | UN2422 |
| Properties | |
| Chemical formula | NH4NO2 |
| Molar mass | 64.06 g/mol |
| Appearance | White or pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1. ammonium nitrite: 1. ammonium nitrite: 1.68 g/cm³ |
| Solubility in water | Very soluble |
| log P | “-3.6” |
| Acidity (pKa) | 4.5 |
| Basicity (pKb) | 4.75 |
| Magnetic susceptibility (χ) | +38.0·10⁻⁶ |
| Refractive index (nD) | 1.387 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -302.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -206 kJ/mol |
| Pharmacology | |
| ATC code | V03AB26 |
| Hazards | |
| GHS labelling | GHS02, GHS03, GHS07 |
| Pictograms | GHS01,GHS03 |
| Signal word | Warning |
| Hazard statements | H272, H302, H315, H319, H335 |
| Precautionary statements | P220, P221, P280, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Lethal dose or concentration | LD50 (oral, rat): 89 mg/kg |
| LD50 (median dose) | 100 mg/kg (rat, oral) |
| NIOSH | TT2945000 |
| PEL (Permissible) | PEL for Ammonium Nitrite: Not established |
| REL (Recommended) | 500 g |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Ammonium nitrate Sodium nitrite Potassium nitrite Ammonium nitrate Sodium nitrate Ammonia Nitrous acid Nitric acid |

