An In-Depth Look at Ammonium Nickel Nitrate
Historical Development
Researchers first began exploring complex nickel compounds about a hundred years ago, during a period when colorimetric analytical chemistry depended on detecting metal ions through their vivid reactions. Ammonium nickel nitrate came into play after scientists noticed some fascinating precipitation reactions, leading to its use in both the lab and industry. Over time, as mining efforts expanded and metallurgists started getting their hands on more nickel ores, more practical nickel salts and complex ammonium salts like this one became common. Early technical literature provided blueprints for manufacturers, and safety guidelines became a necessity after several accidents underscored its reactivity. Now, chemists see it both as a legacy product and a platform for new research.
Product Overview
Ammonium nickel nitrate sits on the shelf as a crystalline, water-soluble salt. It’s most recognized for its ability to deliver nickel ions, which end up in specialty ceramics, catalyst mixtures, and lab synthesis steps. Chemists choose it instead of other nickel compounds when they want higher water solubility or controlled oxidation behavior. This compound usually comes labeled with a unique CAS number, and product sheets often include nickel content percentages, purity by weight, and moisture limits, because these details matter to scientists trying to hit strict process targets.
Physical & Chemical Properties
Standard formulas for this material list it as (NH4)2Ni(NO3)4, and it typically appears greenish or bluish due to nickel’s d-d electron transitions. Its density ranges from 1.7 to 2.2 g/cm³, depending on the hydrate form. The salt dissolves quickly in water and breaks apart into its ammonium, nickel, and nitrate ions. Most batches decompose easily above 160°C, releasing nitrogen oxides which can raise both safety and ventilation challenges. Its decomposition can also yield nickel oxide as a residue, which proves useful for catalysis and ceramics.
Technical Specifications & Labeling
Manufacturers provide these salts with strict labels, covering the batch number, production date, and purity, usually above 98%. The labeling flags all main hazards, including oxidizing power and toxic metal content. Technical spec sheets break down maximum chloride and sulfate contamination, since these affect downstream reactions. Facilities receiving shipments typically run their own spot-check analyses using AAS (atomic absorption spectroscopy) for metals, confirming that what’s on the label lines up with their quality needs. If you’re working under ISO or REACH regulations, you’ll see compliance clearly called out on the outer packaging.
Preparation Method
Most producers start with nickel nitrate hexahydrate and mix it with ammonium nitrate in an aqueous medium. After full dissolution and careful control of acidity and temperature, slow evaporation or cooling yields the crystalline product. The process calls for real attention to sequence and mixing because cross-contamination with transition metals or extraneous anions can throw off the nitrate-to-nickel ratio. At larger scale, plant operators often rely on jacketed reactors to watch temperature precisely—runaways can cause dangerous emissions of nitrogen oxides.
Chemical Reactions & Modifications
Lab chemists can count on ammonium nickel nitrate reacting vigorously with reducing agents, releasing ammonium and nitrate byproducts. If combined with organic fuels, especially in solution, this compound turbocharges redox reactions and generates heat quickly, so small errors can lead to runaway exotherms. Mixing with alkalis gives basic nickel salts or nickel hydroxide, which end up as feeds for battery electrode material synthesis. Acid titrations or additions of chelating agents like EDTA yield intermediate complexes—these intermediates serve as seeds for nanoparticle and advanced catalyst research. At every step, close monitoring of atmosphere and temperature is a must because of the risk of nitrous gas release.
Synonyms & Product Names
In catalogs and safety data sheets, ammonium nickel nitrate shows up under several names. Some call it “ammonium nickel(II) nitrate,” nodding to the nickel oxidation state. Others just list it by its formula. European and Asian suppliers include translations, such as “ammonium-nickel nitrát” or “amonia nikl nitrat.” Some specialty suppliers layer on a product code for their proprietary formulation, especially when selling pre-mixed catalyst precursors or custom hydrate levels. As a rule, buyers should double-check the nickel content and hydrate status to make sure the product matches their intended application.
Safety & Operational Standards
Anyone handling ammonium nickel nitrate spends plenty of time on safety procedures because both nickel and nitrate ions pose real risks. Nickel salts remain notorious skin and respiratory allergens, with some regulatory agencies listing them as Group 2B possible carcinogens. In addition, this salt’s oxidizing power can intensify fires—storage away from organics and combustibles becomes non-negotiable. Workers need gloves, goggles, and preferably a fume hood to limit inhalation exposure. Facilities use airtight containers and enforce spill protocols, since cleanup often means neutralizing agents on site. Fire brigades train for nitrate fires, using plenty of water spray but steering clear of foam or dry chemicals that make matters worse. Waste disposal always runs through authorized hazardous material channels—nobody washes this down the drain.
Application Area
You’ll find ammonium nickel nitrate showing up most often in catalyst research, especially in automotive emissions controls and hydrogenation reactions. Some ceramic glazing operations use it to deliver nickel ions evenly into fired pieces for subtle color and electrical changes. Researchers mixing it into electroplating baths see improved deposit quality in certain cases, though nickel sulfate still dominates in routine electroplating. Laboratory teams rely on it for nickel doping in crystal growth experiments, where the combination of aqueous solubility and clean nickel release saves time. There’s even a niche use as an oxidizer in specialty pyrotechnic mixtures, with strict legal controls in place.
Research & Development
Researchers keep finding creative ways to pull value from this compound. Over the past decade, much of the focus has turned toward using ammonium nickel nitrate to seed nickel oxide nanoparticles, which later end up in energy storage devices like batteries and supercapacitors. Teams refining catalysts for fuel cells often turn to it for homogeneous mixing with other metals before calcination. Some groups experiment with it as a source for two-dimensional nickel-based materials, which attract attention in electronic and optical sensor work. Technical journals show a slow but steady trickle of patents describing tweaks in synthesis and downstream integration, always aiming for cheaper or more sustainable processes.
Toxicity Research
Long-term studies on nickel salts have shown links between exposure and increased cancer risks, particularly for those with chronic skin contact or inhaling fine powders on the job. Ammonium nickel nitrate’s high solubility presents added concern, as the nickel ion enters biological systems more easily. Recent animal and cellular studies emphasize oxidative stress and DNA damage pathways, raising new questions about safe exposure thresholds. Regulatory agencies in Europe and North America have sharply restricted allowable workplace concentrations and made dermal testing a requirement for new users. Several downstream industries—especially those involving ceramic or glass products—have begun switching to alternative coloring or doping agents because of health warnings.
Future Prospects
The long-term outlook for ammonium nickel nitrate balances on two factors: the push for energy storage technology and growing health and safety demands. On the one hand, battery and supercapacitor research brings fresh interest, spurring investment in new production techniques and greener synthesis routes, including attempts to recover nickel ions from electronic waste streams. On the other, the regulatory and market pressure to minimize worker exposure means companies keep developing lower-dust, more stable formulations or move away from nickel altogether for low-risk applications. As automation in chemical processing grows, you’ll probably see smarter process monitoring and better containment solutions, aiming to keep nickel compounds in circulation while keeping workers safe and local environments clean. Ongoing toxicity studies may redraw the lines for acceptable use once again, but for today’s technical challenges, researchers still see value in this versatile compound if safety stays front and center.
What is Ammonium Nickel Nitrate used for?
Digging Into Its Main Applications
Ammonium nickel nitrate sounds technical, but it has a clear place in several industries that drive familiar products and solutions. You won’t find it on the grocery shelf, yet its presence shows up in factories, labs, and workshops. My own background in environmental science and manufacturing gives me a front-row seat to the real-world impact of these chemicals. I’ve stood in classrooms, labs, and on factory tours where ammonium nickel nitrate shifts from an obscure name to a workhorse compound supporting everyday progress.
Life in the Lab and Catalysis
Chemists use ammonium nickel nitrate as a key tool. Think of it as the behind-the-scenes starter for making specific nickel-based catalysts. These nickel-rich catalysts speed up reactions in petroleum refining, food hydrogenation, and chemical manufacturing. Researchers need chemicals that respond predictably and safely. Their trust in compounds like ammonium nickel nitrate stems from years of careful reliability. Take it from my visits to university labs—this chemical helps bridge the gap between raw potential and finished solution.
Electroplating and Surface Treatment
The need for tough, corrosion-resistant coatings on metal parts creates a steady demand for compounds like ammonium nickel nitrate. Nickel plating isn’t just about making things shiny; it’s about extending the life of car parts, electronics, and industrial tools. I’ve watched engineers dip components in plating baths, using careful chemistry to lay down durable nickel coatings. The formula comes together thanks to salts like ammonium nickel nitrate, which deliver nickel ions smoothly and efficiently.
Materials Science and Ceramics
Materials scientists depend on reliable sources of nickel when creating specialty ceramics and advanced glass. Ammonium nickel nitrate plays a part in shaping these hard-wearing materials, which pop up in everything from electronics to advanced medical tools. Sourcing materials that offer tight control over composition allows researchers and engineers to tune performance and quality. During my time collaborating with ceramics engineers, the right precursors—especially those containing nickel—proved essential for breakthroughs in everything from insulation to cutting tools.
Tackling Environmental Solutions
As environmental regulations become stricter, industries look for ways to process waste and clean water more effectively. Some research teams turn to ammonium nickel nitrate as a source of nickel within catalysts or adsorbents aimed at pollution control. In wastewater treatment facilities, for example, tailored nickel compounds can pull harmful chemicals from industrial runoff. Clean water matters everywhere—I've watched plant operators struggle to keep up with contaminants, and seeing new chemical solutions enter the mix brings real hope for communities. Progress often starts with small-scale experiments using chemicals like this one, paving the way toward bigger breakthroughs in clean technology.
Looking Toward Safer Handling and Awareness
Despite all the promise, safety remains non-negotiable. Ammonium nickel nitrate, like many specialty chemicals, calls for strict handling protocols. Nickel compounds can trigger allergies and respiratory issues, so labs and factories don’t cut corners on protective gear and training. Much of what I’ve learned comes from watching teams work with respect for what these chemicals can do—both good and bad. Safety sheets cover every wall, and staff treat each process step with seriousness. Regular audits and revised safety rules protect both workers and the surrounding environment.
Solutions and Responsible Use
Better education and transparency about chemicals in industry would help everyone. Sharing best practices in the workplace and supporting research into safer alternatives remain big pieces of the puzzle. As more people get involved, communities gain insight into what happens inside those locked doors and glass beakers. Responsible use, monitoring, and disposal keep both benefits and risks in balance, so ammonium nickel nitrate remains a valuable resource without becoming a hazard.
Is Ammonium Nickel Nitrate hazardous or toxic?
Understanding the Chemical
Ammonium nickel nitrate turns up in labs and industry thanks to its use in catalysts, ceramics, and even some research projects. Whenever handling anything with the words nickel and nitrate in it, warning bells ring in the back of my mind. Nickel compounds often raise red flags for health, especially with long-term exposure. Mix in nitrate, which can mess with water sources, and now there's a real need for some caution.
Health Risks: Skin, Lungs, and More
It’s easy to overlook small quantities on a lab bench, but working with ammonium nickel nitrate isn’t something to take lightly. Direct contact can irritate skin and eyes pretty quick. Based on both industrial safety alerts and personal habits in chemical handling, gloves and goggles are never optional around it. Breathing in nickel compounds can stir up asthma-like symptoms or worse, and over time, nickel has been tied to cancer, especially for people who inhale dust or fine particles.
Warnings about nickel allergies aren't just bureaucratic overkill. Sensitive folks might break out in a rash just from a stray splash or a little dust. Inhaling it isn’t better, as it can set off coughing fits and wheezing, especially in people already wrestling with breathing problems.
Environmental Concerns and Water Worries
Nitrates don’t just disappear after use. They have a habit of seeping into water supplies, where they can trigger all kinds of trouble for both humans and aquatic life. Drinking nitrate-contaminated water risks methemoglobinemia for babies, a condition where blood stops carrying oxygen right. From my experience near agricultural zones, seeing bright green algae blooms after chemical spills isn’t rare. The nickel adds another layer, with possible buildup in fish and plants, raising hard questions about food safety and long-term soil health.
Facts from the Literature
Digging into public health reports, the European Chemicals Agency notes nickel’s links to cancer and chronic lung disease. The US EPA lists nitrates as concerning when they show up in groundwater, especially near industrial or research facilities. Many lab manuals flag ammonium nickel nitrate as needing secure, labeled storage and careful tracking of usage and waste.
Solutions and Better Practices
No one expects researchers or manufacturers to toss out every substance with risks, but there’s room to do better with storage, usage, and training. Keeping ammonium nickel nitrate inside sealed containers, using proper ventilation, and sticking to personal protective gear can cut exposure to the minimum. Well-marked spill kits should stay within reach instead of in the next building over.
Disposal often ends up as an afterthought, and that's asking for groundwater troubles. Industry veterans recommend keeping contaminated tools and waste in airtight bins until safe disposal. Regular checks on air filters and wastewater output also make a major difference. Teaching people to spot exposure symptoms, and following up on any health complaints, helps keep accidents from turning into full-blown emergencies.
Knowledge Means Safety
Working with chemicals like ammonium nickel nitrate is part of modern life, whether in tech, research, or industry. Understanding hazards and taking them seriously saves lives and cuts down on headaches later. Picking up the right habits early on pays off, protecting people and the environment alike.
What is the chemical formula of Ammonium Nickel Nitrate?
Breaking Down the Components
Ammonium nickel nitrate doesn’t come up in everyday talk, but it draws the attention of chemists and those working in industrial processing or academic labs. Digging into its name gives a hint at its makeup. Ammonium means NH4+. Nickel points to the nickel ion, and nitrate is NO3-. The task is to fit these into a formula that balances out the charges.
Nickel usually takes on a +2 charge, so the nickel(II) ion is Ni2+. Each nitrate ion brings a single negative charge, and ammonium carries a positive charge. To create a neutral compound, the charges have to balance. There’s a bit of algebra to matching everything up. Ammonium and nitrate both bring one charge, just opposites, and nickel comes in with two positives.
The Formula: A Simple Assembly
Combine one nickel(II) ion, two ammonium ions, and two nitrate ions, the chemical formula settles out as (NH4)2Ni(NO3)2. This structure keeps the molecule balanced. Each element matters for getting reactions right in the lab and for any potential applications in industry or science curricula.
Why this Formula Plays a Role in Science and Beyond
Universities usually point to compounds like ammonium nickel nitrate when talking about complex salts and solution chemistry. It’s a favorite for lessons about solubility rules and ionic interactions. The compound stands as an example of what happens when multiple polyatomic ions come together. The structure looks simple, but building it correctly takes understanding how charges work—something that always tripped up students in introductory chemistry classes.
Lab work with ammonium nickel nitrate demands accuracy. Missing out on the correct formula leads to poor results during experiments. I remember my own lab partner back in college confusing this salt with nickel(II) nitrate alone. Our results never matched anyone else’s until we caught our mistake. The ammonium piece changed everything, from how the powder dissolved to its color during reactions.
What Knowing the Formula Means for Real-World Use
Working with any nickel-based compound means handling it with care. Nickel, when dissolved, can present risks to health if someone ignores basic safety steps. Ammonium brings its own hazards, especially around high heat or mixing with certain chemicals. Getting the formula down isn’t just about passing a test—safety data sheets require that same level of detail. Incorrect labeling causes expensive mistakes and potential hazards.
Niche applications for ammonium nickel nitrate show up in electroplating, research projects, or specialty synthesis. Most users look for clear guidance on storage, handling, and disposal. Regulators want manufacturers and labs to be precise. Even a minor error in the formula could throw off compliance paperwork or keep a student from getting full marks.
Looking Toward Smarter Chemistry
Technology may streamline parts of lab work, but grasping the basics like the formula for ammonium nickel nitrate never loses relevance. Chemistry teachers keep drilling students on ionic compounds for a reason. Each formula is a sort of puzzle, and figuring it out gives confidence and practical knowledge—skills that transfer to advanced research, industry tasks, and safe chemical handling every day.
How should Ammonium Nickel Nitrate be stored?
Walking the Lab Floor: Real Risks, Real Solutions
Step inside any lab that handles chemicals, and you’ll see routines that never change. Doors lock, shelves label, warning signs glare at you for good reason. Ammonium nickel nitrate doesn’t care who you are or how long you’ve kept a clean record—if handled carelessly, it can throw a wrench into even the best-managed operation. My years moving chemicals between university and industry benches taught me just how fast a regular day can turn into emergency mode if storage gets sloppy.
Keep It Cool, Keep It Dry
Heat and moisture don’t mix well with this compound. Even one summer’s day with a busted AC taught our team that dry, temperature-controlled storage isn’t a luxury. We use dedicated refrigerators set to an unyielding 2–8 degrees Celsius, far away from any heat source or direct sunlight. Humidity invites clumps and dangerous reactions—think rusted shelving, sticky gloves, or, worst of all, increased risk of accidental releases. Keeping containers sealed tight, with desiccant packs thrown in, made a big difference for both safety and shelf life.
Don’t Store Ammonium Nickel Nitrate Around What It Hates
This stuff reacts badly with organic material, acids, reducing agents and anything remotely flammable. Years back, a neighbor lab lost a week of productivity after a minor spill found its way to an old acetone bottle. Cross contamination and unwanted reactions start small and end with a bang—or a full evacuation. I keep ammonium nickel nitrate on its own sturdy shelf, away from everything else, with big, red “do not stack” reminders taped to the door.
Label Everything—And Mean It
Handwritten scrawls fade. Vague abbreviations confuse even seasoned staff, especially during shift changes. Printed, laminated labels with chemical names, handling precautions, and hazard symbols save time and sanity. During audits, these labels smooth everything. During emergencies, they save lives. Never depend on memory, especially if you handle multiple compounds that sound or look similar.
Contain Spills Before They Start
Nothing quite prepares you for your first spill response drill. Those absorbent liners, sealed bins, secondary containment trays—everyone grumbles about the space they take, right up until a bottle tips. Regular practice, paired with keeping these items nearby, cuts down on panic and lost time when trouble starts. Nobody wants to sweep up shards or mop up leaks after the fact. Prevention and prep win over cleanup every time.
Secure Access and Share Knowledge
Only trained hands should unlock a cabinet holding ammonium nickel nitrate. Too many stories end with an intern reaching for the wrong bottle or an untrained visitor grabbing the nearest jar. Logging all check-ins and check-outs—old-school paper logs, modern inventory software, whatever works—keeps track of who’s doing what. More importantly, it builds a culture where safety becomes habit, not an afterthought.
Discussion Changes Everything
Talking openly about mistakes and close calls keeps everyone honest. Sharing stories of what almost went wrong, not just following every rule because a sign says so, leads to solutions. Swapping tales in a morning safety huddle has, more than once, stopped a bad storage habit from spreading. For ammonium nickel nitrate, and chemicals like it, these conversations offer the best defense against the accidents no manual ever really covers.
Making Safer Labs for Everyone
Proper storage of ammonium nickel nitrate never boils down to just locking it up and checking a box. Consistent, careful practice, open communication, and solid training make all the difference. The chemistry can be dangerous, but respect and routines keep everyone heading home safe.
What are the safety precautions when handling Ammonium Nickel Nitrate?
Why This Chemical Demands Respect
Ammonium nickel nitrate isn’t one of those chemicals you can treat like any old powder stored on the shelf. This greenish compound brings both the explosive potential of ammonium nitrate and the toxicity of nickel salts to the table. Most labs don’t keep it around unless they have a real need. Researchers who’ve worked with it know mistakes can put health and even lives at risk.
Chemicals with oxidizing power raise the stakes quickly. I've seen people underestimate what a splash from a solution can do. Eyes and skin don’t forgive contact with nickel compounds. Repeated exposure to nickel can bring on allergic reactions or even cancer risks. Ammonium nitrate's reputation as an oxidizer adds to the fire hazard—mixing it with the wrong substance or exposing it to the wrong conditions can set off a dangerous reaction.
Real Safety Precautions: Gloves and Beyond
Anyone handling ammonium nickel nitrate wears the proper gloves—not just whatever’s at hand. Nitrile or neoprene gloves stand up much better against nickel compounds than latex or vinyl. Eye protection stays on at all times. Safety goggles aren’t a backup plan either; reactions splash when you least expect it.
Lab coats go on and button up. Open shoes turn into no-go items. I’ve seen situations go sideways just because someone thought they’d just be in the lab for a minute. Even one exposure can set off a nasty reaction.
Working under a fume hood isn’t an option but a rule. Inhaling nickel dust or mist leaves you with far more than just a sore throat. Fume hoods suck that threat right out of the air. Good airflow makes all the difference in keeping lungs clear. Respiratory protection gets extra important if the hood’s not working—or the scale of work gets bigger.
Storage: The Details Matter
Ammonium nickel nitrate needs a cool, dry, and well-ventilated spot. No heat sources or sunlight should sneak in. Even a tiny bit of moisture can lead the chemical to decompose. Keeping it away from flammable or combustible materials avoids accidents. I once saw a bin of ammonium nitrate stored right next to solvents, and the whole lab shivered at the close call.
It never pays to store it in glass containers with ground glass stoppers. The compound has a way of cementing the lid shut over time. Plastic containers or bottles with screw tops cut down the headache. Labeling sits at the center of safe storage. Grabbing the wrong thing in the rush of an experiment brings chaos fast.
Spills, Cleanups, and Emergencies
Every lab using this compound keeps a cleanup plan ready. Small spills mean wetting with water—not dry sweeping. Dry powder in the air travels straight to your respiratory system. Disposable towels and pads go straight into hazardous waste bins after cleanup. Larger spills drive everyone out until evacuation procedures snap into place.
Washing any exposed skin right away cuts down risk, but medical attention shouldn’t wait either. Nickel compounds stick around in the body. Emergency showers and eyewashes can’t be ignored. These showers often gather dust in some labs, but all it takes is one splash to make you glad they’re working.
Disposal with Responsibility
Waste disposal laws around nickel and oxidizers exist for a reason. Pouring waste down the drain turns a personal mistake into a community hazard. Professional hazardous waste services take away the guesswork. People following shortcuts pose risks beyond their own workspace.
Safety with ammonium nickel nitrate comes down to real attention, the right gear, and refusing to skip steps. The stakes always feel higher when personal experience frames every decision in the lab.
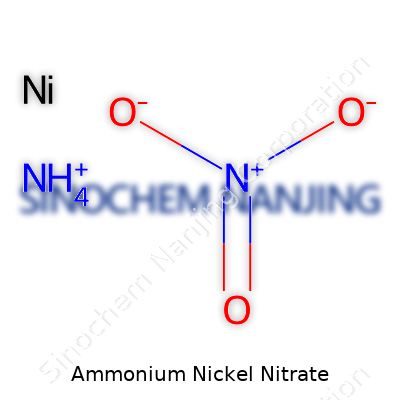
| Names | |
| Preferred IUPAC name | ammonium dinitratonickelate(II) |
| Other names |
Nickel diammonium nitrate Nickel ammonium nitrate |
| Pronunciation | /əˈmoʊniəm ˈnɪkəl ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 15492-38-3 |
| Beilstein Reference | 14620 |
| ChEBI | CHEBI:131262 |
| ChEMBL | CHEMBL4297958 |
| ChemSpider | 23072309 |
| DrugBank | DB14506 |
| ECHA InfoCard | 03b6b7e7-4993-4a51-99ba-066afa8a15e8 |
| EC Number | Ammonium Nickel Nitrate" EC Number: "236-751-3 |
| Gmelin Reference | 80854 |
| KEGG | C18649 |
| MeSH | D017715 |
| PubChem CID | 16211806 |
| RTECS number | QR9650000 |
| UNII | I67O6823NI |
| UN number | UN2726 |
| Properties | |
| Chemical formula | NH4Ni(NO3)3 |
| Molar mass | 182.85 g/mol |
| Appearance | Green crystals |
| Odor | Odorless |
| Density | 1.88 g/cm³ |
| Solubility in water | soluble |
| log P | -2.2 |
| Acidity (pKa) | 10.5 |
| Basicity (pKb) | 5.96 |
| Magnetic susceptibility (χ) | +1,200e-6 cm³/mol |
| Refractive index (nD) | 1.51 |
| Dipole moment | 1.52 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 332.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -365.7 kJ/mol |
| Pharmacology | |
| ATC code | V09DX05 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and serious eye irritation, may cause an allergic skin reaction, may cause cancer, may cause damage to organs through prolonged or repeated exposure, very toxic to aquatic life. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H315, H319, H332, H334, H335, H341, H350, H360D, H373, H410 |
| Precautionary statements | P210, P220, P221, P260, P264, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P321, P330, P337+P313, P370+P378, P405, P501 |
| NFPA 704 (fire diamond) | 2 3 1 OX |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD₅₀ Oral Rat: 175 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 175 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Ammonium Nickel Nitrate: "0.1 mg/m3 (as Ni) |
| REL (Recommended) | 0.01 mg(Ni)/m³ |
| IDLH (Immediate danger) | IDHL: 10 mg Ni/m3 (as Ni) |
| Related compounds | |
| Related compounds |
Nickel Nitrate Ammonium Nitrate Nickel(II) Ammonium Sulfate Nickel(II) Chloride Nickel(II) Sulfate |

