Ammonium Mercury Thiocyanate: A Closer Look at a Legacy Chemical with a Troubling Future
Historical Development
Ammonium mercury thiocyanate brings up a unique mix of curiosity, concern, and nostalgia for anyone who’s ever cracked open an old chemistry set. Those spectacular “Pharaoh’s serpent” demonstrations from school science fairs—columns of ash rising and solidifying in the wake of something almost magical—drew on this compound’s peculiar properties. Scientists discovered its dramatic thermal decomposition in the mid-19th century, and its formation hails from the early efforts of combining mercury and thiocyanate chemistry. As public consciousness about mercury dangers grew, so did the unease around such experiments, reining in what once seemed a harmless scientific curiosity. Throughout decades, what began as educational spectacle and laboratory oddity shifted into a cautionary tale, reminded by old photos and stricter lab protocols everywhere.
Product Overview
In the world of specialty chemicals, ammonium mercury thiocyanate has never been as common as other reagents or salts. Its primary draw comes from its bizarre ability to expand rapidly and form sculptures of carbon and metal ash after ignition, a result of an intricate dance between mercury, sulfur, carbon, and nitrogen within its structure. The sheer drama, mixed with the underlying hazards, makes it stand out from other laboratory reagents. Most chemists who handle it recognize its legacy while eyeing it warily, understanding that its true story reveals as much about changing lab culture as about the compound itself.
Physical & Chemical Properties
With a pale appearance—usually white-to-gray crystalline powder—ammonium mercury thiocyanate doesn’t hint at the spectacle it can produce. Its odor isn’t strong but can remind you of sulfur. The compound is heavy, denser than many other common salts due to the mercury sitting right in its core. Water dissolves it slowly, cold water even less so, and it picks up moisture from the air, clumping before you notice. Heat brings out its most infamous feature: instead of simply melting or burning, it erupts in an exothermic spectacle, breaking down into mercury vapor, carbon nitride, ammonium sulfate, and other byproducts. The decomposition products fill the air with acrid, unpleasant fumes. Any chemist who has observed this reaction once does not forget the sharp, metallic smell or the odd brown-black columns left afterward.
Technical Specifications & Labeling
On a label, you’ll see warnings for poison, environmental hazard, and the familiar crossed bones of acute toxicity. Most sources note purity levels somewhere north of 95%. Safety phrases jump off the bottle: keep away from heat, avoid skin contact, and never breathe in dust or fumes. Transport and storage get a lot of attention, with locked cabinets, corrosion-resistant containers, and frequent inventory checks. Everything hinges on preventing accidental exposure—not just for the person working directly with the material, but for everyone nearby. Users often double-check for clear, unambiguous markings before going any further. Mislabeling of this chemical has led to more than one near-miss in labs that should have known better.
Preparation Method
Preparation usually involves mixing a mercury(II) salt, such as mercury(II) nitrate or chloride, with ammonium thiocyanate in solution. The process isn’t overly complex, but it presents hazards from every angle. You’ve got to control temperature, avoid splashing, and work under a fume hood, or else you risk inhaling fumes or touching mercury compounds. In practice, many labs abandoned small-scale synthesis, preferring to source it from suppliers who follow stricter controls and automated handling. The days when high schoolers mixed up batches in the back room feel very far away, especially with stronger rules and heightened awareness of chemical exposure risks.
Chemical Reactions & Modifications
The most memorable reaction stays the classic thermal decomposition—ignite a pile, and it grows large columns of foamy, gray-black ash. But this only scratches the surface. The thiocyanate group lends a little flexibility for chemical modification, though toxicity restricts widespread experimentation. Most changes come by swapping out the ammonium or mercury for other cations, exploring analogues for slightly safer demonstrations. Even so, the majority shy away from such exploration, especially as disposal of mercury wastes becomes harder and regulations more demanding. Some researchers study complexation or redox chemistry, searching for clues about mercury’s unique behavior; these investigations help illuminate fundamental concepts in coordination chemistry, but rarely lead to practical applications, largely due to the hazards.
Synonyms & Product Names
Common alternate names include ammonio(thiocyanato) mercury, ammonium mercury dithionate, and even “Pharaoh’s serpent powder,” a nod to its cult-like demonstrations. Older textbooks sometimes use outdated notation, like “mercuric ammonium thiocyanate.” Chemical supply firms file it under its CAS number or IUPAC designation, but anyone with a background in science recognizes its spectacular, dangerous side and doesn’t need extra reminders at the point of purchase. Street names or jargon from the era of classic chemistry sets show up sometimes, especially in hobbyist or educational circles reminiscing about less restrictive times.
Safety & Operational Standards
Scientists approach ammonium mercury thiocyanate with a mix of respect, wariness, and sometimes regret. Rules are much tighter compared to three decades ago. Proper fume extraction and full personal protective equipment form the standard baseline. Containment is key: gloves of heavy nitrile, thick lab coats, and face shields stand between users and the potential for fatal absorption. Mercury’s toxicity doesn’t care whether exposure comes through skin, inhalation, or ingestion. Even years after last handling the compound, researchers remember incidents traced to a single careless touch or a tiny drift of dust. Spill kits tuned for mercury emergencies sit nearby, and labs train students repeatedly on emergency response, emphasizing swift decontamination and complete containment. Most labs store this compound in restricted access cabinets with detailed tracking to prevent accidental contact and environmental contamination.
Application Area
Ammonium mercury thiocyanate, beyond its role in classic demonstrations, rarely features in applied chemistry anymore. Fifty years ago, some labs looked at it for specialty mercury analyses or for its weird expansion properties, thinking it might suit pyrotechnicism or novelty products. Times changed. Now, its use narrows down to a few academic settings that need to teach or study properties of mercury or sulfur chemistry in tightly controlled circumstances. Outside the lab, you’re far more likely to run into stories about its hazards than about its usefulness. Chemical museums, historical science exhibits, and regulatory discussions are the main places this chemical shows up.
Research & Development
Research these days treats ammonium mercury thiocyanate with strong suspicion. Projects focus less on discovering new uses, and more on mitigation—studying safe disposal, better containment, and recovery of mercury from compound wastes. Some researchers look for replacement compounds for classic experiments, hoping to retain the theatricality without the danger. In environmental chemistry, the compound serves as a cautionary example. Graduate students pore over old case studies and work out best practices for working with legacy mercury chemicals, drawing lessons from labs that suffered spills or chronic exposure. Journals occasionally publish on environmental controls, chemical remediation, and safer alternatives for demonstrations or teaching tools.
Toxicity Research
Mercury toxicity runs at the heart of every conversation about ammonium mercury thiocyanate. Acute effects range from short-term respiratory problems and skin burns to neurological consequences—tremors, mood changes, and memory lapses. Chronic exposure shadows researchers for years, sometimes revealing itself far after anyone suspected a problem. Health agencies around the world flagged the compound as a major hazard, not just for acute exposure but for its potential to bioaccumulate and leach into groundwater. Animal studies and environmental tracing efforts keep developing, strengthening the link between mercury exposure and systemic harm. Safe handling standards keep climbing higher each year, with fewer institutions willing to accept any risk where staff, students, or the wider environment sit in harm’s way. I’ve seen scientists develop symptoms after decades of careful work, haunted by a moment’s error with a mercury compound. These stories reinforce the importance of strict, sometimes inconvenient protocols.
Future Prospects
The future for ammonium mercury thiocyanate gets narrower every year. While there’s nostalgia for the classic “chemical serpent”, the risks push it further onto restricted lists. Educational uses continue to fade, replaced by safer substitutes or virtual demonstrations. Environmental rules get stricter, with authorities mandating mercury inventories, regular auditing, and heavy penalties for mishandling. Some voices call for total elimination of mercury compounds from non-essential uses, except in rare analytical contexts with unmatched alternatives. Green chemistry and chemical education move away from hazardous examples, seeking demonstrations that keep wonder alive without risking lives or ecosystems. For those of us with years in the lab, ammonium mercury thiocyanate reminds us of how far chemical safety has come—and of the urgent need to keep pushing so that dangerous legacies do not overshadow new discoveries or future generations of chemists.
What is Ammonium Mercury Thiocyanate used for?
Where Curiosity Meets Chemistry
Ammonium mercury thiocyanate triggers memories of crowded science classrooms, where someone set off a fiery-looking reaction dubbed the “Pharaoh’s Serpent.” Kids watched in awe as a grayish mass slowly crawled out of a dish, growing like a monster from a B movie. This compound became a legend in science fairs and chemistry kits, thanks to its dramatic effect when set alight.
The Science Behind the Spectacle
The most famous use of ammonium mercury thiocyanate lies in its role in chemical demonstrations. Light this white powder with a match and it swells, creating snaking columns that seem to defy logic. This reaction isn’t just visually arresting; it stands as a classic example of decomposition—an introduction for many students to how chemicals can change when pushed with heat.
Despite its allure, this compound doesn’t stick around just for the fun of a science show. In the early days, researchers and the curious used it for some analytical chemistry. The decomposition products include carbon nitride and mercury compounds, which show up in chemical analysis. Some analytical labs used this reaction to help spot other elements through the products released.
Hazards Woven into History
Even as the demonstration wows crowds, there’s an uneasy truth dragging behind. Mercury carries a heavy price in toxicity. Breathing in fumes or dust can poison the nervous system, cause kidney failure, and even damage the environment for years. The scorched remains also leave behind tough-to-clean mercury salts. With knowledge about these dangers growing, schools and labs turned away from this demonstration. These days, finding this reaction in classrooms feels rare—safety replaces spectacle.
Chemical Curiosity Meets Policy
Legal controls now shadow ammonium mercury thiocyanate. Many regions ban its use outside of approved research or tightly regulated spaces. My own experience with chemical outreach made me rethink using compounds like this at public events. Teachers opt instead for demos without mercury or cyanide in the picture—safer for both students and the planet.
Beyond demonstrations, little industry still seeks out ammonium mercury thiocyanate. Old patents mention photography or electroplating, but those uses faded as alternatives appeared. With health and ecological worries front and center, few companies or researchers risk storage or transport of a mercury salt with unstable properties.
Better Ways Forward
Solutions call for moving away from toxic reactants and exploring safer chemistry. Green chemistry pushes for demonstrations and experiments that trade spectacle for health and environmental safety. Groups like the American Chemical Society publish guidance and alternatives, focusing on experiments with kitchen ingredients or harmless household supplies.
Despite losing a fascinating crowd-pleaser, the switch to safe science matters. In the end, health and future cleanups win out over ten seconds of classroom wonder. Chemists now tell the story of ammonium mercury thiocyanate as a reminder: even jaw-dropping science must respect its costs.
Is Ammonium Mercury Thiocyanate hazardous or toxic?
Understanding the Compound
Ammonium mercury thiocyanate stands out to anyone who’s spent time in a high school chemistry lab. Teachers and students know it as the chemical behind the old “Pharaoh’s serpent” demonstration. You light it up, and a black, snake-like tendril crawls from a pile of powder. That tiny show sometimes sticks with people, but so do the safety warnings. There’s a reason for that: this compound is loaded with risks that reach beyond a simple burn hazard.
The Hidden Dangers
Mercury, in any chemically-bound form, sparks concern for anyone who takes health seriously. Ammonium mercury thiocyanate breaks down in several nasty ways when heated or burned. It releases mercury vapor, sulfur dioxide, and sometimes even cyanide-containing compounds. Mercury vapor goes straight for your nervous system, harming brain and kidney function over time. The Environmental Protection Agency and Centers for Disease Control both flag mercury as a neurotoxin, meaning no level is really safe to breathe in, especially for kids and pregnant women.
Sulfur dioxide has its own set of problems, irritating your lungs and sometimes triggering asthma attacks. Cyanide’s reputation for toxicity comes from its ability to choke off oxygen in the body. Altogether, these byproducts of a chemical experiment add up to something far more serious than a dramatic classroom trick. They turn a brief chemical curiosity into a possible health emergency, especially if used carelessly or without protection.
Why It Still Raises Concerns
Many schools and hobbyists tossed their stocks of ammonium mercury thiocyanate years ago, but the substance still shows up in old lab kits and on certain online marketplaces. Part of the problem comes from curiosity and tradition—the demonstration is a crowd-pleaser, so people overlook the compound’s risks. Easy online access doesn’t help, either. Some sellers do not provide clear warnings, and folks might assume it’s safe to use without checking the details.
Safety information sometimes falls by the wayside, or people rely on outdated handling methods. I remember watching someone light a “Pharaoh’s serpent” outdoors at a community fair, and no one wore gloves or masks. The wind carried fumes right back through the crowd. No one thought too much about it at the time, but looking back, it feels reckless given everything we know now about these chemicals.
What Can We Do?
Institutions with chemical stockpiles need to take inventories seriously and seek out hazardous leftovers like ammonium mercury thiocyanate. Disposing of old chemicals means working with professionals who know how to deal with mercury. No one should dump this stuff in the trash or try to destroy it at home—the risk isn’t just personal, but community-wide, contaminating air and water supplies.
Teachers need better support: updated training and plenty of resources to find safer demonstrations. Some chemical reactions put on a great show and offer good lessons without endangering health. The science community benefits most from educational tools that inspire curiosity without leaving teachers or students at risk.
Anyone thinking of buying old chemical kits should check for ammonium mercury thiocyanate in the list of ingredients. If it shows up, steer clear, or consult with hazardous waste professionals for safe disposal. Public awareness is key. Informing parents and kids goes a long way to keeping science both fun and safe for everybody.
How should Ammonium Mercury Thiocyanate be stored?
Peculiar Properties That Call For Care
Ammonium mercury thiocyanate doesn’t show up on chemical shelves meant for casual use. Old high school experiments once made it famous for its ‘Pharaoh’s serpent’ reaction, but those who remember handling it know this compound deserves real respect. This stuff brings together ammonium thiocyanate and mercury salts — both hazardous on their own. Combined, they create something that isn’t just toxic but also unpredictable if ignored. Even without immediate danger, people working with it live with a certain unease. Mercury compounds mess with the nervous system, and inhalation or skin contact can create problems that stretch far into the future.
Proper Conditions: Darkness, Dryness, and a Steady Cool
Direct sunlight turns this chemical volatile. One time, a colleague left a reagent bottle near a window ‘for just an hour.’ The label faded, and the crystals inside turned brown and soupy. That’s a harsh reminder: store only in tightly-sealed glass containers, never in plastic, and always in the dark. Reliable chemical suppliers ship them in amber glass bottles for good reason. Excess heat triggers decomposition — sometimes with nothing more than a forgotten lab radiator as the culprit. Keeping it between 2 and 8 degrees Celsius reins in those risks. If storage rooms sit above that temperature zone, the risk of dangerous byproducts sneaking into the air jumps fast.
Moisture: The Silent Enemy
Humidity isn’t just an inconvenience. Water vapor can seep through less-than-tight caps or old seals. I’ve watched a tiny white powder slowly clump into grey sludge after summer storms hit an unventilated storage room. Moisture encourages decomposition and releases noxious gases, so silica gel desiccant packs or well-ventilated, humidity-controlled cabinets aren’t optional luxuries. They’re about keeping leftover chemicals from becoming unknown threats.
Segregation: Chemistry Isn’t Always Friendly
Not every chemical gets along. Most lab accidents I’ve seen start with careless storage — someone thought another label covered ‘just another white solid’ and stacked bottles near acids, fuels, or oxidizers. That’s a recipe for trouble. Ammonium mercury thiocyanate should sit far from strong acids, bases, and oxidizing agents. Mercury compounds love to form more volatile mixes when left near ammonia or chlorine sources. A dedicated, clearly-labeled section makes sense, and chemical organizers must skip the urge to ‘free up shelf space’ by shuffling things together.
Attention to Access: Not for Curious Hands
Easy access corners and unlocked cabinets tempt curious minds. Stories circulate about lab visitors or students drawn by strange names or chemical myths. Key locks and sign-in procedures keep track of usage. People I trust with such keys usually hold specific training on mercury handling, so we can avoid accidental poisonings or environmental breaches. Oversight from safety officers draws grumbles, but every mishap avoided matters. Safety data sheets printed and taped to the inside of doors speed up responses if spills or exposures happen.
Disposal: Never Down the Drain
No amount left over belongs in the trash or sink. Environmental agencies — like the EPA or local waste authorities — demand hazardous material pickup for a reason. Dumping mercury salts ruins water tables and sets off clean-up bills no institution wants. Double-bagging leftovers, labeling clearly, and calling professional chemical disposal companies saves more than just time later. It also means peace of mind at the end of every lab session, knowing nothing toxic waits for an unlucky custodian.
What are the safety precautions when handling Ammonium Mercury Thiocyanate?
Not Your Ordinary Chemical
Ammonium mercury thiocyanate isn’t something you leave lying around. Even as a science geek who has seen and handled my share of unusual lab substances, this one demanded a pause. It looks a bit like white powder or fine crystals, but don’t let that fool you. This stuff turns downright hazardous if approached with a casual attitude.
Why So Dangerous?
People often talk about the mesmerizing “Pharaoh’s Serpent” demonstration, where this compound puffs up and creates sinuous ash when ignited. The wow factor is big, but so’s the risk. The dust and fumes can load your system with mercury — and mercury poisoning stays for life. Even small exposures build up in the body, damaging nerves, kidneys, and other organs in ways that medicine usually can’t fix.
Protecting Yourself Starts with Awareness
Nobody should even think about opening a jar of ammonium mercury thiocyanate without gloves and serious protection. I’ve seen smart folks skip that step and regret it. The powder absorbs through skin. After just a short contact, the hands are a conduit straight to trouble.
Go for thick nitrile or butyl rubber gloves. Thin latex doesn’t hold up. Long sleeves offer a good barrier and will stop droplets from landing where you can’t see them. Gown and lab coat are essentials, not extras.
I remember a day in the lab when somebody cracked open a container out of curiosity, and the stuff managed to get airborne. Eye protection got tested that day. Goggles beat glasses every time because particles bounce in through the sides. Face shields add another layer, especially if there’s any chance of splashing.
Breatheful of Poison? Not Today
The fumes and dust are ruthless. Mercury vapor puts lungs on the front line. Good ventilation offers some safety, but there’s value in sticking with a certified chemical fume hood. The simple fan on a countertop doesn’t cut it. A respirator rated for mercury vapor gives peace of mind when the process kicks up lots of dust.
Keep It Contained
Shortcuts with containment invite disaster. Use only sealed, labeled containers. Store this compound away from acids and heat. It doesn’t ask for much — just a cool, dry, well-ventilated space, locked so kids or visitors won’t stumble across it.
Never use glassware or tools that might later touch food or drink. Cross-contamination travels on shoes, clothes, and hands, so leave lab gear at work. If anything spills, sweep it up with a damp disposable cloth and never a dry broom, since sweeping throws powder into the air.
Cleaning Up Isn’t Just About Neatness
After working with it, every surface and tool deserves a wipe-down with specialized cleaning agents made for mercury compounds. Wash up, scrub under nails, and leave gloves in the hazardous waste. Disposal makes a difference: all materials, even the wrappers from the gloves, go into a designated hazardous waste bag.
Solutions and Smarter Moves
Colleges and industry labs have started favoring digital or virtual alternatives for old demos with ammonium mercury thiocyanate. Safer substitutes lead to lessons without the risk and still bring science to life. Education about dangers needs to match hands-on excitement. Proper training keeps people curious, but also careful.
What is the chemical formula of Ammonium Mercury Thiocyanate?
Formula and Structure
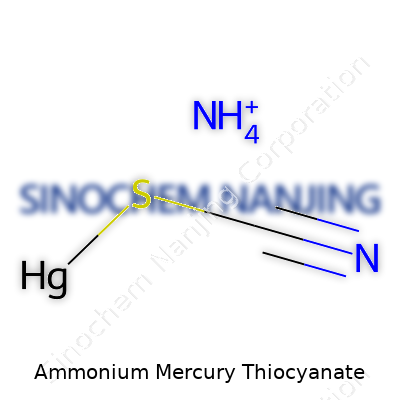
Ammonium mercury thiocyanate, known by its chemical formula NH4[Hg(SCN)3], stands out in the chemical community for its strange reaction to heat and its place in classic science demonstrations. The molecule contains an ammonium ion (NH4+), a mercury ion coordinated with three thiocyanate ions (Hg(SCN)3-). This formula isn’t something that shows up in standard chemistry classes, but the result of mixing mercury(II) salts with ammonium thiocyanate forms these light tan crystals, quietly hiding a shockingly active nature.
What Happens During Decomposition?
I remember sitting in a dusty school lab, watching in awe as a teacher showed us the “Pharaoh’s Serpent” demonstration. He placed a bit of this compound on a fire brick, then touched a match to it. The pile didn’t just burn — it bubbled and writhed as a massive, ashy snake seemed to crawl from the tiny mound. Beneath the fun, the reaction involved serious chemistry: once heated, ammonium mercury thiocyanate decomposes into carbon nitride foam, along with releases of mercury vapor, sulfur oxides, and other hazardous substances.
Dangers Often Overlooked
Many of us only saw the dramatic foam. Later, I learned that the mushrooming mass masks a toxic brew. Mercury vapor is highly poisonous. So are many byproducts of the breakdown. The demonstration has a dark side, responsible for its ban in many countries. Hundreds of cases of mercury poisoning and environmental damage have been linked to careless handling of this and other mercury compounds. Children and adults alike have developed chronic health issues from inhaling those vapors.
Environmental Impact and Safety
Compounds collecting mercury lead to contamination that stays for decades. Once released, mercury sticks around — in rivers, soil, and living bodies. In the US, strict storage and disposal laws aim to stop these problems at the source, yet old stocks of ammonium mercury thiocyanate sometimes turn up in forgotten stores or old science labs. The situation shows that chemistry isn’t just what happens on a workbench; sometimes, it spills far beyond.
Better Approaches and Safer Choices
As a chemist who values safe, hands-on science, I see real lessons here. Fascination with chemical demonstrations can be turned toward safer experiments. Teachers and enthusiasts now use baking soda, sugar, or even paraffin wax for fiery science tricks, skipping deadly mercury. Safer chemicals mean no toxic fumes. Students get the same sense of scientific awe, minus the hidden health risks.
Managing mercury compounds responsibly begins with awareness and honest conversation about their danger. Substituting safer materials, securing storage, and following hazardous waste disposal guidelines protect people and the environment. Today, with better access to safety data, nobody has to trade education for health. Chemistry is about discovery, but every experiment worth doing is worth doing safely.
| Names | |
| Preferred IUPAC name | Ammonium tetrathiocyanatomercurate(II) |
| Other names |
Mercuric ammonium thiocyanate Ammonium mercuric thiocyanate Mercury ammonium thiocyanate |
| Pronunciation | /əˈmoʊniəm ˈmɜːrkjuri ˌθaɪ.oʊˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 12125-15-6 |
| Beilstein Reference | 3534433 |
| ChEBI | CHEBI:22379 |
| ChEMBL | CHEMBL38710 |
| ChemSpider | 15351 |
| DrugBank | DB11428 |
| ECHA InfoCard | 03e96d5a-7468-4a10-ab7b-2d9fecfd3426 |
| EC Number | 215-630-3 |
| Gmelin Reference | 83277 |
| KEGG | C01086 |
| MeSH | D000687 |
| PubChem CID | 24634 |
| RTECS number | XZ1925000 |
| UNII | 2G2H6AFX8X |
| UN number | UN1649 |
| CompTox Dashboard (EPA) | DB07424 |
| Properties | |
| Chemical formula | NH4[Hg(SCN)4] |
| Molar mass | 284.43 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.95 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -3.46 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 4.46 |
| Magnetic susceptibility (χ) | −85.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.618 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -96.6 kJ/mol |
| Pharmacology | |
| ATC code | S51BC02 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes burns, may cause sensitization by skin contact, releases toxic fumes under fire conditions. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P210, P222, P273, P281, P302+P352, P305+P351+P338, P310, P370+P378 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Autoignition temperature | 160 °C |
| Lethal dose or concentration | LD50 oral rat 40 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-Rat 41 mg/kg |
| PEL (Permissible) | PEL: 0.1 mg(Hg)/m³ (as Hg) |
| REL (Recommended) | No recommendation provided |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Mercury(II) thiocyanate Thiourea Potassium thiocyanate Ammonium thiocyanate |

