Ammonium Hydrogen Sulfate: The Workhorse Compound in Industry and Research
Historical Development: From Byproduct to Backbone
Ammonium hydrogen sulfate tells a real story about chemistry’s ability to turn a waste product into essential feedstock. Years ago, most chemists saw this compound only as a leftover from ammonia scrubbing or caprolactam production. Early references pop up as far back as the 19th century, mostly in the context of fertilizer runoff and sulfur recovery. Over the decades, as industries became sharper about resource recovery, ammonium hydrogen sulfate shifted from being unwanted sludge to a key resource. Real growth followed World War II’s chemical boom; engineers realized that containing and utilizing every bit of reactive nitrogen and sulfur could mean less waste, tighter margins, more responsible stewardship. Academics followed, digging into its structure, properties, and reaction pathways—especially as environmental scrutiny increased.
Product Overview: Ubiquity Under the Radar
This salt might not grab headlines, but you’ll find it shaping countless processes behind the scenes. Firms in the textile, mining, and specialty fertilizer worlds use it by the ton, often without drawing attention to its role. It comes as a colorless to white crystalline solid with strong hygroscopic traits. Although often overshadowed by its cousins ammonium sulfate and ammonium bisulfate, its presence pops up in more technical corners: acidification reactions, catalyst preparation, water treatment, and specialized lab protocols. I remember visiting a fertilizer plant and seeing bins of ammonium hydrogen sulfate tucked between far more glamorous products—yet engineers said they’d be lost without it for pH adjustment and precipitation steps.
Physical & Chemical Properties: Substance With an Edge
Solid at room temperature, this salt clings to water and packs a strong acidic punch. Formed from stoichiometric amounts of ammonia and sulfuric acid, its molecular structure makes it more acidic than ammonium sulfate, giving it broader reactivity. It dissolves quickly in water, releasing both ammonium and bisulfate ions. In concentrated form, it gives off ammonia fumes, which any chemist with a sensitive nose will pick up instantly. You won’t find it burning or exploding in the lab, but let’s not gloss over the corrosive potential—it can eat away at copper and some steels if maintenance gets ignored. Its melting point hovers just below 150°C, handy for those wanting a low-temp source of acidity.
Technical Specifications & Labeling: No-Nonsense Communication
Regulatory expectations for this salt run tight. Industrial supplies face purity standards, usually 98% or better, with maximum thresholds for heavy metals, free acid, and insoluble matter. Labeling rules in most regions require hazard pictograms relating to skin and eye irritation. Safety data sheets must follow GHS conventions, but I don’t see anybody buying it in small bottles at the hobby store. Most procurement teams review specs like particle size and bulk density, because both affect handling; too dusty, and the product clings everywhere, too chunky and it won’t dissolve evenly in solution tanks. Exact requirements depend on application—fertilizer blends need less purity than electronics or labwork.
Preparation Method: Getting Down to Chemistry
Industries produce ammonium hydrogen sulfate by neutralizing sulfuric acid with a controlled dose of ammonia gas or liquid ammonia. Trained operators monitor temperature and pH closely, since overshooting the acid side leaves too much free sulfuric acid, while excess ammonia tips the balance toward less acidic ammonium sulfate. Slurry settles out as a crystalline mass, and after centrifugation or filtration, the salt dries down to a stable solid. Some plants tweak reaction parameters to optimize particle size, especially if downstream users want free-flowing material. Old-school chemists might remember small-scale prep in glassware, but modern output reaches thousands of tons every month. Much of the process uses waste streams—proof that industrial chemistry doesn't have to be a zero-sum game.
Chemical Reactions & Modifications: Beyond the Basics
What stands out about ammonium hydrogen sulfate isn’t just where it starts, but where it can go. It acts as an intermediate in synthesizing ammonium sulfate, ammonium alum, and other ammonium salts through secondary reactions with bases or additional acid. Environmental engineers trust its strong acidity for pH adjustment and dissolving scale. Lab chemists sometimes use it to decompose organic matter or carry out mild oxidations; it synergizes well with hydrogen peroxide in particular. The compound can release ammonia under heat—useful for generating gaseous ammonia in situ or scrubbing unwanted acid vapors. I've met process chemists who tweak residual sulfuric acid or add trace elements to create custom blends, targeting everything from soil improvement to rare element extraction.
Synonyms & Product Names: Multiple Faces, Single Identity
Ammonium hydrogen sulfate appears in texts and catalogs under a handful of names: ammonium bisulfate, ammonium acid sulfate, and even monoammonium sulfate (though that label often causes confusion with a different compound). Product codes vary, but anybody in industry will recognize the NH4HSO4 formula as shorthand. Most reference books and regulatory listings skip the fanciful trade names and stick with IUPAC conventions—clarity matters, especially in hazardous goods logistics. This chemical doesn’t have the glamorous branding seen with high-value specialty reagents.
Safety & Operational Standards: Respect Goes a Long Way
Nobody survives long by slacking off on chemical safety, and ammonium hydrogen sulfate keeps you honest. Exposure risks fall into the “respect, don’t fear” category—serious but manageable. The compound stings eyes and skin and can trigger coughing fits if dust becomes airborne. I’ve seen old bins in neglected warehouses eating through the paint and rusting steel bins. Ventilation stands out as a must, especially in storage and mixing areas. Full PPE (gloves, goggles, aprons) stays non-negotiable, and regular ventilation checks reduce long-term risk. OSHA and EU standards treat it as a hazardous corrosive, requiring tight labeling and secure transport. The waste stream must follow local environmental disposal codes, often passing through neutralization tanks before discharge.
Application Area: Quiet Influence on Major Processes
The influence of ammonium hydrogen sulfate stretches beyond its reputation. In fertilizer manufacture, it helps manage pH and acts as a source of both ammonium and sulfur—two essential elements in agriculture. Mine engineers use it to leach valuable metals from low-grade ores. Textile plants rely on its acerbic bite to clean and soften processing baths. In water treatment, it helps control alkalinity and support chlorine dioxide sanitation regimes. Analysts harness it for sample digestion protocols when high sulfur content is required. I’ve also watched chemists use it to regenerate exhausted ion exchange resins, giving the filters a second life and slashing operational costs. Lab kits and reagent sets often include it as a buffer or reactant, especially in protocols demanding mild acidity without excess bulk minerals.
Research & Development: New Tricks for an Old Salt
R&D teams keep pushing this compound into new territory. Academic labs are probing its role in battery technology, using ammonium hydrogen sulfate as an ionic conductor or pH buffer in industrial electrolytes. Environmental chemists test its effectiveness for remediating metal-polluted soils. Some research programs dig into safer and more efficient preparation routes, driven by the need to cut emissions and reclaim even more secondary raw materials. Process safety experts are validating dust management techniques, given concerns about chronic exposure. There’s work on film-forming blends for flame retardants or agricultural sprays, harnessing the compound’s hygroscopic properties for slow nutrient release. Scientific journals document catalysis applications too—especially for processes pushing toward green chemistry benchmarks.
Toxicity Research: Balancing Use and Health
No responsible chemist ignores health risks, and the story here is nuanced. Acute exposure mainly causes irritation; long-term risks tie back to dust inhalation, prolonged skin contact, or contaminated water discharge. Animal studies show low systemic toxicity compared to more aggressive acids, but researchers caution against downplaying chronic effects. Wastewater scientists monitor ammonium releases, concerned about eutrophication and downstream nitrate conversion. Hospitals and poison control centers see few cases, mostly accidental worker exposure; prompt rinsing often prevents damage. There’s a real incentive to tighten handling practices, install better ventilation, and train plant operators, minimizing the odds of both acute and chronic impact.
Future Prospects: Ready for What’s Next
Looking at coming decades, the story hinges on resource recycling, clean production, and value-added chemistry. Stricter regulations around sulfate and ammonia emissions push plant managers to come up with tighter processes, reducing both input and waste. R&D directors have their eyes on smarter usage—turning ammonium hydrogen sulfate into feedstock for new materials, water purification agents, or chemically tailored fertilizers. The rise of precision agriculture and circular economy models gives this compound a new relevance. It’s not chasing headlines like lithium or rare earths, but I see more engineers and scientists revisiting its potential, especially as industries hunt for every possible efficiency. The journey from byproduct to backbone continues, fueled by practical innovation—not empty hype.
What is Ammonium Hydrogen Sulfate used for?
Not Just a Lab Chemical
Ammonium hydrogen sulfate rarely shows up in public conversations, but its presence stays steady in industries that feed our everyday needs. As a white, crystalline powder, this compound steps up in jobs ranging from cleaning refinery equipment to fertilizing crops. Its combination of sulfate and ammonium brings both acidity and nitrogen into play, and these features shape its wide set of uses.
Descaling in Industry
Many refineries and industrial plants deal with mineral buildup. These deposits threaten to slow machines down and even break them over time. From my own work in industrial maintenance, scale removal eats up budgets and makes scheduling tougher, especially if you need to shut things down to clean. Here’s where ammonium hydrogen sulfate comes in. Mixed into cleaning solutions, it reacts with hard mineral scales like calcium carbonate, dissolving deposits and scraping away years of buildup. Machinery runs smoother, and unplanned shutdowns become less of a nightmare. Using it in closed systems limits worker exposure and makes disposal easier, so the safety track record holds strong.
Boosting Crop Yields
Modern agriculture runs on inputs. As someone who spent summers working near fertilizer plants, the giant stockpiles of raw materials left an impression. Ammonium hydrogen sulfate contributes by delivering both nitrogen, a growth-defining nutrient, and sulfur, which is crucial for plant protein formation. Some soils, especially those farmed intensively or exposed to lots of rain, lose sulfur quickly. Farmers often mix this compound into blended fertilizers when other sources can’t deliver enough. The effect? Healthier crops, greener leaves, and higher yields in fields that otherwise turn pale and stunted from lack of nutrition. Since food costs matter to all of us, the knock-on impact lands right on dinner tables across the world.
Helping the Environment
Ammonium hydrogen sulfate also finds a role in curbing industrial air pollution. In the world of smokestack “scrubbers,” pollution control technology depends on chemicals that bind or neutralize gasses before they escape. Power plants and processing facilities often produce ammonia or sulfur-based emissions. By using this compound, they catch two birds with one stone: neutralizing acidic gases and capturing ammonia in a form that's easier to store. Some companies have embraced closed-loop systems where this waste stream is fed back into fertilizer production. This kind of recycling not only saves money, it keeps harmful pollutants out of the atmosphere.
Challenges and Smarter Handling
Working around ammonium hydrogen sulfate isn’t without risk. The compound’s acidity can eat through skin and textiles, and breathing its dust leaves lungs irritated. In the fertilizer world, dust management and safe storage matter as much as the chemistry. Over the years, stricter guidelines have dropped the rate of on-site accidents. From my perspective, tighter education for workers and community-facing transparency build trust and keep things running well. Automated handling and better ventilation cut down on exposures while letting the industry balance productivity with worker safety.
The Path Forward
As factories and farms face pressure to grow cleaner and more efficient, the search for better tools continues. Ammonium hydrogen sulfate’s story isn’t just about chemistry labs; it’s tied to the challenge of feeding a growing population, shrinking waste, and keeping equipment running. Investing in greener production methods, sharing up-to-date research, and listening to local community concerns keep this compound on the responsible edge of resource management. Transparency from producers, steady improvements in safety, and ongoing research give everyone a stake in smarter solutions.
Is Ammonium Hydrogen Sulfate hazardous or toxic?
Everyday Encounters With Chemical Risk
Plenty of chemicals find their way into daily life, from cleaning supplies at home to fertilizers out in the field. Ammonium hydrogen sulfate doesn’t jump out at most people unless they work in a lab, chemical plant, or the fertilizer industry. Even though it stays behind the scenes, understanding what it can do to the human body and environment matters.
Direct Health Risks
Ammonium hydrogen sulfate irritates skin, eyes, and the respiratory system. The salt’s low pH draws water out of cells fast, which leads to burning and pain. A small handful of the compound won't threaten life outright, but touching it with bare hands results in red, itchy patches that stick around for hours. Splashing it in the eye means you’re reaching for an eyewash station fast. Breathing in the dust sends sharp discomfort down the throat, making people cough, sneeze, or wheeze.
Working with the compound, even in a school lab, sheds a little dust into the air. Inhaling a small amount catches up with people fairly quickly, as the sensation mimics mild smoke inhalation. Larger accidents—like a broken bag in an industrial setting—expose more workers and raise health stakes. If someone already has asthma or other respiratory trouble, a lungful triggers attacks far faster than pollen season ever could.
Is It Poisonous?
Toxicity often means a substance directly poisons cells, attacks organs, or builds up in the system. Ammonium hydrogen sulfate doesn’t slip into people’s bloodstreams easily, and eating a small amount once does not put lives in danger. Swallowing a large dose, though, brings heavy irritation, vomiting, and diarrhea. In animal testing, doses have to get pretty high to become life-threatening, but that doesn’t mean it's safe to swallow.
If the salt breaks down, part of it can release ammonia, which adds another layer of discomfort, especially indoors or in hot weather. Ammonia vapor stings eyes and airways, so proper ventilation in any workspace counts for a lot.
Environmental Footprint
Spilling ammonium hydrogen sulfate around fields or waterways releases a heavy shot of acid. That drops the pH of soil and water, making growing tough for many plants and aquatic animals. The salt’s ammonium chunk ends up feeding algae and upsetting river or lake ecosystems if it washes downstream. Fish kills or sudden weed booms trace back to run-off events like this.
A spill at a factory or transport accident means a cleanup crew suits up in chemical-resistant gear, scooping the powder carefully. They know that letting it seep into groundwater inflames both public health and natural cycles, making regulation and prevention a constant battle.
Managing Risk for Workers and the Public
Respecting the hazards of ammonium hydrogen sulfate looks like strong gloves, safety goggles, good air flow, and regular training for workers. Schools and factories lock down the chemical in marked, dry storage areas. Quick clean-up for any spill, and a clear response plan for accidental exposure, helps everybody avoid surprises.
Communities living near fertilizer or chemical plants deserve honest conversation about what’s stored on-site and plans for spills or leaks. Laws in many countries demand regular reporting, emergency planning, and neighborhood alerts—because being transparent about chemical hazards builds trust and helps keep people safe.
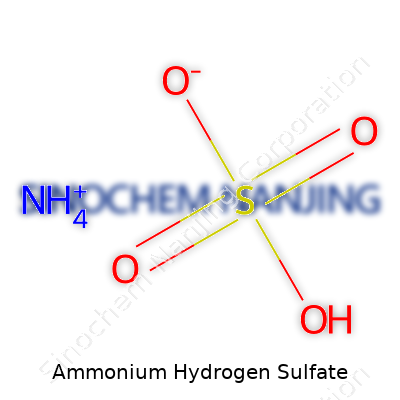
What is the chemical formula of Ammonium Hydrogen Sulfate?
Ammonium Hydrogen Sulfate: NH4HSO4
Every time I see a question about a chemical formula, I think back to long days spent in the lab, chalk streaks on my hands, trying to keep track of acids, bases, and the odd salts that came up on exams. Ammonium hydrogen sulfate, NH4HSO4, doesn't just turn up out of nowhere. It's the sort of compound you meet in practical situations: fertilizer manufacture, some sorts of cleaning products, and even a handful of industrial reactions. The formula might look simple, just a mash-up of an ammonium ion and a hydrogen sulfate anion, but its real importance comes into play outside the textbook.
Breaking Down the Chemical Bonds
NH4HSO4 is made from the ammonium ion (NH4+) and the hydrogen sulfate ion (HSO4-). Anyone who's ever mixed an acid with a base and watched a new salt form—maybe with a little fizz or color change—can appreciate the chemistry here. There’s nothing mysterious about it; it's the result of mixing ammonia and sulfuric acid. The ammonium brings nitrogen and hydrogen, the hydrogen sulfate brings more hydrogen, sulfur, and oxygen. The mix looks like this: one nitrogen, five hydrogens, one sulfur, and four oxygens. Knowing what’s in the formula helps folks in agriculture feed soil better and anyone in industry work more safely around chemical stocks.
Fertilizers, Safe Handling, and Real Risks
As someone who’s seen the damage that careless chemical handling can do, I can’t overlook real risks in labs and fields. Ammonium hydrogen sulfate often carries the “irritant” label. Industrial safety isn’t just about reading safety sheets; it comes from understanding consequences. Sulfates interact with water in the air to make acidic solutions, and careless contact stings skin and eyes. Handling it without gloves or a mask can land you in trouble. Yet, the reason for widespread use comes from its reliability. It acidifies soil in places where crops just won’t thrive otherwise, which helps farmers grow food in regions where the soil pushes back against modern agricultural methods.
Agricultural Benefits and Environmental Concerns
Using NH4HSO4 in fertilizer blends keeps certain plants healthy in difficult ground. Wheat and corn fields depend on the extra punch of sulfate. It’s not just about boosting yield, though. Excess runoff from overuse can strain local waterways and disrupt ecosystems. Decades of experience with fertilizer runoff made me pay close attention to riverbanks in the Midwest, where algae blooms affect water supplies and hit small town economies. Over-applying any ammonium or sulfate-based fertilizer sets off a chain reaction of unintended side effects, from groundwater acidification to toxic algae.
Toward Smarter, Safer Chemistry
I’ve learned to ask questions about any compound that moves from the lab bench to the farm field. Simple formulas like NH4HSO4 come with big responsibility. Better application methods, soil sensors, and smarter regulation of storage and transport would cut down on accidents and keep fertilizers effective. The formula itself hasn’t changed in a century, but how we handle it always can. From agriculture classrooms to messy tool benches, understanding these details doesn’t just keep you safer: it sharpens practical thinking, helps avoid waste, and gives soil a chance to recover for the next season. Knowledge, not just formulas, always makes the difference.
How should Ammonium Hydrogen Sulfate be stored and handled?
Pitfalls That Come With Storing Ammonium Hydrogen Sulfate
Ammonium hydrogen sulfate, a pale, granular solid you often find in chemical labs and industrial setups, doesn’t look all that scary at first glance. Anyone who’s worked with chemicals knows looks tell only half the story. This compound reacts sharply to moisture and can corrode metal over time. I’ve watched a once orderly storage closet turn into a rusty mess thanks to a few ignored guidelines—and half-open sacks left sitting over a damp weekend.
Dryness always tops the list for storage needs. Humid air kicks off clumping and turns bags of ammonium hydrogen sulfate into immovable bricks. Anyone handling supply logistics in a factory or school knows daily weather can defeat the best of intentions. Chemical-resistant shelves and bags with real seals, not those weak twist-ties, make life easier. Storage off the ground—on pallets or racks—keeps the bottom row dry during accidental spills or floods.
Personal Safety in the Workplace
Grabbing a scoop bare-handed seems quick, but skin quickly reminds you ammonium hydrogen sulfate irritates. It gives off ammonia and sulfur oxides if heated or mixed wrong. I once watched a new lab tech ignore gloves and end up nursing a rash for a week. Rubber gloves, goggles, and long sleeves seem like overkill until you have to wash acid out of your favorite jeans. Industrial experts put strong exhaust fans near their chemical cupboards, because even slow leaks can spoil the air. If your eyes start to itch or the back of your throat gets scratchy, don’t try to tough it out—open the windows, get upwind, and check for loose powder.
Rules That Matter on the Shop Floor
Label everything, always. It sounds basic, but people forget that white powder can mean sugar, salt, or something hazardous. Slapping a weatherproof label with both the chemical name and a hazard warning saves much grief. On a big site, the chemical safety sheet ought to live near the stock: clear, easy to read, and not buried under months of paperwork.
Bulk storage tanks sound efficient but hide big risks. Mild steel may last years, then fail with little warning, sending acidic runoff across the floor. Fiberglass and high-density plastic win here, though the upfront cost gives budgets a jolt. Routine inspection—monthly walkarounds, not just annual reports—finds small leaks before they make the local paper.
Responding When Things Go Wrong
Spills bang on the panic button. Fine powder drifts, gumming up floors and making everything slippery. Dry sweep or vacuum systems make cleanup safer; water sometimes just spreads the mess. Emergency eyewash stations and simple signs matter during long hours in the warehouse: I remember the look on a coworker's face after an unexpected splash hit his cheek—he found relief because he knew right where to go.
Working Smarter With Reliable Information
Every team works better with training that sticks. Blunt talk from supervisors and clear, real-world examples build habits that keep people safe. Health authorities and chemical associations publish details updated each year—staying current is more than compliance, it’s about peace of mind for everyone who wears the uniform. Information translates to action: posters, daily reminders, hands-on drills. A smooth-running storeroom or lab doesn’t happen by accident; people pull together because they’ve seen what happens if they don’t.
What are the safety precautions when working with Ammonium Hydrogen Sulfate?
Working With a Difficult Chemical
I remember my first semester in a university lab, getting handed a bag of white crystals and a stern lecture on safety. Ammonium hydrogen sulfate seemed harmless at a glance, almost like table salt, but that sense of comfort vanishes after the first sharp breath of sour air. This compound deserves careful respect because it burns the eyes, irritates skin, and turns even a routine spill into a minor crisis.
Understanding the Real Dangers
Ammonium hydrogen sulfate stings when it hits bare skin or eyes. The dust floats easily and, once inhaled, can choke off easy breathing or bring a cough that lingers all afternoon. I have seen students come in with nothing more than a dust mask and leave with eyes streaming and complaints of a raw throat.
The risk doesn’t end there. Mixed with the wrong chemicals, this compound releases toxic fumes or acids that eat away at lungs or equipment. Water turns small spills into slippery patches. Even in dry rooms, electrostatic charges can send dust into the air or onto other surfaces, making accidental exposure easy.
The Basics: Gloves, Goggles, and Good Sense
At the root of safe handling, it starts with solid habits. Nitrile gloves beat latex every time for chemical protection. Fit those tightly and change them at the first sign of damage. I double-glove in labs with unpredictable students. Goggles stay strapped on, not perched on the head. One unconscious hand-rub in the middle of a task can lead to hours in the campus doctor’s office.
Lab coats do more than signal seriousness. Thick cotton holds off splashes and keeps unsafe dust away from day clothes. I have seen chemical burns spread from shirt sleeves because someone decided short sleeves were easier to move in. Covered skin always wins.
Good Ventilation Saves Health
Working in a room designed for chemicals keeps the air clear and safe. Fume hoods aren’t luxuries. The cloud that rises during a transfer isn’t always visible, but I trust the slight whoosh of airflow keeping those vapors contained. One time, during a maintenance sweep, failing fan motors let the fumes drift back into the lab room. That mild tang in the air became a sign to shut down activities until real safety returned.
Housekeeping Stops Accidents
Keep benches clear. Cap bottles before moving a step. Label everything, even temporary jars. More than once, an unlabeled beaker looked like water, only for the story to end in confusion, extra clean-up, and more paperwork. Bags of spill-absorbent powder, neutralizing agents, and eye-wash stations should be within arm’s reach any time ammonium hydrogen sulfate shows up.
Planning and Emergency Practice
Nobody remembers where the emergency shower is until it matters. Walk through the steps out loud every session. The best time to know the path between workstation and eyewash is before a mistake. Post the relevant safety data on the wall, not hidden in a binder. I still find students who never check that binder until an incident occurs.
Trained response beats panic every time. With any chemical exposure, quick action prevents long-term problems. Every spill, inhalation, or splash needs direct, confident action—a rinse, a call for help, the right antidote.
The Commitment Behind Every Lesson
This group of safety habits shapes the work culture around any dangerous compound. Ammonium hydrogen sulfate punishes carelessness, but it rewards careful routine. For every task, I carry the memory of past close calls. They remind me that the real skill isn’t just knowing the science. It’s keeping people healthy enough to keep learning.
| Names | |
| Preferred IUPAC name | azanium hydrogen sulfate |
| Other names |
Ammonium bisulfate Ammonium hydrogen sulphate Ammonium acid sulfate Ammonium acid sulphate |
| Pronunciation | /əˈmoʊniəm ˈhaɪdrɪdʒən ˈsʌlfeɪt/ |
| Identifiers | |
| CAS Number | 7803-63-6 |
| Beilstein Reference | 1698915 |
| ChEBI | CHEBI:29940 |
| ChEMBL | CHEMBL1201579 |
| ChemSpider | 22816 |
| DrugBank | DB16471 |
| ECHA InfoCard | 100.028.266 |
| EC Number | 016-041-00-8 |
| Gmelin Reference | 1245 |
| KEGG | C13786 |
| MeSH | D000648 |
| PubChem CID | 222472 |
| RTECS number | BS9625000 |
| UNII | 5F8YO2D9DK |
| UN number | UN2967 |
| CompTox Dashboard (EPA) | DTXSID0041484 |
| Properties | |
| Chemical formula | NH4HSO4 |
| Molar mass | 115.13 g/mol |
| Appearance | White crystalline solid |
| Odor | odorless |
| Density | 1.78 g/cm³ |
| Solubility in water | soluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 0.76 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | '1.527' |
| Dipole moment | 3.4 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1178.4 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes serious eye damage. |
| Precautionary statements | P264, P270, P280, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3250 mg/kg (oral, rat) |
| NIOSH | WN4725000 |
| PEL (Permissible) | Not established |
| Related compounds | |
| Related compounds |
Ammonium sulfate Ammonium bisulfite Ammonium thiosulfate Ammonium sulfite |

