Unlocking the Story of Ammonium Hexanitrodiphenylamine
Historical Development
Looking through the pages of military science, synthetic chemistry, and safety engineering, ammonium hexanitrodiphenylamine, often called “Explosive D,” landed at a turning point in the early 20th century. Wars demanded more powerful and stable explosive materials, which led scientists to dig deeper into nitroaromatic compounds. Early research circled around improving on classical mixtures by finding explosives less sensitive to shocks but still potent. Ammonium hexanitrodiphenylamine quickly gained notoriety for these qualities. As a result, piles of papers and books from the world’s defense laboratories chronicle a substance that, while not as sexy as TNT, became reliable in cartridges and armor-piercing shells. The research wasn’t just academic curiosity. In times of world wars and shifting global landscapes, advancing this field could spell the difference between technological edge and obsolescence.
Product Overview
For anyone focused on energetic materials, ammonium hexanitrodiphenylamine sits on a short list of explosives that blended power with reliability. The chemical formula C12H5N13O12·NH4 sketches just a hint of its complexity. Scientists in the munitions industry liked it not only for its brisance but for how it handled day-to-day bumps — less prone to detonation from accidental jolts on the factory floor. Folks on production lines could breathe a tad easier when loading shells or mixing batches. Over the years, its real pull was this balancing act between punch and safety, helping to keep it relevant even as other technologies emerged.
Physical & Chemical Properties
Most people wouldn’t recognize ammonium hexanitrodiphenylamine on sight: it’s a yellowish or brownish crystalline powder, not flashy, not particularly memorable until you learn what it can do. It resists melting until it faces strong heat, and its dense chemical structure means it won’t dissolve in water, avoiding degradation from humidity in old storage bunkers. Its detonation velocity falls just shy of RDX, but better insensitivity gives handlers a little less to worry about. The compound’s high nitrogen and oxygen content primes it for rapid redox reactions during detonation, releasing considerable energy and a distinctive smoke signature. Over time, chemists began to measure it not just for explosive power, but for reliability in different weather and storage conditions, giving military planners more control over long campaigns.
Technical Specifications & Labeling
Anyone who walks into a storage magazine or an old defense stockroom sees heaps of regulations—and with good reason. Ammunium hexanitrodiphenylamine’s technical specs match those of most secondary explosives in terms of testing for sensitivity to heat, friction, and impact. Packaging needs tough, sealed drums. Proper marks include hazard symbols, UN codes, and shelf-life indicators per international destructive materials’ agreements. Specialists keep close track of batch numbers due to occasional shifts in purity between manufacturers, given that small contaminants can bump up sensitivity. These technical details aren’t red tape: they’re the direct result of decades of learning the hard way.
Preparation Method
Synthesizing ammonium hexanitrodiphenylamine isn’t backyard chemistry. Chemists usually start from diphenylamine, running it through staged nitration using concentrated nitric and sulfuric acids. The process requires slow additions, external cooling, and careful control of ratios to avoid runaway reactions. Adding ammonium salts pulls the desired explosive out as a precipitate from the acidic bath. This whole dance must follow strict protocols, as a small slip causes thermal runaway or dangerous byproducts. In historical labs, workers hand-stirred vats with face shields and maintained detailed logs. Modern plants bring automation and closed systems, but oversight remains constant to avoid catastrophic accidents.
Chemical Reactions & Modifications
People in energetic chemistry soon realized that small tweaks to ammonium hexanitrodiphenylamine’s structure shift its stability and power. Substituting ammonium for other cations, like sodium or potassium, periodically enters the literature, but ammonium’s stability remains a standout. Nitration depth can be tuned, yielding analogs with less or more nitro groups which then shift sensitivity and energy output. Researchers run compatibility tests to see if D can safely blend with other explosives or plasticizers, improving load flexibility. All these modifications come with a trade-off between handling safety and power, so old-timers in the field often say that sticking close to the original formula brings the least headaches on the production floor.
Synonyms & Product Names
A material this old travels with aliases. Some documents call it “Explosive D,” others stick to the mouthful of ammonium hexanitrodiphenylamine. Technical papers toss in “NHDA” or “Dipicrylamine ammonium salt.” Toxicology studies sometimes refer back to trade names that crop up only in dusty military archives. It takes a practiced hand to sift through reports and catch which label matches which material—an important step, because mixing up standards or hazard profiles could turn a routine delivery into a headline-grabbing accident.
Safety & Operational Standards
Safety doctrines around ammonium hexanitrodiphenylamine come after decades of too-close calls. Crews use remote handling where possible, grounded equipment, and tight limits on static discharge. Dust control matters since any airborne material raises the risk of accidental ignition. Regulatory bodies require environmental monitoring: vapors, water runoff, and even soil residues near old plants get sampled to prevent long-term exposure. Personnel training drills don’t let up, as mistakes linger in the community’s memory. Some nations demand tracking from synthesis through disposal, not just to prevent accidents but also to track proliferation in unstable regions.
Application Area
The main headlines for ammonium hexanitrodiphenylamine trace through heavy artillery, armor-piercing shells, and certain shaped-charge warheads. Its reputation as a secondary explosive grows from tough-to-detonate bulk that still responds strongly under intentional initiation. This allowed militaries to stuff larger shells without as much fear of cook-off in fires or from near-misses. In ammunition depots and old ordnance recyclers, D’s stubbornness to detonate from stray sparks or mechanical bumps turns potential disasters into near-misses. In some countries, it shows up as a specialty additive in mining or demolition, where reliable, predictable blasts matter. Over the years, civilian uses faded out, edged aside by environmental rules and better alternatives, but in defense stockpiles, it still fills a real need.
Research & Development
Lab notebooks and government grant summaries for explosive D stack high in the world’s scientific repositories. Ongoing experiments try to bind it in polymer matrices for safer handling, or tweak the synthesis to trim costs and waste. Environmental scientists scan for degradation products, exploring pathways that might make disposal safer and less polluting. Increasing attention in the past decade follows interest in legacy munitions cleanup and reversal of environmental impact near storage sites and training ranges. Even so, the pace of innovation slows as budgets tighten and new explosives like CL-20 or HNIW attract the spotlight—with higher power and (sometimes) safer handling profiles. Yet, specialist journals return to D for its unique spot between old-fashioned grit and modern sensitivity standards.
Toxicity Research
Long-term effects of ammonium hexanitrodiphenylamine’s residues in water and soil prompted closer looks as environmental regulations gained teeth. Animal studies and real-world sampling report low acute toxicity from simple skin contact but highlight possible chronic issues from inhaled dust or prolonged water exposure. Degradation yields several aromatic nitro compounds that resist easy breakdown, turning cleanup into a multigenerational task. Risk management often means strict workplace limits for air concentration and frequent health monitoring for workers, some of whom carry memories of colleagues sickened by careless past practices. Transparency in reporting occupational exposure has improved outcomes, with illness rates dropping as standards toughened across the industry.
Future Prospects
Looking ahead, ammonium hexanitrodiphenylamine stands at a crossroad. On one hand, global defense forces keep stocks as insurance, relying on battle-tested safety backed by solid statistics. On the other, material science pushes forward, searching for higher-performing, less polluting compounds. Governments invest in cleanup technologies and tighter controls, while niche research groups keep chasing modifications that recycle old D-based munitions into more benign substances. In some regions, the focus now lies with remediation, converting buried legacy waste into less-toxic forms. For all the talk of new energy carriers and green explosives, plenty of specialists still bookmark ammonium hexanitrodiphenylamine as a reliable workhorse until those futuristic chemistries finally clear the last hurdles of production, handling, and long-term safety.
What is Ammonium Hexanitrodiphenylamine used for?
History and Purpose
For many people, the name Ammonium Hexanitrodiphenylamine doesn’t roll off the tongue easily. Chemists know it more as “Explosophore” or by its bombastic abbreviation, HND. This chemical earned its place in history during the twentieth century, as research teams were looking for alternatives to TNT.
Anyone familiar with explosives or military history will tell you: scientists kept searching for new, better explosives, chasing that extra bit of safety or raw power. HND came up as one of these alternatives, poised to fill in as a primary charge or booster for munitions. It’s not your backyard chemistry set—manufacturing and handling require strict protocols.
Use in Explosives
HND shows up in the world of high explosives because it combines stability with brisance. It stands tough under rough storage conditions, where older explosives like picric acid could sweat or degrade. That kind of dependability made HND prized in detonators and boosters by military engineers. In some cases, manufacturers blended it with other compounds for specialty charges.
Unlike modern plasticizers or propellants that go into fireworks or small arms rounds, HND occupies a more serious spot. Think tank shells, demolition charges, and deep-mining applications. Its high detonation velocity gives it that needed force to initiate less sensitive explosives, making sure the main charge goes off as planned.
Health and Environmental Risks
Chemicals used in explosives rarely come risk-free. The toxicity of HND, both in handling and in environmental terms, raised flags over the years. Factory workers who produced or packed HND in past decades faced exposure. Happily, modern industry follows tighter safety standards. Full-body PPE, forced ventilation, and careful training cut down on dangerous accidents.
HND doesn’t belong in rivers, aquifers, or the global food chain, either. Research shows it can break down into toxic byproducts if left in the open or exposed to moisture. No surprise, then, that disposal and remediation efforts follow strict guidelines. Dump sites and leftover stockpiles sometimes haunt military testing grounds, creating headaches for local communities and governments trying to clean up contaminated land.
Why Alternatives Matter
In recent decades, alternative explosives gained traction, both for performance and environmental reasons. Military organizations in Europe and North America began switching to compounds that burn cleaner and pose fewer health risks. Each new chemical gets vetted hard: Does it pack the desired punch? Will it sit safely in barrels or shells under tough conditions? Can it be disposed of without polluting soil and water?
Switching to safer compounds comes from real lessons—the stories of explosions at munitions factories, survivors battling rare illnesses, and families coping with chemical spills. If new variants lower accident rates or ease the cleanup, they make their way into supply chains.
Perspective from Industry and Research
Having worked with hazard assessments, I notice push and pull between tradition and innovation. Old hands sometimes rely on legacy compounds because they know how they behave. Younger chemists, though, push for cleaner, more sustainable choices. This tension pushes research forward.
Nobody wants to see the mistakes of the past repeated. The challenge revolves around building something that keeps people safe—on the front lines and at home, at the test range and in the factory. For future generations, it makes sense to keep searching for solutions that leave less of a mark on health and on the world.
What are the safety precautions when handling Ammonium Hexanitrodiphenylamine?
Understanding What’s at Stake
Ammonium Hexanitrodiphenylamine isn’t one of those compounds sitting quietly on a shelf. Anyone who has stepped into a lab where energetic materials get mixed or stored feels the air change when someone mentions its name. This substance, with its long chemical title and history in explosives, demands respect far beyond reading labels and checking from a safety manual.
Why Gloves and Goggles Don’t Cover Everything
There’s no way around the fact that proper personal protective equipment forms the first line of defense. Nitrile or neoprene gloves mean business. Safety goggles close off the most vulnerable entry to the body. A lab coat—preferably antistatic—to minimize any chance of errant sparks from synthetic fibers. That’s just walking in the door.
Yet handling these chemicals asks for something deeper than gear: experience and a sharp focus on every movement. A single careless gesture—slamming down glassware or letting dust drift by—can ruin a whole day or worse. Years back, a senior chemist used to say that if your workspace looks neat, you reduce the risk of accidental mixing or spill. I’ve seen near misses traced back to clutter and distraction.
Controlling the Environment
Working with Ammonium Hexanitrodiphenylamine indoors asks for a good ventilation system and spark-free, grounded work surfaces. No sources of static or open flames allowed. In shared workplaces, grounding all tools and using explosion-proof electrical fittings beats rolling the dice with luck.
The general rule: store this compound in a cool, dry place, away from flammable substances and strong acids. There’s no reason for it to share company with anything that likes to start a chain reaction. Even sunlight has no business heating things up— keeping it in opaque, clearly labeled containers helps lower unplanned surprises.
Don’t Treat Spills Lightly
I remember a summer intern reaching for a paper towel during a small spill. Old-timers almost dove across the aisle to stop him. With Ammonium Hexanitrodiphenylamine, dry sweeping or using organic materials (like paper towels) can mean disaster. Spark-risk goes up fast. Instructing everyone to only use damp, non-sparking cloths and avoiding friction makes a world of difference.
Immediate isolation of the spill area works better than waiting for a supervisor. Alerting coworkers, ventilating, and keeping open flames far away usually keeps panic out of the equation. Clean-up should only start once everyone nearby understands the plan.
Preparation Cuts Down Mistakes
Complacency stands out as the silent enemy. From my experience, all staff, whether new or seasoned, benefit from drills on proper use and emergency response. Real scenarios beat theoretical training every time. Simple steps like double-checking that containers remain closed and free of residue, or making sure that hands get washed after use, go a long way.
Documentation and regular review of safety data sheets help to reinforce the limits and dangers. If something gets confusing, it's always best to ask questions and wait for clarity before touching the product.
Building a Safety Culture
People working with hazardous chemicals, especially energetic ones, often learn that trust is built every day, not just signed off in orientation. Respect for Ammonium Hexanitrodiphenylamine’s risks means everyone watches out for one another, double checks procedures, and walks away from shortcuts. Nothing matches looking out for the person working across the bench and expecting them to do the same.
In the end, taking these precautions seriously makes sure that both the material and those handling it go home safely. That’s where the real value of strict safety practices lies—protecting lives, not just ticking off a checklist.
What is the chemical formula of Ammonium Hexanitrodiphenylamine?
Beyond the Letters and Numbers
People often overlook the stories behind chemical names. Ammonium Hexanitrodiphenylamine doesn’t roll off the tongue, but its formula—C12H5N13O16—carries weight in the world of energetic materials. Back in college, it was the jawbreaker chemicals like this that got folks whispering about lab accidents, blackened table legs, and the unassuming power locked inside a few crystals.
What the Formula Really Means
C12H5N13O16: this isn’t just a collection of atoms. With twelve carbons, five hydrogens, thirteen nitrogens, and sixteen oxygens, it's built for a very specific purpose. That formula informs how it behaves—volatile, oxygen-rich, nitrogen-heavy. This compound made a name for itself in the military and explosives industry, known there as “HND” or “Erinit.” The large number of nitro groups signals instability and reactivity. That’s a double-edged sword.
Real-world Impact
As a student, I saw safety drills focused as much on ammonium hexanitrodiphenylamine as on the more notorious TNT. It pops up in textbooks as an ingredient in some World War II era munitions. The point of these chemicals isn’t just that they explode—their value comes from reliability and controlled detonation. Mishap rates drop when you use a compound with predictable chemistry. Soldiers and engineers don’t get that luxury with homemade mixes, as we saw in many tragic accidents.
Facts that Stand Out
- Ammonium hexanitrodiphenylamine packs more available oxygen than many legacy explosives. This is a trait chemists chase because it delivers a more complete blast, with less toxic by-products left behind.
- The high nitrogen content reduces the smoke and residue, an important factor in battlefield conditions where concealment matters almost as much as destructive power.
- Despite theoretical advantages, production requires tight temperature controls and skilled handling. Any slip—wrong solvent, static spark, poor storage—has consequences far beyond what most people imagine from a simple powder.
Why This Matters Now
Safety and sustainability—the world’s asking more of chemicals than ever before. Ammonium hexanitrodiphenylamine demonstrates how careful synthesis still can’t solve every challenge. Disposal practice troubles me most. Old stockpiles make headlines as environmental hazards, leaking nitrates and posing blast risks long after their intended use. Educational campaigns and tighter regulations help, but economic corners still get cut in low-budget operations.
Potential Paths Forward
Chemists work on greener explosives that mirror the power of compounds like HND while breaking down harmlessly after use. Tracking and accountability in every stage—synthesis, storage, disposal—must get stricter, even though that means more paperwork and less profit for some companies. Open-source data on incidents and improved safety equipment can save both lives and reputations.
As chemical formulas go, C12H5N13O16 isn’t just a bunch of elements—it's a signpost showing where safety, science, and real-world consequences meet. Every bottle stored in a dusty warehouse has a story, and often, a lesson behind it.
How should Ammonium Hexanitrodiphenylamine be stored?
The High Stakes of Storage
Ammonium hexanitrodiphenylamine isn’t the kind of chemical that gets forgiving if handled carelessly. Even the name flashes a warning in my mind, drawing memories from my work in both university labs and short stints at a munitions plant. Mishaps rarely end with just a mess on the floor. You face real risks—fire, explosion, toxic fumes—and they don’t hit in slow motion. In pressurized conversations with more seasoned colleagues, two themes always came up: treat energetic chemicals with respect, and never underestimate the environment in which they sit.
Official reports point to several high-profile storage accidents. In old Soviet stockpiles and wartime factories, improper storage of similar compounds led to catastrophic fires and injuries. Once, I assisted with an investigation following a minor blast; the lesson came down to neglecting placement and humidity control.
What Needs Locking Down
Direct sunlight heats things up fast, which can destabilize ammonium hexanitrodiphenylamine. Even small changes in temperature upset this compound. Nearby electrical equipment, soldering stations, or even a winter heater can set off a chain reaction. Every chemist I’ve met will tell you: keep it cool, keep it dark, let nobody improvise.
Humidity deserves special mention. Water vapor sparks slow breakdown, producing gases that build up inside containers. Pressure rises, seals burst, and, if you’re unlucky, there’s ignition. In one lab I worked at, an old air conditioning failure nearly caused a disaster because nobody noticed a slow climb in moisture. Only a routine check saved the inventory.
Get the Room Right, Get the Containers Right
Storing this chemical starts with location. Steel cabinets painted to resist corrosion, grounded floors, and isolating the chemical from other reactive materials—these steps never get skipped in my experience. Checklists matter. Thick plastic bins or specially lined containers give an added measure of protection. No cracked seals, no worn gaskets. All openings close tight, and containers get inspected before anyone signs off for the day.
Fire suppression is another reality check. Older buildings often rely on generic sprinkler systems, which fail with energetic compounds. Dry powder extinguishers and sand buckets work better. In my last facility, a full fire drill ran every month. Supervisors double-checked that nothing blocked cabinet doors or escape routes.
Training, Labels, and Security
Labeling compounds with clear warnings and date codes keeps confusion at bay. In one internship, my mentor demanded every team member review the paperwork before handling even a gram. Security adds another layer. Some compounds attract theft, sometimes for dangerous reasons. Industrial sites use sign-in sheets, camera monitoring, and badge access to prevent unauthorized touches.
Education sits at the core of safer storage. Every worker gets training—not a dry policy readout, but hands-on exams, walks through storage rooms, spotting what’s out of place. Enforcement isn’t about rules on a wall; it’s about repetition, reminders, and consequences for cutting corners.
The Role of Regulation and Technology
National and international chemical safety rules set minimum standards, but they don’t always keep pace with science or human error. Digital tracking, real-time environment monitoring, and smart alarm systems now bolster physical storage. I’ve seen these gadgets pay off—alarms catch minor leaks long before they turn dangerous.
Learning from the past, keeping vigilance in the present, and upgrading systems to match new threats form the backbone of safe ammonium hexanitrodiphenylamine storage. Lax handling puts lives and entire communities at risk. Caution, not habit, keeps everyone coming back in one piece after a shift.
Is Ammonium Hexanitrodiphenylamine explosive or hazardous?
What Ammonium Hexanitrodiphenylamine Really Means for Safety
Every so often, someone brings up an exotic-sounding chemical with a name so long it barely fits on a label: ammonium hexanitrodiphenylamine. The concern about safety jumps right to the front. This stuff falls under the umbrella of high explosives, a group well-known for risky business. From my time looking into chemical safety, one thing stays clear—chemicals that don’t look familiar usually get handled with extra care for a reason.
You find ammonium hexanitrodiphenylamine in certain propellant mixes and sometimes in military-grade explosives. It shows up because of its power—its explosive force runs high, which made it a favorite during research for new energetic materials in the mid-20th century. It breaks down in a way that lets off a lot of gas in an instant, so folks in defense R&D thought it could punch up the performance of old recipes.
The Risks in the Room
The moment a material ends up labeled as a high explosive, it gets grouped with other substances to treat with respect. Ammonium hexanitrodiphenylamine comes with a catch: it’s sensitive. Drop it, shock it, or expose it to too much heat and you’re rolling the dice. At a molecular level, its structure wants to break apart into smaller gases, and the energy packed inside doesn’t take kindly to rough handling. People sometimes think, "Maybe if you don’t light it on fire, it can’t hurt you." That’s wishful thinking here. Friction, static electricity, and even some kinds of impact have set off similar compounds.
Chemicals with nitrogen-rich structures or lots of nitro groups almost always call for careful storage and dedicated facilities. Workers in chemical plants handling these compounds suit up and follow strict rules for a reason. The last thing anyone wants is to wind up on the news because of a bang that rattled the street. It’s not just about workers—the risk follows the material wherever it travels, whether in trucks or storage bunkers.
Why We Should Care—And What Can Be Done
Hazards from high explosives don’t stay in one lane. Besides the potential for accidental detonations, ammonium hexanitrodiphenylamine could pose environmental and health risks. Many nitro-based compounds create toxic byproducts. Getting these substances into water or soil won’t do ecosystems any favors, which tells me public health must factor into every part of the equation, not just workplace safety.
Looking around, you see good solutions start with respect for protocols. Reliable training makes a difference. Regulations for storage are built from the ground up through lessons written in hard experience— you might remember headlines about fertilizer plants or factories that went wrong. Inspection schedules, proper temperature controls, and clear record-keeping all put a lid on risk. On a bigger scale, research keeps chugging away to find less dangerous alternatives whenever possible.
In my own work with hazardous materials, double-checking policies and double-gloving aren’t habits—they’re life insurance. Sometimes, the best move involves swapping out a risky ingredient for something less touchy. As scientists tinker with new ideas for propellants and explosives, the industry’s future might lean toward “greener” chemistry. Until then, ammonium hexanitrodiphenylamine stays a reminder: just because something promises power, doesn’t mean it comes without demands for care.
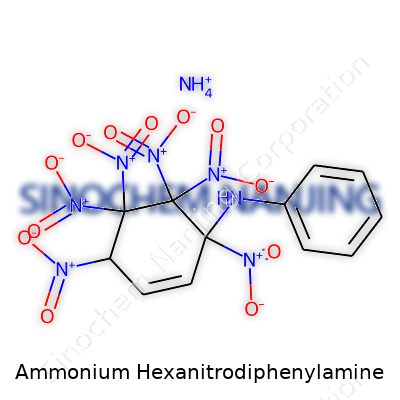
| Names | |
| Preferred IUPAC name | Ammonium 2,4,6-trinitro-N-(2,4,6-trinitrophenyl)anilinide |
| Other names |
Hexyl Hexogen HND Oktogen |
| Pronunciation | /əˈmoʊniəm ˌhɛksaɪˌnaɪtroʊˌdaɪfɪˈnɪlˌæmin/ |
| Identifiers | |
| CAS Number | 15862-58-7 |
| Beilstein Reference | 146881 |
| ChEBI | CHEBI:76267 |
| ChEMBL | CHEMBL1234812 |
| ChemSpider | 22553 |
| DrugBank | DB11435 |
| ECHA InfoCard | 03e6e6a7-7f09-4a82-bc43-850857b8b7cb |
| EC Number | 229-347-8 |
| Gmelin Reference | **79246** |
| KEGG | C18827 |
| MeSH | D000687 |
| PubChem CID | 22261 |
| RTECS number | JF0825000 |
| UNII | Q3JTX2Q7TU |
| UN number | UN0147 |
| CompTox Dashboard (EPA) | DTXSID5020158 |
| Properties | |
| Chemical formula | C12H12N10O12 |
| Molar mass | 438.16 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1.73 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -1.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.10 |
| Basicity (pKb) | 4.75 |
| Magnetic susceptibility (χ) | -58,0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.642 |
| Dipole moment | 2.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -87.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −2547 kJ/mol |
| Pharmacology | |
| ATC code | N05CM17 |
| Hazards | |
| Main hazards | Explosive, harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS03,GHS09 |
| Signal word | Danger |
| Hazard statements | H200: Unstable explosives. |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P404, P501 |
| NFPA 704 (fire diamond) | 3-3-1-OX |
| Autoignition temperature | > 290 °C (554 °F; 563 K) |
| Lethal dose or concentration | LD50 oral (rat) 175 mg/kg |
| LD50 (median dose) | 340 mg/kg (rat, oral) |
| NIOSH | RN136 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.01 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Hexanitrodiphenylamine Tetryl Ammonium Nitrate Nitroguanidine |

