Ammonium Dichromate: Insight Into a Controversial Industrial Chemical
Historical Development
Ammonium dichromate has roots reaching back into the earliest days of industrial chemistry. Discovered in the late 18th century, it came into broader use across photography, pyrotechnics, and laboratory education during the Industrial Revolution, thanks to its bold color and strong oxidizing power. By the early 20th century, the chemical's castle-orange hue and its ‘volcano’ reaction often amazed science students, but the industrial world cared less about spectacle and more about utility. As the demand for chromium compounds grew, methods of production scaled up. Factories turned out barrels of this oxidizer to fuel the needs of pigment production, metal finishing, lithography, and tanning. Over time, as awareness of worker safety and environmental hazards sharpened, regulatory pressure carved out new boundaries for its use and storage, reshaping its market presence but not erasing its historical footprint.
Product Overview
The compound comes as bright orange, sand-like crystals—impossible to mistake on a lab bench. Chemists use it because of its ability to release oxygen, especially in processes where strong oxidation counts. It finds its way into advanced photography, certain pyrotechnic mixtures, and some synthetic organic chemistry reactions. Despite those uses, major industries have shifted away from it because of safety and environmental risks. Companies still supply the chemical for specialized applications, academic research, and select manufacturing processes, but it no longer enjoys the widespread spread seen a few generations ago.
Physical & Chemical Properties
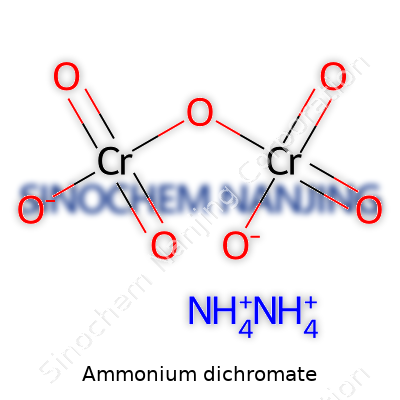
Ammonium dichromate’s chemical formula, (NH4)2Cr2O7, reflects its dual role as a source of both ammonium and powerful hexavalent chromium ions. At room temperature, it comes as hard, orange-red crystals with a melting point near 180°C, after which it swiftly decomposes. This decomposition doesn’t mean gentle breakdown; heat drives the formation of nitrogen gas, water vapor, and solid chromium(III) oxide, which results in the dramatic foaming “volcano” effect. Highly soluble in water, the compound remains stable in a cool, sealed container, but even minor contact with organic material or heat can touch off a fire. Large-scale users take care to account for its strong oxidizing abilities and store it well away from any combustible stock.
Technical Specifications & Labeling
Bags and bottles of ammonium dichromate carry warning labels that stand out from the pack. Labels point out the strong oxidizer classification, skin and respiratory hazard flags, and reminders of strict disposal regulations. Technical sheets provide purity figures—usually 98% or better among reputable suppliers—and particle size grading. They spell out clear storage guidance and shelf-life expectations, often giving a maximum storage temperature, compatible container materials, and recommendations for storing with spill containment. Transport regulations, influenced by GHS and UN codes, require durable, sealed packaging that resists both physical damage and moisture intrusion, with clear hazard diamonds on every pallet and drum.
Preparation Method
Production of ammonium dichromate generally starts with sodium dichromate, itself a byproduct of chromite ore and sodium carbonate. Manufacturers react sodium dichromate with ammonium sulfate under carefully controlled temperatures in aqueous solution. As the solution cools, ammonium dichromate forms and then drops out as striking orange crystals. Most production facilities recycle or treat spent mother liquors, aiming to reduce toxic wastewater output. Smaller-scale labs replicate the process in glassware, sometimes for demonstration or teaching purposes, though ventilation and gloves matter at every scale.
Chemical Reactions & Modifications
The most iconic reaction involving ammonium dichromate plays out in chemistry classrooms, where heating a pile of the crystals with a flame bursts forth clouds of green chromium(III) oxide and steam, imitating a tiny volcanic eruption. This reaction doesn’t just look dramatic—it points to the compound’s readiness to hand off oxygen to nearby substances, burning up most organics and reducing itself to less toxic chromium(III). In synthetic chemistry labs, it oxidizes alcohols and other organic molecules with precision, though other agents (like chromium trioxide or permanganate) have started replacing it because they are less hazardous. Chemists can adjust reaction conditions by adding acids, buffers, or co-catalysts to bust out subtle changes to efficiency or product outcome. Chromate-based reactions linger in photographic processes and organic syntheses that haven’t transitioned to greener approaches.
Synonyms & Product Names
In the business of chemicals, names can throw off buyers unless they read up. Ammonium dichromate goes by a list of alternative designations, including Vesuvian Fire, ammonium bichromate, and orange dichromate. Industrial catalogs tag it with registry numbers like CAS 7789-09-5. Certain suppliers shorthand the title as ADC or use “ammonium pyrochromate” for historical clarity. Regardless of the tag, every name points to the same bright-orange, hazardous oxidizer.
Safety & Operational Standards
Handling ammonium dichromate demands respect, not just compliance. Every form of contact—skin, eyes, or inhalation—proves dangerous. The hexavalent chromium in the mix tears up lung tissue, compromises kidney function, and carries a hefty cancer risk given long-term exposure. Operations need gloves, goggles, dedicated lab coats, dust masks, and fume hoods. In industry, strict environmental controls run alongside personal protective equipment: closed-system handling, certified ventilation, and strict lockout/tagout for maintenance. Waste streams with even trace amounts enter hazardous waste collection, not regular garbage. Regulatory bodies—OSHA, EPA, and their global equivalents—treat ammonium dichromate as a substance demanding regular training, incident tracking, and periodic safety audit. Emergency showers and eyewash stations remain non-negotiable at any facility working with this chemical.
Application Area
Despite sliding out of favor for general use, ammonium dichromate holds on in a few tight niches. Its powerful oxidizing properties still prove valuable in the synthesis of specialty organic chemicals and some inorganic chromates. In bygone decades, it was routine in photoengraving, fireworks (especially for vivid color and smoke effects), and early textile dyeing. Today, chemists pull the compound into reaction schemes where alternatives underperform, or for research on chromium chemistry. Universities limit its use to small-scale teaching—mostly under tightly controlled, one-time displays—highlighting both the science and the risk.
Research & Development
R&D around ammonium dichromate has moved in two directions. The first involves reimagining safer alternatives for oxidation, trying to move manufacturing and labs to less toxic, less persistent chemicals with similar effectiveness. Green chemistry labs worldwide explore catalysts and processes that keep chromium out of waste streams. The other direction sticks with refining how to contain and neutralize hexavalent chromium in industrial waste. Techniques such as chemical reduction, encapsulation, and advanced filtration all seek to cut the danger, both for workers and for people living downstream from production sites. Some academic groups also look for ways to repurpose spent oxidizers, reducing raw material demands and lessening hazardous output.
Toxicity Research
Decades of lab and epidemiological study hammer home the dangers of ammonium dichromate. Hexavalent chromium compounds storm across cell membranes and bind to DNA, triggering mutations and opening the door to multiple forms of cancer, especially in lungs and the upper airway. Animal studies connect chronic exposure to kidney and liver malfunction. Even a spill on bare skin can lead to painful ulcers or slow-healing wounds, sometimes dubbed “chrome holes” in older literature. Long-term occupational health studies back up these risks, highlighting the urgent need for airtight safety measures, regular health monitoring among users, and environmental cleanup at contaminated sites. Nations have responded by tightening permissible exposure limits, clamping down on improper disposal, and pressing for substitution by safer chemicals wherever possible.
Future Prospects
Pressure continues to mount for industries to leave behind chromium(VI) chemistry entirely. More chemical producers now offer green alternatives that replicate much of ammonium dichromate’s oxidative power without its legacy of harm. Regulators lean on these trends, rewriting approval guidelines and boosting incentives for innovation in both chemicals and containment technologies. For all its fire and color, ammonium dichromate has reached a crossroads: researchers now treat it as a relic in most mainstream applications, keeping it around for heritage demonstrations and very select industrial roles. The smart money backs research that promises the oxidizer’s power without the environmental or health bill, paving the way for safer, cleaner chemistry.
What are the main uses of Ammonium Dichromate?
Fireworks, Science Demos, and Lessons
Growing up, my first encounter with ammonium dichromate came during a chemistry lesson. Our teacher pulled out a small pile of orange crystals, set a match to it, and within seconds we watched as a spiky green “volcano” grew, sparked, and filled the lab with smoke. Behind its dramatic spark, ammonium dichromate played a role in many science demonstrations for decades. That reaction—the famous “ammonium dichromate volcano”—left more than a cloud of ash; it inspired curiosity about chemistry for generations of students. The effect relies on the compound’s ability to decompose rapidly, releasing nitrogen gas, water vapor, and chromium(III) oxide. Besides grabbing students’ attention, this property pushed its use in old-school pyrotechnics and early photography.
Photography and Lithography
Before digital, the story of photography ran through darkrooms filled with chemicals. Ammonium dichromate helped transfer images to plates, especially in a style known as “gum bichromate” printing. The compound reacts to light, turning soluble binders like gelatin or gum arabic into insoluble films where light strikes. This allowed photographers, especially artists chasing special effects or alternative techniques, to create complex images with their own hands. Printers also used this chemical in lithography, where it hardened the surface of printing plates in intricate patterns, enabling precise reproduction of artwork and text.
Industry in the Age of Safety
Factories once relied on ammonium dichromate for dyeing textiles, tanning leather, and acting as a strong oxidizer. Textile dyeing used it as a mordant—a fixer that locks powerful colors into fibers. Leather workers treated hides with chromium salts, including this one, to toughen and preserve them. Industrial research tapped into its oxidizing ability for creating pigments and jumpstarting reactions in organic chemistry labs, especially across the 20th century. At the same time, everywhere this salt appeared, the hazards weren’t far behind. Dust from ammonium dichromate poses real risks: it damages the lungs, touches off cancer concerns, and harms skin. After learning more about these effects, many companies turned to safer alternatives like less-toxic chromium compounds or iron salts, but some artists and craftspeople still use ammonium dichromate for its unique chemistry.
Handling the Hazards
The picture gets complicated with the health side of ammonium dichromate. Contact can burn the skin, breathing it can scar the lungs, and repeated exposure can cause cancer. These dangers forced schools to stop using it in classroom demos. Countries added strict rules about handling, storing, and disposing of it—good news for people and the planet, but it slowed down opportunities for hands-on chemistry lessons. Adults who use ammonium dichromate at work need gloves, masks, and plenty of ventilation. Art groups and labs stick to small batches, tightly controlled, to dodge the risks. The global push toward green chemistry continues to press for less hazardous alternatives, signaling the end of ammonium dichromate’s heyday outside of very specialized uses.
Looking Ahead
Old chemistry books still praise ammonium dichromate’s flexibility, but health facts changed the landscape. For photography, digital technologies now offer easier, safer methods. Science teachers steer toward safer demonstrations, using videos or innocuous substances to spark that same awe I felt in class. Industry and research have notched down reliance, searching for chemicals that do the job without the threat. This tough orange salt had a starring role in chemistry’s past, though today, its use reads more like a cautionary tale about balancing innovation with health.
Is Ammonium Dichromate hazardous or toxic?
Spotlight on a Dangerous Orange Crystal
Bright orange crystals like ammonium dichromate have always caught my eye in a lab—it’s hard to miss something that glows neon in a dish. Many people know it from high school "volcano" demonstrations: the pile of orange powder erupts like a miniature mountain, spewing ash and steam. There’s a thrill in seeing chemistry come to life, yet there’s real risk lurking in that spectacle.
Ammonium dichromate carries a heavy danger label for good reason. It releases toxic fumes, including chromium(VI) compounds, during decomposition. Any form of chromium(VI) raises red flags in terms of safety. My chemistry safety training pounded this into my mind: breathe in even a bit of this dust, and it can damage your lungs, liver, and kidneys. Ingesting it is much worse. Skin contact can cause ulcers or long-lasting burns, as hexavalent chromium works deep into tissue.
Beyond the Classroom: Real-World Impact
Chromium(VI) doesn’t just affect lab workers or students. This chemical leaks into the environment during industrial use and improper disposal. Cradle to grave, chromium(VI) can persist in soil and groundwater. There’s a direct link between exposure and increased cancer risk. The International Agency for Research on Cancer lists chromium(VI) compounds as known human carcinogens.
There was a period in the mid-20th century when safety precautions looked more like afterthoughts than real guidelines. I’ve met older lab techs who worked barehanded. Today, strict rules surround ammonium dichromate. Even small amounts demand gloves, goggles, and a hood. Used materials need proper hazardous waste disposal—not tossed in the trash or rinsed down the drain.
Protecting Ourselves and Our Planet
Some schools have ditched the volcano demonstration for good. It seems like a loss, but the long-term health risks call for safer alternatives. Teachers and hobbyists now reach for harmless compounds that give the same visual pop without the toxins.
Factories still use ammonium dichromate in limited roles: photography, textile dyes, or specialty synthesis. Big companies stay compliant by containing dust, filtering exhaust, and monitoring waste streams. Strong laws pressure companies to clean up after themselves. In Europe, the REACH regulation heavily restricts the use of hexavalent chromium; the United States has its own strict OSHA limits.
Alternatives exist for many processes. Green chemistry has become more than a slogan—it’s an actual movement. Researchers develop catalysts or dyes with little or no toxicity. This shift comes from people understanding what’s at stake. Environmental contamination is tough to reverse, and health effects last a lifetime.
Personal Responsibility in the Lab
I treat any chemical that can burn through skin and stain hands with suspicion. Tidy habits and good storage help. I double-check labels and always store ammonium dichromate in sealed containers, far from incompatible chemicals. If a spill happens, the entire area gets cleaned up with respiratory protection, regardless of how small it looks.
Curiosity and fascination draw people to science, but they shouldn’t come at the cost of safety. Anyone working with ammonium dichromate owes it to themselves and those nearby to respect its hazards and follow every precaution. Better to safeguard your health and the environment than put either at risk.
How should Ammonium Dichromate be stored safely?
Risk Lurks in the Bright Orange Bottle
Few chemicals attract attention in the lab quite like ammonium dichromate. That bright, striking powder commands respect, mostly because the risks jump out as soon as you read the label: toxic, oxidizing, and carcinogenic. Handling this stuff in college labs taught me early about respecting the power of chemistry. It’s not just a matter of storing it and walking away — a careless move could throw a wrench in safety and health.
Real Hazards Demand Real Planning
Ammonium dichromate does not forgive sloppy storage. Accident reports shed light on what goes wrong. Exposure to skin and inhalation leads to lasting harm. The vivid orange color does not hint at its invisible risks. Moisture triggers the slow build-up of toxic chromium compounds. Spilled powder can track into spaces you wish were untouched. The substance sits in the same league as some of the best-known cancer-causing agents. For those running labs or chemical supply rooms, overlooking these facts has never ended well.
Storage Tips Proven Over Time
Every instructor or occupational safety officer offers advice, though the best ideas are born from trial and error. Ventilation tops the list. I’ve seen rooms thick with chemical odors — that’s a red flag. Never store ammonium dichromate with flammable materials. Its oxidizing power turns an innocent pile of paper into a threat. If the label rubs off or wears away, confusion starts, so using clear, chemical-resistant labeling keeps everyone in the loop, even during a stressful emergency drill.
Getting temperature right makes a difference. Hot spots near radiators or sunny windows will challenge the integrity of the bottle and the powder. Store containers in cool, dry, locked cabinets dedicated to oxidizers. Add a couple of airtight bags. Not glamorous, but it blocks moisture and, by extension, those corrosive reactions. Corrosion-proof shelving protects from the accidental leaks that eat through paint and metal.
Personal Safety Never Runs Out of Style
Gloves, goggles, and lab coats pay for themselves in a single incident. Splashes and accidental dust clouds surprise even experienced workers. Emergency eye wash stations nearby guarantee a faster response. Washing hands thoroughly before stepping out or eating — a small break in routine once allowed traces to reach lunch tables. Those lessons don’t leave you.
The Role of Rules and Training
Laboratories that last take rules seriously. Documented standard operating procedures tell workers exactly what to do. Regular safety drills transform rules from theory into instinct. Labeling waste clearly and arranging for professional disposal beats the false economy of down-the-drain shortcuts. State and federal guidelines exist for good reason. Skipping one checklist item — a leaky bottle, an open cabinet, a missing glove — can carry lifelong consequences.
Smart Alternatives and Solutions
Not every experiment requires ammonium dichromate. Over the years, many labs swapped in safer alternatives. Universities now rotate through greener options for demonstration purposes. This wisdom comes from repeated exposure to hazards, not just regulations. Practicing substitution changes the story entirely for the people in the room.
What are the handling and disposal procedures for Ammonium Dichromate?
Recognizing the Real Hazards
Ammonium dichromate doesn’t get the same attention as mercury or cyanide, but its risks can cause just as much harm. This orange crystalline powder looks harmless until you remember it carries hexavalent chromium, one of the top carcinogens out there. Skin contact can lead to ulcers; inhaling the dust does severe damage to the lungs. Even brief exposure causes irritation in most people. I once worked in a university chemistry lab where this chemical never left the locked poison cabinet. We checked our gloves, lab coats, and goggles twice before going near it.
Using the Right Tools and Mindset
Handling this stuff takes more than a rush-job or quick hands. Wear gloves made for chemical resistance, such as nitrile or neoprene. Standard latex can degrade or let the dust through much faster. Eye protection isn’t optional. Goggles that seal, not just splash guards, give the best defense. Fume hoods aren’t just fancy furniture — they trap the dust and vapors before anyone breathes them. Closed shoes, lab coats, and even work aprons give protection against accidental spills.
Keeping the workspace tidy helps more than most people think. Ammonium dichromate crystals can spread as dust if left out, so only handle the chemical in small amounts, far from other chemicals, open flames, or heat. I learned to triple-check labeling and secure containers with screw tops, so nobody stumbles into trouble hunting for something else.
Spill Response: Fast Action, Not Panic
If a spill happens, treat it like a fire drill. Nobody should try to sweep up crystals or dust clouds with a brush or their own hands. Wet paper towels or HEPA-filtered vacuums do the trick, making sure not to create fine airborne particles. All spilled material, used cleaning supplies, gloves, and lab paper go straight into a separately marked hazardous waste bag — never the regular garbage bin. I remember one incident in college: a small crystal spill led to a full shutdown of the building’s lab for the afternoon. It took three people in full gear to clean up every trace.
Responsible Disposal: Not Down the Drain
Dumping ammonium dichromate into the sink is a short path to environmental disaster. Most municipal water systems can’t filter out hexavalent chromium with regular treatment. Any leftover chemical goes into properly labeled, leakproof containers, often with a solid absorbent if it’s a liquid mix or spilled slurry. I once talked to a waste management supervisor who said that keeping waste containers sealed until pickup saves hours of headache and stops leaks before they start.
Hazardous waste teams trained under environmental law handle the rest — including the final step where the chromium is reduced to a safer form before disposal. Anyone generating this waste needs proper documentation, following local and national laws (like RCRA rules in the US). Half of the EPA’s fines I read about come from labs or schools skipping paperwork or mixing wastes by mistake.
Fixing Missteps and Building a Culture of Safety
A strong safety routine shapes how a lab, school, or plant works with materials like ammonium dichromate. Annual training matters far more than written rules stashed in a binder. Safety officers or department heads should watch for shortcut habits and update equipment before it fails. Inspections with practical feedback, not just checkboxes, make all the difference. From firsthand experience, I know that a relaxed attitude about this chemical opens the door to injuries, fines, and sometimes far worse outcomes. Investing time and resources into training, secure storage, and waste management pays off in safety and peace of mind.
What is the chemical formula and physical appearance of Ammonium Dichromate?
The Basics Behind Ammonium Dichromate
Ammonium dichromate carries the chemical formula (NH4)2Cr2O7. Over the years, working in laboratories and teaching high school chemistry, I’ve come across this substance on more than one occasion. It usually shows up as a bright orange crystalline powder. That bold color stands out immediately in a lab full of white samples and beige chemicals, so there’s little chance of confusing it with other compounds.
A Substance With a Striking Look
There’s something almost theatrical about ammonium dichromate. Pour it into a shallow dish and the orange crystals catch the light. It has a bit of a glistening quality, almost like coarse sugar. If you’ve ever seen the famous “volcano demonstration,” that’s this substance in action. When ignited, it decomposes and produces a greenish ash and thick clouds of gas, so it goes from vibrant orange to dusty green in a matter of seconds. Seeing this change up close hits home how dramatic chemical reactions can get and why respecting proper safety procedures makes a difference.
Importance and Risks in Everyday Use
That flaming volcano isn’t just for show. Ammonium dichromate continues to be used as a source of chromium compounds, in pyrotechnics, and in laboratory settings. Over the last decade, regulations on this chemical have tightened. The main concern comes from the chromium(VI) it contains. Studies from both the Occupational Safety and Health Administration (OSHA) and the International Agency for Research on Cancer (IARC) identify chromium(VI) compounds as human carcinogens.
Just handling this chemical brings health risks like skin irritation, breathing difficulties, and cancer after long-term exposure. In my early teaching days, extra care meant sealed containers and fume hoods—no one wants students inhaling toxic dust or getting exposed through contact. Seeing how safety improved over the years provided a firsthand lesson on why transparency, clear labeling, and restricted access in schools and workplaces matter.
Solutions for Safer Chemistry
No shortage of alternatives exists for the classic volcano experiment now. Many educators use safer props or simulations. In industrial settings, companies shift toward replacing hexavalent chromium chemicals with less hazardous options. Across the globe, regulations like REACH in Europe and tighter U.S. EPA guidelines force manufacturers to phase out the most dangerous compounds wherever possible. Working with chemical safety teams, I’ve seen how risk assessments, material safety data sheets, and personal protective equipment protect workers in ways that didn’t exist a generation ago.
People still use ammonium dichromate in certain applications, but continuous education and innovation limit the risks. With regulations and growing awareness, exposure to hazardous chemicals like this no longer happens so casually. Watching that shift in real time, I believe that science and industry can find safer ways to achieve the same ends. Careful handling, regular training, and strict environmental controls make a real difference.
| Names | |
| Preferred IUPAC name | diammonium dichromate |
| Other names |
Potassium bichromate Dichromic acid ammonium salt Ammonium bichromate Ammonium pyrochromate |
| Pronunciation | /əˌmoʊniəm daɪˈkroʊmeɪt/ |
| Identifiers | |
| CAS Number | 7789-09-5 |
| Beilstein Reference | 3587150 |
| ChEBI | CHEBI:31206 |
| ChEMBL | CHEMBL1354 |
| ChemSpider | 53177 |
| DrugBank | DB14029 |
| ECHA InfoCard | ECHA InfoCard: 026-062-00-1 |
| EC Number | 232-143-1 |
| Gmelin Reference | 190665 |
| KEGG | C01459 |
| MeSH | D002876 |
| PubChem CID | 25515 |
| RTECS number | AH4025000 |
| UNII | 7V6W8QKK56 |
| UN number | UN1439 |
| Properties | |
| Chemical formula | (NH4)2Cr2O7 |
| Molar mass | 252.06 g/mol |
| Appearance | Orange-red crystals |
| Odor | Odorless |
| Density | 2.15 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.24 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.5 |
| Basicity (pKb) | 3.86 |
| Magnetic susceptibility (χ) | +1200·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.614 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.0 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -752.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -482 kJ/mol |
| Pharmacology | |
| ATC code | V09AX10 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes severe skin burns and eye damage, may cause cancer, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H272, H302, H312, H314, H317, H334, H341, H350, H360Df, H372, H410 |
| Precautionary statements | P261, P273, P280, P301+P312, P302+P352, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 3-1-3-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 125 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 125 mg/kg |
| NIOSH | MV2150000 |
| PEL (Permissible) | PEL: TWA 0.05 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit): 0.001 mg Cr(VI)/m³ (as 8-hour TWA) |
| IDLH (Immediate danger) | 15 mg Cr(VI)/m3 |
| Related compounds | |
| Related compounds |
Sodium dichromate Potassium dichromate Chromic acid Chromium trioxide Ammonium chromate |

