Ammonium Cerium Nitrate: A Closer Look at a Unique Chemical Compound
Historical Development
Chemists in the late 19th century found themselves fascinated with rare earth elements, and cerium stood out among them. The discovery of Ammonium Cerium Nitrate came after cerium first appeared in Sweden in 1803; scientists refined extraction techniques as industrial demand for rare earth metals grew. Many academic labs in Europe began using Ammonium Cerium Nitrate to probe oxidation reactions decades before industries picked up the process. By the mid-20th century, chemical companies improved the purity and yield of Ammonium Cerium Nitrate, using it in research and specialty chemistry. Today’s technical progress traces back to that early push for new oxidizers and separation processes, when postwar demand pressed researchers to reimagine how rare earth salts played a role in chemical synthesis.
Product Overview
Ammonium Cerium Nitrate carries a strong reputation among chemists as a robust oxidizing agent, showing up in both academic and industrial laboratories. Its deep red crystals set it apart visually, but the real story comes down to its versatility. Whether the goal involves organic synthesis, microscopy staining, or battery component manufacturing, Ammonium Cerium Nitrate holds value thanks to its stable properties and reliable results. Its distinct advantages—water solubility, room temperature stability, and ease of handling—have built strong demand in fields that require potent yet manageable oxidants, while its compatibility with other rare earth salts expands its reach into material science and analytical chemistry.
Physical & Chemical Properties
The bright orange-red hue of Ammonium Cerium Nitrate powder points to its cerium(IV) state, and this compound needs only moderate conditions to maintain stability. It dissolves easily in water, forming solutions that can be filtered or titrated without much trouble. Its molecular structure features cerium at the center surrounded by nitrate ions and ammonium, all working together to give the compound its oxidative strength. In my own lab work, drying and storing Ammonium Cerium Nitrate in an airtight container—shielded from light and moisture—has helped preserve its crystal form, keeping clumping and hydrolysis to a minimum over time.
Technical Specifications & Labeling
Users typically receive Ammonium Cerium Nitrate in sealed plastic containers, labeled clearly with the chemical name, molecular formula ((NH4)2Ce(NO3)6), net weight, and lot number for traceability. Purity often exceeds 99% for analytical grade material, and safety data sheets come packed with hazard warnings, storage instructions, and waste disposal guidance. Labels highlight the oxidizing nature, offering details like melting point, solubility, and recommended storage temperatures to keep users well informed about safe and proper handling.
Preparation Method
Producers start with cerium oxide extracted from monazite sands or bastnasite minerals. After dissolving cerium oxide in nitric acid, a selective oxidation step shifts cerium to its tetravalent state; ammonium nitrate then reacts in solution to form Ammonium Cerium Nitrate. The process demands careful control of temperature and acidity to avoid contamination by other rare earth ions or lower-valence cerium compounds. Once precipitation occurs, filtration and crystallization refine the product, yielding pure, high-grade crystals. Some labs even recycle spent solutions to recover unreacted cerium, reducing costs and environmental footprint.
Chemical Reactions & Modifications
Ammonium Cerium Nitrate grabs attention in organic chemistry labs where strong oxidizers drive otherwise sluggish reactions. The compound bumps up alcohols to aldehydes and ketones, or clears out organic contaminants from analytical samples. Its use in cerimetric titrations speaks to its accuracy and reproducibility. Researchers looking to tweak reactivity often substitute the ammonium ion with sodium or potassium, giving rise to closely related cerium(IV) nitrates suited for particular niche applications. Its reactivity extends into the development of catalysts, where cerium supports fuel cell development and the cleaning of exhaust gases in environmental technology.
Synonyms & Product Names
Across catalogs and research papers, chemists have coined several names for Ammonium Cerium Nitrate. Common synonyms include ammonium ceric nitrate, cerium ammonium nitrate, and CAN. Some product lines refer to it simply by its acronym, while international labels sometimes translate or abbreviate the chemical differently based on local language conventions or regulatory codes. In the global marketplace, this web of names sometimes creates confusion, but CAS number lookup services and supplier certifications help users match products with technical requirements.
Safety & Operational Standards
Anyone who’s handled Ammonium Cerium Nitrate can tell you the compound must be treated with care, especially given its oxidizing power. Direct contact with skin or eyes can cause irritation, and inhaling its dust proves harmful. Laboratories post prominent hazard symbols, provide gloves and goggles, and store it away from organic solvents to prevent unintended reactions. Regulatory agencies like OSHA and REACH define rules for shipment, labeling, and emergency treatment. Waste solutions, too, require neutralization or specialized disposal routes, preventing the release of harmful cerium or nitrates into the broader environment.
Application Area
Most organic chemists reach for Ammonium Cerium Nitrate while working on complex molecule synthesis, especially during challenging oxidation steps that demand selectivity and control. Mineralogists and materials scientists employ the compound in staining and etching procedures, using its unique reactions to reveal structural features under a microscope. Electronics industries depend on it during rare earth metal refinement, helping separate cerium from its lanthanide cousins. Environmental laboratories measure pollution levels with equipment that uses Ammonium Cerium Nitrate for redox titration of metal ions, providing insight into water and soil contamination.
Research & Development
Academic and industrial labs worldwide continue exploring how Ammonium Cerium Nitrate streamlines synthesis or drives greener technology. Hydrogen fuel cell research investigates new cerium-based catalysts to increase efficiency and durability. In my own university years, researchers tested different ammonium salts as precursors for luminescent nanomaterials, with Ammonium Cerium Nitrate standing out thanks to its clean reactivity and reliable supply chain. Ongoing R&D projects focus on improving purification strategies, recycling spent solutions, and searching for new analytical techniques that better harness cerium’s unique redox properties.
Toxicity Research
Though cerium compounds appear in nature, human exposure to high levels carries risks. Animal studies show kidney and liver effects with prolonged ingestion, while acute toxicity from inhalation or direct contact primarily affects mucous membranes and skin. Regulatory bodies closely review all available evidence, setting occupational exposure limits and restricting discharge into waterways. Many labs run detailed risk assessments and mandate training before working with the material—a practical safeguard based on experience, since errors often stem from poor planning or inattention to detail.
Future Prospects
Looking forward, Ammonium Cerium Nitrate stands poised for continued relevance as industries chase higher performance and sustainability. Advancements in rare earth recycling reduce both environmental impact and reliance on limited mining sources; improved safety protocols and real-time monitoring make handling less hazardous. Applications in advanced battery chemistries, fuel cells, and green oxidation methods promise new growth opportunities. Sharing knowledge through conferences and open-access journals brings diverse global teams together, building a foundation for smarter, safer, and more sustainable use of cerium chemicals in the years ahead.
What is Ammonium Cerium Nitrate used for?
The Chemistry Behind Everyday Breakthroughs
Ammonium cerium nitrate lives on the shelves of many chemical labs for good reason. Most people probably haven’t heard of it, yet its impact stretches far beyond the textbook. This compound serves as an oxidizing agent, which means it helps kick-start chemical changes in other substances—often by giving up oxygen or grabbing electrons. Anyone who’s spent time around a lab bench knows the transformation power of a good oxidizer. Ammonium cerium nitrate steps up when something needs transforming without letting things get out of control.
Fine Chemicals and the Push for Precision
Industries rely on ammonium cerium nitrate to support selective chemical reactions. Take organic synthesis, for example. Pharmaceutical chemists prefer tools that don’t make a mess of delicate molecules since they need the pieces to snap together in just the right way. Here, ammonium cerium nitrate brings finesse, helping to build complex structures—from new drugs to future agrochemicals—one small step at a time. That kind of control doesn’t come easy. Doctors and patients see the payoff when a drug works without too many side effects.
Lights, Camera, Cerium
It’s not all about medicine and chemistry sets, either. The electronics world looks different thanks to ammonium cerium nitrate. Engineers use it for photoengraving, which builds the tiny masterpieces inside circuit boards. The compound helps shape microscopic patterns, letting manufacturers paint fine details onto silicon chips and glass. These patterns run inside everything from smartphones to MRI scanners. Without this kind of chemical, shrinking electronics might grind to a halt. These innovations don’t just make gadgets smaller—they also spur everything from better medical imaging to faster computers.
Cleaning Up Stubborn Messes
Sometimes you see ammonium cerium nitrate cleaning glass in laboratories. Lab glassware can gather residue you can’t scrub away with soap and water. Dirt and organic stains come off more quickly if you dunk them in a solution containing this compound. Nobody wants cross-contamination during an experiment—the results can mislead researchers or waste time. I've worked with glass cleaned this way; you notice the difference when you handle delicate measurements and sensitive samples. A streak-free beaker keeps the next experiment honest.
The Environmental and Safety Angle
Handling any strong oxidizer, including ammonium cerium nitrate, means respecting the risks. Exposure can cause harm through inhalation or skin contact. Accidents don’t just stay at the bench. A chemical like this, if mishandled, can trigger spills or reactions with other substances. Safety practices rely on real-world knowledge: eye protection, proper ventilation, and responsible disposal methods protect people and the environment. Factories must pay attention to these details—regulators and workers count on it.
Looking Ahead: Opportunities and Better Practices
Emerging research pushes for greener chemistry. Scientists now look for ways to recycle oxidizing agents and reduce hazardous byproducts. Some teams use ammonium cerium nitrate in catalytic methods so that it works over and over instead of winding up as waste. Such ideas matter because they save money and cut down on pollution. I’ve seen university labs wrestle with both cost and cleanup. Passing down safer habits and smarter use of chemicals builds trust in products people use every day—and keeps science moving forward.
What is the chemical formula of Ammonium Cerium Nitrate?
Understanding Ammonium Cerium Nitrate at a Glance
Anyone curious about chemistry, especially those tinkering in labs or supporting manufacturing, runs into chemicals like Ammonium Cerium Nitrate now and then. This bright orange compound goes by the chemical formula (NH4)2[Ce(NO3)6]. It's not some rare curiosity reserved for academic articles. In fact, almost every chemistry teacher or industrial lab tech has handled it at some point.
What Makes This Compound Special?
Ammonium Cerium Nitrate stands out for its strong oxidizing power. The cerium atom here holds a +4 oxidation state, which means a lot for anyone doing work that involves electron transfer. This oxidizing ability gets a workout in organic chemistry, analytical testing, even in the etching of precision glass. For someone running an experiment, using the wrong formula can tank results and sometimes put safety at risk. Knowing this chemical's identity—exactly what elements it brings to the table—isn't just trivia. It's vital for accurate measurements and safe handling.
Real-World Problems Tied to Knowledge Gaps
Easy to imagine how a mix-up might happen: a new research assistant might think any yellow-orange crystal in the cupboard is Ammonium Cerium Nitrate, but certainty only comes from matching the formula. I’ve seen seasoned chemists double-check because a small substitution—say, using Sodium instead of Ammonium—changes solubility, reactivity, even the safety profile.
This particular compound often plays a role in syntheses where precision isn’t a nice-to-have, but a requirement. Manufacturing teams working with specialty glass lean on it for etching patterns into surfaces like microscope slides, camera lenses, and scientific glassware. The ammonium part of the formula isn’t a passive piece—it impacts how the compound dissolves, what byproducts get left behind, and how waste disposal needs to be managed. My own experience around high school lab benches taught me early that chemical spills and mishandling rarely end well, especially with nitrates. Clean-up can blossom into a whole afternoon’s work if even minor mistakes happen.
Implications for Safety and the Environment
Nitrates don’t just disappear down the drain without causing ripples. Runoff carries a risk of promoting algae growth in waterways—a process called eutrophication that chokes out fish and upsets ecosystems. Cerium compounds, used without care, add up over time, especially in older facilities with questionable disposal routines. Regularly updating staff on safe procedures, and investing a little extra effort in tracking chemical inventories, helps nip these problems in the bud.
Practical Solutions from the Ground Up
Training stands out as the real game-changer. I’ve found the best labs—even the ones running out of a high school basement—spend time walking new folks through every common reagent, including Ammonium Cerium Nitrate. Putting chemical formulas, common uses, and up-to-date material safety sheets front and center on shelves cuts down on confusion.
Some bigger operations install digital inventory systems, flagging whenever someone checks out a bottle. Simple? Yes, but it stops a lot of mix-ups. Regular waste audits, plus good habits around labeling, pay off in fewer accidents and smoother audits.
Getting Details Right Builds Trust—and Results
So much in science and manufacturing comes down to knowing the basics backward and forward. The formula (NH4)2[Ce(NO3)6] isn’t just something to write on a quiz, it’s a ticket to successful experiments, safe workplaces, and cleaner communities. My years in the lab have shown me that getting the details right—starting with the formula—sets up everyone for better outcomes.
Is Ammonium Cerium Nitrate hazardous or toxic?
Why This Compound Cuts Both Ways
Ammonium cerium nitrate does not make headlines in everyday life, but chemists and industries rely on it for strong oxidation reactions and unique lab work. The orange-red crystals make certain chemical transformations possible, from organic synthesis to photoengraving. If you have never worked with it, you might wonder whether it is really as hazardous as safety sheets suggest or if some of the warnings are hype.
Direct Health Risks Stand Out
Coming in contact with ammonium cerium nitrate brings a sting you don’t forget. During grad school, a few drops splashed near my glove cuffs, reddening my skin in minutes. Skin and eye exposure often lead to irritation. The real worry settles in on inhalation or ingestion—both open a door to nausea, headaches, or even damage to internal organs. Many sources confirm these points: both the cerium and the nitrate pieces are involved in the toxicity. Nitrates can convert to nitrites in the body and shake up oxygen transport, causing headaches or more dangerous symptoms if exposure is high. Cerium salts, less common in daily life, gather in the liver and lungs after long-term exposure. For those working in cramped labs or production lines, repeated handling builds risk.
Environmental Risks: What Happens After Discarding?
Dumping solutions or crystals down the sink brings its own set of problems. Cerium, like most rare earths, can disrupt aquatic ecosystems. Fish and smaller water species show signs of stress or reproductive issues after cerium exposure. While the world pays more attention to mercury, the slow creep of rare earths deserves more eyes. The nitrate ions feed rapid algae growth, robbing water of oxygen and tipping the balance for entire ecosystems.
Ways To Work Safer
Every chemist knows to glove up, wear protective glasses, and run reactions in a fume hood. For ammonium cerium nitrate, these habits become mandatory routines. Ventilation stands at the center of safe practice. Watch out for powder spills—once airborne, the dust can enter both eyes and lungs in seconds. Some companies invest in automatic dosing machines or extra-long pipettes. That hands-off approach keeps skin farther away from the crystals and protects airways. Rinsing any exposed skin immediately trims reaction time, but no one wants to end up at that step.
Better Storage Means Fewer Surprises
Moisture brings out the worst in ammonium cerium nitrate. Crystals pick up water fast, turning sticky and hard to handle. In the lab, sealed containers lined with desiccant powders cut down on risk. Labeling and inventory checks seem dull, but they save headaches when students or co-workers cycle through the benches. Everyone finds what they need—without nasty surprises from old, leaking bottles.
Handling Waste Responsibly
Pouring leftover solutions down the sink is not a shortcut anyone wants to take. Waste buckets labeled for oxides and oxidizers make recycling and neutralization possible. Working with a disposal company means this chemical does not slip into community water tables. Most universities have training every year, so new workers know how to file disposal tags and call for pickups. Industry can go another step by installing water treatment for effluent lines or switching to greener oxidants if the synthesis allows.
Takeaways from the Real World
No one working with ammonium cerium nitrate would call it harmless. The hazards show up both in single spills and slow leaks. Training, engineering controls, and accountability matter more than any regulatory checklist. For anyone building procedures around this compound, a smart mix of vigilance and forward-thinking design saves both employees and the environment from unnecessary harm.
How should Ammonium Cerium Nitrate be stored?
Recognizing Risks in Everyday Labs
Ammonium cerium nitrate works as a strong oxidizer. That means this compound reacts with common materials like paper, solvents, and even some metals. Even without a big background in chemical safety, anyone who spent time in a high school or university lab has likely heard stories of small accidents—maybe spilled acid, mislabeling, or a forgotten bottle. Substances like ammonium cerium nitrate deserve more attention because their risks carry bigger consequences. Once, I saw a bottle stored too close to a pile of rags; there was no problem that day, but the danger felt real. Simple mistakes can do serious harm if this chemical is handled without respect for its properties.
Getting Storage Right: Practical Steps
To prevent problems, I always keep strong oxidizers like ammonium cerium nitrate in a cool, dry cabinet away from sunlight, with the temperature staying as stable as possible. Labs often use chemical storage cabinets with strong ventilation because they vent any built-up fumes and reduce fire risk. If someone stores this chemical next to flammable solvents or combustible organic materials, a small spark or spill could start a fire. Every instructor or lab manager I know checks their inventory for this exact risk during monthly safety reviews.
Humidity matters. Damp air can make ammonium cerium nitrate clump or degrade over time. I saw one storage room in an old building where moisture from a leaky pipe caused labels to peel and chemicals to get sticky, leading to wasted money and time. Diligent storage means checking seals, using desiccators if needed, and writing the last inspection date on bottles. A dry environment not only protects the chemical but also keeps storage areas safe for everyone working there.
Labeling and Limiting Access
Clear labeling helps everyone in a workspace know what they are handling. Labels should list the substance name, the date it arrived, and hazard symbols. During routine lab tasks, fatigue or stress sometimes cause people to grab the wrong bottle. Labels cut confusion, adding one more barrier between a regular day and a dangerous accident.
Only trained team members should handle or move ammonium cerium nitrate. This isn’t gatekeeping or making life difficult—it’s investing in safety. Training covers how to clean up a spill, what to do if skin contact happens, and how to use fire extinguishers suitable for oxidizing chemicals. A locked storage cabinet, secured at all times, adds another layer of protection. Every chemist I know has a story about an intern or visitor poking around where they shouldn’t, which shows the value of a good lock and thoughtful policies.
Preparing for the Unexpected
It’s easy to get complacent if nothing dramatic ever happens. Still, planning for emergencies feels like common sense. Spill kits belong near storage areas, stocked with non-combustible absorbents. Fire extinguishers rated for chemical fires should hang nearby, not just in unsafe corners. Emergency phones or alarms keep help close at hand, just in case a reaction gets out of control.
I’ve seen experienced researchers run through emergency drills, often catching small details others missed. Practicing these scenarios makes the difference between a manageable cleanup and a disaster. Keeping careful logs—dates opened, who handled what, and storage checks—also builds the culture of shared responsibility that keeps labs running safely.
Moving Forward with Responsibility
Safe storage for ammonium cerium nitrate means more than following a checklist. It’s about understanding how a single chemical links to the well-being of colleagues and the success of research. Every small change—better labeling, keeping cabinets dry, limiting who handles dangerous materials—builds a safer workplace. Sharing stories and lessons learned keeps new users alert to hazards and ensures fewer mistakes happen. Each time someone sets up a safe storage space, they help science move ahead without the fallout from avoidable accidents.
What are the safety precautions when handling Ammonium Cerium Nitrate?
Why Extra Care Matters
No one loves paperwork after an accident in the lab. Few things end a productive week faster than a chemical burn or lung scare. Ammonium cerium nitrate doesn’t always look dangerous, but it carries risks that should keep any scientist on their toes. This powerful oxidizer can easily react in ways that surprise even seasoned researchers, especially when mixed with organic compounds or exposed to the wrong materials.
Personal Experience: Small Mistakes Make Big Problems
One semester, a colleague stored a tiny bottle near a heat source. It didn’t take long before the entire cabinet smelled off. Turns out, even moderate warmth led to gradual decomposition, which fouled our whole air system for a week. Up close, skin irritation caught me off guard more than once during grad school. Glove breaks and bare wrists never seem like a big deal in the moment. They turn painful fast when dealing with ammonium cerium nitrate.
Key Precautions in Day-to-Day Use
Solid rules give peace of mind in unpredictable situations. Start with eye protection. Splashes and accidental aerosols make goggles standard gear, not optional. Nitrile gloves beat latex every time for this compound. Lab coats should cover sleeves, and no one should skip a thorough wash after finishing up.
Keep this chemical separate from anything flammable or easily oxidized. Even a tiny spill on a messy benchtop can trigger a fire. Planning the day’s work before opening the bottle saves more trouble than any other step. Knowing where to place waste, how to clean up, and which neutralizer works best keeps panicked sprints to the eyewash station rare.
Ventilation and Storage Keep Trouble Low
Speak to any lab manager who’s handled ammonium cerium nitrate for years, and you’ll hear stories about stealthy fumes. Fume hoods aren’t overrated in this case; they keep trace vapors away from lungs and reduce the risk of accidents. I’ve watched experienced chemists handle weighing under clunky acrylic shields, simply because a few stray crystals react so unpredictably when exposed to the tiniest bit of water or unintended heat.
Store the bottle in a dry, cool, and well-ventilated cabinet. Materials like wood and cardboard don’t mix with strong oxidizers. Metal cabinets, clearly labeled and away from acids or bases, give extra protection if something leaks or breaks. Inventory checks each month catch small cracks or clumps that point to stability problems down the line.
Waste Disposal: Not an Afterthought
Don’t toss leftovers or spills into a regular trash bin or down the drain. Collect all waste in a clearly labeled, dedicated container and work with the hazardous waste handler at your site. Some labs keep a spill kit on the counter, complete with absorbent pads, neutralizer, and instructions for dealing with the worst-case scenario. Quick response reduces both mess and exposure.
Better Training and Culture
Training and reminders from supervisors make a big difference. No one stays alert on instinct alone all the time. Regular refresher courses and an open-door policy for safety questions drop accident rates. Peer checklists before and after using chemicals like ammonium cerium nitrate sound tedious, but building these habits into the daily workflow protects both new researchers and seasoned veterans.
Putting real experience ahead of shortcuts forms the backbone of safe chemical work. Reliable procedures, a healthy respect for risk, and sharing horror stories openly all push safety from something we read in manuals to something that guides every hand in the lab.
| Names | |
| Preferred IUPAC name | ammonium tetranitrate cerate(IV) |
| Other names |
Ammonium Cerium(IV) Nitrate Ceric Ammonium Nitrate CAN Cerium Ammonium Nitrate |
| Pronunciation | /əˈmoʊniəm ˈsɪəriəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 16774-21-3 |
| Beilstein Reference | 17115 |
| ChEBI | CHEBI:78033 |
| ChEMBL | CHEMBL1201807 |
| ChemSpider | 64952 |
| DrugBank | DB14532 |
| ECHA InfoCard | ECHA InfoCard: 100.004.306 |
| EC Number | 234-742-3 |
| Gmelin Reference | 10985 |
| KEGG | C18680 |
| MeSH | D002523 |
| PubChem CID | 24858256 |
| RTECS number | GF9590000 |
| UNII | DX6BXX38QF |
| UN number | UN1477 |
| Properties | |
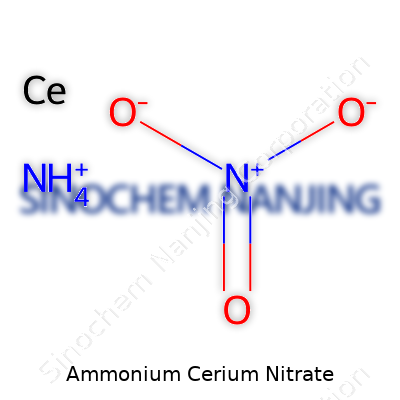
| Chemical formula | (NH4)2Ce(NO3)6 |
| Molar mass | 548.22 g/mol |
| Appearance | Red-orange crystals |
| Odor | Odorless |
| Density | 1.68 g/cm³ |
| Solubility in water | soluble |
| log P | -4.5 |
| Vapor pressure | Negligible |
| Basicity (pKb) | pKb: 4.0 |
| Magnetic susceptibility (χ) | +72.0e-6 cm³/mol |
| Refractive index (nD) | 1.505 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 254 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye damage, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS07, GHS08, GHS09 |
| Pictograms | GHS01, GHS03, GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H272, H302, H315, H319, H335, H410 |
| Precautionary statements | Precautionary statements: "P210, P220, P221, P264, P280, P301+P312, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-3-OX |
| Autoignition temperature | > 220 °C (428 °F; 493 K) |
| Lethal dose or concentration | LD50 (oral, rat): 850 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 5,500 mg/kg |
| NIOSH | Not Listed |
| PEL (Permissible) | No OSHA PEL established. |
| REL (Recommended) | fume hood |
| Related compounds | |
| Related compounds |
Ceric ammonium sulfate Cerium(IV) sulfate Cerium(IV) oxide Cerium(IV) chloride Cerium(III) nitrate |

