Ammonium Carbamate: The Hidden Backbone of Industrial Chemistry
Historical Development
Ammonium carbamate often flies under the radar in conversations about important chemical compounds, but its story traces back to the era when modern chemistry began taking shape. In the nineteenth century, scientists chipped away at mysteries by piecing together how ammonia, carbon dioxide, and related compounds behaved. Ammonium carbamate emerged in those studies, discovered as both a natural by-product and a stepping stone to other valuable chemicals. For decades, its intermittent appearance in laboratory glassware made it more of a curiosity, but its value grew rapidly with the scaling of nitrogen-based industries. The ramp-up in fertilizer demand after World War II drew more attention toward the compound, as industrial processes required intermediates that could bridge the gap between raw ammonia and finished urea. This compound’s rise isn’t a tale of overnight innovation but rather a slow realization that its properties serve a practical purpose.
Product Overview
Anyone who works with chemistry or agriculture probably sees ammonium carbamate as a powdery, sometimes crystalline substance—usually white, sometimes faintly colored by impurities. In industry, it sits in the shadow of names like urea or ammonium nitrate, but its utility extends into making those big players. Hardly anyone outside of chemical plants notices it, because it rarely leaves the factory floor in its pure form, but that doesn't diminish its role. As a fertilizer precursor, it helps sustain the food supply chain by quietly feeding into larger processes.
Physical & Chemical Properties
You can think of ammonium carbamate as unpretentious but reliable—soluble in water, able to decompose with a gentle nudge from heat or by just sitting out in the open. At room temperature, it’s stable enough if kept dry, but given any moisture or warmth, it starts breaking down into carbon dioxide and ammonia gas. The compound’s molecular dance is straightforward: one molecule of ammonia pairs with carbon dioxide to form it, and the reverse can easily happen. This see-sawing behavior makes it both a challenge and an opportunity for chemical engineers. Keeping it packed away in airtight containers is crucial, or else operators dealing with it risk it slowly evaporating away without much fuss.
Technical Specifications & Labeling
Regulators and suppliers rarely find themselves writing out long, elaborate specification sheets for pure ammonium carbamate because most of it doesn’t travel far from where it’s made. Still, clarity matters. The standard commercial product should contain a high percentage of the compound itself, with moisture and impurities like free ammonia kept to a minimum. Operators need to know what they’ve got, so clear labeling with chemical formulas and hazard warnings becomes non-negotiable. The most important details center on dryness, purity, and storage conditions, since overlooked moisture leads to product loss and safety risks.
Preparation Method
Manufacturing ammonium carbamate doesn’t require rocket science, but it does reward careful control. Factories typically react anhydrous ammonia with carbon dioxide, often under pressure and lower temperatures, to yield the white solid. The reaction happens fast but doesn’t reach perfect completion, so technicians cycle gases, collect the product, filter out water, and sometimes re-combine what’s left unreacted. There’s a fine balancing act between squeezing out enough yield and keeping the process affordable. In my experience, process engineers seldom get the luxury of textbook conditions—they juggle temperature, pressure, and flow rates with an eye toward what really works on the ground.
Chemical Reactions & Modifications
If someone asks what ammonium carbamate’s main chemical trick is, the answer is its ability to slip easily back into ammonia and carbon dioxide. That trait gets used in making urea, where ammonium carbamate forms the critical bridge between raw materials and the end product. Heat it a bit and you release gases, cool and combine, and you’re back at solid state again. It also stands out as a source of ammonia in other syntheses—its breakdown provides a steady stream of gas for further reactions. Modifications don’t extend far, since chemists value its simplicity, but in complex multi-step setups, being able to shunt between its different chemical forms opens up flexibility.
Synonyms & Product Names
Science often revels in having too many names for the same thing, and ammonium carbamate proves that point. Old texts and supplier catalogs might call it "ammonium carbamic acid" or just abbreviate it as "NH2COONH4" based on chemical shorthand. These alternative names dot patents and journal articles, so mixing them up can trip up the unwary researcher.
Safety & Operational Standards
No one should gloss over safety with ammonium carbamate, even if it doesn’t pack the dramatic hazards of stronger chemicals. Long-term exposure to ammonia and carbon dioxide fumes—what you end up with if the product breaks down—can irritate the lungs and eyes, sometimes badly. Tight handling protocols keep product dry and limit airborne dust. Ventilation becomes the frontline defense in production areas, and staff use gloves and goggles as a routine part of the job. Plant engineers set up gas detection and train teams relentlessly, since a little negligence leads to chronic issues or sudden releases. Over the years, I’ve seen how a strong safety culture pays dividends, especially during maintenance or unexpected cleanups. Regulatory agencies don’t let up, either, setting maximum exposure limits and calling for regular safety reviews.
Application Area
Farmers and food producers largely benefit downstream from ammonium carbamate, though they may not recognize the name. The stuff rarely sits on store shelves, but it serves as a linchpin in nitrogen fertilizer production, feeding straight into the manufacture of urea. Synthetic chemistry research leans on it as a testbed for developing new ammonia- or carbamate-based products. Some niche uses have cropped up in pharmacy and as a blowing agent, but bulk use stays rooted in the fertilizer supply chain. The tightrope walk of manufacturing efficiency depends on having this compound behave predictably in reaction vessels, which keeps costs stable and the market supplied. Research plants sometimes use it for controlled studies that need a slow-ammonia-release source.
Research & Development
Recent years have brought a wave of innovation in how chemists approach compounds like ammonium carbamate. Researchers want to tighten the efficiency of urea plants further, minimizing energy loss and waste. Some teams are experimenting with catalysts that coax the formation and breakdown of ammonium carbamate at gentler temperatures, or that tweak yields under industrial conditions. More advanced sensors and process algorithms keep tabs on how much is present at any stage, helping bring predictability. I’ve watched grant-funded labs search for new uses, whether in environmental science or cleaner chemical syntheses. Still, most real impact lands in improving fertilizer manufacturing by shaving off a few percent in carbon emissions or boosting throughput.
Toxicity Research
Toxicity data on ammonium carbamate helps set workplace safety levels and guides emergency response. The breakdown gases—ammonia and carbon dioxide—get most scrutiny due to their established risks. Ammonia exposure causes immediate irritation at low levels and more severe lung problems higher up. Ammonium carbamate itself doesn't linger long in the body or environment, but accidental release exposes handlers to those harmful by-products. Animal studies set benchmarks and safety guidelines, but for most people, real risk comes from inhaling dust or fumes over time. Training, monitoring, and strict maintenance of storage conditions do more to keep people safe than short-term fixes. As far as broader environmental harm, the compound breaks down so fast it doesn’t usually build up, but bad practices in disposal or storage can nudge local air or water quality in the wrong direction.
Future Prospects
Looking at where ammonium carbamate fits in tomorrow’s world, a few trends stand out. The pressure is on to make fertilizer production greener and less wasteful, so even mid-step intermediates like this compound face scrutiny. Companies are retooling plants and pushing for closed-loop systems to trap and reuse as much material as possible. Researchers chase catalysts and new reactor designs to wring out every bit of efficiency, not just to cut costs but to lower environmental impact. New process controls give a more precise handle on how ammonium carbamate behaves in real time, promising steadier yields and quicker troubleshooting. A wildcard remains the steady rise of alternative nitrogen sources—if new green ammonia or biological processes catch on and scale up, the role of compounds like ammonium carbamate could shift again. For now, though, its mix of reliability and reactivity keeps it right in the thick of the chemical industry’s daily work.
What are the main uses of Ammonium Carbamate?
The Backbone of Modern Fertilizers
Step onto any commercial farm, odds are you’ll bump into the impact of ammonium carbamate. At its core, this compound helps supply nitrogen to the soil—a nutrient that pretty much keeps crops alive. Farmers face pressure to meet the demands of a growing population, and fields must deliver high yields year after year. Adding nitrogen helps plants grow fast and strong, and ammonium carbamate often serves as a building block for creating urea, which makes up a huge percentage of the world’s fertilizers.
Why does ammonium carbamate matter so much? It acts as an essential intermediate in the chemical process that turns ammonia and carbon dioxide, both plentiful, into urea. Urea’s popularity doesn’t surprise anyone who has watched harvest tally up, thanks to its efficiency at getting nitrogen into soils. Without this middleman, many fertilizer plants would grind to a halt.
Supporting Chemical Manufacturing
It’s not just fields that benefit. Chemical plants rely on ammonium carbamate for making urea-formaldehyde resins and plastics, which later turn into adhesives or bonding agents. These products end up holding together particleboard, plywood, and laminated furniture. Many of us have these things in our homes—maybe without ever thinking about what’s in the glue.
Outside the building sector, ammonium carbamate also plays a role in textile treatment, surfacing in garments, carpets, and draperies as resins harden and finish the weave. If you’ve marveled at a new shirt’s crispness, resin chemistry probably deserves a nod. This application doesn’t grab headlines, but the everyday convenience comes from smart use of chemistry.
Environmental and Safety Concerns
The value of ammonium carbamate doesn’t gloss over its downsides. It can decompose easily, giving off ammonia—a gas that harms the lungs and causes headaches in closed spaces. I remember walking through an old warehouse where ammonia leaks left a strong chemical sting in the air. The experience made it clear how important proper ventilation and storage are. Chemical spills aren’t just news stories; they’re risks that real people face while handling these substances.
Nitrogen runoff worries a lot of people, including scientists and local fishers. Fast growth in crops sometimes brings unintended side effects—algae blooms choking rivers and lakes after fertilizer leaches from the soil. Solutions exist: precision application, slow-release formulations, and tighter safety standards can all cut waste and shield waterways. Some regions already use sensors and satellite imagery to match fertilizer doses closely to what the crops need.
Pushing for Smarter Solutions
Changing the status quo takes work, but new technology is reshaping the landscape. Controlled-release fertilizers, for example, keep nitrogen locked in the soil longer, which means less runoff and more bang for the buck. Research drives the push for alternatives that work just as well but leave a lighter mark on the environment.
In my own family’s small garden, experimenting with organic compost showed firsthand how local nutrient cycling can help the soil without heavy Reliance on chemicals—though on a scale like commercial agriculture, safe and smart use of ammonium carbamate and related compounds still fills an important gap.
Between keeping global food supplies stable and supporting the industries around us, ammonium carbamate stands as a quiet powerhouse, but one that demands respect for both its benefits and its risks.
What is the chemical formula of Ammonium Carbamate?
Why Ammonium Carbamate Matters in Agriculture and Industry
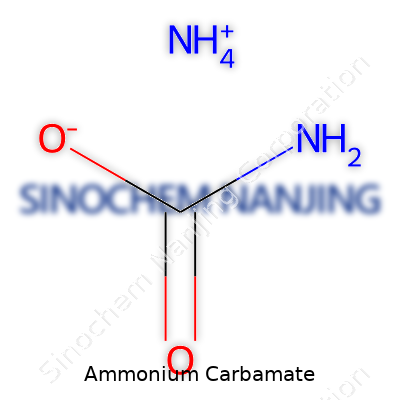
Ammonium carbamate is more than just a line in a chemistry textbook. Every time I talk to a farmer concerned with soil nutrition or an engineer designing a cleaner industrial process, the topic often circles back to essential compounds like ammonium carbamate. With the formula NH4CO2NH2, this salt finds real jobs on farms and in factories. The history of synthetic fertilizers and nitrogen cycling owes a lot to chemicals like this one. For good or for bad, it forms the backbone of countless urea and fertilizer manufacturing processes.
Behind the Formula: Simple Structure, Big Role
NH4CO2NH2 may look like an unassuming string of letters and numbers, but this formula represents the salt that forms directly from ammonia and carbon dioxide. When working with fertilizer manufacturers, I’ve seen how they rely on this compound because it breaks down quickly and feeds plants with both nitrogen and carbon. In plants, nitrogen stays in high demand, and ammonium carbamate—by its nature—supplies it efficiently. Urea plants use the reaction between ammonia and carbon dioxide to first create ammonium carbamate before converting it into urea. This means the formula sits at the heart of huge, global operations feeding millions.
Practical Chemistry: Making and Using Ammonium Carbamate
From firsthand experience in both research labs and field research, ammonium carbamate acts almost like a bridge. Its presence links simple raw ingredients to more complex end products. Chemically, it forms under specific temperatures and pressures. I’ve watched teams adjust conditions in real time during ammonia and carbon dioxide reactions to get the highest yield possible. This isn’t just academic work; the stakes are tangible. More efficient production here means cheaper, more available fertilizers for people everywhere.
Risks and Responsible Handling
Every chemical offers benefits and brings risks. Ammonium carbamate breaks down into ammonia and carbon dioxide if left at room temperature, especially in open air. During a stint at a pilot plant, we constantly measured its stability because ammonia gas poses clear risks if released. Proper storage—cool, sealed environments—keeps both workers and surroundings safe. Large-scale use often means there’s always vigilance regarding air quality and occupational safety.
Environmental Considerations and Future Solutions
The environmental side of things can’t be ignored. Ammonium carbamate plays a part in the larger nitrogen cycle concerns, including runoff and greenhouse gas production. Scientists keep looking for sharper ways to capture emissions during its use and to recycle carbon dioxide and ammonia more efficiently. In my consulting work, companies have experimented with closed-loop systems that cut losses and reduce overall environmental loads. Tracking the full life cycle of this compound—from raw material to final application—guides smarter policy and innovation. Solutions start with thoughtful research and the willingness to adapt old practices in light of new science.
Trustworthy Handling and Continuous Improvement
With decades of combined public research and lived professional experience backing up its safety and efficiency record, ammonium carbamate keeps its place in many industries. Reliable sources like peer-reviewed studies and industry best-practice guidelines remain essential for anyone involved in its use or regulation. Chemical knowledge, practical skill, and a commitment to transparent safety measures make all the difference when dealing with such a fundamental compound. As new tools and regulations emerge, staying updated helps protect both people and the environment.
Is Ammonium Carbamate hazardous or toxic?
What Actually Is Ammonium Carbamate?
Ammonium carbamate comes into play as a white, crystalline solid. It pops up as an intermediate in the production of urea, especially in fertilizer plants. Anywhere synthetic urea is made, this compound runs through the pipes, reacting easily with water, carbon dioxide, and ammonia. Its use does not usually raise eyebrows in the daily work of agriculture or industry, but any chemical like this deserves a closer look.
Health and Safety Risks
I’ve worked near industrial settings, and there’s always an undercurrent of concern with anything carrying the “ammonium” label. Ammonium carbamate can give off ammonia gas, especially in warm or humid conditions. Ammonia gas irritates eyes, nose, and throat fast. Even at low concentrations, exposure in a poorly ventilated room can cause coughing and watery eyes. Larger concentrations, which aren’t hard to reach in a small spill or unsealed bag, can trigger real respiratory issues or burns to skin and eye tissue.
Getting it on your hands or in your eyes leads to stinging or possible burns. If swallowed, it can bring on pain, nausea, or worse if enough gets in. Long-term inhalation in workplaces where there’s dust or repeated gas exposure may cause chronic irritation or breathing trouble. Workers need to treat powdered ammonium carbamate with the same respect given to caustic cleaning agents.
Environmental Concerns
This chemical breaks down rapidly, releasing ammonia and carbon dioxide. Once released, ammonia does not simply disappear. It finds its way into the air, and those strong-smelling vapors affect lungs, eyes, and even sensitive plants nearby. Laying ammonium carbamate on fields, letting it build up in soil or wash into streams, adds unnecessary ammonia loads to the environment, which can spur on algae blooms and upset the balance of local water bodies. Fish and aquatic life start to struggle when ammonia creeps up.
Facts from Studies and Practice
Research has pointed out that single exposures don’t usually create long hospital lists, but the chemical has a history of contributing to workplace incidents. One CDC record showed episodes of cough and disorientation from ammonium carbamate dust, especially in older, cramped facilities. In 2008, an industrial safety review in North America flagged a leak that sent a handful of workers to urgent care. No tragic headlines, yet the issues trace back to improper handling and storage.
It’s not listed as a “major hazard” like some heavy metal or cyanide, but it earns respect in safety training. Ammonium carbamate’s data sheets lay out PPE requirements—gloves, goggles, and, for airborne dust, a mask rated for ammonia. Facilities need exhaust fans and spill kits ready, not tomorrow, but every morning.
Better Practices and Personal Responsibility
Every industry wants to speed up processes, but a single shortcut making contact with ammonium carbamate could cost health and money. If I were advising a new plant supervisor, I'd push for tight training on ventilation, clear labeling, and daily checks for leaks. Environmental monitoring, like regular air sampling for ammonia, keeps surprises off the schedule.
Access to information matters, too. Safety Data Sheets should be posted, not buried in a folder. People respond better when they see, not just hear, instructions—eye wash stations in plain sight, pictograms on containers, routines for spill cleanup practiced, not only described.
With ammonium carbamate, it’s not dramatic disaster that’s looming—just routine, avoidable harm to lungs, eyes, or water. Respecting what’s on the label keeps everyone safer at home, work, or out in the field.
How should Ammonium Carbamate be stored and handled?
Understanding the Real Risks
Ammonium carbamate might look harmless on the outside—a white solid—yet once you spend time in any lab or plant, you start to see there’s more to it. The chemical isn’t just a basic fertilizer intermediate. It breaks down easily to ammonia and carbon dioxide, both of which can cause real headaches if they get loose in a workplace. Breathing in ammonia will sting your nose and eyes and make it hard to catch a breath. I've worked in spaces where a few careless minutes with storage led to a harsh, eye-watering smell spreading through the air. That stays with you, both the lesson and the discomfort.
Why Temperature and Ventilation Really Matter
Heat speeds up the breakdown of ammonium carbamate. Even a warm garage can nudge it along, raising the pressure inside closed containers and creating a risk of sudden gas release. I remember one small facility using metal drums without paying close attention to the temperature—and after a stretch of summer days, a few barrels started to swell. The point was clear: cool, dry storage isn’t just a guideline; it prevents both spoilage and dangerous leaks.
Good airflow does more than replace stale air. It dilutes any gases that escape. Trying to get by with closed, stuffy stockrooms almost always leads to someone getting a whiff of something they shouldn’t. Every facility I’ve trusted with chemicals runs ventilation fans in storage rooms and uses warning monitors for ammonia. These features keep both air and conscience clear.
The Importance of Containers and Labels
Corroded, cracked, or unlabeled containers tempt fate. Sealed, corrosion-proof drums work best. Plastic or stainless steel stands up to the chemical over time—no rust, no leaks. With the right labels and hazard markings, nobody grabs the wrong barrel or mistakes its contents. More than once, I’ve seen small teams abandon original packaging for “just a few days,” forgetting that moisture in the air creeps in. That chalky, clumped powder is not what the safety sheet promised. Keeping chemicals in their original, sealed containers saves a lot of panic and waste.
Protecting People: Training and Emergency Steps
What’s written on a safety data sheet means nothing unless people read and remember it. I’ve watched newcomers work around chemicals and learn the hard way why we use goggles, gloves, and a good apron. Ammonium carbamate doesn’t burn like acid, yet it eats away at skin and eyes if given the chance. Proper personal protective equipment stops most injuries before they start. Regular training, plain talk, and job-specific demos cut down on carelessness.
The smart approach also includes knowing the quickest way out, the spot of the emergency eyewash, and the number to call. Anyone who’s seen ammonia splash understands why rinsing and quick medical attention count far more than reports after the fact.
Protecting the World Outside the Lab
Chemicals rarely stay put if stored or handled poorly. Spills end up in the ground, water, or air, and those effects reach beyond any single workspace. After a poorly contained leak many years ago, our town water supply registered chemical traces. Remediation cost more than proper handling ever would have.
Smart storage and careful handling don’t just keep workers safe. They protect local communities and the environment. It doesn’t take complex gear—just common sense, sturdy containers, and respect for the risks. Reliable practices prevent accidents and build trust in every facility, no matter its size.
What is the solubility of Ammonium Carbamate in water?
Understanding How Ammonium Carbamate Mixes with Water
Walk into a fertilizer plant or a college chemistry lab and you’re likely to find ammonium carbamate on a shelf. The stuff grabs attention because of its part in the urea production process and some of the ways it can break down under the right conditions. One thing people often ask about is how much of this compound actually dissolves in water, since solubility means everything for folks mixing solutions, figuring out dosing, or trying to avoid unwanted precipitation during manufacturing.
A Look at the Numbers: Solubility Facts
At room temperature—around 25°C—ammonium carbamate dissolves in water up to about 120 grams per 100 milliliters. That’s a lot; think about dumping three large handfuls of table salt into a cup of water without hitting the limit. This solubility drops as water gets cooler, which becomes a headache in storage rooms during winter.
Solubility flips fast when things start heating up. The compound breaks apart above 60°C, giving off ammonia and carbon dioxide. That’s more than just an academic tidbit: workers in plants handling bulk ammonium carbamate have to keep a close eye on both temperature and ventilation because that sudden boost in gas can lead to pressure problems, not to mention safety hazards from ammonia vapor.
Why Solubility of Ammonium Carbamate Matters
In agriculture, getting the right mix into irrigation water or fertilizer blends hinges on making sure the salt actually dissolves. Clumps left in tanks mean uneven crop nutrition. In industrial urea production, solubility decides how efficiently processes run and whether scaling or blockages form in pipes.
Pharmaceutical labs and research teams have their own headaches. Ammonium carbamate sometimes becomes a precursor or an intermediate in small-scale reactions, and knowing the limit before you end up with unplanned crystals matters for yield and purity. Still, I remember neighbors who ran an organic test plot being surprised at cloudy irrigation water, which turned out to be precipitation from mixing a little too much of this compound.
Risks and Environmental Concerns
Dissolving large quantities of ammonium carbamate means dealing with ammonia fumes if water gets too warm. Toxicity isn’t much of a worry for plants, as ammonium and carbamate ions are part of natural nitrogen cycles, but for human health, ammonia inhalation should not be taken lightly. Farmers and technicians sometimes ignore that thin line between a stable mixture and a chemistry mess. In some facilities, I saw warning signs near storage tanks about keeping temperatures steady and rooms well-aired.
Making Best Use of Solubility Knowledge
For anyone working with ammonium carbamate, it helps to keep solutions cool and capped. If working in a busy plant, monitoring room temperature and ventilation becomes about more than local comfort—it’s about safety. For educators, bringing a sample into the lab and letting students watch crystals vanish in water can drive home those numbers from the textbook. Watching a real reaction helps ground lessons in reality.
Solubility stands at the center of user experience with this compound, from guaranteeing the complete absorption of raw material into a fertilizer tank all the way to ensuring smooth, safe operation in industry. Knowing the upper limit—120 grams per 100 milliliters at room temperature—lets users make smart decisions about mixing, storage, and safety, all grounded in practical, everyday concerns.
| Names | |
| Preferred IUPAC name | azanium;carbamate |
| Other names |
Diammonium carbamate Carbamic acid diammonium salt Ammonium hydrogen carbamate |
| Pronunciation | /əˈmoʊniəm kɑːrˈbeɪmət/ |
| Identifiers | |
| CAS Number | 1111-78-0 |
| Beilstein Reference | 505203 |
| ChEBI | CHEBI:62998 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 54754 |
| DrugBank | DB11349 |
| ECHA InfoCard | ECHA InfoCard: 100.011.685 |
| EC Number | 208-058-0 |
| Gmelin Reference | 15980 |
| KEGG | C01343 |
| MeSH | D000646 |
| PubChem CID | 62668 |
| RTECS number | BO0875000 |
| UNII | TZ4LOL8K3J |
| UN number | UN3077 |
| Properties | |
| Chemical formula | NH4CO2NH2 |
| Molar mass | 78.07 g/mol |
| Appearance | White crystalline solid |
| Odor | Ammonia odor |
| Density | 1.527 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -5.0 |
| Acidity (pKa) | pKa ≈ 8.3 |
| Basicity (pKb) | 3.40 |
| Magnetic susceptibility (χ) | -41.5e-6 cm³/mol |
| Refractive index (nD) | 1.434 |
| Dipole moment | 3.8 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −426.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –629.4 kJ/mol |
| Pharmacology | |
| ATC code | NH4HCO2 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H319, H315 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Autoignition temperature | > 130 °C (266 °F) |
| Lethal dose or concentration | LD50 oral (rat) 1300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1300 mg/kg |
| NIOSH | SU8375000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Ammonium Carbamate: "15 mg/m³ (total dust), 8-hour TWA (OSHA) |
| REL (Recommended) | 4 mg/m³ |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium carbonate Ammonium bicarbonate Ammonium cyanate Ammonium carbazate Urea |

